Abstract
BACKGROUND:
CDT1 is the essential regulator of the initiation of DNA replication. Overexpressed CDT1 can cause DNA damage through re-replication. However, the function of CDT1 in prostate cancer (PCa) development has not been established.
METHODS:
Through bioinformatics, expression levels of CDT1 were found to be higher in metastatic PCa when compared to primary PCa. Then, immunohistochemical staining confirmed that the expression of CDT1 was significantly correlated with the occurrence of distant metastasis. For PCa cells, we established a stable clones knockdown CDT1. MTT was used in analyzing the proliferation ability of cells. Migration as well as invasion assays were performed. Effects of CDT1 knockdown on the cell cycle were evaluated by flow cytometry. Expression levels of EMT-associated markers in PCa cells were determined by Western blotting. And PI3K/AKT/GSK3
RESULTS:
Over expression of CDT1 in PCa cells enhanced cell migration, invasion, tumor metastasis and was correlated with cell cycle regulation. Our results showed that knockdown of CDT1 inhibited G1 to S phase transition and induced the G1 phase cell cycle arrest in PCa cells. Moreover, it upregulated the expressions of epithelial markers (E-cadherin) and down-regulated mesenchymal markers (including Slug, N-cadherin, MMP2, vimentin, Snail, and MMP9) via regulating the phosphorylation level of PI3K, AKT and GSK3
CONCLUSIONS:
CDT1 promotes PCa cell metastasis by promoting cell cycle and PI3K/AKT/GSK3
Introduction
Globally, prostate cancer (PCa), the most prevalent tumor among men, is the 2
Cell cycle regulator and DNA replication factor 1 (CDT1) is a central regulator of DNA replication initiation [3]. It regulates cell transition into the S phase and stops the cell cycle in the mitotic phase when there is a lack of replication [4]. In normal mammalian cells, when DNA is damaged, CDT1 inhibits the cell cycle into S phase through the P53 signaling pathway [5]. In tumor cells, this pathway is dysfunctional, and CDT1 accumulates at the site of DNA damaged [5]. Overexpressed CDT1 can cause DNA damage through re-replication. It affects cell cycle progression as well as responses to cellular DNA damage. High expression of CDT1 has a positive correlation with malignancy degree and a negative correlation with the survival cycle of many tumors, such as colon cancer, breast cancer, non-small-cell lung carcinomas and hepatocellular carcinoma [6, 7, 8, 9]. It has also been reported that CDT1 plays a role in PCa occurrence and progression, however, its role in cell cycle progression remains unclear [10, 11].
According to current research, wnt/
We found that, in mPCa patients, CDT1 is overexpressed and has a positive correlation with high Gleason scores (GS) as well as poor prognostic outcomes. The results support the key role of CDT1 in PCa progression and metastasis. We found that CDT1 acted on the initiation of cell cycle replication and promoted cell proliferation. We also found that CDT1 can induce EMT via PI3K/AKT/GSK3
Materials and methods
Gene expression levels and survival analyses
Four data sets [GSE35988,GSE74367,GSE6752,and GSE27616] [17, 18, 19, 20] were evaluated using GEO2R (
Online tools were performed to clarify the gene expression levels. Expression of CDT1 in TCGA_PRAD based on nodal metastasis status have been featured in UALCAN (
Immunohistochemical staining and scoring analyses
PCa diagnosis was independently performed by two pathologists. The TNM stage as well as Gleason scores of PCa samples were verified. Approval was attained from Affiliated Hospital of Chifeng University’s Committees for Ethical Review of Research Involving Human Subjects.
Immunohistochemical analyses (IHC) of CDT1 expressions were done on needle biopsy samples at the Affiliated Hospital of Chifeng University Department of Pathology.
PCa tissue paraffin sections were deparaffinized, then, hydrated. Antigen retrieval was done through the microwave method. Blocking of endogenous peroxidase activities was done by incubation of slides in 0.3% hydrogen peroxide. After incubation in the presence of primary as well as secondary antibodies, tissue sections were developed using peroxidase as well as 3,3
CDT1 expressions in PCa samples were blind determined by two independent pathologists. The immunostaining intensities for each sample were distributed into: negative
Cell culture, transfections, and regents
PCa cell lines (LNCaP, PC3, DU145) were acquired from the American Type Culture Collection (ATCC, Manassas, VA, USA), cultured in RPMI-1640 medium supplemented with 10% fetal bovine serum (BI, Cromwell, CT, USA) and 1% penicillin-streptomycin solution. Cell incubation was under the condition of 37
The siRNA sequences
The siRNA sequences
The primer sequences
The Trizol reagent (Invitrogen) was used to isolate RNA as instructed by the manufacturer. Then, cDNAs were synthesized by using a reverse transcription kit (Roche) and analyzed by quantitative real-time PCR (qRT-PCR) using the Applied Biosystems 7900 Real-Time PCR System (Thermo Scientific) and the SYBR Green PCR Master Mix (Roche) as instructed by the manufacturers. The internal control was GAPDH. Primer sequences are shown in Table 2.
CDT1 expression was elevated in mPCa, compared to primary PCa, and was significantly associated with metastasis, Gleason score and shorter survival. In all four data sets, CDT1 expression was elevated in mPCa, compared to primary PCa (A-E). In the TCGA database, CDT1 expressions were significantly elevated in patients with lymphatic metastasis compared to those without metastasis (F). In TCGA database, CDT1 expression was positively correlated with Gleason score (G). In the TCGA database, CDT1 expression was positively associated with shorter survival (H).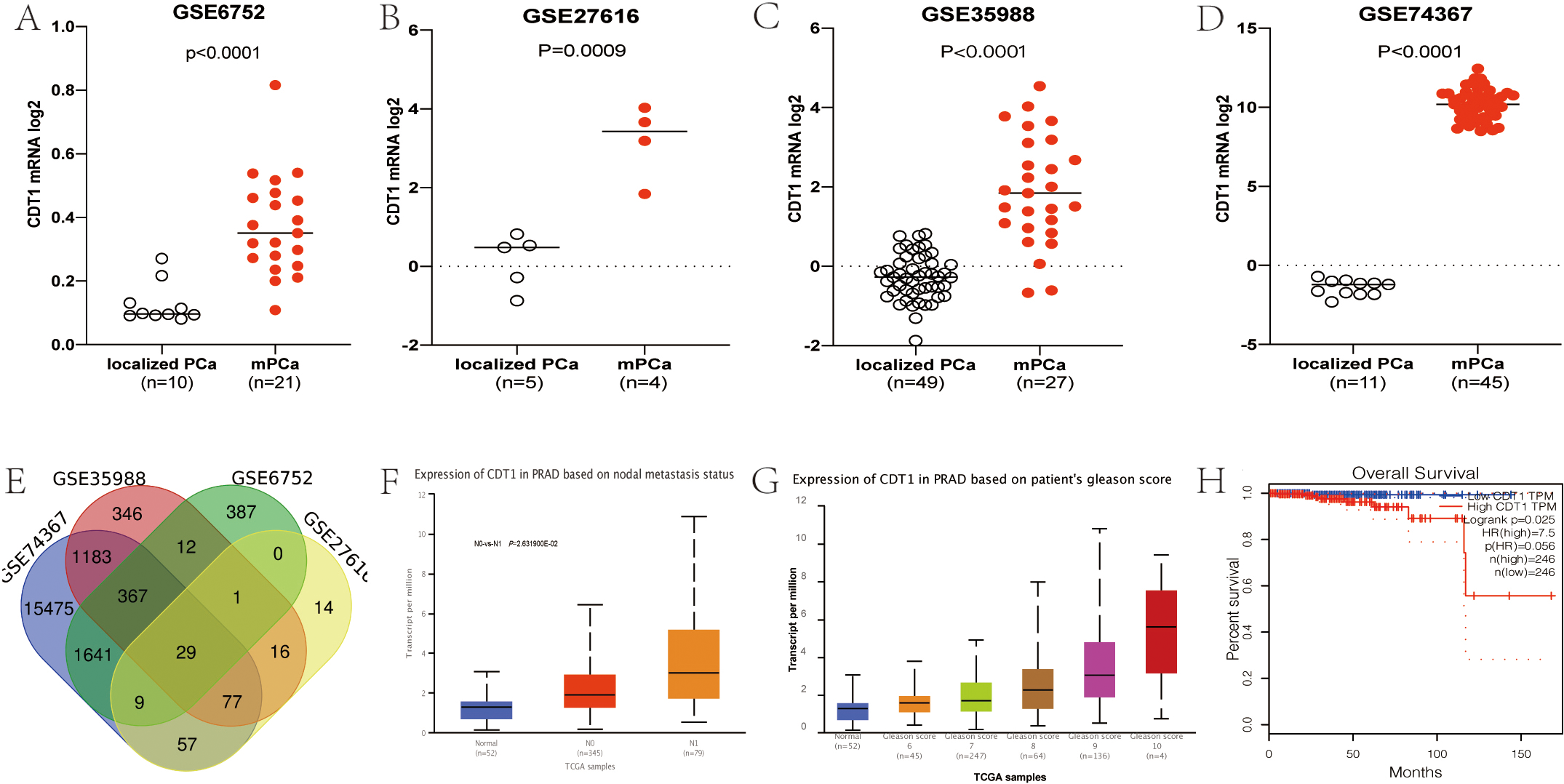
Cold PBS (3 ml) was used to wash the cells, after which they were lysed with RIPA lysis buffer. Protein samples were separated on 10% SDS/PAGE gel and transferred to PVDF membranes. The membranes were incubated for 1 h in fat-free milk (5%) at room temperature, washed three times using the lysis buffer followed by overnight incubation at 4% in the presence of specific primary antibodies. Then, incubation of membranes was done for 1 hour in the presence of secondary antibodies at room temperature. After TBST washing, the membrane was prepared for exposure and enhanced chemiluminescence (ECL) reagent (cat. no. 32106; Thermo Scientific, USA) was employed for band visualization. Primary antibodies were: CDT1 (Cell Signaling Technology, Catalog: 8064), E-cadherin (Cell Signaling Technology, Catalog: 14472), N-cadherin (Cell Signaling Technology, Catalog: 13116), Slug (Cell Signaling Technology, Catalog: 9585), Snail (Cell Signaling Technology, Catalog: 3879), MMP2 (Cell Signaling Technology, Catalog: 40994), Vimentin (Cell Signaling Technology, Catalog: 5741), MMP9 (Cell Signaling Technology, Catalog: 13667), GAGDH (Cell Signaling Technology, Catalog: 5174), PI3K (abcam, Catalog: ab179463), p-PI3K (abcam, Catalog: ab278545), p-AKT (abcam, Catalog: ab38449), AKT (abcam, Catalog: ab8805), p-GSK3
MTT assay
Cells culture was done in 96-well plates. MTT solution (5 mg/ml) was prepared in advance and stored at 4 degrees refrigerator. Cell incubation was at 37
Cell migration and invasion assays
Migration assays were conducted in 24-well culture plates using Transwell chambers (Corning). Cells culture was done in chambers with RPMI-1640 medium and 10% fetal bovine serum (FBS). The medium with 20% FBS was placed in each well under the chambers. After 48 hours, Transwell chambers were collected to observe the migrated number of cells under a microscope. Polyethylene terephthalate (PET) membranes with pore diameters of 8-
Cell cycle assay
Cells seeding at 1
Statistical analyses
Comparisons of means was done using one-way or two-way ANOVAs followed by post hoc for multiple comparisons test and unpaired two-tailed Student’s
Results
CDT1 is highly expressed in metastatic tissues than in primary tissues and correlates with poor prognostic outcomes for PCa patients
Through GEO2R analysis of the four data sets, CDT1 expressions were found to be significantly elevated in metastatic lesions, when compared to the primary tumor (Fig. 1A–E). Further verified by TCGA-PRAD database, its expression level in lymphatic metastasis was significantly elevated when compared to the primary tumor (Fig. 1F), with positive correlations with the GS (Fig. 1G). Patients whose CDT1 expressions were elevated exhibited significantly shorter overall survival outcomes (Fig. 1H), implying that elevated expressions of CDT1 correlate with PCa progression.
Representative image of Immunohistochemical staining. Immunostaining intensities for each sample were allocated into negative = 0 (A), weak 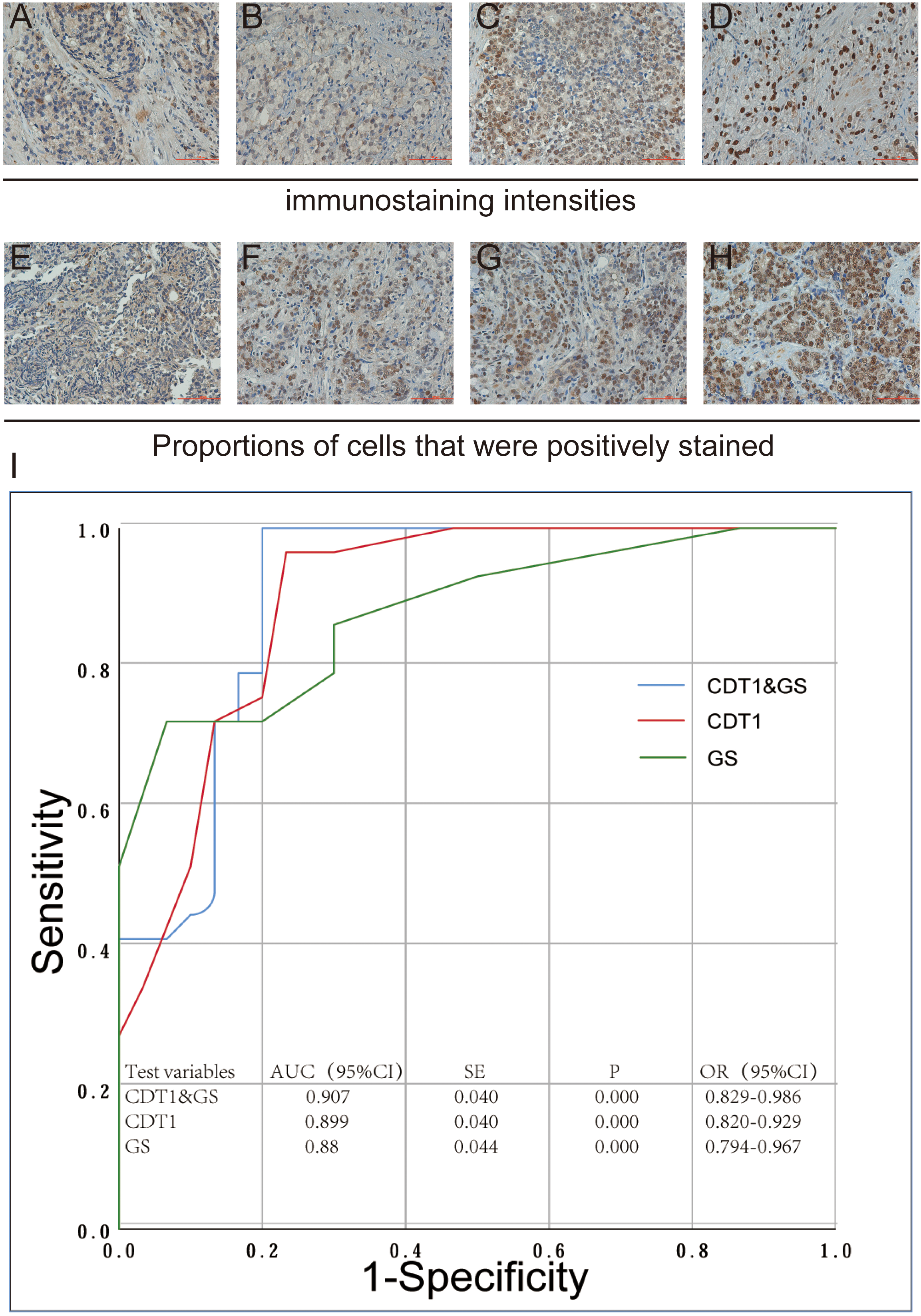
We investigated the relationship between CDT1 and metastasis by IHC staining. We selected 60 cases of PCa patients from Affiliated Hospital of Chifeng University, and collected prostate biopsy specimens for IHC staining. There were 30 cases of primary PCa and 30 cases of mPCa. The results of IHC staining indicated that the factors related to staining intensity and proportion were GS, T stage, lymph node metastasis and distant metastasis (Table 3).
Bivariate analysis of CDT1 immunoreactivity in human prostate cancer samples
Bivariate analysis of CDT1 immunoreactivity in human prostate cancer samples
Furthermore, we made logistic regression and receiver operating characteristic (ROC) curves with IHC staining score and GS as independent variables and distant metastasis as dependent variables. The results demonstrated that CDT1 immunoreactivity could indeed be a predictive marker of distant metastasis and the cut-off value of IHC Staining score was 7.5 (AUC: 0.899;
To establish the function of CDT1 in PCa, qRT-PCR and western blots were performed. PCa cell lines, such as LNCaP, which is an androgen-sensitive PCa cell line, as well as PC3 and DU145, which are androgen-insensitive cell lines with a high malignant potential were evaluated.
All suggested that the expression of CDT1 was low in the primary tumor LNCaP cell lines, and high in PC3 as well as DU145 cell lines (Fig. 3A). Furthermore, CDT1 protein expression was dramatically upregulated in PC3 as well as DU145 cell lines compared to LNCaP cell lines by Western blotting (Fig. 3B). CDT1 knockdown in PC3 as well as DU145 cells were established by lentiviral transfection. The transfection efficiency was verified by QRT-PCR (Fig. 3C) and Western blot (Fig. 3D). Cell proliferation levels and viabilities were assessed by the MTT assay (Fig. 3E) and clone formation experiments (Fig. 3F). The results showed that cell proliferation levels and viabilities to be significantly low in CDT1 knockdown cells. In addition, CDT1 knockdown cells had significantly suppressed migration abilities (Fig. 3G). Besides, invasion activities and adhesion abilities of CDT1 knockdown cells were significantly suppressed when compared to the control group (Fig. 3G). Therefore, CDT1 is majorly involved in PCa cell proliferation as well as metastasis.
CDT1 has a regulatory role on the expressions of cell cycle-associated genes in PCa cells
To investigate molecular mechanisms involved in CDT1-associated PCa cell proliferation, GSEA was performed to evaluate the effects of CDT1 expressions in the TCGA-PRAD database. The result shows that CDT1 involved in cell cycle, with normalized enrichment score (NES)
CDT1 promotes EMT in PCa cells
We used western blot analysis to detect EMT-associated markers. Compared to the control group, PC3 cells that had been transfected with si-CDT1 exhibited weaker migratory capabilities (Fig. 3G). Treatment of PC3 cells with combined si-CDT1 significantly suppressed cell migration and invasion abilities. These findings were comparable to those found in DU145 cells (Fig. 3G). Furthermore, expressions of EMT proteins (Vimentin and N-cadherin) as well as other migration-associated genes (Slug, MMP2, Snail and MMP9) were suppressed in PCa cell lines treated with si-CDT1. Expression levels of these proteins were significantly suppressed after combined treatments of PC3 cells and DU145 cells with si-CDT1. Moreover, expression levels of E-cadherin in PCa cells were elevated when compared to control cells (Fig. 5A–C). Further research showed the phosphorylation level of PI3K, AKT and GSK3
CDT1 knockdown can inhibit cell proliferation, invasion and metastasis. Expression levels of CDT1 in LNCap, PC3 and DU45 cell lines were determined by qRT-PCR (A) and western blot (B). Knockdown efficiency was verified by qRT-PCR (C) and Western blot (D). The MTT assay was done to evaluate cell proliferation after CDT1 knockdown (E). Transwell assays were done to evaluate cell migration as well as metastasis after CDT1 knockdown (F).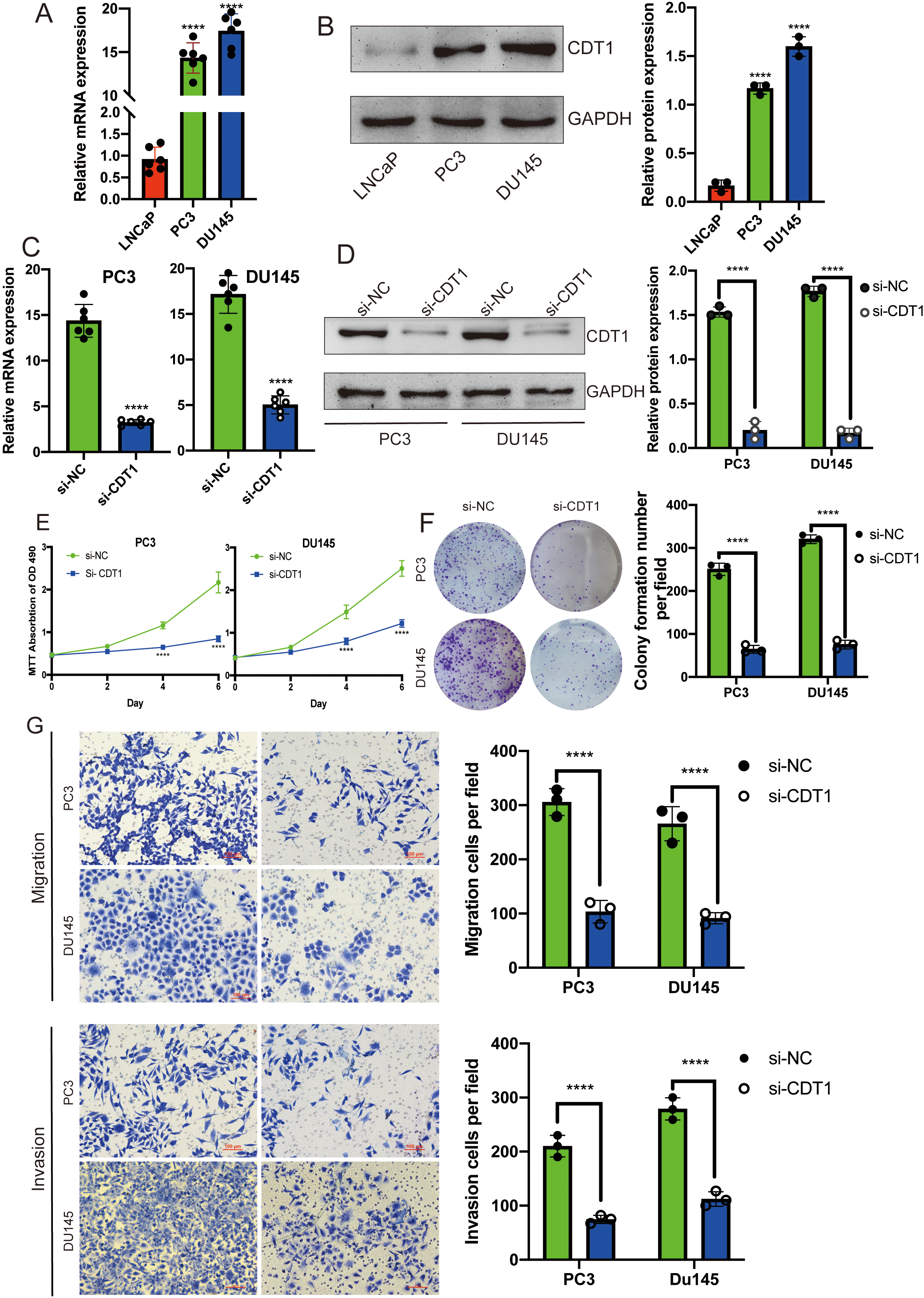
Cell cycles were measured by flow cytometry. When CDT1 was knocked down in PC3 (A) as well as DU145 (B) cell lines, the proportion of G1 phase increased while that of S phase decreased. Data are shown as mean 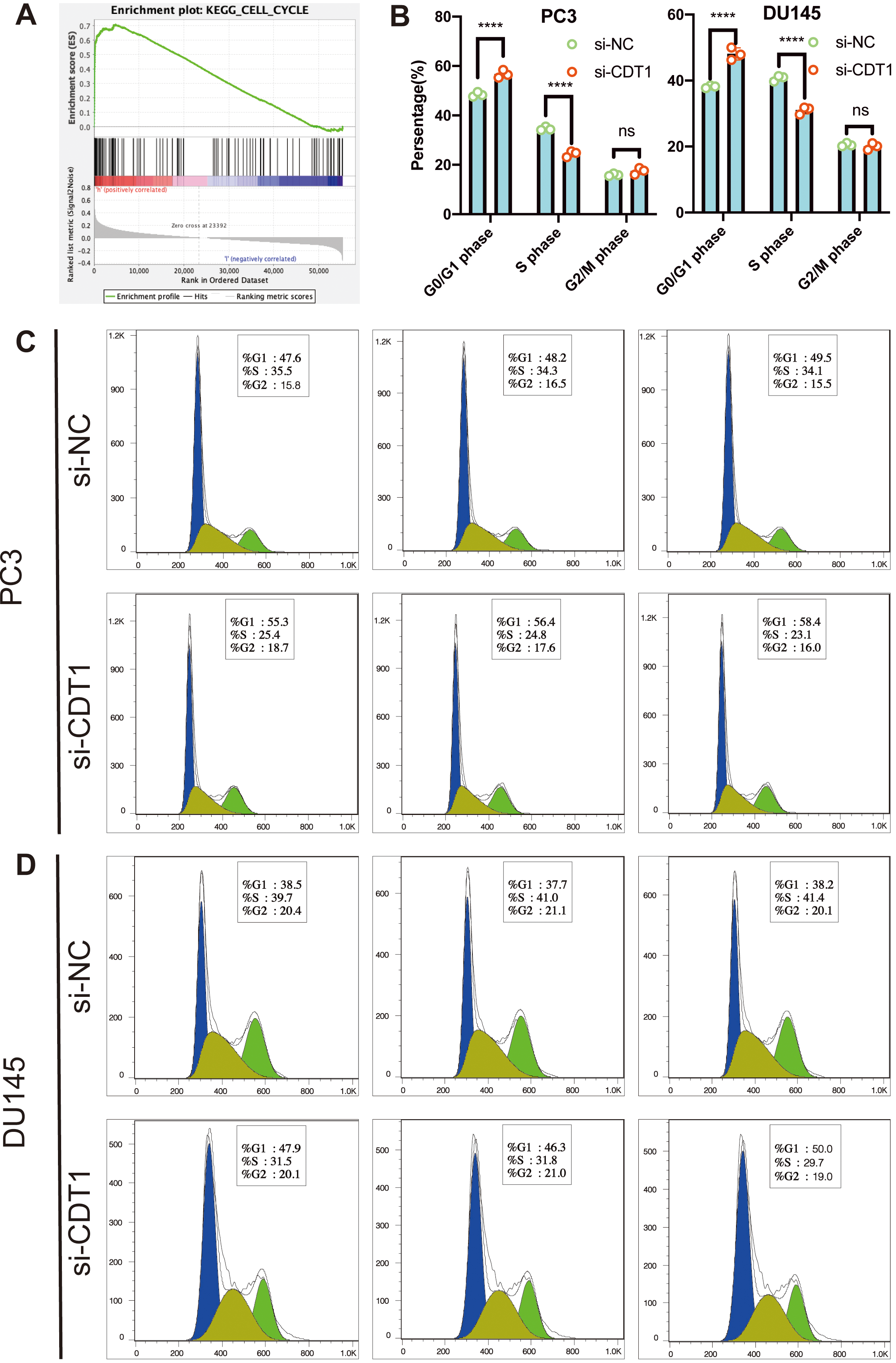
EMT-associated proteins and PI3K/AKT/GSK3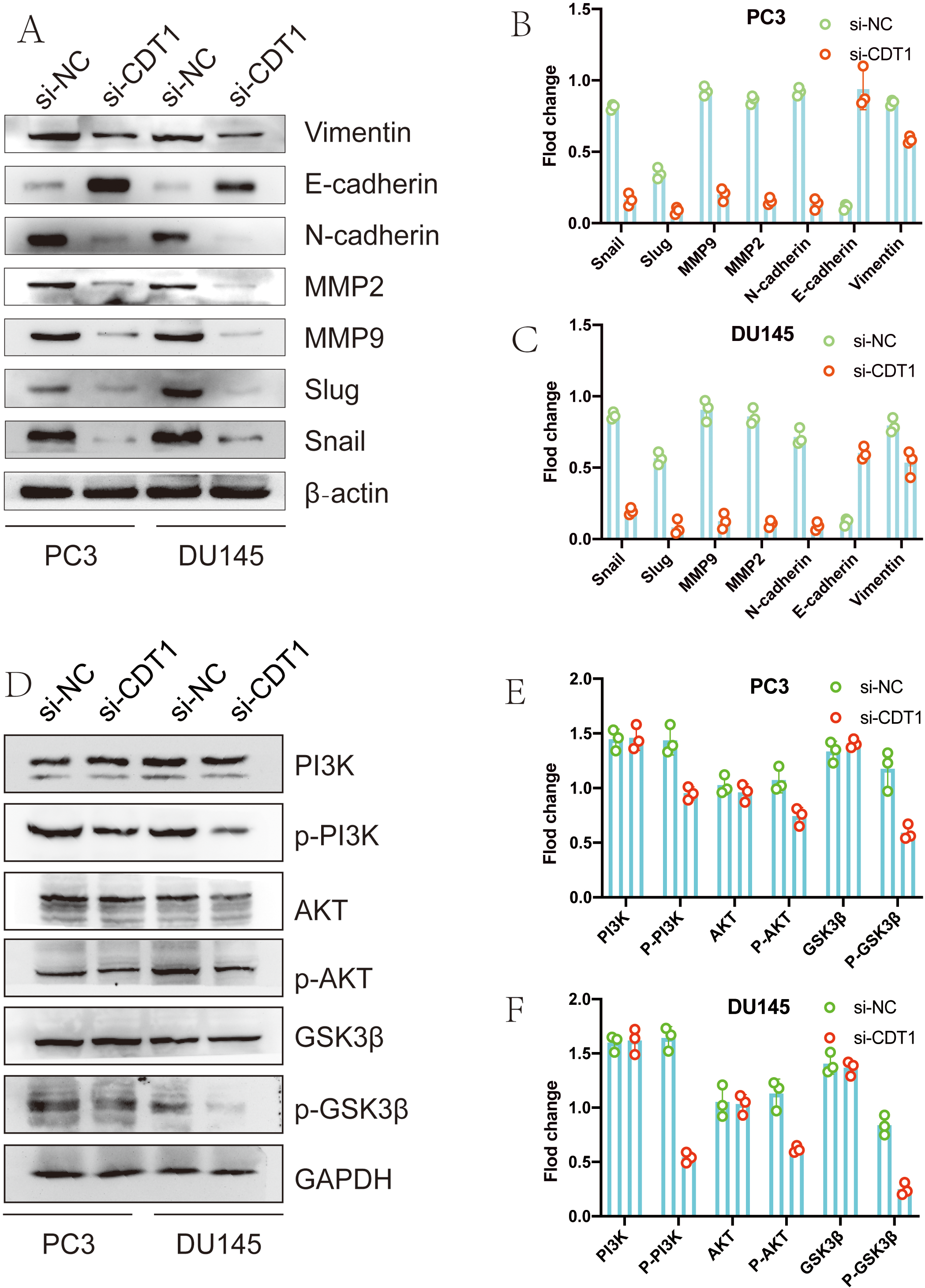
In may tumors, such as breast cancer, lung cancer, hepatocellular carcinoma and colon cancer, CDT1 expressions are highly elevated and are associated with poor survival [6, 7, 9, 24]. Our study also confirmed that CDT1 expressions are elevated in PCa and are correlated with Gleason score as well as poor survival. According to the TCGA database, CDT1 expression in patients with lymphatic metastasis of PCa was significantly elevated than in non-metastatic patients. We also performed immunohistochemical verification with our specimens, and the results were consistent with the TCGA data.
According to the results of GSEA, CDT1 was related with the cell cycle. Cell cycle studies using flow cytometry and demonstrated that knockdown of CDT1 in PCa inhibited cell cycle transformation from G1 phases to S phases. Alexandra Kanellou et al. [4] showed that CDT1 is important for exclusively loading the minichromosome maintenance 2–7 helicase complex onto chromatin during the G1 cell cycle phase, thereby regulating DNA replications to once in each cell cycle. Yiru et al. [25] reported that CDT1 is regulated by FOXO3 to make sure DNA is only replicated once in each cell cycle. Expressions of CDT1 leads to re-replication, which leads to tumor genesis and progression. Pegah et al. [26] showed that FBXO31 could promote the ubiquitination of CDT1 and further degrade it.
Five-year survival rates for non-metastatic PCa have been reported to be about 98%, but only 28% for mPCa [1, 27]. Elucidating the PCa metastasis mechanism will improve the survival rate of patients. High expressions of CDT1 were found in patients with mPCa, and high expression of CDT1 was also closely associated with metastasis in other tumors [6, 28]. This could be a gateway to control prostate cancer metastasis. EMT, a gradual cellular process that occur in several states, plays vital roles in cancer metastasis [29]. Subsequently, we examined the effect of knockdown CDT1 on EMT and PI3K/AKT/GSK3
Conclusion
In summary, we found the high expression of CDT1 in PCa by bioanalysis, and did the analysis related to survival, and did the mutual verification of multiple databases. Subsequently, we performed immunohistochemical validation with our own samples, with the findings being consistent with those of the bioanalytical data. We then conducted cell experiments to confirm that CDT1 can promote cell cycle and EMT process, explaining the possible mechanism of its promotion of PCa metastasis. So far, therapeutic strategies targeting CDT1 for cancer treatment have been inefficient. Therefore, CDT1 is a potential target for PCa metastatic therapy.
Funding sources
Fund program: Scientific research projects of the Inner Mongolian higher educational system (NJZY 20197).
Author contributions
Conception: Chunhui Wang, Jizhong Che and Ying Jiang.
Interpretation or analysis of data: Chunhui Wang, Jizhong Che and Ying Jiang.
Preparation of the manuscript: Chunhui Wang.
Revision for important intellectual content: Chunhui Wang, Jizhong Che, Ying Jiang and Guochang Bao.
Supervision: Chunsheng Li.
Footnotes
Acknowledgments
The authors are deeply thankful to Dr. Yi Shao of Tianjin Medical University (Tianjin, China) for his earnest guidance
