Abstract
BACKGROUND:
Although lysyl-tRNA synthetase (KARS1) is predominantly located in the cytosol, it is also present in the plasma membrane where it stabilizes the 67-kDa laminin receptor (67LR). This physical interaction is strongly increased under metastatic conditions. However, the dynamic interaction of these two proteins and the turnover of KARS1 in the plasma membrane has not previously been investigated.
OBJECTIVE:
Our objective in this study was to identify the membranous location of KARS1 and 67LR and investigate if this changes with the developmental stage of epithelial ovarian cancer (EOC) and treatment with the inhibitor BC-K01. In addition, we evaluated the therapeutic efficacy of BC-K01 in combination with paclitaxel, as the latter is frequently used to treat patients with EOC.
METHODS:
Overall survival and prognostic significance were determined in EOC patients according to KARS1 and 67LR expression levels as determined by immunohistochemistry. Changes in the location and expression of KARS1 and 67LR were investigated in vitro after BC-K01 treatment. The effects of this compound on tumor growth and apoptosis were evaluated both in vitro and in vivo.
RESULTS:
EOC patients with high KARS1 and high 67LR expression had lower progression-free survival rates than those with low expression levels of these two markers. BC-K01 reduced cell viability and increased apoptosis in combination with paclitaxel in EOC cell xenograft mouse models. BC-K01 decreased membranous KARS1 expression, causing a reduction in 67LR membrane expression in EOC cell lines. BC-K01 significantly decreased in vivo tumor weight and number of nodules, especially when used in combination with paclitaxel.
CONCLUSIONS:
Co-localization of KARS1 and 67LR in the plasma membrane contributes to EOC progression. Inhibition of the KARS1-67LR interaction by BC-K01 suppresses metastasis in EOC.
Keywords
Abbreviations
Introduction
Among gynecologic cancers, epithelial ovarian cancer (EOC) is the leading cause of death [1]. Approximately 70% of patients show a favorable initial response to primary treatment including surgery and chemotherapy. However, successful management of EOC is difficult because most cases are diagnosed at advanced stages and recur with distant metastases [2, 3]. Identification of alternative therapeutic approaches and targets for controlling metastases is urgently required to improve the prognosis of patients with EOC.
Because metastasis is directly related to cancer mortality [4], methods to control metastasis are important. Invasion, which is the initiation step of cancer metastasis, requires proteolytic degradation of surrounding tissues and the extracellular matrix (ECM) and altered cancer cell adherence. Laminin is a major constituent of the ECM and has critical roles in cell adhesion, differentiation, and migration [5] by interacting with receptors such as integrins and the 67-kDa laminin receptor (67LR). High expression of 67LR in breast, lung, ovary, colon, and prostate carcinomas and lymphomas has been reported and is positively correlated with cancer progression and malignancy [6, 7, 8, 9, 10]. In an EOC study published by our group, positive 67LR expression was associated with advanced EOC stages and shorter progression-free survival than negative 67LR expression (PFS) [9]. In addition, treatment of human EOC cell lines with 67LR siRNA inhibited tumor cell proliferation and invasion while enhancing apoptosis [9].
Lysyl-tRNA synthetase (KARS1) relocates to the plasma membrane after a laminin signal and stabilizes 67LR, which is implicated in cancer metastasis [11]. BC-K01 interacts with KARS1 to inhibit association with 67LR and suppress metastasis, as shown in a previous study of mouse models of breast and lung cancer [12]. BC-K01 decreases the amount of membranous 67LR and suppress cell morphology changes, actin rearrangements, FAK activation, and cell invasion.
Both KARS1 and 67LR are predominantly located in the cytosol of cells and are involved in protein synthesis [11, 13]. KARS1 charges lysine to empty tRNA to produce lysyl-tRNA and forms multi-synthetase complexes with other tRNA synthetases [14]. 67LR originates from 37LRP/p40 RPSA, a ribosome subunit [15]. The precursor is dimerized by acetylation and embedded in the plasma membrane [16]. Laminin attachment of a cancer cell triggers physical interaction between KARS1 and 67LR, and this linkage significantly enhances the metastatic ability of cancer cells [11]. Protein expression levels and the subcellular location of proteins are functionally associated with clinical symptoms. Although previous studies have investigated the 3D structures of KARS1 and 67LR in conjunction with BC-K01, dynamic changes in the interaction between KARS1 and 67LR and their localization have not been investigated [12]. Asynchronous movement of KARS1 and 67LR from the cytosol to the membrane and the membrane to cytosol was observed in this study. EOC metastasis can potentially be suppressed by inhibiting the interaction between KARS1 and 67LR.
Materials and methods
Patient and tissue specimens
Fifty-two patients who underwent surgery for EOC at the Department of Obstetrics and Gynecology at Samsung Medical Center between October 2003 and November 2005 were included in this study. This study was reviewed and approved by the Institutional Review Board of Samsung Medical Center, Seoul, Korea. Formalin-fixed, paraffin embedded tissues were obtained from primary ovaries obtained during surgery of women with EOC (
Immunohistochemical analysis
Immunohistochemical staining was performed with the standard peroxidase/DAB method (Dako Cytomation, Glostrup, Denmark) on formalin-fixed, paraffin-embedded tissue sections. KARS1 expression was detected by primary rabbit polyclonal KARS antibody (NMS-01-0005; Neomics, Gyeonggi-do, Korea). 67LR expression was detected by primary rabbit polyclonal 67LR antibody (NBP1-33002; Novus Biologicals, Littleton, CO, USA), as described previously [9]. Immunohistochemical procedures were performed as described previously [18]. Antigen-antibody reactions were detected using the Dako REAL
Western blot analysis
Cells were lysed using the Membrane Protein Extraction Kit (K268; BioVision, Milpitas, CA, USA). Protein lysates were separated on 6–10% acrylamide gels by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to Hybond-ECL nitrocellulose filter paper (GE 10600002; Amersham, Buckinghamshire, UK). Membranes were blocked with 5% BSA in Tris-buffered saline containing 0.1% Tween-20 for 1 h at room temperature. Protein bands were probed with KARS antibody (NMS-01-0005; Neomics, Gyeonggi-do, Korea), 67LR antibody (NBP1-51270; Novus Biologicals, Colorado, USA), pan-cadherin antibody (sc-59876; Santa Cruz Biotechnology, Inc., Dallas, TX, USA), or Hsp90 antibody (ab13492; Abcam, Cambridge, UK). Horseradish peroxidase-conjugated anti-rabbit (ab6721; Abcam, Cambridge, UK), anti-mouse antibody (ab6728; Abcam, Cambridge, UK), or anti-goat antibody (ab205723; Abcam, Cambridge, UK) were used as secondary antibodies. Bands were visualized by enhanced chemoluminescence using an ECL kit (32106; Thermo Fisher Scientific, Waltham, MA, USA) according to the manufacturer’s protocol.
Immunofluorescence staining
For immunofluorescence staining, cells were fixed with chilled methanol at
Fluorescence-activated cell sorting (FACS)
EOC cells were seeded in 60-mm plates for FACS analysis. A CytoFLEX flow cytometer was used (Beckman Coulter, California, USA). Laminin (final concentration 10
Cell culture
HeyA8 and SKOV3ip1 cell lines were gifts from Dr. Anil K. Sood, Department of Cancer Biology, University of Texas M.D. Anderson Cancer Center, TX, USA. H460, H1299, and H596 cell lines were purchased from the Korean Cell Line Bank, Korea. A2780 and SK-Lu-1 were purchased from ATCC. Human EOC cell lines were maintained in complete media (RPMI 1640) supplemented with 10% fetal bovine serum (FBS) and 0.1% gentamicin sulfate (Gemini Bioproducts, Calabasas, CA) in 5% CO
WST-based cell viability assay
WST-based cell viability assay was performed using the WST-1 assay kit according to the manufacturer’s protocol (ab65475; Abcam, Cambridge, UK). To determine the cell viability of drug-treated cells, cells were seeded on 96-well plates (1
Animal care and development of in vivo models, including established cell line xenografts
Female BALB/c nude mice were purchased from Orient Bio, Seongnam, Korea. This study was reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) of Samsung Biomedical Research Institute (protocol no.H-A9-003), which is accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care International (AAALAC International) and abides by the guidelines of the Institute of Laboratory Animal Resources (ILAR). To establish orthotopic models, HeyA8 (1
TUNEL assay
Apoptotic cell death was assessed using a terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling (TUNEL) assay with a commercially available apoptosis detection kit (Promega, Fitchburg, WI) according to the manufacturer’s instructions. Briefly, after routine deparaffinization, rehydration, and blocking of endogenous peroxidases with 3% hydrogen peroxide in PBS for 10 min at room temperature, tissue sections were digested with 20
Cell impedance analysis
To assess the metastatic ability of cells, real-time cell attachment to plates was evaluated. The iCELLigence (380600970; ACEA Biosciences, California, USA) Label-Free Real-Time Cell Analysis System (ACEA Biosciences, USA) was used for this experiment. All setting procedures were performed following the manufacturer’s protocol. In brief, cell attachment was detected using electric cell culture plates. When cells attach to a plate, the resistance of the bottom cell plate changes. After seeding 1
Statistical analysis
The Mann-Whitney U test was used to evaluate significance and compare differences among groups in both in vitro and in vivo assays. All statistical tests were two-sided, and
Epithelial ovarian cancer (EOC) patient characteristics. EOC patients were divided by expression level of 67LR and KARS1. The group that expressed both 67LR and KARS at high levels were assigned to the combined index 1 group, while the remaining patients were assigned to the combined index 0 group. This table shows the differences in general characteristics of these two groups of EOC patients
Epithelial ovarian cancer (EOC) patient characteristics. EOC patients were divided by expression level of 67LR and KARS1. The group that expressed both 67LR and KARS at high levels were assigned to the combined index 1 group, while the remaining patients were assigned to the combined index 0 group. This table shows the differences in general characteristics of these two groups of EOC patients
Prognosis of patients with ovarian cancer. (A) Representative immunohistochemistry (IHC) staining to evaluate 67LR and KARS1 expression in EOC tissue. Serous epithelial ovarian cancer were scored as positive or negative for 67LR and KARS1 expression. (B-C) KARS1 and 67LR combined index-based progression-free survival (PFS) curves (B) and overall survival (OS) curves (C) of 52 patients with EOC.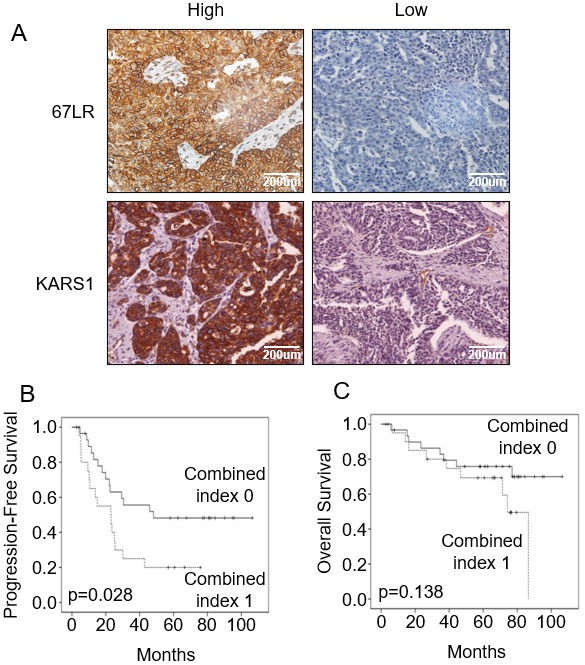
Expression of KARS1 and 67LR and prognosis of EOC patients
KARS1 and 67LR expression levels were assessed using immunohistochemistry in 52 EOC cases including 40 serous, 10 endometrioid, and 2 mucinous tissue subtypes (Table 1). Median follow-up time of the study cohort (
Multivariate Cox proportional hazards analysis of progression-free survival (PFS) and overall survival (OS). This table shows PFS and OS according to several classifications related to the prognosis of EOC patients
Multivariate Cox proportional hazards analysis of progression-free survival (PFS) and overall survival (OS). This table shows PFS and OS according to several classifications related to the prognosis of EOC patients
PFS: progression-free survival, OS: overall survival, HR: hazard ratio, CI: confidence interval, PDS: primary debulking surgery.
Survival rates of patients with ovarian cancer based on 67LR and KARS1 expression. (A-B) Progression-free survival based on expression of KARS1 (A) and RPSA (B). (C) Survival probability according to the combined group (high expression of both KARS1 and RPSA, low expression of both KARS1 and RPSA).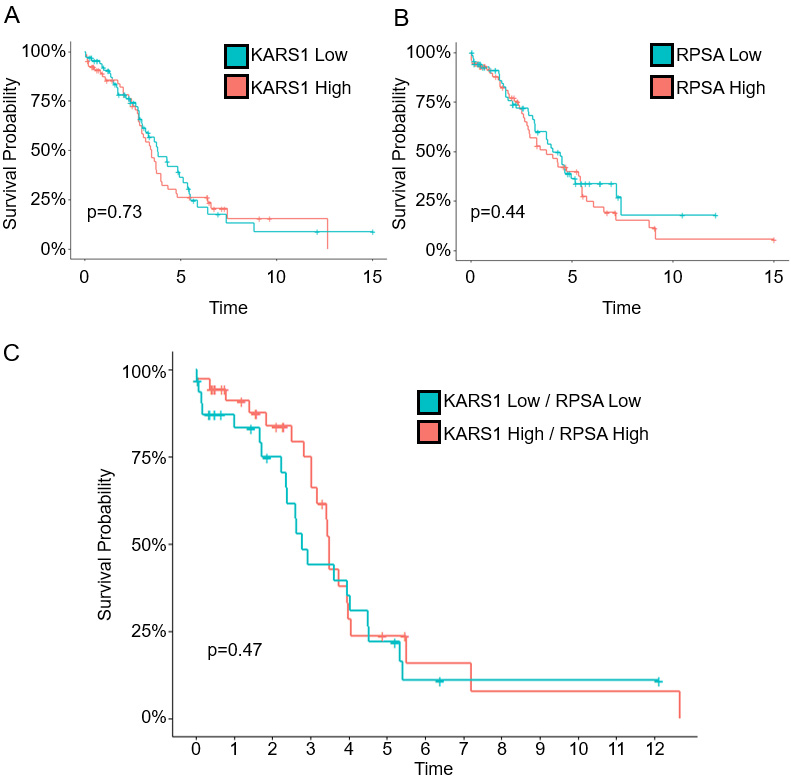
Changes in KARS1 and 67LR localization in response to BC-K01 treatment of the epithelial ovarian cancer cell line Hey A8. (A) Changes in the expression of KARS1 and 67LR over time at the plasma membrane after laminin (10 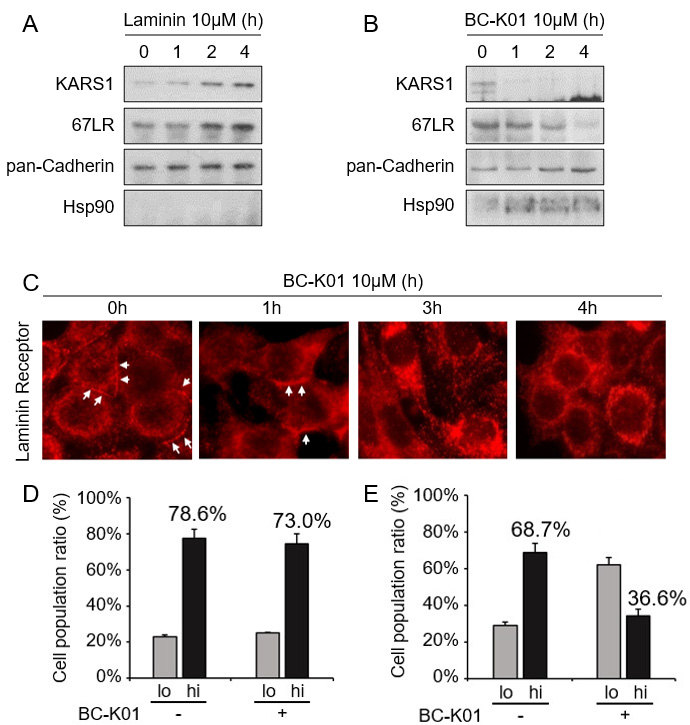
Highly adhesive EOC cells are more sensitive to BC-K01 than less adhesive EOC cells. (A) Western blot to assess ERK phosphorylation in Hey A8 cells after BC-K01 treatment for 4 h under laminin (LN) coated conditions. (B) Graph indicates relative attachment force. 1 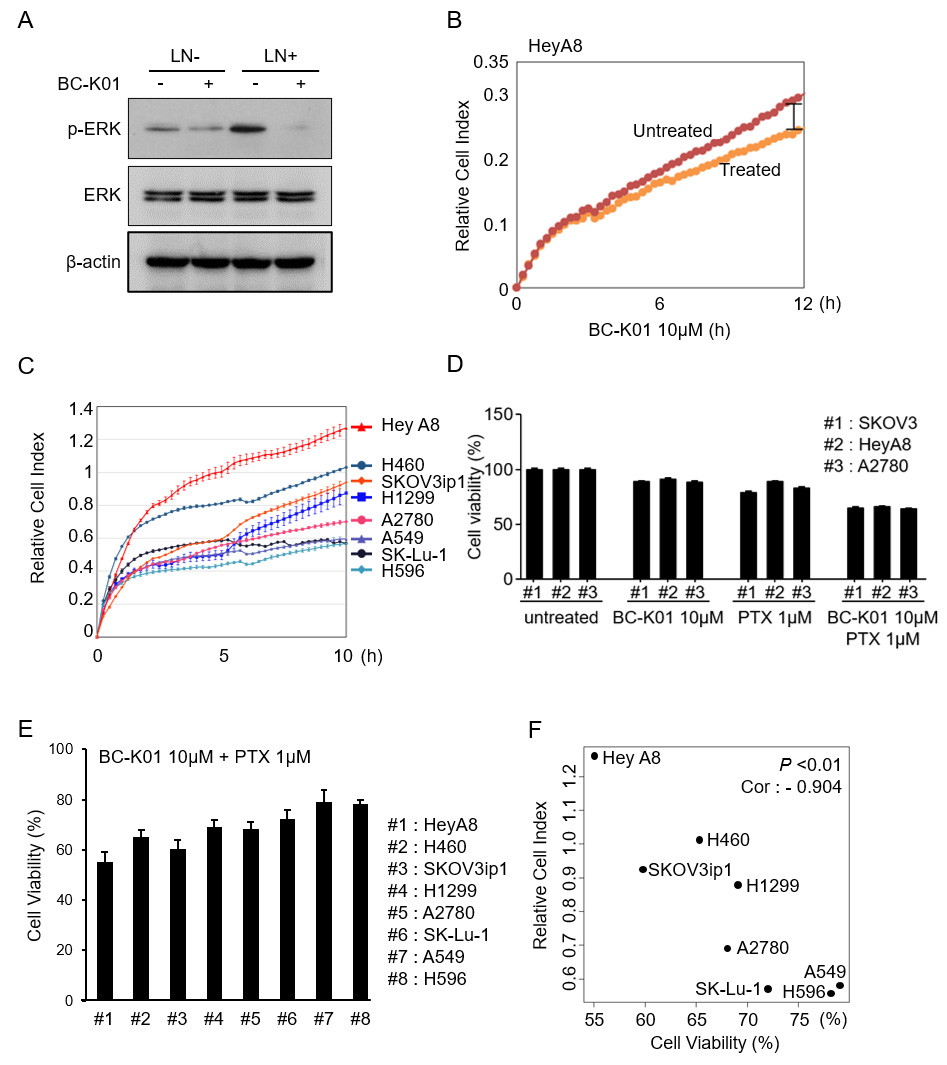
In vitro effects of BC-K01 in orthotopic mouse models of EOC. (A-B) Combined BC-K01 and paclitaxel treatment decreased tumor weight in the Hey A8 model (A) and SKOV3ip1 model (B). (C-D) The effect of combined treatment on tumor weight was assessed in the Hey A8 xenograft model (C) and SKOV3ip1 xenograft model (D). Significant decreases in tumor weight (A-B) and nodules (C-D) were observed. (E) Apoptotic activity in harvested tumor tissues (SKOV3ip1) was significantly increased in BC-K01 and paclitaxel groups based on the TUNEL assay (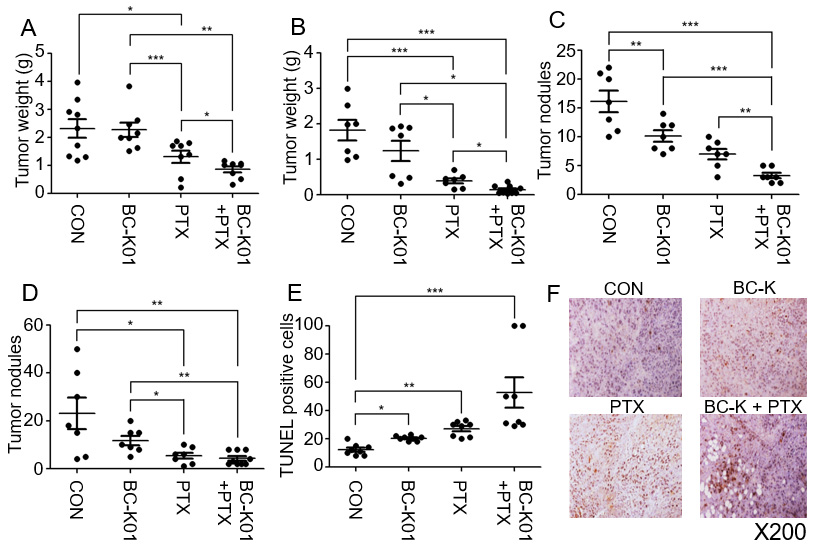
For combined index-based classification of patients with EOC, tissues were stained by immunohistochemistry for KARS1 and 67LR (Fig. 1A). KARS1 and 67LR intensities were categorized as low or high (Table 1). Briefly, for 67LR, the presence or absence of membrane localization was used as the standard to distinguish between low and high categories. In KRS, the pathologist designated the basal histological level (right lower panel of Fig. 1A) as low tissue expression. KARS1 and 67LR expression were significantly correlated with poor progression-free survival. Patients with high KARS1 and 67LR expression by immunohistochemistry (combined index 1) showed lower progression-free survival rates than other patients (combined index 0) (
TCGA data were used to investigate the clinical significance of KARS1 and 67LR RNA expression levels. EOC patients with higher KARS1 expression showed no significant difference in survival compared to those with lower KARS1 gene expression (Fig. 2A,
Both 67LR and KARS1 expression in the membrane increased in a time-dependent manner upon laminin treatment (Fig. 3A). Upon BC-K01 treatment, however, different patterns of expression were seen for these two proteins (Fig. 3B). KARS1 expression decreased more rapidly after BC-K01 than 67LR expression. To understand dynamical delocalization of 67LR in response to BC-K01 treatment, 67LR expression was analyzed in a time-dependent manner by immunofluorescence staining (Fig. 3C) and fluorescence activated cell sorting (Fig. 3D and E). At 2 h after laminin treatment without BC-K01, 78.6% of cells expressed 67LR and 73.0% expressed BC-K01 (Fig. 3D). Under the same conditions, 68.7% of cells expressed membranous KARS1 (Fig. 3E). Treatment with BC-K01 decreased the number of cells that expressed high levels of KARS1 to 36.6% (Fig. 3E). This result indicates that BC-K01 decreased membranous KARS1 expression. Thus, BC-K01 prevents recruitment of KARS1 to the membrane during laminin activation.
Decreased cellular adherence and viability with BC-K01 treatment
We next investigated the effect of BC-K01 on ERK phosphorylation. BC-K01 decreased phosphorylation of ERK in both the presence and absence of laminin (Fig. 4A). To determine the effect of BC-K01 effect on cancer metastasis, cellular adherence was measured. Using impedance measurements (relative cell index), we demonstrated that BC-K01 also reduced cell adhesion to laminin-coated surfaces (Fig. 4B). To compare the adhesion ability of various cell lines, adhesive force was tested in various lung cancer and ovarian cancer cell lines (Fig. 4C). Experiments were also performed with paclitaxel (PTX) and BC-K01 using a WST-based cell viability assay to assess if paclitaxel complemented the effects of BC-K01. To assess whether the combination treatment had a synergistic or additive effect, the viability of cells treated with BC-K01 alone, paclitaxel alone, and the combination of BC-K01 with paclitaxel was compared with that of control cells. BC-K01 was not cytotoxic towards the cell lines we evaluated. However, in combination with paclitaxel, treatment with BC-K01 enhanced cytotoxicity (Fig. 4D). To measure adherence against resistance, a WST-based cell viability was conducted (Fig. 4E). Interestingly, the responsiveness to BC-K01 (in combination with paclitaxel) was significantly correlated with laminin attachment (Fig. 4E and F). The correlation coefficient factor was
Combination effects of BC-K01 and paclitaxel in EOC cell line xenograft models
To evaluate the effect of BC-K01 on in vivo tumor growth, experiments were performed using intraperitoneally established orthotopic HeyA8 or SKOV3ip1 EOC models. We assessed the combination effects of BC-K01 and paclitaxel treatment in these mouse models as well as the effects of treatment with BC-K01 alone and paclitaxel alone. BC-K01 and paclitaxel dual treatment was more effective than BC-K01 or paclitaxel treatment alone in orthotopic tumor tissues (Fig. 5A and B). In the HeyA8 model, combined BC-K01 and paclitaxel treatment significantly decreased tumor weight compared with BC-K01 alone (
Our in vivo and in vitro data demonstrate that combination treatment with BC-K01 and paclitaxel inhibits tumor growth and metastatic nodules while inducing apoptosis of cancer cells.
Discussion
In this study, survival analysis of EOC patients revealed that KARS1 and 67LR are involved in EOC progression. Their co-localization on the plasma membrane can be used to predict patient survival. Combination treatment with BC-K01 and paclitaxel reduced the viability, migration, and invasion of EOC cells (HeyA8 and SKOV3ip1) and increased apoptosis. Furthermore, combined BC-K01 and paclitaxel treatment significantly decreased in vivo tumor weight and the number of nodules in xenograft models of EOC. These phenomena are likely due to inhibition of the interaction between KARS and 67LR.
Proteins such as glutamyl-prolyl-tRNA synthetase have been suggested to have possible utility in the treatment of inflammatory diseases and cancer [20, 21, 22, 23]. Moreover, inhibition of the interaction between HIV type 1 capsid protein and KARS1 with cyclic peptides has been considered for treating HIV infection [24]. KARS1 targeting has potential advantages in cancer treatment. Targeting of the LR-binding activity of KARS1 is different from LR expression knockdown, which adversely affects translation and cell viability [6]. BC-K01 did not inhibit the catalytic activity of KARS1, protein synthesis, or decrease cell viability [12]. Therefore, BC-K01 appears to be a specific metastasis inhibitor without fatal effects on normal cells.
Previous studies revealed that suppressing 67LR with specific antibodies or siRNAs reduced the invasion of metastatic cancers [25, 26]. Nonetheless, targeting 67LR or its precursor 37LRP may also interrupt the normal functions of 67LR. KARS1 is overexpressed in various cancers [27, 28, 29, 30]. We reasoned that inhibition of the metastasis-promoting interaction of KARS1 with 67LR could reduce metastasis without affecting the normal functioning of 67LR. BC-K01 inhibits the pro-metastatic association of KARS1 with 67LR in two ways. First, BC-K01 binds residues that are critical for interaction with 67LR to directly block the interaction between these two proteins. Second, BC-K01 suppresses membrane localization of KARS1 by restraining flexible N-terminal extension. In addition to BC-K01’s ability to reduce cell viability, BC-K01 also increases apoptotic activity. 67LR has been shown to be related to apoptotic activity in a number of cancers, including EOC [9, 31, 32]. Down-regulation of 67LR by BC-K01 inhibited the interaction between KARS1 and 67LR and may have induced apoptosis in EOC cell lines.
One limitation of our study was the relatively small number of patients with EOC that we investigated. Survival analysis based on immunohistochemistry determination of KARS1 and 67LR expression levels was only significant when the combined index was used, possibly due to an insufficient number of samples examined. HeyA8 and SKOV3ip1 cells, which were used in in vitro and in vivo experiments to evaluate the role of BC-K01 in invasion, migration, and apoptosis of EOC cells via western blot, express KARS1 and 67LR at only high levels. Another limitation of this study is the lack of in vivo experiments with patient-derived xenografts, which would have provided more clinically translatable results. Furthermore, we did not explore the mechanism(s) underlying the apoptotic activity of BC-K01; further research is warranted.
In summary, we demonstrated that BC-K01 can be used as an anticancer agent in EOC and provided evidence that KARS1 and 67LR are involved in apoptosis, migration, and invasion of EOC cells. Our data suggest that specific modulation of the KARS1-67LR interaction by BC-K01 is a promising potential treatment option for EOC.
Author contributions
Conception: J-W.C., and J-W.L.
Interpretation or analysis of data: D.H.L., E.S.P., Y-J.C., Y-Y.L., B.L., J-J.C., and C-H.C.
Preparation of the manuscript: D.H.L., E.S.P., Y-J.C., J-W.C., and J-W.L.
Revision for important intellectual content: D.H.L., E.S.P., Y-J.C., S.L., J-W.C., and J-W.L.
Supervision: J-W.C., and J-W.L.
Footnotes
Acknowledgments
His work was supported by the National Research Foundation of Korea (NRF) Grant (NRF-2022R1A2C2009281 and 2020R1C1C1007482), a Korea Medical Device Development Fund grant funded by the Korean government (KMDF_PR_20200901_0153-2022) and a grant (21153MFDS601 in 2022) from Ministry of Food and Drug Safety.
Conflict of interest
The authors have no competing financial interests to declare.
