Abstract
BACKGROUND:
Recent reports indicated the importance of chemotractant CXCL-13 in solid tumors and lymphoid malignancies. However, the prognostic value of the mentioned cytokines as biomarkers in chronic lymphocytic leukemia patient’s remains to be identified. Therefore; this study was designed in order to address the relation between CXCL-13 concentrations levels and markers of severity in CLL patients.
METHODS:
Our study included 150 CLL patients and 20 controls. Serum CXCL-13 was determined by ELISA for CLL patients at diagnosis as well as controls.
RESULTS:
The serum CXCL-13 levels were significantly higher in CLL patients as compared to controls. The high CXCL-13 concentration levels was significantly associated with high number of smudge cells; high LDH; high grade of Rai stage, short time to first treatment (TTT). Cox regression analysis was conducted for prediction of TTT, using age, gender, WBCs, smudge cells, CXCL-13, LDH, ZAP70, CD38,
CONCLUSION:
High CXCL-13 serum levels at CLL diagnosis is correlated with other markers of disease activity; and could be served as biomarkers that predict CLL patient’s outcome.
Introduction
Chemokines are a group of small molecular weight proteins has the power to act as chemoattractants, thereby functioning to induce the migration of nearby responding cells. These chemokines, together with extracellular mediators, including growth factors, are key modulators of inflammation by controlling complex interaction networks via an autocrine and a paracrine mechanisms. Dysregulation of chemokines has been reported in certain diseases including autoimmune disorders, CNS infections; and lymphoma. CXCL-13 serum levels was suggested as a biomarker for angio-immunoblastic lymphoma (AITL) [1, 2, 3, 4].
CXCL-13 is the ligand for CXCR-5, also known as the Burkitt lymphoma receptor-1 (BLR1), and the CXCL-13/CXCR-5 axis is necessary for B cell homing to lymph node follicles and for the production of immunoglobulin, which coordinates the humoral immunity of the body [5]. There is also growing evidence that CXCL-13 and CXCR-5 participate in the pathogenesis of multiple subtypes of lymphomas [6]. Malignant cells in follicular lymphoma were shown to express CXCR5, secrete CXCL13, and migrate in response to CXCL-13, suggesting that CXCL-13 recruits malignant B-cells to ectopic germinal centers and contributes to their development [7]. Elevated CXCL-13 levels have been reported in CNS B cell lymphomas [9, 10], patients with idiopathic multicentric Castleman Disease [10, 11], cutaneous B and T-cell lymphoproliferative disorders, intraocular lymphomas, and chronic lymphocytic leukemia [12].
CXCL-13 and its receptor CXCR-5 have emerged as key players of cancer initiation and progression. The identification of an autocrine and a paracrine interactions between the tumor microenvironment and cancer cells mediated by CXCL-13 highlights how autonomous and non-autonomous mechanisms contribute to the development of the cancer phenotype and the dissemination of cancer cells to metastatic sites [13].
The CXCL13 as chemoattractant organizes the cellular architecture of B-cell follicles and germinal centers. CXCL13 plasma concentrations function as a biomarker for normal germinal center activity and increases during adaptive immune response [14]. Previous studies reported that CXCL13 plasma levels was correlated with CLL B-cell expansion [12, 13, 14]. Therefore; we hypothesized that CXCL13 plasma concentrations could correlate with CLL disease activity and progression.
The aim of this study is to evaluate soluble CXCL-13 concentration as a biomarker for refining CLL severity.
Patients and methods
This study included 150 CLL patients and 20 controls. CXCL-13 was determined by ELISA at time of inclusion in parallel with well-established prognostic markers including time to first treatment (TTT). The diagnosis of CLL cases was according to WHO 2016. For each one of the patients the followings were carried out; blood counts; blood smear; immunophenotyping by flowcytometry using the following panel (CD5/CD19; CD23; FMC7, sIg, CD79b) as well as Cytogenetic findings (17pdel; 11qdel; 13pdel; trisomy 12) by FISH techniques. All included patients and controls had given informed consent and the study had been approved by local ethical committee at Mansoura University Oncology Centre. The patients were included in our study between January 2016 to January 2019.
Inclusion Criteria
CLL patients at diagnosis before start of therapy.
Exclusion Criteria
CLL patients Under therapy.
CLL patients combined with other malignancy.
Serum CXCL13 quantification by enzyme-linked immunosorbent assay (ELISA)
Blood samples were obtained at CLL diagnosis and from controls in blank tubes. Serum samples were separated and preserved in aliquot 1.5 ml in
Statistical analysis
Categorical variables such as Rai stage risk, Cytogenetics abnormalities, CD38 and ZAP-70 expression were tabulated with frequency and percentage. While; continuous variables, such as white blood counts (WBC), LDH levels,
The Fisher’s exact test was used to evaluate the association between dichotomized CXCL-13 levels and categorical variables; the Mann Whitney U test or Kruskal-Wallis test was applied to compare between two groups or more than 2 groups regarding the continuous variables. Spearman rank correlations coefficients were used to evaluate the association between two continuous variables. The time to treatment (TTT) was defined as the time from sample collection to the date of the therapy; if patients have received any or censored on the last visit date. TTT was estimated by the method of Kaplan and Meier and assessed by the log-rank test to calculate the median TTT.
Cox models using univariate and multivariate were applied to evaluate the association of TTT with covariates. The clinical or laboratory variables which were significant in univariate analysis were retained in the multivariate analysis. The
Results
Clinical and laboratory characteristics in patient’s vs control
The present study was conducted on 150 CLL patients beside 20 healthy controls. Individuals in the CLL group were 105 males and 45 females; aged from (61.12
Patients characteristics
Patients characteristics
Categorization of patient’s characteristics and laboratory parameters among CLL patients according to the patients age
Comparison of patient’s characteristics and laboratory parameters among studied groups
Mann-whitney tests*, Chi-square test, independent sample
Stratification of CLL laboratory findings according to Ria stages indicated that high WBCs, increased smudge cells, high LDH, and CXCL-13
Comparison of patient’s characteristics and laboratory parameters among studied groups
Mann-whitney tests*, Chi-square test, ANOVA **
Comparison of patient’s characteristics and laboratory parameters namely WBCs, smudge cells, and LDH were significantly elevated in CLL patients group with CXCL-13
comparison between Clincopathological paramaters in patients with high vs those with low CXCL13
comparison between Clincopathological paramaters in patients with high vs those with low CXCL13
Mann-whitney tests*, Chi-square test, independent sample
ROC analysis was conducted to identify the optimal Cut off levels for prediction of TTT. The analysis revealed that best cut-off value of CXCL-13 that could predict TTT was 65.5. The area under the curve (AUC) was 0.846 (
Performance characteristics of CXCL13 for prediction of TTT
ROC analysis was conducted to identify the optimal Cut off levels for prediction of shorter TTT. CXCL13 best cut-off value was 65.5. The area under the curve (AUC) was 0.846 (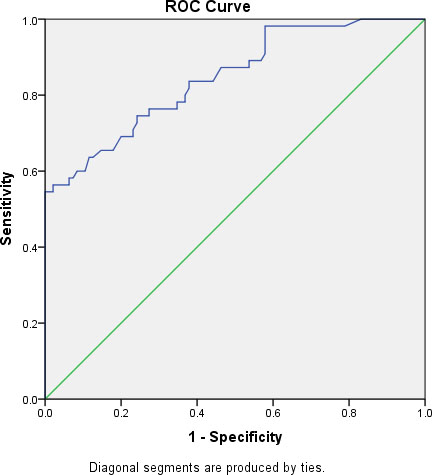
Of the 150 patients studied 110 progress and required treatment. Cox regression analysis was conducted to identify the best parameter that could predict TTT, using age, gender, WBCS, smudge cells count, CXCL-13, LDH, ZAP70, CD38,
Impact of CXCL-13 serum levels on CLL TTT was evaluated by Kaplan-Meier curve
To determine the differential impact of CXCL-13 serum levels on TTT, we categorized patients into 2 subgroups based on their CXCL-13 serum levels (CXCL-13
As regard CXCL13, TTT estimates 82.7% at 12 months interval and 82.7% at 25 months interval in CXCL13 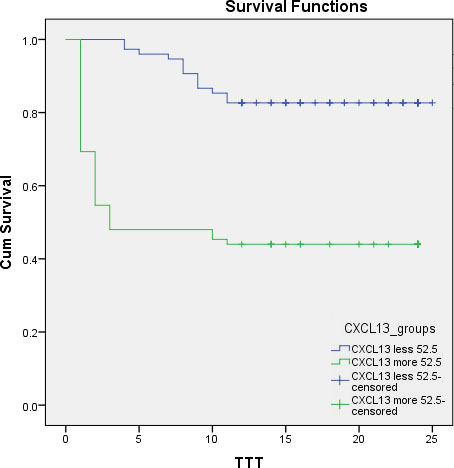
CXCL-13/CXCR-5 axis plays central role in cancer development and progression. The interaction between cancer cells and its micro-environment through the an autocrine as well as a paracrine interactions which is mediated by CXCL-13 highlights different pathways that contribute to development and dissemination of cancer cells to the sites of metastasis [13]. CXCL-13 was identified as the most up regulated cytokine in plasma from patients with idiopathic multicentric Castleman disease [10].
The present study was conducted on 150 patients with CLL beside 20 healthy controls. Individuals in the study were 105 males and 45 females aged from (61.12
Comparison of patient’s characteristics and laboratory parameters among studied subgroups, Male patients were frequently detected in group age
In the present study CXCL-13, WBCs and smudge cells were significantly higher in patients as compared to controls. Also, other laboratory parameters among studied groups namely, WBCs, smudge, LDH were significantly elevated in high risk Rai staging compared to other groups. On the other hand TTT was significant reduced in high risk Rai stage when compared to other groups. Also; there were significant difference as regard ZAP-70, CD38,
Cox regression analysis was conducted for prediction of TTT, using age, gender, WBCs, smudge cells, CXCL-13, LDH, ZAP-70, CD38,
Survival analysis studies was evaluated by Kaplan Miere curve in order to address the impact of CXCL-13
Conclusion
Our findings indicated that CXCL-13 serum concentrations levels at CLL diagnosis is a valuable biomarkers for CLL disease staging as well as a marker for disease activity.
Funding statement
This work was not supported by a grant.
Author contributions
Salah Aref: Conception and supervision.
Ahmed Ramez: Interpretation and analysis of data.
Doaa Atia: Laboratory work; Preparation of Manuscript.
Tarek Abou Zeid: Interpretation and analysis of data.
Enas Gouda: Lab Work; Preparation of the manuscript; Revision for important intellectual.
Footnotes
Conflict of interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
