Abstract
BACKGROUND:
This study aimed to investigate the efficiency of combining tumor-associated antigens (TAAs) and autoantibodies in the diagnosis of lung cancer.
METHODS:
The serum levels of TAAs and seven autoantibodies (7-AABs) were detected from patients with lung cancer, benign lung disease and healthy controls. The performance of a new panel by combing TAAs and 7-AABs was evaluated for the early diagnosis of lung cancer.
RESULTS:
The positive rate of 7-AABs was higher than the single detection of antibody. The positive rate of the combined detection of 7-AABs in lung cancer group (30.2%) was significantly higher than that of healthy controls (16.8%), but had no statistical difference compared with that of benign lung disease group (20.8%). The positive rate of 7-AABs showed a tendency to increase in lung cancer patients with higher tumor-node-metastasis (TNM) stages. For the pathological subtype analysis, the positive rate of 7-AABs was higher in patients with squamous cell carcinoma and small cell lung cancer than that of adenocarcinoma. The levels of carcinoembryonic antigen (CEA) and cytokeratin 19 fragment 211 (CYFRA 21-1) were significantly higher than that of benign lung disease and healthy control groups. An optimal model was established (including 7-AABs, CEA and CYFRA21-1) to distinguish lung cancer from control groups. The performance of this model was superior than that of single markers, with a sensitivity of 52.26% and specificity of 77.46% in the training group. Further assessment was studied in another validation group, with a sensitivity of 44.02% and specificity of 83%.
CONCLUSIONS:
The diagnostic performance was enhanced by combining 7-AABs, CEA and CYFRA21-1, which has critical value for the screening and early detection of lung cancer.
Introduction
Lung cancer is one of the most common malignant tumors, as well as the leading causes of cancer-related death worldwide [1, 2]. The average 5-year survival rate of lung cancer is approximately 17.4%, but this decreases dramatically as the cancer becomes more advanced. Early screening, detection and diagnosis are crucially important to reduce the mortality and improve the overall survival rate of patients with lung cancer [3, 4, 5].
For the early diagnosis of lung cancer, low-dose computed tomography (LDCT) screening is recommended in patients with high risk, demonstrating a sensitivity of 93.8%. The application of LDCT reduces a mortality of 20% in lung cancer patients [6]. However, LDCT cannot distinguish benign nodules from early malignant tumors accurately, which leads to high rate of false-positive results. The repeated radiation exposure and substantial costs also limit the widespread application of CT as a screening procedure in clinical practice [7, 8]. Histopathology is typically used to diagnose lung cancer, but it is invasive. Current serum diagnostic biomarkers for lung cancer focused on tumor-associated antigens (TAAs), such as carcinoembryonic antigen (CEA), carbohydrate antigen 125 (CA-125), cytokeratin 19 fragment 21-1 (CYFRA 21-1), squamous cell carcinoma related antigen (SCC-Ag) and Progastrin releasing peptide (ProGRP). These TAA markers showed an increased positive rate in patients with advanced stages, but are rarely used as early biomarkers because of their low sensitivity and specificity [9]. Recent studies reported that detection of serum tumor-associated autoantibodies (TAAbs), which are produced by host cells against TAAs, may be a promising cancer screening method [10]. TAAbs are more stable in peripheral blood than TAAs, and exhibit better sensitivity and specificity in the previous studies [11, 12]. For single antibody detection, the sensitivity was low and panels of different autoantibodies showed better diagnostic performance, with the sensitivity ranging from 30.0% to 94.8%, specificity ranging from 73% to 100.0%, and auras under ROC curve (AUCs) ranging from 0.630 to 0.982 [12]. The combination of multiple autoantibodies had potential efficacy used as diagnostic tools in tumors [13, 14]. Currently, a panel of seven autoantibodies (7-AABs), including p53, PGP9.5, SOX2, GAGE7, GBU4-5, MAGEA1 and CAGE are more prevalent and proved to be useful blood biomarkers for the diagnosis of early lung cancer or distinguishing benign nodules from malignant tumors [5, 12, 15, 16]. However, the diagnostic sensitivity of 7-AABs varied greatly in different studies, and whether the combination of 7-AABs with TAAs could improve the diagnosis of lung cancer still needs further investigated. Correspondingly, it’s essential to explore more effective combination of serum markers for the screening and early diagnosis of lung cancer.
This study aimed to investigate the clinical value of 7-AABs combined with TAAs, including CEA, CYFRA21-1, SCC-Ag and ProGRP for the identifying of lung cancer not only from healthy controls but also from benign lung diseases.
Materials and methods
Patients
This was a retrospective study and the subjects were recruited from Tongji hospital, Tongji Medical College, Huazhong University of Science and Technology, from June to October 2020. The lung cancer and benign lung disease groups were included according to the pathology results after surgery. In the training set, a total of 199 patients with lung cancer and 72 patients with benign lung diseases (lung benign disease and non-tumor benign disease) according to the pathology results were included, Tumor histology was classified according to the World Health Organization guidelines for histologic type of lung tumors. Tumor-node-metastasis (TNM) stage was determined according to the American Joint Committee on Cancer staging manual (seventh edition). In addition, 172 age and sex matched healthy controls (HCs) were also recruited and determined by interview, physical examination and with normal CT results. The exclusive criteria include HIV and HCV positive, and patients who had antineoplastic therapy, radiotherapy or chemotherapy before surgery or cancer diagnosis. To validate this study, the following subjects were recruited from Sino-French New City of Tongji hospital, including 258 lung cancer, 81 benign lung disease and 170 healthy controls. The study protocol was approved by the ethical committee of Tongji hospital, Tongji Medical College, Huazhong University of Science and Technology (TJ-IRB20210331). All of the subjects provided written informed consent.
Specimen collection and processing
Serum from 3–5 ml fasting blood was separated by centrifugation at 3000 rpm for 10 min within 4 hours of sample collection. Samples that could not be processed immediately were stored at
Quantitation of autoantibodies in serum samples
The serum concentrations of autoantibodies were quantitated by a commercial enzymelinked immunosorbent assay (ELISA)-based test kit (Hangzhou Cancer Probe Biotech Co., Ltd, Hangzhou, China). The ELISA kit was tested according to the manufacturer’s recommendations. In brief, the detection kit components were equilibrated to room temperature and diluted based on the instructions. We washed the antigen-coated wells with 200–300
Statistical analysis
The data were analyzed using SPSS 22.0, Graphpad Prism 6.0 and MedCalc (Version 18.2.1). The results are presented as mean
Results
This study enrolled 443 participants for the training group and 509 participants for the validation group. The basic characteristics of the included population were shown in Table 1. A total of 199 pathologically confirmed lung cancer (including 162 adenocarcinoma (AD), 31 squamous cell carcinoma (SCC), 5 small cell lung cancer (SCLC) and 1 large cell lung cancer (LCLC)), 72 benign lung disease (including 6 lung benign disease and 66 non-tumor benign disease) and 172 healthy controls were included in the training group. The validation group comprised 258 lung cancer (including 222 AD, 27 SCC, 8 SCLC and 1 LCLC), 81 benign lung disease (17 lung benign disease and 64 non-tumor benign disease) and 170 healthy controls. According to the TNM stage, 65.3% and 75.6% of the patients with lung cancer were classified as stage I in the training and validation cohorts, respectively.
Clinical characteristics of all study participants and positive rates of autoantibodies
Clinical characteristics of all study participants and positive rates of autoantibodies
Data were presented as median or numbers (%).
The positive rate of combined autoantibody detection in different groups of lung cancer patients
The serum concentrations of the 7-AABs (including p53, GAGE7, PGP9.5, CAGE, MAGEA1, SOX2, and GBU4-5) were determined by ELISA. By using the commercial assay cutoffs, positivity is defined as having an elevated AABs assay signal. The positive rate of 7-AABs was higher than the single detection of antibody (
Comparisons of single autoantibody biomarker levels in the lung cancer, benign lung disease and healthy control groups were conducted. In the training group, only serum levels of PGP9.5 were markedly higher in lung cancer patients compared with that of benign lung disease and healthy control groups (Fig. 1A and B). In the validation group, the concentrations of CAGE, MACE A1, PGP9.5 were significantly higher in lung cancer group (Fig. 1C). Both in the training and validation cohorts, the levels of CEA and CYFRA21-1 in lung cancer group were significantly increased, but not for SCC-Ag and ProGRP (Fig. 1B and D).
Comparisons of each tumor-associate autoantibodies and antigens among lung cancer, benign lung disease and healthy controls in the training and validation groups. Data were shown as mean 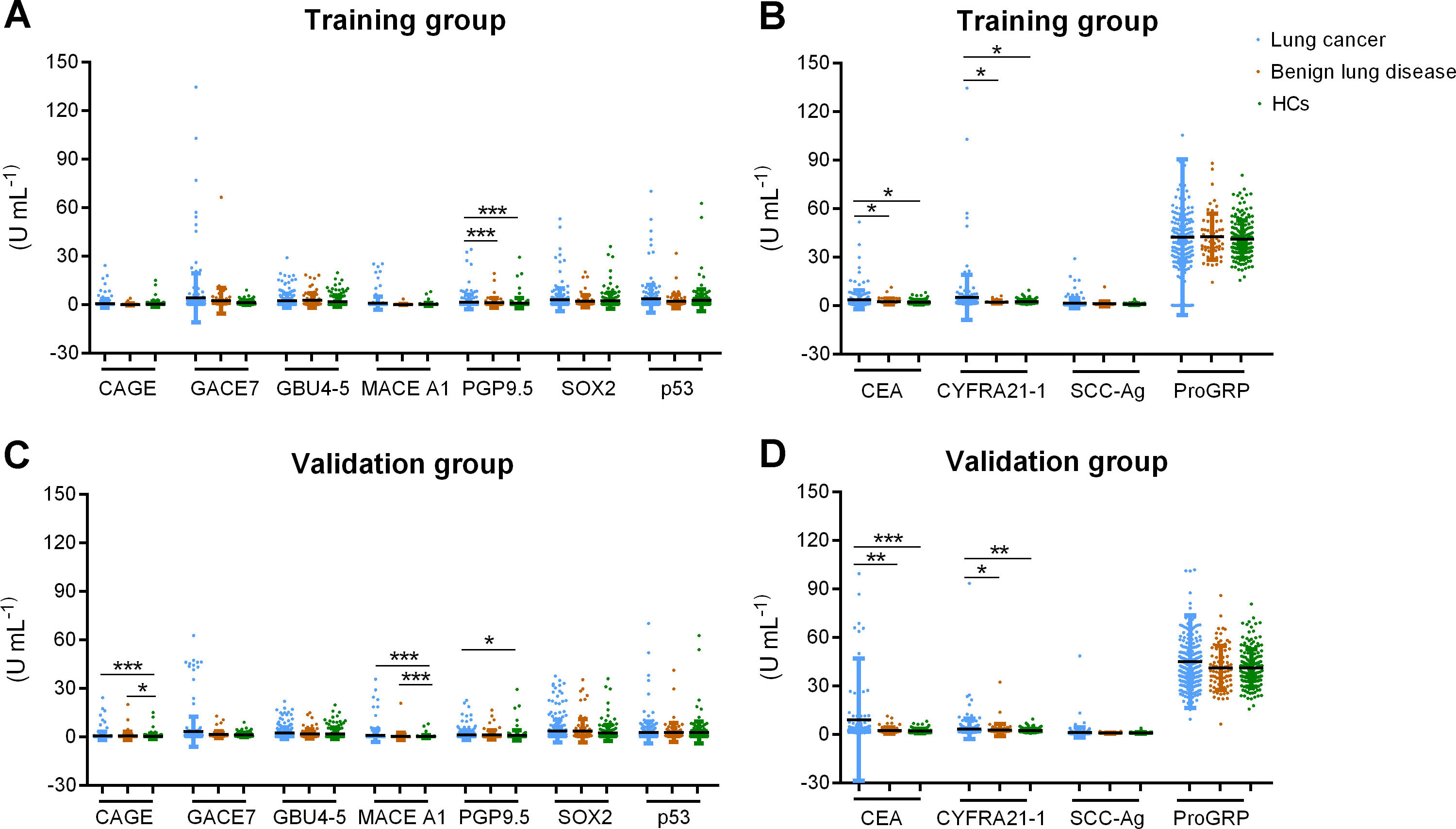
ROC analysis was conducted to estimate the performance of 7-AABs and traditional tumor biomarkers using the data of training group (Fig. 2A). The performance of 7-AABs seemed to be unsatisfied for discriminating lung cancer from benign lung disease (AUC 0.547, 95% CI: 0.485–0.607) or from healthy controls (AUC 0.566, 95% CI: 0.514–0.617). New diagnostic panels were established according to the logistic regression analysis. When benign lung disease used as control group, only CEA and CYFRA21-1 were significant variables for predicting the diagnosis of lung cancer. The probability of lung cancer was calculated based on the equation from the logistic regression model:
We compared the diagnostic value of the model (AUC: 0.678, 95%CI: 0.606–0.721) and found that the comprehensive performance of the diagnostic model was better than CEA (AUC: 0.607, 95%CI: 0.546–0.666) and CYFRA21-1 (AUC: 0.638, 95%CI: 0.578–0.696) (
The performance of the diagnostic model (AUC: 0.681, 95%CI: 0.631-0.728) was superior than 7-AABs (AUC: 0.566, 95%CI: 0.514–0.617), CEA (AUC: 0.651, 95%CI: 0.546–0.666) and CYFRA21-1 (AUC: 0.592, 95%CI: 0.540–0.642) (
At last, this model exhibited an AUC of 0.686 (95%CI: 0.640–0.729) and was statistically better than the single markers, with a sensitivity of 52.26% and specificity of 77.46% (Table 3).
In the validation group, ROCs of the 7-AABs panel and TAAs were analyzed (Fig. 2B). Between lung cancer and benign lung disease groups, the diagnostic model was validated with an AUC of 0.657 (95%CI: 0.604–0.708), sensitivity of 56.03% and specificity of 69.14%. The diagnostic model for discriminating lung cancer from healthy controls also showed a better diagnostic efficiency with an AUC of 0.663 (95%CI: 0.616–0.708), sensitivity of 47.47% and specificity of 73.26%. In the whole control group, the performance of diagnostic model exhibited an AUC of 0.668 (95%CI: 0.626–0.709), with a sensitivity of 44.02% and specificity of 83.00% (Table 3).
Comparison of different markers in distinguishing lung cancer from benign lung disease and healthy controls
LC: lung cancer, HCs: healthy controls.
Receiver operator characteristic (ROC) curves for each single tested biomarkers and diagnostic models in different cohorts. (A) Comparison of the diagnostic ability of the model and each biomarker in the training group. (B) Comparison of the diagnostic ability of the model and each biomarker in the validation group. LC: lung cancer, HCs: healthy controls.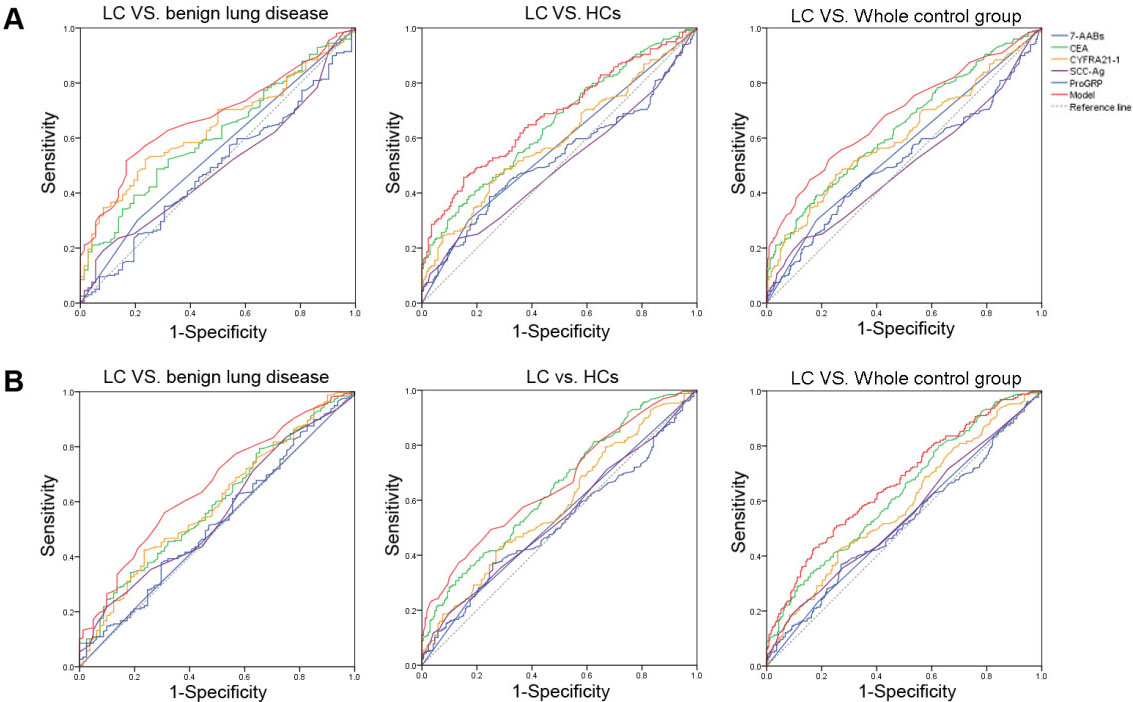
Lung cancer is one of the most common cancers as well as the leading cause of cancer-related death worldwide [17]. In the early stages, patients with lung cancer are asymptomatic or atypical, and most patients have local or distant metastasis at the time of diagnosis, leading to the low 5-year survival rate (16%) [18]. CT scanning could reduce the incidence of patients with late stage lung cancer, but low specificity of CT might expose patients to radiation and unnecessary further examination [6]. Various blood biomarkers have been reported for the diagnosis of lung cancer, such as circulating tumor cells, tumor DNA, microRNA, traditional TAAs and tumor-associated autoantibodies [18, 19, 20]. Autoantibodies are produced in the process of oncogenesis and can be detected before the presence of TAAs [21, 22]. Combined detection of autoantibodies have been reported to have potential efficacy as diagnostic and prognostic tools in lung cancer [12]. The sensitivity of a single autoantibody is very low due to the complexity and heterogeneity of lung cancer as well as the expression of TAAs [23]. Previous studies have reported that the performance of TAAbs combination detection have been evaluated, and the specificity was high but with low sensitivity [12, 16, 21, 22]. Therefore, to improve the clinical value in early screening and diagnosis of lung cancer in combination of TAAs and TAAbs still needs to be developed.
In the present study, a panel of seven autoantibodies (including p53, PGP9.5, SOX2, GAGE7, GBU4-5, MAGEA1 and CAGE) were assessed and appeared to more frequently present in the serum of lung cancer than that of healthy controls, but not for benign lung disease. Our results show that the positive rate of 7-AABs was higher in SCC and SCLC than AD [16], and had an increased tendency in patients with progressed TNM stages. However, some studies presented that no obvious differences of positive rates were observed in lung cancer patients with different TNM stages, which might be due to the different participants and needs to be investigated in larger populations [5, 24].
The traditional tumor biomarkers, such as CEA, CYFRA21-1, SCC-Ag and ProGRP have been widely applied as diagnostic and predictive markers of lung cancer [25]. CEA was reported to be highly associated with primary tissue, lymphatic metastasis, and distant metastasis [26]. CYFRA21-1 was expressed in respiratory epithelial cells and could be detected in NSCLC tissue [27]. The expression of SCC-Ag was correlated with differentiation degree of squamous cell carcinoma tissue but had no additional value when used in combination with CYFRA21-1 due to its low sensitivity [28]. ProGRP was a useful biomarker for the diagnostic and evaluation of SCLC [29]. In the current study, the levels of CEA and CYFRA21-1 were observed significantly increased in lung cancer group both in the training and validation cohorts, which suggested to be useful markers to establish a well optimized model with strong predictive value.
Further multiple logistic regression analysis was performed to select biomarkers for a mixed model. Previous studies showed that combination detection of 7-AABs and serum tumor markers could improve the diagnostic accuracy for lung cancer [30]. Unexpectedly, only CEA and CYFRA21-1 were included to yield a final model for distinguishing lung cancer from benign lung disease. When distinguishing lung cancer from healthy controls, 7-AABs, CEA and CYFRA21-1 were significant variables. Moreover, benign lung disease and healthy controls were used as a whole control group, the diagnostic performance was evaluated using a new model including 7-AABs, CEA and CYFRA21-1, with a sensitivity of 52.26, specificity of 77.46%. Further validation was also conducted among different groups and similar results were obtained in the validation group. Accordingly, 7-AABs could contribute the early diagnostic of lung cancer, especially when differentiating from healthy controls and combination of 7-AABs, CEA and CYFRA21-1 could improve the whole diagnostic performance. The performance of 7-AABs in the current study was not as superior as reported, which might be due to the heterogeneity of cohorts and detection platform within different studies [31, 32]. Therefore, larger numbers of samples to validate the efficiency of TAAs and autoantibody combination detection should be done and further non-invasive markers still need to be investigated for the early diagnostic of lung cancer, especially when differentiated from benign lung disease.
Conclusion
This study showed that the positive rate of 7-AABs was higher in lung cancer group than that of healthy controls. A new panel of detecting CEA, CYFRA21-1 and 7-AABs was established and could contribute the early detection of lung cancer in some extent. Biomarkers for the screening and early diagnostic of lung cancer still need to be explored.
Funding
This study was supported by the National Mega Project on Major Infectious Disease Prevention (2017 ZX10103005-007).
Author contribution
Hongyan Hou designed the project and wrote the original draft; Renren Ouyang, Shiji Wu, Ting Wang, Wei Wei, Min Huang collected the data; Bo Zhang, Botao Yin, Jin Huang and Minxia Zhang analyzed the data; Yun Wang and Feng Wang reviewed and edited the manuscript.
Ethics statement
The study protocol was approved by the ethical committee of Tongji hospital, Tongji Medical College, Huazhong University of Science and Technology.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Footnotes
Acknowledgments
This study was supported by the National Mega Project on Major Infectious Disease Prevention (2017 ZX10103005-007).
Conflict of interest
The authors have no conflicts of interest to declare.
