Abstract
BACKGROUND:
Isoform-specific function of doublecortin-like kinase 1 (DCLK1) has highlighted the key role of the DCLK1-S (short isoform) in the maintenance, progression, and invasion of the tumor.
OBJECTIVE:
This study was designed to produce an anti-DCLK1-S polyclonal antibody to evaluate DCLK1-S in human colorectal cancer (CRC) specifically.
METHODS:
The expression pattern and clinical significance of DCLK1-S were assessed in a well-defined tissue microarray (TMA) series of 348 CRC and 51 adjacent normal tissues during a follow-up period of 108 months.
RESULTS:
Expression of DCLK1-S was significantly higher in CRC samples compared to adjacent normal samples (
CONCLUSIONS:
Our findings strongly supported that high cytoplasmic expression of DCLK1-S compared to its moderate expression could be considered an independent prognostic factor influencing DSS.
Introduction
Colorectal cancer (CRC) is the fourth most frequently diagnosed cancer and the leading cause of global death [1]. CRC also makes up the third most frequent cancer in males and second in females in Iran, with the increase of almost twice the mortality rate from 2.87 in 1990 to 6.8 in 2017 in 100,000 in both sexes [2, 3]. Despite developments in anticancer treatments, most CRC cases are resistant to conventional cancer therapies. Surgery, radiotherapy, chemotherapy, and targeted therapy are used as the current treatment for patients with CRC. Combining these traditional methods with new targeted therapy strategies could increase the survival rate of patients with CRC [4]. Therefore, identifying biomarkers can be considered an essential step for improving targeted molecular therapies (immunotherapy) and increasing the survival of patients with cancer. Considering the crucial role of cancer stem cells (CSCs) in tumorigenesis, tumor invasion, and drug-resistance, many efforts have been made to discover specific markers to target CSCs in various cancers, including CRC [5, 6, 7, 8]. Among various markers used for identification of CRCCSCs, doublecortin-like kinase 1 (DCLK1), as a putative CSC marker has been shown to have a crucial oncogenic role in various in-vivo and in-vitro experiments, particularly in pancreatic and colon cancers [7, 9, 10, 11, 12] so that, DCLK1 is called as a well-known CRCCSC marker distinguished cancer cells from normal cells in Apc Min/
In the recent decades, many experiments have focused on the biological function of two different transcripts of DCLK1; long isoform (DCLK1-L, NM_004734.4) and short isoform (DCLK1-S, NM_ 001195415.1) with a molecular weight of
In our previous research, expression pattern and prognostic significance of DCLK1 have been studied in colorectal, gastric, and bladder carcinomas [19, 20, 21, 22, 23] using available commercial anti-DCLK1 antibodies that can detect C-terminal domain of both DCLK1-L/S isoforms or only N-terminal end of DCLK1-L isoform. In contrast, the role of the main isoform to overexpress DCLK1 has remained unknown. To the best of our knowledge, there is no commercial anti-DCLK1-S antibody. Also, no study has been previously performed to investigate the prognostic significance of DCLK1-S on tumor tissues, particularly in CRC by immunohistochemistry (IHC). Therefore, for addressing these gaps, the present study was conducted to generate an anti-DCLK1-S-specific polyclonal antibody elicited by six amino acids synthetic peptide that has been reported as the only different form in the sequence homology epitopes of both DCLK1 isoforms (DCLK1-L and DCLK1-S) [12].
In the current study, for the first time, membranous, cytoplasmic, and nuclear localization of DCLK1-S elicited by polyclonal anti-DCLK1-S antibody, generated in our laboratory was evaluated in a large number of formalin-fixed, paraffin-embedded (FFPE) series of CRC samples by tissue microarray (TMA)-based IHC analysis. The prognostic significance of short isoform of DCLK1 (DCLK1-S) was also investigated in this series of patients with CRC.
Materials and methods
Production of anti-DCLK1-S-specific polyclonal antibody
Polyclonal antibody to DCLK1-S was generated based on the protocols were described previously [24, 25]. DCLK1-S specific antibody was produced and the reactivity of rabbits sera as well as the purified antibodies was evaluated by ELISA. A synthetic peptide corresponding to the published amino acid sequence of DCLK1-S isoform (MLELIE) was selected as immunogen [12]. A cysteine residue was added to C-terminal of peptide (NH2-Met-Leu-Glu-Leu-Ile-Glu-Cys-COOH) for conjugation with Maleimide-activated keyhole limpet hemocyanin (mcKLH) (Thermo Scientific, Rockford, IL, USA) and used for immunization of two female New Zealand White Rabbits purchased from Pasteur Institute of Iran. Two rabbits were immunized with 250
To assess conjugation efficacy, the peptide was also conjugated with Imject Maleimide-activated bovine serum albumin (BSA) (Thermo Scientific, Rockford, IL, USA). Conjugation efficacy was investigated by analysis of the electrophoretic pattern of the BSA-conjugate [24]. Production of DCLK1-S specific antibodies was evaluated by enzyme-linked immunosorbent assay (ELISA) Anti-DCLK1-S specific antibodies were purified by the peptide coupled to CNBr-activated Sepharose-4B. The purity and reactivity of the purified antibodies, were assessed by sodium dodecyl-sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and ELISA techniques, respectively. The immunoreactivity pattern of the generated antibody was compared with available commercial anti-DCLK1 antibody; ab31704 (which is commonly used to detect long and short DCLK1 isoforms; Abcam, UK) in CRC tissues by IHC method.
Indirect ELISA
Reactivity of rabbits sera, as well as the purified anti DCLK1-S antibodies, was tested by ELISA Briefly, the coating was performed using 10
Sample collection and patients characteristics
FFPE series of 385 CRC samples and 70 matched adjacent normal tissues were collected from university hospitals including Hasheminejad, Rasool Akram, and Firoozgar during 2010–2019 in Tehran, Iran. Clinicopathological information including age, gender, tumor size, tumor differentiation, vascular and neural invasion, lymph node involvement, tumor, nodes, and metastases (TNM) staging, distant metastasis, and tumor recurrence was collected from the patients’ documents. None of the patients with CRC in this study had received neoadjuvant treatment before surgery. Size cutoff and TNM stage of the tumors were determined according to the American joint committee on cancer/union for international cancer control (AJCC/UICC) and the TNM staging system, respectively [26]. The patients’ data were kept anonymous in all steps of the study. The interval from the date of surgery to the date of death by CRC was used for disease-specific survival (DSS) analysis.
Moreover, progression-free survival (PFS) was defined as the interval between the primary surgery and the last follow-up visit if the patient showed no evidence of disease, tumor recurrence, or distant CRC metastasis. The current study was approved by the Human Research Ethics Committee of the Iran University of Medical Sciences, Tehran, Iran (Ref No: IR.IUMS.REC 1396.32186). All the procedures performed in this study were following the 1964 Declaration of Helsinki and its later amendments. Informed consent was obtained from all participants, parents or legally authorized representatives of participants at the time of sample collection with routine consent forms.
TMA construction
As described previously, colorectal tissue arrays were prepared and constructed by a TMA instrument (Minicore; ALPHELYS, Plaisir, France), as described previously [6, 27, 28]. Pathologist pointed three representative areas of the tumors on hematoxylin & eosin (H & E) slides of CRC samples. Different regions of each tumor were punched into TMA recipient blocks in three copies. Based on TMA studies, each core would denote the whole tissue’s staining pattern with 90% accuracy, while using two cores or more could increase this accuracy by 95–99%, respectively [29]. In this study, three cores were evaluated from each tissue sample to overcome heterogeneity in antigen expression and increase the accuracy and validity of the analysis. The mean score of the three cores was calculated as the final score for IHC analysis of DCLK1-S.
Immunohistochemistry
Immunostaining of CRC slides (whole tissue and TMA slides) was performed by standard chain polymer-conjugated antibody (EnVision; standard EnVision-HRP kit, Bio pharmadx), as described previously [6, 21, 30]. Briefly, following de-waxing at 60
Evaluation of immunostaining
The scoring step was performed semi-quantitatively in a coded manner by two pathologists (MA and MR) who were blinded to clinical and pathological parameters (as described previously) [32], and a consensus agreement was achieved. Given a deal to the final score of DCLK1-S expression, the general distribution of the tumor cells was assessed at 10
Different localization pattern of DCLK1-S expression in CRC tissues by IHC staining: considering our findings A) cytoplasmic expression, B) membranous, and C) nucleus expression of DCLK1-S were observed in CRC samples (All images were taken at 400 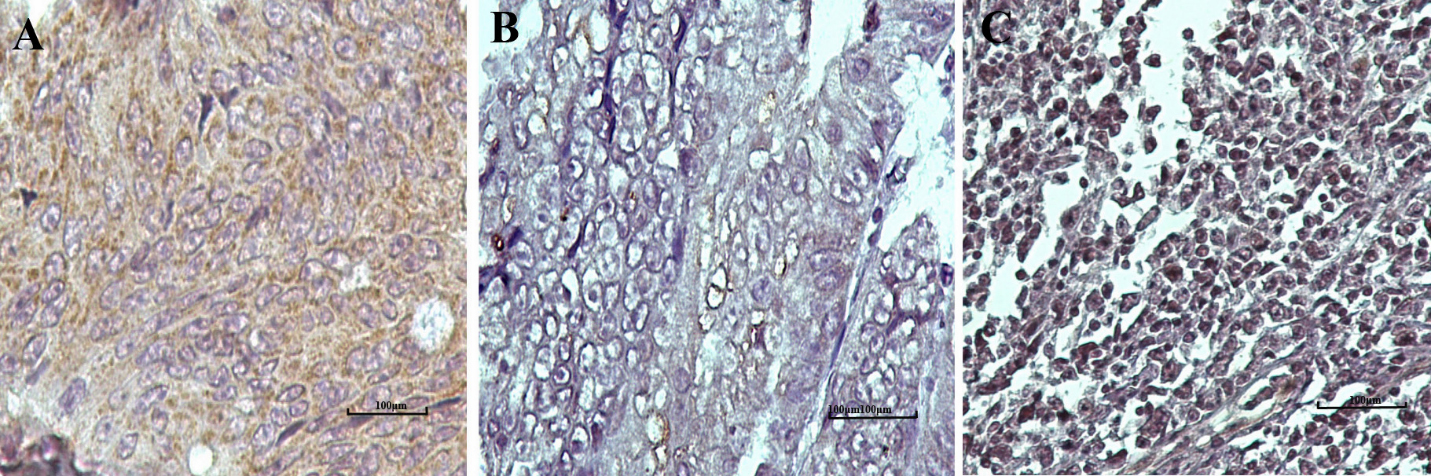
Immunohistochemical staining of DCLK1-S in CRC tissues and negative controles. (A) Negative, (B) low, (C) moderate, and (D) high expression of DCLK1-S in CRC tissue. Replacement of the primary antibodies by (E) pre-immune purified rabbit IgG (isotype control) and (F) Tris Buffer Saline, and (G) primary antibody saturated with immunizing peptide, as the negative reagent controls. (H) Negative expression of DCLK1-S in adjacent normal tissue. The results showed that increased cytoplasmic expression of DCLK1-S in CRC samples in comparison to adjacent normal samples (All images were taken at 200 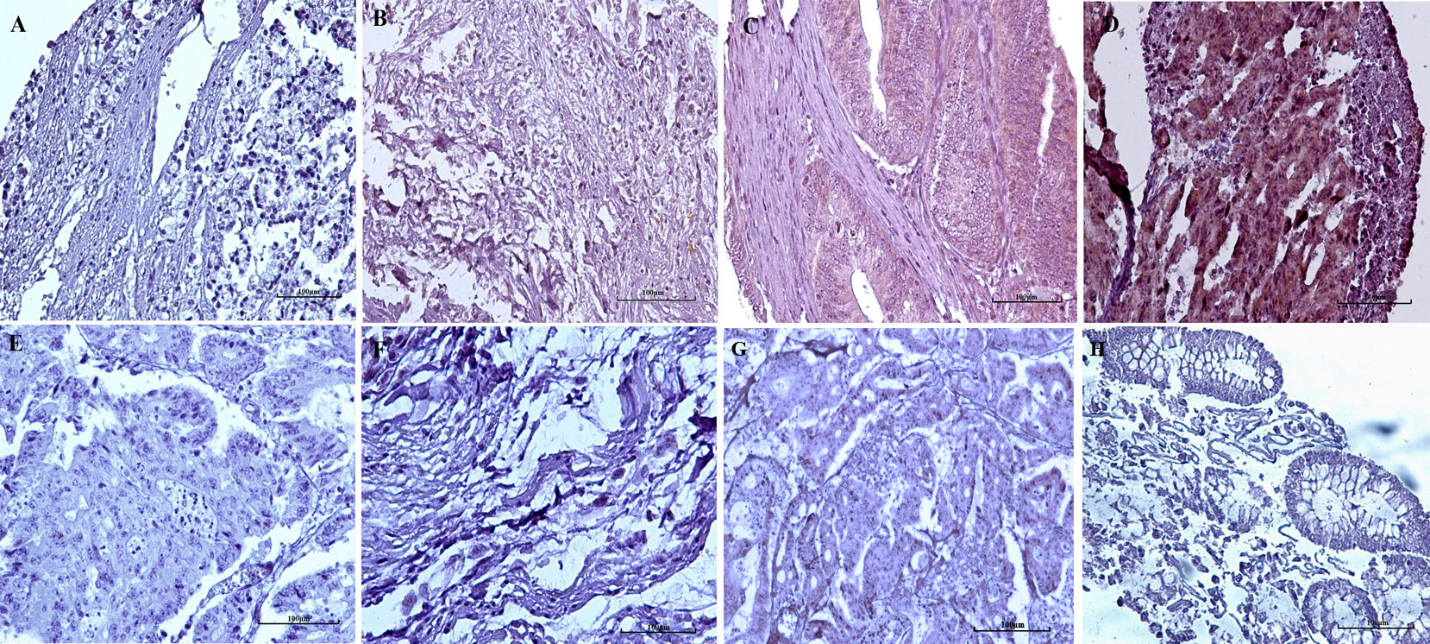
SPSS software version 22 (IBM Corp, USA) was used for statistical analysis. Pearson’s
Results
Production of antibody and characterization
Production of anti-DCLK1-S antibody
Efficacy of conjugation and immunization was evaluated by SDS-PAGE analysis of the BSA-conjugate and ELISA test, respectively. The smear pattern of SDS-PAGE and the absence of free peptide showed effective conjugation of BSA-peptide (supplementary Fig. 1A). The immunoreactivity of the antibodies was confirmed by the ELISA test. The results showed high immunoreactivity of the antibodies reaching to plateau as low as an antibody concentration of 1250 ng/ml (Supplementary Fig. 1B).
Validation of anti-DCLK1-S antibody
Immunoreactivity of anti-DCLK1-S was evaluated by the IHC method on CRC whole tissues as positive control samples and humans normal and cancerous tissues of testis, ovary, and skin as negative control samples. In addition, all the experiments used isotype control and primary antibody saturated with immunizing peptides as negative reagent controls. The results clearly showed a specific pattern of immunoreactivity in CRC tissues compared to negative control tissues (Fig. 2 and supplementary Fig. 2). The antibody could strongly recognize DCLK1-S in CRC samples, and localization was restricted to the cytoplasmic area and partially in the cell membrane and nucleus area of CRC samples (Fig. 1). No staining was observed in humans in normal and cancerous tissues of testis, ovary, and skin, demonstrating specific immunoreactivity of the produced anti-DCLK1-S antibody (supplementary Fig. 2).
The commercial anti-DCLK1 antibody, which can recognize the C-terminal end of both long and short isoforms of DCLK1 (anti-DCLK1-L/S antibody; ab31704) was used to compare the expression pattern of DCLK1-L/S vs. DCLK1-S. Results showed no difference in the expression pattern using the generated anti-DCLK1-S antibody and the commercial anti-DCLK1-L/S antibody, as shown in supplementary Fig. 3. No staining was also observed in negative control sections incubated by pre-adsorbed or substituted by pre-immune-purified rabbit IgG as primary antibody (Fig. 2).
Immunohistochemical evaluation of anti-DCLK1-S antibody in CRC tissues
Study population
Considering technical problems during TMA construction and IHC staining, among all 385 collected CRC samples and 70 adjacent normal tissues, 348 CRC and 51 adjacent normal tissue samples remained for statistics analysis of DCLK1-S expression. Patients had a mean age of 60
Expression of DCLK1-S in the patients with CRC and adjacent normal samples
IHC analysis results showed that most CRC samples (293/348, 84.2%) had a positive DCLK1-S expression. All the positive samples represented cytoplasmic localization of DCLK1-S in tumor cells area (293/293, 100%), which 9.5% (28/293) of them showed both cytoplasmic and nuclear expressions and 4.7% (14/293) of them showed both cytoplasmic and membranous expression of DCLK1-S (Fig. 1). The findings of the intensity of staining, percentage of positive tumor cells, and
Box plot analysis of DCLK1-S expression levels in CRC specimens versus adjacent normal tissues. The results of Mann-Whitney U test showed that there is a statistically significant differences in the median expression levels of DCLK1-S among CRC and adjacent normal tissues (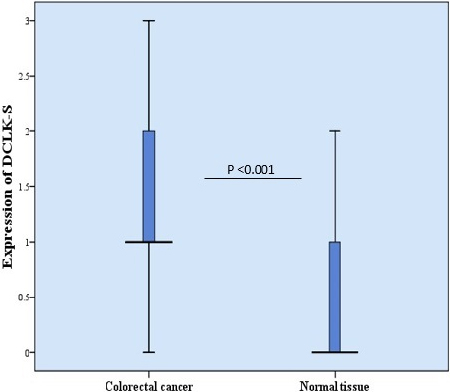
The results of Pearson’s
The association between cytoplasmic DCLK1S expression and clinicopathological parameters of colorectal cancer (CRC) samples (intensity of staining and h-score)
The association between cytoplasmic DCLK1S expression and clinicopathological parameters of colorectal cancer (CRC) samples (intensity of staining and h-score)
other clinicopathological characteristics. All findings are available in Table 1.
Cytoplasmic expression of DCLK1s in colorectal cancer (CRC) and adjacent normal tissue samples
H-score histological score.
Among 348 CRC samples included in this study, 235 (67.5%) samples had a history of tumor recurrence, distant metastasis, or cancer-related death events. Distant metastasis and tumor recurrence occurred in 67 (28.5%) and 72 (30.6%) of patients, respectively, while in 80 (34%) of patients, metastasis and recurrence were not observed. During the follow-up period, cancer-related death and the other causes of death were documented in 67 (90.5%) and 7 patients (9.5%), respectively. Mean and median follow-up times were equal to 43.5 (SD
The results of Kaplan-Meier survival analysis represented that the CRC patients with high cytoplasmic expression of DCLK1-S had shorter DSS compared to those with moderate and low cytoplasmic DCLK1-S expression (log-rank test,
Kaplan-Meier curves for disease-specific survival (DSS) and progression-free survival (PFS) according to the expression levels of DCLK1-S in CRC. (A, B). The mean time was (48 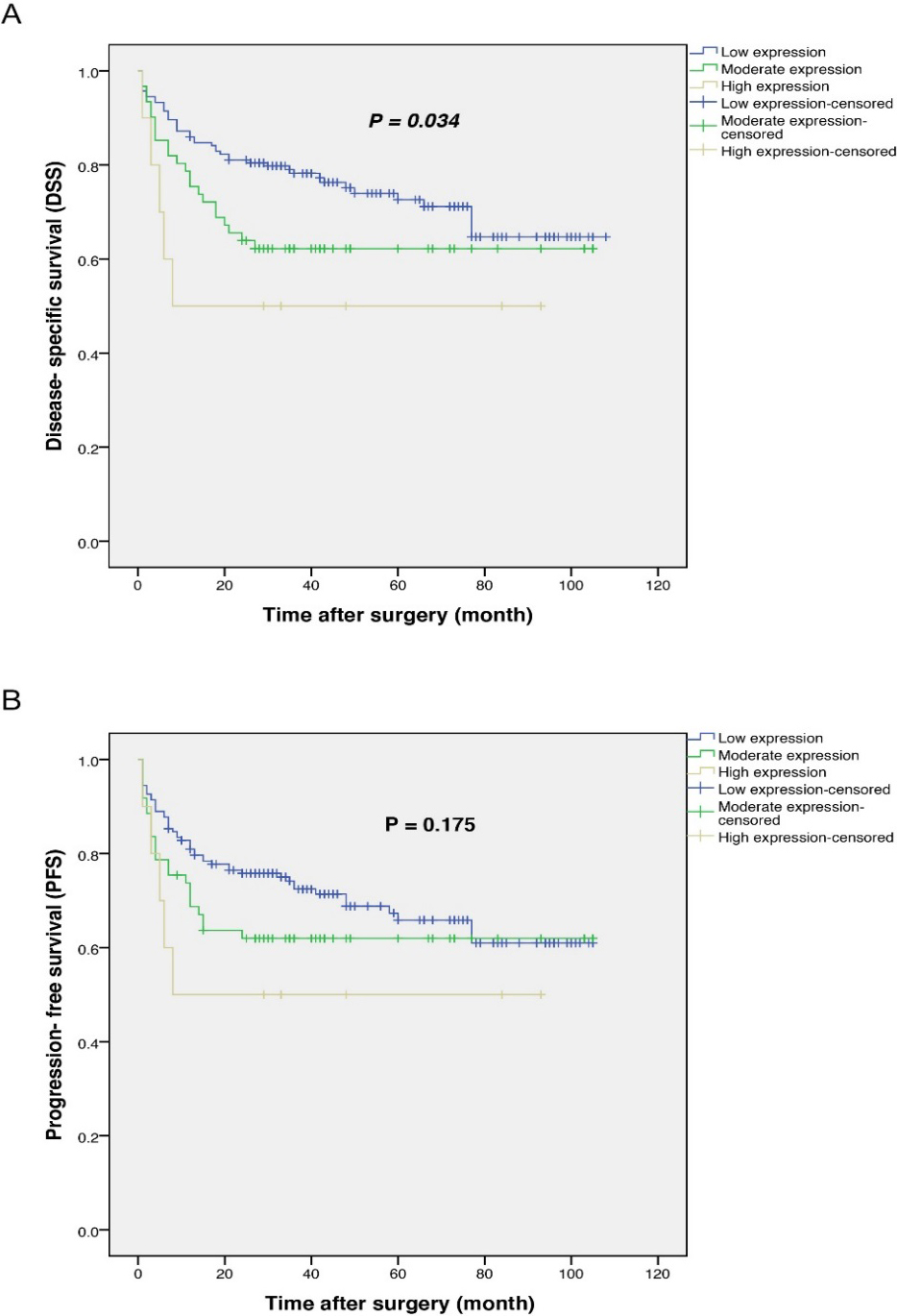
Univariate and multivariate Cox regression analyses of potential prognostic factor for disease-specific survival (DSS) in patients with colorectal cancer (CRC)
HR: hazard ratio, CI: confidence interval. The variables with
As demonstrated in Table 3, the results of the univariate analysis showed that cytoplasmic expression of DCLK1-S, age, tumor differentiation, and TNM stage were significant risk factors influencing DSS. Moreover, the prognostic value was increased in the high cytoplasmic expression of DCLK1-S vs. moderate expression of DCLK1-S (HR: 2.70, 95% CI: 0.98–7.38;
DCLK1, a putative CSC marker is a member of microtubule-associated proteins whose biological function has been identified in different human cancers, particularly in colorectal and pancreatic carcinomas [10, 12, 34]. The previous studies have also reported that suppressing DCLK1 had a significant direct correlation with tumor invasiveness and drug resistance in renal clear cell cancer and pancreatic cancer [35, 36]. In CRC, upregulation of DCLK1 is significantly associated with tumor progression, aggressiveness, metastasis, and poor prognosis [37]. Therefore, the oncogenic role of DCLK1 in a wide range of biological studies has suggested the prominence of the DCLK1 marker as a promising molecular target in the treatment of CRC. From this point, finding distinguished CSC markers which have not been identified on normal stem cells play a fundamental role in targeted therapy strategies and tumor eradication. Nakanishi et al. revealed CSC characteristics of DCLK1 in intestinal tumor cells but not in normal stem cells in-vivo [9]. Vreugdenhil et al. cloned different transcripts of DCLK1 and discovered two distinguishing alternative splicing variants of DCLK1 transcripts; DCLK-short and DCLK-long [38]. The previous studies have also highlighted these transcripts’ isoform-specific function and localization profile in Wistar rats’ brains [14]. Interestingly, epigenetic differences have discovered that DCLK1-S isoform (
Although many preliminary studies have confirmed the clinical value of DCLK1 expression in tumor progression and aggressiveness, the available commercial anti-DCLK1 antibodies widely used in the previous studies could detect sequence homology epitopes of both isoforms, DCLK1-L and DCLK1-S, or only DCLK1-L isoform; hence, the specific implication of DCLK1-S has remained largely unknown.
In our previous works, the oncopathological value of DCLK1 expression was evaluated in various cancers, including colorectal, bladder, and gastric carcinomas; results were in line with other studies [19, 21, 35, 39, 40, 41, 42]. In a comprehensive study on 472 bladder cancer cases, an increase was found in the expression of DCLK1 (DCLK1-L/S) that associated with more aggressive tumors, advanced stages, and poorer DSS in the patients with bladder cancer [21]. Additionally, results of our previous study on local and circulating DCLK1 (DCLK1-L) in 58 fresh CRC tissues and their correspondent normal margins demonstrated significantly higher expression of DCLK1 in the patients with CRC having advanced-stage and higher tumor grade in both protein and mRNA expression levels [40]. We have recently reported a highly significant expression of DCLK1 (DCLK1-L/S) in CRC cases compared to adenomatous and non-adenomatous colonic polyps and a positive significant correlation between DCLK1 expression and tumor size, tumor differentiation, and lymph node involvement [22]. Other clinical investigations have also represented that higher expression of DCLK1 (DCLK1-L/S) had a significant positive correlation with tumor invasion, metastasis, and shorter survival in patients with renal clear cell carcinoma and bladder cancer [35, 42]. However, it is important to state that the commercial anti-DCLK1 antibodies used in these studies can only recognize the C-terminal end of DCLK1-L/S protein or only the N-terminal end of DCLK1-L isoform; thus, further studies are required to investigate the biological role of DCLK1-S in tumor progression and invasiveness.
Since the absence of the commercial anti-DCLK1-S antibody was the primary obstacle for assessing this molecule at the protein level, in the present study a polyclonal specific antibody against DCLK1-S was successfully developed in our laboratory. This will provide further opportunities to investigators to obtain more accurate and valuable findings related to DCLK1-S localization and its clinical significance as the unique DCLK1 isoform responsible for invasiveness and aggressive behavior in various tumors by the IHC method.
Our findings demonstrated that most CRC samples with positive expression of DCLK1-S had pronounced cytoplasmic localization, besides partially nuclear and very low membranous expression patterns of S-isoform. In parallel, the results of immune electron microscopy (IEM) have confirmed the presence of S-isoform in the cytosol, mitochondrial, and cell membrane fractions of HCT-116 and COLO205-S-GFP cell lines, unlike L-isoform that was present mainly in cell membrane fractions of HEK293 cells only expressing L-isoform [12]. Other clinical studies that used anti-DCLK1-L/S or anti-DCLK1-L antibodies have also reported mainly cytoplasmic and membranous localization of DCLK1 in gastric, colorectal, and bladder carcinoma tissues [20, 21, 32, 42]. Meanwhile in the current study, the nucleus localization of DCLK1-S was shown in tumor cells area.
Besides, O’Connell et al. suggested that DCLK1-S was the main DCLK1 isoform expressing in humans CRC cell lines vs. DCLK1-L markedly expressing in normal colorectal cell lines; thus, DCLK1-S can be used as a promising prognostic and targeted-therapeutic marker in the treatment of CRC [11]. Other studies have demonstrated higher expression of DCLK1-S in polyps from high-risk vs. low-risk patients and downregulation of DCLK1-L/FOXD3 in polyps from high-risk compared to low-risk patients, indicating the potential suppressor function of FOXD3 in the regulation of DCLK1-S [18]. The existence of FOXD3 in normal or low-risk polyps inhibited expression of DCLK1-S, while methylation of FOXD3 in cancerous cells can cause overexpression of DCLK1-S in high-risk cases.
Our comparative immunostaining findings showed a similar pattern of immunoreactivity by anti-DCLK1-S antibody and antiDCLK1-L/S antibody, which both represented strong staining of DCLK1 in CRC tissues. A similar staining pattern in both antibodies may be due to the fact that DCLK1-S is the predominant isoform in CRC tissues. Although, further studies are required to show different expressions of these two isoforms.
Evaluating the circulatory DCLK1-S could lead to a better understanding of its biology and function in the tumor. However, this retrospective study aimed to investigate the DCLK1-S local protein expression as a novel isoform in the colorectalcancer tissues and its association with CRC patients’ outcome therefore, FFPE series of CRC samples were collected. The current study provided novel and interesting insights into the expression pattern and prognostic significance of DCLK1-S in a well-defined series of CRC tissues. Results of IHC analysis revealed a significant upregulation of cytoplasmic DCLK1-S expression in CRC samples compared to adjacent normal tissues. Furthermore, a significant association was found between high expression of DCLK1-S and advanced TNM stage, the increased tumor differentiation, and poor DSS in these cases. Consistent with our study, only one study pointed to a significant clinical value of DCLK1-S expression associated with tumor aggressiveness at mRNA level in the patients with CRC [11]. Additionally, our results showed that 5-year DSS was equal to 50, 62, and 74% for the patients with high, moderate, and low cytoplasmic expression of DCLK1-S, respectively.
Conclusions
Taken together, to better understand the specific role of the main DCLK1 isoform in the aggressiveness of CRC tumors, a specific polyclonal anti-DCLK1-S antibody was produced identified only by six particular amino acids related to short isoform and its specificity was validated by IHC method in CRC samples. Our findings strongly supported the critical role of DCLK1-S as the DCLK1 isoform, which led to tumor aggressiveness and worsened DSS in CRC samples. Significantly, high cytoplasmic expression of DCLK1-S protein compared to moderate expression was considered an independent poor prognostic factor of DSS in these series of CRC cases. Hence, additional investigations and larger trials on the potential therapeutic role of DCLK1-S as a novel CRC agent can shed new light on the treatment of patients with CRC.
Authors’ contributions
Z.M. and R.Gh. designed and supervised the work; E.K. wrote the manuscript, did the literature search, data acquisition, and statistical analysis constructed TMA blocks, and performed all experiments. L.S. interpreted the data, helped to prepare the Tables, and contributed to edit the manuscript. M.R. scored TMAs slides after immunohistochemically staining and helped to prepare the Fig. 2. L.E. helped to prepare the blood sampling of the rabbits in the animal experiment section. M.R. helped with the literature search and prepared Fig. 1. M.A. contributed to manuscript edit and data analysis. All authors read and approved the final manuscript.
Funding
Research reported in this publication was supported by Elite Researcher Grant Committee under award number [982625] from the National Institutes for Medical Research Development (NIMAD), Tehran, Iran. The current study was part of a PhD thesis with Grant Number: 96-03-126-32186, which was carried out at the Oncopathology research center at the Iran University of Medical Sciences.
Ethics approval and consent to participate
The current study was approved by the Human Research Ethics Committee of the Iran University of Medical Sciences, Tehran, Iran (Ref No: IR.IUMS.REC 1396.32186). All the procedures performed in this study were under the 1964 Declaration of Helsinki and its later amendments. Informed consent was obtained from all participants, parents or legally authorized representatives of participants at the time of sample collection with routine consent forms.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-210330.
sj-pdf-1-cbm-10.3233_CBM-210330.pdf - Supplemental material
Supplemental material, sj-pdf-1-cbm-10.3233_CBM-210330.pdf
Footnotes
Acknowledgments
We are grateful to our colleague at Iran University of Medical Sciences who provided insight and expertise that greatly assisted the research. Apart from this, it must be declared that the authors received no specific funding for this work.
Conflict of interest
The authors declare no competing interests.
