Abstract
Background:
Cryoablation, an effective means of ablating cancer, is often used in conjunction with adjuvants that target cancer cells in a specific cell cycle stage to increase treatment efficacy. The objective of this study was to investigate the impact of cell cycle stage on cancer freeze response as well as investigate the potential cellular kinetic effect of calcitriol, the active metabolic of vitamin D3, when used as a cryosensitizing adjuvant in order to maximize prostate cancer cell death.
Methods:
Cell cycle distribution of PC-3 cells was analyzed via flow cytometry to compare gap 1, synthesis, and gap 2/mitosis phase subpopulations pre- and postfreeze as well as changes elicited by calcitriol pretreatment. Distinct gap 1, synthesis, and gap 2/mitosis phase populations were obtained through fluorescence-activated cell sorting and synthesis phase thymidine synchronization. Posttreatment viability was assessed using alamarBlue and fluorescence microscopy to assess live, apoptotic, and necrotic subpopulations.
Results:
A small but statistically significant increase in synthesis phase and decrease in gap 2/mitosis phase populations was noted at 6 hours postfreeze in asynchronous samples. Synchronization in synthesis phase yielded an increase in cell death when combined with freezing to both −15°C and −20°C. Calcitriol pretreatment increased the gap 1 phase population by 20% and a synergistic decrease in viability following freezing. However, gap 1–sorted populations combined with calcitriol treatment did not exhibit this synergistic effect. Fluorescence microscopy of fluorescence-activated cell sorting-sorted cells revealed necrosis as the predominant form of cell death in all phases, though apoptosis did play a role.
Conclusion:
Although initial results suggested a potential sensitivity, PC-3 cells exposed to freezing as sorted populations did not reveal significant differences in cell death. As such, the data from this study suggest that there is no difference in cell cycle stage sensitivity to freezing injury.
Introduction
In 2015, an estimated 233 000 new cases of prostate cancer were diagnosed in the United States. 1 As the most highly diagnosed nonskin cancer in men, treatment for both early stage and advanced disease remains critical. Cryosurgery has become a primary treatment option for prostate cancer and yields disease-free survival similar to other therapies. 2,3 One of the challenges of prostate cryosurgery is the determination of positive freeze margins necessary to minimize recurrence risk while avoiding damage to the prostatic neurovascular bundle and rectal wall. Advancements in cryosurgical technology have improved the overall efficacy of treatments and minimized postoperative complications. 4,5 However, often it is a challenge to achieve the necessary temperature for total cellular destruction (>40°C) throughout the tumor; as such, adjunctive treatments aim to increase the lethality of mild freezing by combining agent/drug exposure with cryosurgery. 6 –8
Previous studies have investigated conventional chemotherapeutic agents’ efficacy in conjunction with freezing. 9 –12 The majority of chemotherapy drugs affect the DNA synthesis or cell division processes, which acts as a cellular stress-inducing event and thus causes activation of cell cycle checkpoints, DNA damage responses, and the initiation of cell death cascades. Similarly, ionizing radiation induces a reproducible effect whereby cells exhibit sensitivity based on cell cycle phase; specifically, gap 2/mitosis (G2/M) phase populations are the most radiosensitive, synthesis (S) phase the most radioresistant, and gap 1 (G1) phase demonstrates intermediate sensitivity. 13 Similar cell cycle–dependent responses have also been reported with various chemotherapeutic agents wherein the drug targets cancer cells in specific cell cycle stages. 14,15 Although the role cell cycle plays in therapeutic outcome in radiation and chemotherapy is understood, its role in cell response to cryoablation remains unknown. The literature suggests that the multitude of stressors initiated during freezing, including ischemia, hypoxia, apoptotic induction, and metabolic uncoupling, all have a cell cycle–related dependence when applied individually. 16 –18 The combinatorial action of these coupled with the physical and biochemical stressors occurring during cryoablation may amplify or potentially negate the cell cycle dependence. As such, this study examined postfreeze viability and cell cycle phase distribution as well as the responses of individual phases to freezing injury as a first step in determining the role of cell cycle kinetics in cell death following freezing. Previous studies have highlighted the synergistic effect of the addition of vitamin D3 (VD3) metabolite calcitriol prior to freezing androgen-sensitive and androgen-insensitive prostate cancer. 19,20 As such, we also investigated the impact of cell cycle dependence on combinatorial calcitriol/freeze strategies. We hypothesized that calcitriol cryosensitization occurs through cell cycle–regulated pathways.
Materials and Methods
Cell Culture
PC-3 cells (ATCC CRL-1435) were cultured in T-75 flasks (Cell Treat, Shirley, Massachusetts) in RPMI-1640 (Caisson, North Logan, Utah) with 10% fetal bovine serum (Atlas Biologicals, Fort Collins, Colorado) and 1% penicillin/streptomycin (Corning Life Sciences/Mediatech Inc, Manassas, Virginia). Subcultures were plated into 35-mm dishes (Cell Treat) or Costar 96-well Stripwell plates (Corning Inc, Tewksbury, Massachusetts) for experimentation.
Vitamin D3 (Calcitriol) Treatment
1,25α Dihydroxyvitamin D3 (Calbiochem/EMD Millipore, Billerica, Massachusetts) was reconstituted to a (200 μmol/L) stock in ethanol, stored at −80°C, and diluted to a final working concentration of 50 nmol/L in culture medium immediately prior to application to experimental cultures for 24 hours prior to freezing (drug was left on for 24 hours postfreeze recovery). Vitamin D3 and calcitriol will be used interchangeably to describe 1,25α dihydroxyvitamin D3.
Freezing Protocol
Costar 8-well strips (75 µL medium/well) or 35-mm dishes (1.5 mL medium/dish) were exposed to freezing temperatures (−10°C, −15°C, −20°C, or −25°C) in refrigerated baths (Neslab/Thermo Scientific, Waltham, Massachusetts) for 5 or 10 minutes. Ice nucleation was initiated using liquid nitrogen vapor when samples cooled to −2°C to prevent supercooling. Sample temperature was recorded using a type T thermocouple (Omega, Stamford, Connecticut). Samples were thawed passively at room temperature for 10 minutes and then returned to 37°C for recovery.
Viability Assessment
Viability was assessed using the alamarBlue (Life Technologies, Grand Island, New York) metabolic activity assay diluted 1:20 in Hanks balanced salt solution (Corning/Mediatech). Medium was replaced with 100 µL/well alamarBlue, incubated in the dark for 60 minutes at 37°C, and analyzed with a TECAN SpectraFluor Plus plate reader (excitation λ = 530 nm and emission λ = 590 nm; Tecan Austria GmBH, Grödig, Austria). Assessment was repeated for at least 3 days of recovery.
Cell Cycle Synchronization
A double thymidine block was utilized to achieve S-phase synchronization. Thymidine (Sigma-Aldrich Corp, St Louis, Missouri) was reconstituted to a (50 mmol/L) stock solution in sterile water and in media to a final working solution dilution of 2 mmol/L. Cells were exposed to thymidine for 22 hours, released for 8 hours, followed by a second thymidine exposure for 18 hours. Three hours following the second thymidine removal yielded >85% S-phase synchronization.
Cell Cycle Distribution Analysis
Samples were collected from 35-mm dishes using TrypLE Express (Life Technologies), pooled and pelleted by centrifugation (450g for 5 minutes), washed with cold 1× phosphate-buffered saline (Corning), fixed in 15-mL conical tubes using ice-cold 70% ethanol dropwise while vortexing, and then stored at 4°C (>12 hours). For analysis, samples were centrifuged at 300g for 5 minutes, ethanol removed, and resuspended in propidium iodide/RNase staining buffer (BD Pharmingen, Franklin Lakes, New Jersey). Samples were assessed in triplicate using microcapillary flow cytometry (Guava Technologies/EMD Millipore, Billerica, Massachusetts) and analyzed using ModFit LT (Verity Software House, Topsham, Maine).
Fluorescence-Activated Cell Sorting
Samples were stained with Hoechst 33342 (Molecular Probes, Eugene, Oregon) for 1 hour at 37°C and sorted at Cornell Biomedical Sciences Flow Cytometry Core Laboratory (Cornell University, Ithaca, New York) using a FaCSARIA (BD, Biosciences, San Jose, California). Unstained controls were used for gating. Following sorting into G0/G1, S, or G2/M, cells were returned to 37°C and plated for experimentation. Fluorescence-activated cell sorting (FACS) results are presented as 1 representative experiment.
Microscopy
The fluorescent probes Hoechst 33342 (living cells, 0.06 µg/µL), propidium iodide (necrotic cells, 0.007 µg/µL), and Yo-Pro-1 (apoptotic cells, 0.8 µmol/L; Thermo Fisher Scientific) were applied to samples and incubated for 15 minutes at 37°C in the dark. Phase contrast and fluorescent images were acquired with a Zeiss Axiovert 200 (Carl Zeiss Microscopy, Thornwood, New York) using 3 × 3 MosaiX at 10× magnification. Fluorescence images were quantified based on color (blue = living, green = apoptotic, red = necrotic, respectively) using ImageJ software (National Institutes of Health, Bethesda, Maryland).
Data Analysis
Raw fluorescent units were converted to percentage survival (± standard error of mean) based on 37°C controls following background subtraction. Experiments were repeated a minimum of 3 times with an intraexperimental repeat of n = 7 and each condition had an n of 7 (N = 21). Statistical analysis was performed by Student t test; statistically significant when P < .05.
Results
Impact of Freezing on PC-3 Cell Survival
PC-3 cells were frozen to 10°C, −15°C, −20°C, or −25°C for 5 or 10 minutes to establish a baseline in vitro freeze response (Figure 1). At the mild subfreezing temperature of −10°C, no difference was observed in postfreeze viability compared to controls. Increasing freeze duration from 5 to 10 minutes at −15°C increased cell death by 27% (86.6% [±4.6] vs 63.4% [±3.0], respectively) and 54% at −20°C (27.8% [±1.6] vs 13.0% [±0.9]; Figure 1). Similar improvement was noted in the −25°C samples of 5 and 10 minutes (19.3% [±1.8] vs 4.1% [±0.6], respectively). These findings corroborate other studies on prostate cancer response to freezing. 21,22

PC-3 response to thermal insults. PC-3 cells were seeded into Costar strip well plates at 3.75 × 104 cells/cm2 and grown for 48 hours to reach confluency (85%-90%). Media exchange (75 μL/well total volume) was performed 30 minutes prior to freezing. Cells were exposed either a 5- or 10-minute freeze to nadir temperatures of −10°C, −15°C, −20°C, or −25°C. AlamarBlue metabolic activity indicator was used to determine relative fluorescent units from which background values were subtracted. Resultant viability was compared to a prefreeze control set at 100%.
Impact of Freezing on Cell Cycle in Asynchronous Populations
With the differential response of PC-3 cells to freezing observed, asynchronous populations were examined to assess any changes in cell cycle distribution. Cell cycle analysis revealed small but statistically significant changes in distribution following freezing (Table 1). Specifically, a single 10-minute freeze at −15°C yielded a significant increase in the S-phase population compared to controls at 6-hour postthaw (49.3% [±2.01] vs 38.9% [±1.8], P < .01; Table 1). By 24 hours postfreeze, the kinetics of −15°C treated samples had returned to normal cycle distributions (Table 1). Exposure to −20°C for 10 minutes resulted in a decrease in the percentage of G2/M phase cells and subsequent increase in G1 throughout the postfreeze assessment period in both the adherent and lifted populations (Table 1, P < .01 in all conditions).
Cell Cycle Distribution of Asynchronous PC-3 Cells Following Freezing.

Abbreviations: G1, gap 1 phase; G2, gap 2 phase; M, mitosis.
Analysis of Freezing Impact on Synchronous Cell Cycle Populations
Given the potential for cell cycle dependence in freeze sensitivity in asynchronous populations (Table 1), PC-3 cells were synchronized in S phase using a double thymidine block yielding a 90% homogenous population (Figure 2) and then frozen to −10°C, −15°C, and −20°C (Figure 3). In synchronized samples, a single −15°C exposure resulted in a 34% decrease in viability compared to nonfrozen synced controls versus a 23% decrease in asynchronous samples (76.4% [±1.9] vs 98.6% [±1.3], P < .01 at 24 hours postfreeze). Freezing to −20°C yielded an 88% reduction in viability in synchronized samples versus 63% in asynchronous population viability (21.8% [±1.7] vs 38.6% [±0.8], respectively; Figure 3).

Confirmation of synthesis phase synchronization. PC-3 cells were synchronized using a double thymidine block (2 mmol/L thymidine exposure for 22 hours, release for 8 hours, second 2 mmol/L thymidine exposure for 18 hours). Cells were fixed in 70% ethanol, stained with propidium iodide/RNase, and 10 000 events acquired using a Guava microcapillary flow cytometer. Analysis was performed using ModFit LT.

Asynchronous versus synchronous PC-3 cell response to freezing—alamarBlue metabolic activity data. PC-3 cells were synchronized using a double thymidine block prior to a 5-minute freeze event to −10°C, −15°C or, −20°C and compared to asynchronously growing populations. Viability was measured using a metabolic activity indicator for 3 days postfreeze and compared to prefreeze controls.
As the thymidine synchronization data suggested potential S-phase susceptibility to freeze injury, PC-3 samples were sorted into G1, S, or G2/M phase subpopulations, allowed to adhere to culture plates, and then frozen within 2 hours to assure synchrony. Samples were exposed to freezing conditions and assessed for viability, apoptosis, and necrosis at 1, 4, 8, and 24 hours postfreeze. Fluorescent microscopy analysis revealed that following freezing to −15°C for 10 minutes, S-phase populations resulted in a 67.13% decrease in viability 24 hours postfreeze compared to S-phase controls (Table 2). Interestingly, the same freeze exposure in asynchronous cells yielded only a 37% decrease (Figure 1). G1–sorted populations exposed to −15°C resulted in a 62% decrease in viability at 24 hours compared to sorted G1 controls. Similarly, sorted G2/M populations yielded a 60.16% decrease compared to G2-sorted controls at 24 hours postthaw. Sample viability assessment with alamarBlue corroborated the fluorescence microscopy data at 24 hours postfreeze as well as demonstrated surviving populations were able to repopulate over a 10-day postthaw interval (data not shown). Overall, the 24-hour viability data for the sorted populations revealed similar levels of cell death among each of the 3 groups (∼60% decrease) following a −15°C freeze, which was elevated from that of the asynchronous groups following freezing to −15°C that resulted in a ∼40% decrease in viability. In addition to postfreeze viability, fluorescence microscopy analysis was conducted to quantify apoptotic and necrotic subpopulations (Table 2). Following a −15°C freeze, G1 and G2/M phase populations exhibited similar levels of necrotic cell death at 1, 8, and 24 hours postfreeze. Interestingly, a decrease in necrosis was observed at 4 hours postthaw in the −15°C samples. Apoptotic assessment revealed a small but significant increase in the −15°C samples over the initial 24-hour postfreeze interval in both the G1 and G2/M populations. In S-phase samples, necrosis peaked at 8 hours postfreeze, whereas apoptosis continued to increase over the 24-hour assessment period. Overall, the sorted population data revealed that necrosis played a predominant role in cell death following freezing in all cell cycle–phase subpopulations, whereas apoptosis played a lesser, yet significant, role. Further, the data revealed no significant difference between G1, S, and G2/M phase sensitivity to freezing (−15°C 24-hour viability: 38.2% vs 43.7% vs 40.9%, respectively).
Mode of Cell Death Analysis of Sorted Cells Postfreeze.
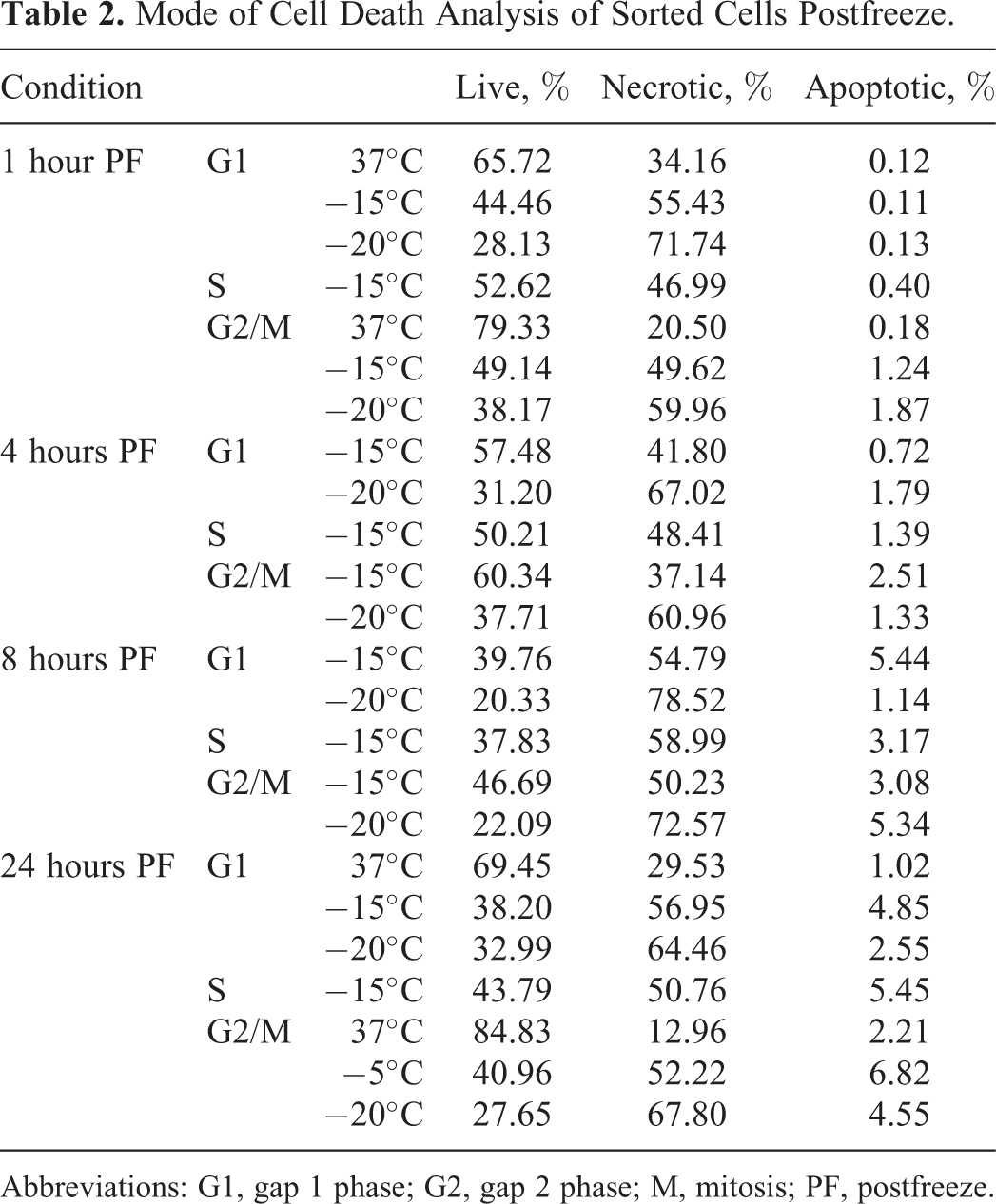
Abbreviations: G1, gap 1 phase; G2, gap 2 phase; M, mitosis; PF, postfreeze.
Impact of Calcitriol (VD3) and Freezing on Cell Cycle
We have previously reported on the ability of calcitriol to synergistically decrease postfreeze prostate cancer cell viability when combined with freezing. 19,20 To this end, investigations were conducted to determine whether this was a result of cell cycle alterations. Samples were exposed to calcitriol (50 nmol/L) prior to freezing to −15°C for 10 minutes, given that in asynchronous samples ∼70% survival was found following a 10-minute freeze and much lower viability observed at −20°C (∼20%). Analysis of exposure times revealed no significant impact on cell cycle distribution between control asynchronous and calcitriol-treated samples at 1, 3, or 6 hours of treatment. Following 24-hour exposure, a 20% increase in the G1 subpopulation was observed, which correlated with an overall 10% decrease in each of the S and G2/M populations (Figure 4).
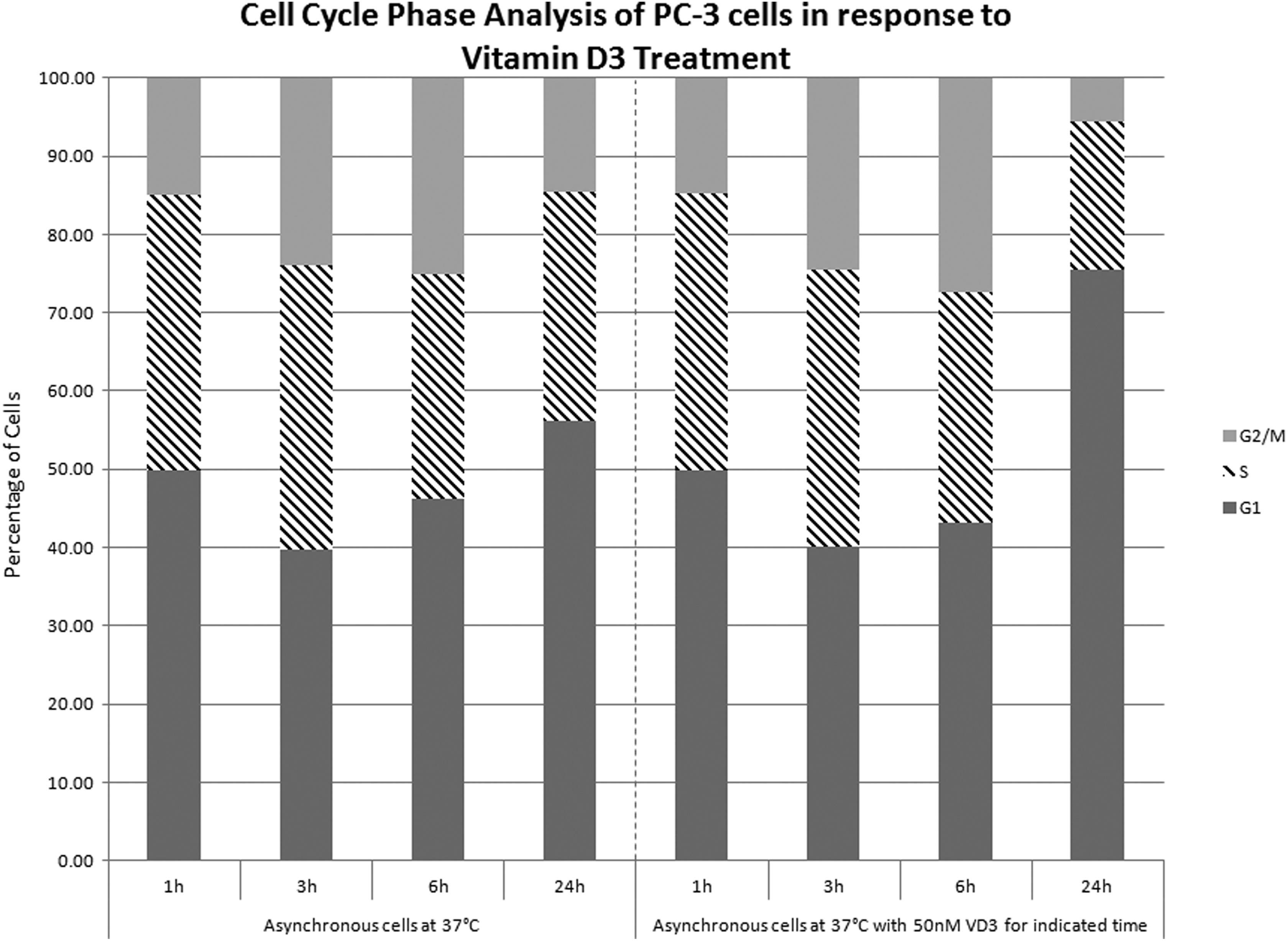
Cell cycle phase analysis of PC-3 cells in response to vitamin D3 treatment. Asynchronous PC-3 cells were fixed at the indicated time points (1, 3, 6, or 24 hours) and cell cycle phase distribution was analyzed using a Guava microcapillary flow cytometer. Calcitriol was added for a total duration of 1, 3, 6, or 24 hours. Analysis was performed using ModFit LT.
When asynchronous samples pretreated with calcitriol were frozen to −15°C, overall cell viability decreased to 39.1% (±1.9) compared to 72.2% (±1.5) in nontreated −15°C samples (Figure 5). A similar trend was also noted following the combination of calcitriol pretreatment and −20°C with postfreeze viabilities of 6.3% (±0.3) versus 18.4% (±1.3) in −20°C freeze alone samples. With the observed increased freeze susceptibility of calcitriol-pretreated samples, studies were conducted on G1-sorted samples to evaluate the impact of calcitriol pretreatment. Interestingly, in G1-sorted samples, the combination of G1 selection and calcitriol pretreatment did not yield an increase in cell death compared to nontreated sorted G1 samples (Table 3). G1 samples frozen with 50 nmol/L calcitriol did not exhibit a synergistic effect (57%) that differed from G1-untreated cells. Although no difference was observed in the G1, G1 calcitriol, and asynchronous calcitriol viability, the overall viability of each of these populations was found to be significantly lower than nontreated asynchronous samples frozen to −15°C (38%, 29%, and 39% vs 72%, respectively).

Viability analysis of PC-3 cells following freezing with or without calcitriol pretreatment. PC-3 cells were seeded into Costar strip well plates at 3.75 × 104 cells/cm2 and grown for 24 hours. A media exchange was performed after 24 hours, and calcitriol (50 nmol/L) was applied to appropriate samples for an additional 24 hours. Media exchange (75 μL/well total volume) was performed 30 minutes prior to freezing. Cells were exposed either a 5- or 10-minute freeze to nadir temperatures of −15°C or −20°C. AlamarBlue metabolic activity indicator was used to determine relative fluorescent units from which background values were subtracted. Resultant viability was compared to a prefreeze control set at 100%.
Mode of Cell Death Analysis of Calcitriol Pretreated G1 Cells.

Abbreviations: G1, gap 1 phase; VD3, vitamin D3.
Discussion
This study investigated the response of synchronized prostate cancer cell subpopulations in comparison with asynchronous populations following exposure to mild subfreezing conditions. These studies were conducted to assess the impact of cell cycle stage on prostate cancer cell susceptibility to freezing. The mild subfreezing temperatures of −15°C and −20°C were focused on, as a significant level of survival has been reported in this range. 7 –12,22,23 We hypothesized that if cell cycle stage susceptibility was observed, that new strategies involving agents to synchronize cells within a tumor could be combined with freezing to improve cell destruction in the margin of the freeze zone.
The attainability of synchronization in vivo is complicated by the inherent heterogeneity of tumors. However, the combination of multiple treatment regimes that target different cell cycle phases may represent a viable treatment option. Postfreeze phase distribution analysis revealed a decrease in G2/M phase cells between 1 and 3 hours following a −20°C thermal insult (Table 2). The −15°C condition followed suit in a delayed manner beginning at 6 hours postfreeze accompanied by a significant increase in S-phase cells. This subsequent increase in S phase may indicate a delayed reentry into G2/M following −15°C exposure in surviving cells. To model this S-phase accumulation, PC-3 samples were synchronized in S phase using thymidine prior to freezing. These studies revealed a significant decrease in viability following freezing exposure, indicating that a first-line treatment modality that results in a shift to S phase may have beneficial effects on cell response to mild freezing (>−15°C). These findings may provide insight into the benefits of the cryo–chemo combination strategy reported by Le Pivert et al 12 and Clarke et al, 9 among others, where it was hypothesized that the combination of sublethal doses of 5-fluorouracil (5-FU) and mild freezing (−20°C) results in increased cell death by halting the cell cycle at the S/G2 transition. In the current study, a similar result found with synchronization of PC-3 populations in the S phase prior to freezing resulted in increased cell death (Figure 4).
With the identification of S-phase synchronized samples being more susceptible to freeze injury, experiments on FACS-sorted sample populations (G1, S, and G2/M) were conducted to determine whether the increased sensitivity observed was phase related. Viability analyses of frozen-sorted populations revealed no detectible preferential cell cycle phase–based susceptibility following freezing to −15°C or −20°C (Table 2). Although no substantial effects were noted between phase sorted samples, it was observed that sorted populations had increased freeze susceptibility compared to matched asynchronous samples (∼2-fold increase). These findings were at odds with the thymidine synchronization experiments as well as the 5-FU literature reports that suggested the S-phase samples should have been more susceptible. Studies are under way to further understand these observations.
We have previously reported on the utilization of calcitriol, the active metabolite of VD3, as a cryosensitizing agent in prostate cancer cells. 19,20 Given one of the responses elicited by calcitriol is the upregulation of cell cycle regulatory proteins, including p21 and p27, in a variety of cell systems, 24,25 experiments on the combination of calcitriol pretreatment and freezing were conducted. Although PC-3 cells are relatively unresponsive to the antiproliferative effects of calcitriol alone, an increase in the G1 subpopulation following exposure for 24 hours was noted (Figure 4). Further, a synergistic decrease in cell viability was observed following combinatorial treatment with freezing (Figure 5). Given these observations, we exposed G1-sorted samples to calcitriol followed by freezing to −15°C, yet no difference in postfreeze viability with or without calcitriol treatment in G1 samples was observed. However, pretreatment with calcitriol in these experiments was limited to 2 hours due to experimental constraints. In previous studies, a pretreatment period of 24 hours was utilized. Although no difference was seen between the G1 and G1 calcitriol samples, the overall level of cell death was found to be similar to that of asynchronous samples when exposed to calcitriol for 24 hours (Table 3 and Figure 5). As such, it is possible that the preselection of a sorted G1 population mimicked the effect of calcitriol cryosensitization in an asynchronous population. This suggests that the cryosensitization effects are not occurring via a rapid, nongenomic signaling pathway but rather require a traditional genomic transcription factor response.
It has recently been reported that the ability of calcitriol to regulate target genes differs between cell cycle phase in both normal and malignant prostate cells. 26 Specifically, normal prostate cells were the most responsive to calcitriol in G1, whereby multiple genes involved in antiproliferative effects were positively regulated. PC-3 cells, in contrast, were shown to exhibit reduced or negative regulation of the same genes. The authors postulated that a significant increase in methylation patterns at certain vitamin D response elements on the DNA may contribute to this differential regulation. Thus, the mechanism of cryosensitization is possibly occurring through a novel pathway independent of P53 or retinoblastoma protein (phospho-RB) status. Epigenetic modifications may also be an important factor in calcitriol’s ability to impart freeze sensitization without the typical antiproliferative response. Further studies are necessary to elucidate the pathways involved in the cryosensitization effects of calcitriol.
The results obtained from this study suggest that cell cycle phase is likely not a critical factor in sensitivity to cell death following freezing. However, the data also support the potential of adjuvant-based treatment strategies to further improve outcome, given that while freeze exposure of sorted cell cycle populations did not show increased sensitivity of one phase over another, each phase alone was individually more sensitive than asynchronous populations. Further, the use of sublethal synchronization agents including thymidine, 5-FU, and VD3, among others, results in an increased lethal outcome when combined with mild freezing. This is in stark contrast to when any of these agents or mild freezing is utilized as a “monotherapy.” In summary, our findings suggest that the effects of cryoablation are cell cycle independent, and as such, cryo provides for a highly effective treatment modality that targets all cells of a heterogeneous tumor equally.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: NIH Grant 1R43CA183265-01 (PI John M. Baust). NIH Grant 1R43CA123993-01A1 (PI Kristi K. Snyder). CPSI Biotech.
