Abstract
BACKGROUND:
CD10 and CD15 expression has been reported in several tumors. Whether CD10 and CD15 have a role in colorectal mucinous and signet ring adenocarcinoma (MSA) tumorigenesis is not yet known.
OBJECTIVE:
We aimed to investigate the role of CD10 and CD15 expression in mucinous colorectal adenoma-carcinoma sequence (ACS) and determine if there is any clinical and prognostic significance associated with their expression.
METHODS:
Seventy-five cases of colorectal MSA, and 9 cases of adenoma samples were collected. Manual TMA blocks were constructed and immunohistochemistry for CD10 and CD15 was done.
RESULTS:
Compared to adenomas, CD15 expression was significantly higher in MSA (
CONCLUSIONS:
CD15 expression seems to have a role in mucinous colorectal ACS, with significant impact on the survival of MSA patients. Further studies are suggested to identify any genetic alterations that may underlie a potential association with disease progression.
Introduction
Colorectal carcinoma (CRC) is the third most common cancer and the third most deadly malignancy worldwide based on data from GLOBOCAN 2018 [1]. CRC arises from adenomatous polyps in 80% of cases [2]. Histologically, 90% of CRC are adenocarcinoma not otherwise specified (ANOS). Less common are certain subtypes with mucinous activity, namely mucinous and signet ring adenocarcinomas (MSA) [3]. MSA distinctively show more than 50% mucinous component, either extracellularly (mucinous carcinoma), or intracellularly (signet ring adenocarcinoma) [4]. Although MSA is uncommon accounting for only 5% to 15% of CRC cases, it is usually associated with larger primary lesions, deeper tumor invasion, and higher chances of both lymph node and distant sites involvement. Regarding prognosis, patients with MSA are almost similar to those with ANOS in stages I and II. However, in stages III and IV, MSA patients show significantly worse disease outcome [5].
Matrix metalloproteases (MMPs) play an important role in the interaction between normal epithelial cells and stromal cells, thus defining the potential for tumor invasiveness and metastatic spread. CD10 is a zinc-dependent metalloprotease of 100 kDa that is expressed on the surface of many normal cells such as precursor B and T cells, epithelial cells of the breast, kidney, intestine, and salivary glands myoepithelial cells [6]. It is not expressed by the epithelial cells of the normal colorectal mucosa [7]. CD15 is a cell adhesion molecule which is normally expressed on the surface of human leukocytes and responsible for embryogenesis, neural stem and progenitor cells development [8]. CD10 and CD15 are both known to be expressed in many different types of tumors as melanomas, esophageal, gastric, lung, breast, prostate, and colorectal carcinomas [9, 10, 11]. Melanoma and breast carcinoma expressing dysregulated CD10 were shown to have more aggressiveness and worse prognosis [9]. In contrast, CD10 expression in primary bladder tumors was correlated with good prognosis [12]. On the other hand, higher expression of CD15 in primary liver and bile duct tumors was correlated with invasiveness [13]. Moreover, CD15 mediates leukocyte extravasation at inflammatory sites, which can also lead to easier metastasis of tumor cells [14].
In CRC, few studies linked CD10 and CD15 expression to both clinical and prognostic aspects of the disease. The correlation between both markers was suggested to be an indication of poor prognosis and associated with disease progression [15]. Furthermore, they were associated with increased invasion and metastasis especially liver metastases [16]. However, previous studies were concerned with CD10 and CD15 expression in ANOS with exclusion of MSA cases [15, 17]. In this study, we aimed to examine the expression of CD10 and CD15 in colorectal adenomas and MSA using immunohistochemical (IHC) staining, and to investigate any association of CD10 and CD15 with clinicopathological features or prognosis in this group of patients.
Methods
Samples
We reviewed slides and archived paraffin blocks of colorectal adenomas and adenocarcinomas for patients attending Gastroenterology Center, Mansoura University, Egypt during the period from January 2007 till January 2011. Clinicopathological data and follow-up data were retrieved from the patients’ electronic records and by contacting patients through telephone calls. Cases with incomplete clinical data or tissues with few epithelial cells containing mucous were excluded from the study. Seventy-five cases of colorectal MSA, and nine cases of adenomas were included in this study.
The slides were examined for tumor type, grade, depth of invasion (T), tumor margins (infiltrating or pushing), lymphatic, vascular, and perineural invasion. The slides were also examined for associated schistosomiasis, and development of the tumor in an underlying adenoma. The TNM classification and staging were reviewed for each case. The demographic data of patients including gender, age, along with the size, shape, location, and multiplicity of the tumor were also retrieved.
Tissue microarray construction
Modified mechanical pencil tip method was utilized to prepare three manual tissue microarrays (TMA) [18]. From each block, 0.8 mm diameter core of tissue was taken for section preparation, minding the representativeness of the tissue. Both normal and charged slides were coated with 4
Immunohistochemistry (IHC) procedure
Sections of 4
Evaluation of IHC slides
The slides were reviewed for CD10 and CD15 expression independently by two investigators (Foda AA and Alamer HA). The result was described as negative or positive. Most of the positive cases showed focal staining. Any case with no visible tissue was excluded from the study.
Statistical analysis
SPSS, version 24.0 (SPSS Inc, IBM, Chicago, Illinois) was utilized for data analysis. The significance of histological and clinical parameters was evaluated using
Results
The pathology Lab (Gastroenterology Center) at Mansoura University received 75 cases of MSA out of 341 CRC cases (22%) during the period 2007 to 2011. Fifty-six (74.7%) of these cases consisted of mucinous adenocarcinomas, while 19 (25.3%) cases were signet ring adenocarcinomas.
There were 45 (60%) male and 30 (40%) female cases. Most of the cases (70.7%) occurred in patients aged 40 years or older. In 39 cases (52%) the size of the tumor was
Clinicopathological features of 75 MSA cases
Clinicopathological features of 75 MSA cases
Representative images of H&E stained slides (a) A case of colorectal adenoma (b) A case of mucinous adenocarcinoma (x200).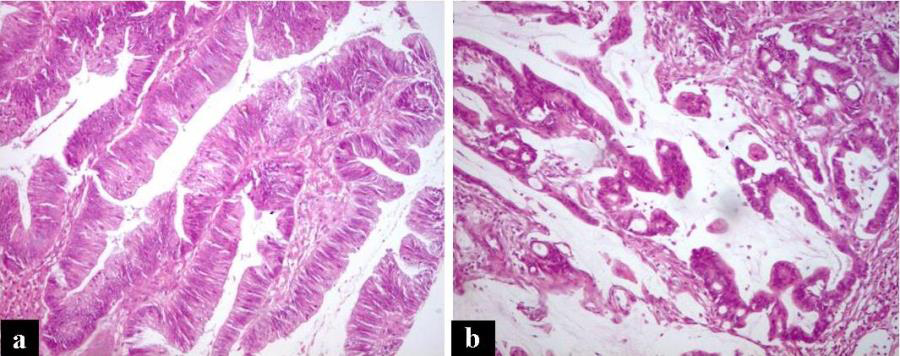
Representative images of IHC staining of studied cases (a) Negative CD10 staining in a case of colorectal adenoma (b) Positive CD10 cytoplasmic staining in a case of mucinous adenocarcinoma (c) Negative CD10 staining in a case of mucinous adenocarcinoma (d) Negative CD15 staining in a case of colorectal adenoma (e) Positive CD15 cytoplasmic staining in a case of signet ring adenocarcinoma (f) Negative CD15 staining in a case of mucinous adenocarcinoma (x200).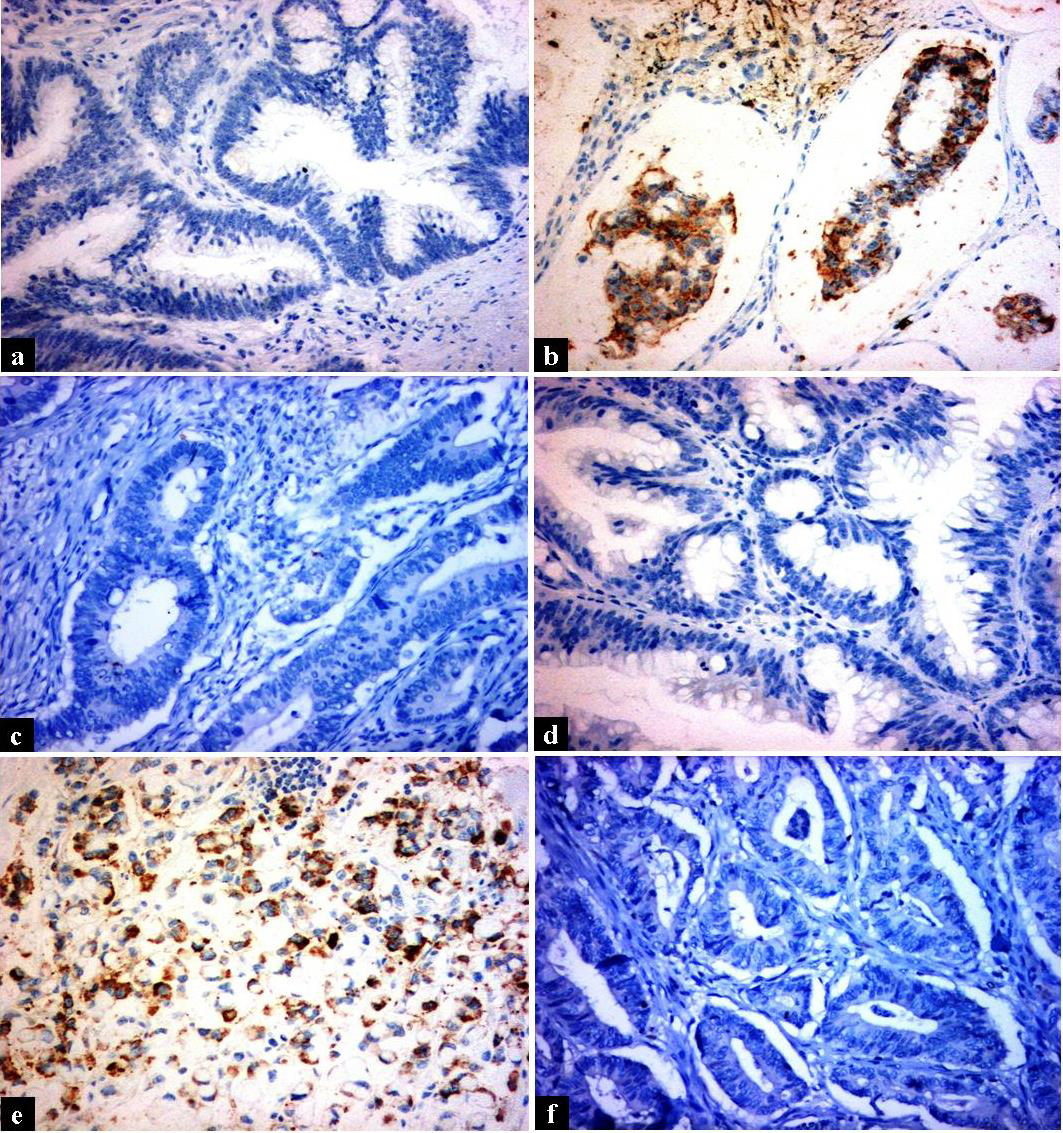
While all adenomas were negative for both CD15 and CD10, more than half of MSA cases (53.7%) showed CD15 positivity and only 14.5% of these cases showed CD10 positivity (Figs 1 and 2). This translated into statistically significant (
Studying the association between different clinicopathological features of MSA cases and either CD10 or CD15 expression revealed that CD15 positivity was significantly associated with microsatellite stable (MSS) tumors (
Relation of CD10 and CD15 expression to clinicopathological features of MSA cases
1. Fischer exact test. 2. Chi-square test.
Correlation between CD10 and CD15 expression in MSA cases
1. Chi-square test.
Regarding disease outcome, after median follow-up duration of 25.6 months (interquartile range
Relation of CD10 and CD15 expression to disease-free and overall survival in MSA cases
1. Log rank test.
Relation of CD10 (a&b) and CD15 (c&d) expression to disease-free (DFS) and overall survival (OS) in MSA patients.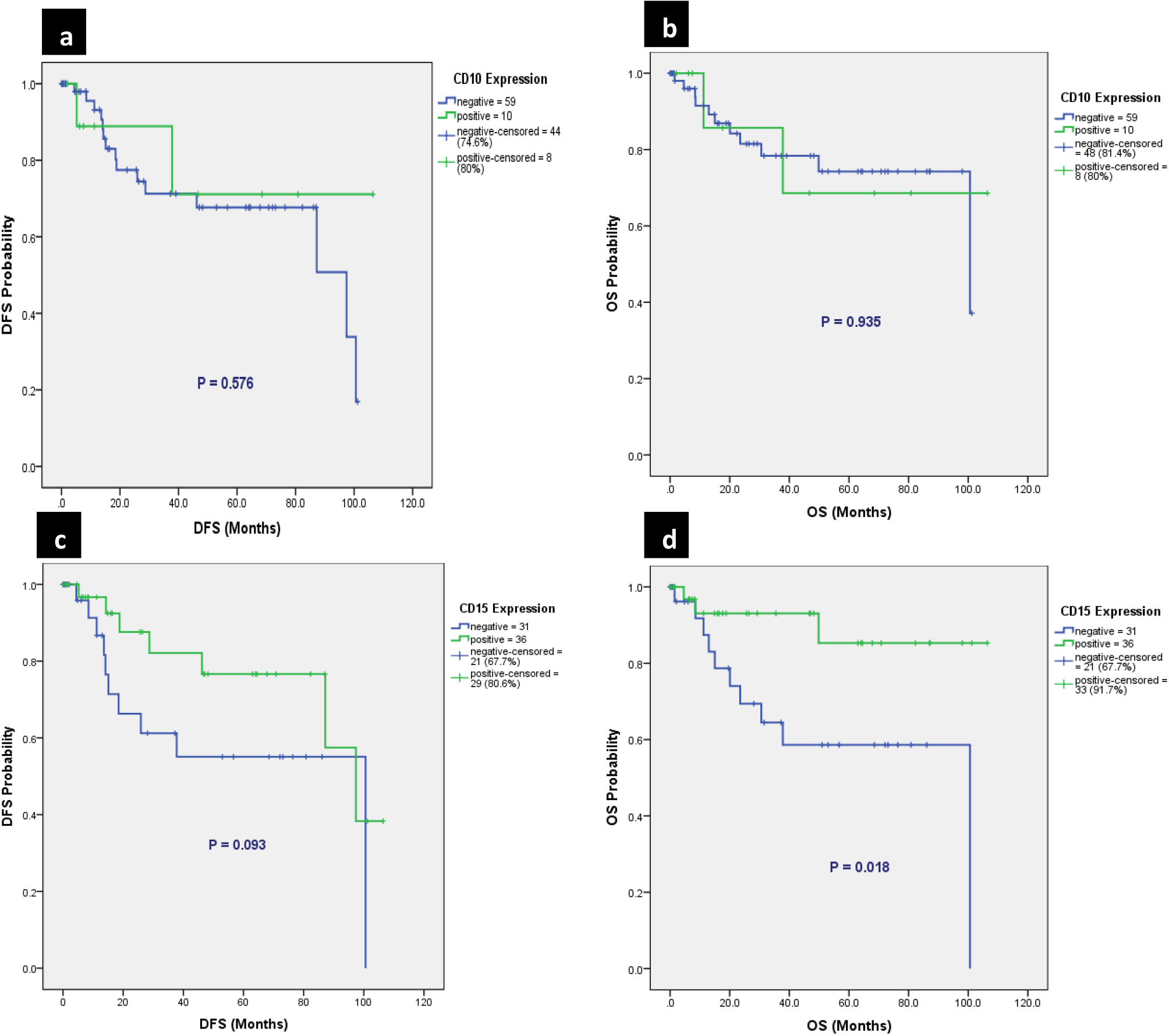
Adenoma-carcinoma sequence (ACS) is a predominant pathogenic pathway of colorectal carcinogenesis. It follows a step-up pattern in which transformation of normal colorectal epithelium to adenoma occurs initially, followed by development of invasive carcinoma. Genetic instability starts in the adenoma phase followed by changes occurring within the tumor histologically [19]. Previous studies have suggested that CD10 expression may be associated with the development or progression of CRC [15, 17]. CD10 enzymatically cleaves polypeptides leading to the accumulation or loss of peptides which alters cellular proliferation and differentiation. It also functions by interfering with intracellular signaling pathways [6].
In our study, although CD10 expression was higher in MSA compared to adenomas (14.5% vs. 0%), the difference was not statistically significant (
Regarding the correlation between CD10 expression and clinicopathological features of MSA cases, we found no significant relation with a variety of tested parameters. Noteworthy that CD10 expression was more in fungating tumors with marginal significance. This goes in line with findings of Oliveira et al. [21] who reported higher CD10 expression in exophytic CRC lesions (
CD15 expression in cancer is involved in adhesion between tumor cells and endothelial cells and is linked to metastatic potential [24, 25]. Very few studies addressed CD15 expression in CRC. Of these, Jang et al. [15] reported an increasing CD15 expression during CRC development; 23% in low grade adenoma compared to 50% in intramucosal CRC and 44% in invasive CRC. Whether CD15 expression is related to progression of mucinous CRC or not is not yet fully investigated. Expression of CD15 was also investigated in various epithelial cancers other than CRC with contradicting evidence about its relation to disease behavior. Its expression appears to signify aggressive behavior in some tumors and benign behavior in others. For instance, it highly predicts poor survival when expressed in patients with hepatocellular carcinoma [26]. Similarly, in breast carcinoma, it showed association with invasiveness and metastatic potential [11]. In contrast, clear cell renal carcinoma was associated with poor prognosis in absence of CD15 expression [27].
To the best of our knowledge, this is the first study to examine CD15 expression in colorectal MSA. Unlike CD10, the expression of CD15 in MSA was significantly more frequent compared to adenomas (53.7% vs. 0%,
Several studies have identified CD10 as a prognostic marker for worse survival in different tumor types such as breast cancer, prostate cancer, and melanoma [9]. Data on prognostic value of either CD10 or CD15 expression in CRC are scarce. One study by Khanh and colleagues [29] found that CD10 expression in infiltrating immune cells, but not tumor cells, was a useful predicting marker for high recurrence risk and mortality in stage II CRC. Concerning the impact of CD10 and CD15 expression on our patients’ prognosis, only CD15 positivity was associated with better OS (
We recognize an important aspect in studying metastatic tumors which is assessment for molecular markers such as RAS expression. We are still working on RAS expression in metastatic/recurrent tumors, although a small number of these tumors were included in this study. Once we are ready with these data, they can be published in the future.
Conclusion
We conclude that CD15 expression seems to have a role in mucinous colorectal ACS. Although CD10 expression was linked to CRC progression in other studies, its role in mucinous and signet ring carcinomas is unclear. Unlike CD10, CD15 positivity has a significant impact on the survival of colorectal MSA patients. Further research with more patient numbers of this rare entity is suggested to confirm the role of CD10 and CD15 in mucinous colorectal ACS and to identify any genetic alterations that may underlie a potential association with disease progression.
Footnotes
Acknowledgments
None.
Author contributions
Conception: Abd AlRahman Foda
Interpretation or analysis of data: Abd AlRahman Foda, Hadi Abdulhadi Helali, Fayza Sami Fayad, Sara Waleed Hussian
Preparation of the manuscript: Abd AlRahman Foda, Ziad Emarah, Ahmed M. Ramez
Revision for important intellectual content: Khaled Abdelwahab, Tamer Akl
Supervision: Haitham Abdulkarem Alamer, Nadeem Ikram
