Abstract
Background:
In this study, we investigated the impact of chemotherapy on breast cancer-specific survival (BCSS) in patients with mucinous carcinoma using the surveillance, epidemiology, and end results (SEER) database.
Methods:
A large-scale SEER-based retrospective analysis was conducted; 13,329 patients with mucinous carcinoma from 1994 to 2014 were identified. Clinicopathological characteristics were compared using the chi-square test. BCSS curves were generated using the Kaplan–Meier method. The prognostic significance of all demographic and clinicopathological characteristics and treatment patterns were calculated using univariate and multivariate regression analyses.
Results:
Mucinous carcinoma was demonstrated to be less aggressive than invasive ductal carcinoma and predicted a better prognosis in the Kaplan–Meier analysis (hazard ratios = 0.336, 95% confidence interval: 0.308–0.368, p < 0.001). Univariate and multivariate analyses revealed that chemotherapy did not provide any additional benefit for patients with mucinous carcinoma. Predictors for receiving chemotherapy were younger age, estrogen receptor-negative, progesterone receptor-negative, and human epidermal growth factor receptor 2-positive status, higher grade, larger tumor size, lymph node involvement, radiation reception, and mastectomy. Further subgroup analysis verified that regardless of the hormone receptor (HR) and lymph node (LN) status, patients did not benefit from chemotherapy.
Conclusion:
Our study showed that patients with HR+/LN– mucinous carcinoma did not benefit from chemotherapy and that chemotherapy could not improve the survival of all subtypes of mucinous carcinoma based on large-scale SEER data. These results support that patients with mucinous carcinoma could be exempt from chemotherapy. Additional research is needed to further evaluate the impact of adjuvant treatments, particularly in patients with favorable histology.
Introduction
Breast cancer is recognized as the leading cause of death in women worldwide. Breast cancer is a heterogeneous disease and its heterogeneity can be observed at multiple levels, from classical histopathological characterizations to modern molecular classifications. 1 In order to better classify different breast cancer subtypes and develop more specific and individualized therapies, histological type has long been recognized as an important indicator of prognosis. According to the latest version of the World Health Organization classification, 2 breast cancer can be categorized into 21 distinct histological types based on cell morphology, growth, and architecture patterns. The most common histological type is invasive ductal carcinoma (IDC), and other special types account for up to one-quarter of all breast cancers, including mucinous carcinoma.
Mucinous carcinoma is a special histological type of breast cancer, and its incidence rate fluctuates between 1% and 6% of all primary breast cancers.3–9 Consequently, there are scant population-based descriptive data, and the low incidence rate has impeded the implementation of large-scale prospective studies. Treatment guidelines for systematic and regional control of mucinous carcinoma were mostly extrapolated from data based on IDC. However, IDC has different features from mucinous carcinoma, 10 which has a significant level of mucin production, with mucin generally accounting for at least 50% of the tumor composition. Mucinous carcinoma usually occurs in elderly postmenopausal women and is characterized by favorable characteristics, including a higher expression of hormone receptor (HR), lower expression of human epidermal growth factor receptor 2 (HER2),11–15 lower incidence of lymph node metastasis, and better differentiation.5,16 Patients with mucinous carcinoma have better prognoses than IDC patients in the majority of studies, and the 10-year overall survival rate has been reported to be over 80%.17–19 However, other studies reported that the long-term survival curves of mucinous carcinoma were almost identical to those of IDC, or there was no survival difference between mucinous carcinoma and IDC even after adjusting for clinicopathological factors.5,15,20,21
Breast cancer can be classified by HR status, specifically estrogen receptor (ER) and progestogen receptor (PR) status, and HER2 status. In clinical practice, endocrine treatment is a cornerstone for HR+ patients, and chemotherapy is the only choice for triple-negative breast cancer patients. However, we should be aware that these parameters and molecular subtypes are mostly based on gene expression profiling and high-throughput sequencing of IDC of no special type and did not include uncommon subtypes. Weigelt et al. 9 reported that each type of breast cancer fell in one molecular subtype only, suggesting a high homogeneity at the transcriptome level of each special type. This finding could be further applied at the genomic level, and similar patterns of copy number variations could be detected in samples of the same special type.8,9 Moreover, some special types, such as adenoid cystic carcinoma, 22 are classified as triple-negative and basal-like breast cancer, but they have good prognoses in practice. These results imply that unnecessary treatments may erroneously be recommended to patients with good prognoses when the histology is not taken into account. 1 San Antonio Breast Cancer Symposium suggested that endocrine therapy should be considered first in patients with HR-positive disease, such as mucinous carcinoma, because its more favorable molecular factors may make it less sensitive to chemotherapy. 21 However, there are limited available data on the outcomes of patients with mucinous carcinoma receiving chemotherapy.
The objective of this study was to compare the demographic and clinicopathological characteristics and treatment patterns of mucinous carcinoma and IDC, evaluate the significance of available prognostic factors for patients with mucinous carcinoma, and to appraise the outcomes of patients receiving chemotherapy in a retrospective analysis of the SEER database.
Materials and methods
Ethics statement
Our study was approved by the independent ethics committee/institutional Review Board of Qilu Hospital of Shandong University (IRB #KYLL-2016-350). The data released by the SEER database do not require informed consent from patients since breast cancer is a reportable disease in the United States.
Patient selection and data processing
We used SEER*Stat version 8.3.2 to generate a case-listing file. We included 463,112 patients who met the following criteria: female sex, year of diagnosis from 1994 to 2014, age at diagnosis >20 years old, breast cancer as the first and only malignant cancer diagnosis, diagnosis not from autopsy, patients without distant metastasis at diagnosis, pathologically confirmed as IDC (ICD-O-3 8500/3) or mucinous carcinoma (ICD-O-3 8480/3). Among these patients, 13,329 were diagnosed with mucinous carcinoma and 449,783 were diagnosed with IDC.
Demographic characteristics included age at diagnosis, race, marital status, and insurance status. Age at diagnosis was treated as a categorical variable and classified into three groups: 20–49 years, 50–69 years, and 70+ years. Tumor characteristics included histological grade; tumor size; lymph node (LN) status; ER, PR, and HER2 status; and breast subtype.
Statistical analysis
Clinicopathological characteristics were compared between IDC and mucinous carcinoma using Pearson’s chi-square test. Breast cancer-specific survival (BCSS) was calculated from the date of diagnosis to the date of death from breast cancer. Survival curves were generated using the Kaplan–Meier method, and the log-rank test was used to evaluate differences in survival between groups. Univariate and multivariate Cox proportional hazard models were applied to estimate the association of covariates with BCSS and calculate the hazard ratios and 95% confidence intervals (CIs).
In order to account for the differences in baseline characteristics between groups, we performed 1:1 propensity score matching (PSM) between mucinous carcinoma and IDC patients using the following predetermined factors: age at diagnosis; race; marital status; insurance status; grade; tumor size; lymph node status; ER, PR and HER2 status; breast subtype; surgery type; radiation; and chemotherapy treatment.
When the two-sided p value was less than 0.05, the difference was considered statistically significant. Analyses were performed using SPSS version 18.0 (IBM SPSS Statistics, Chicago, IL, USA).
Results
Demographics and clinical characteristics of the study population
There were 13,329 patients with mucinous carcinoma and 449,783 with IDC meeting the inclusion criteria of our study.
The demographic, clinicopathological, and treatment characteristics are summarized and compared between the two cohorts in Table 1.
Clinicopathological and demographic characteristics of breast cancer patients diagnosed with IDC or mucinous carcinoma.
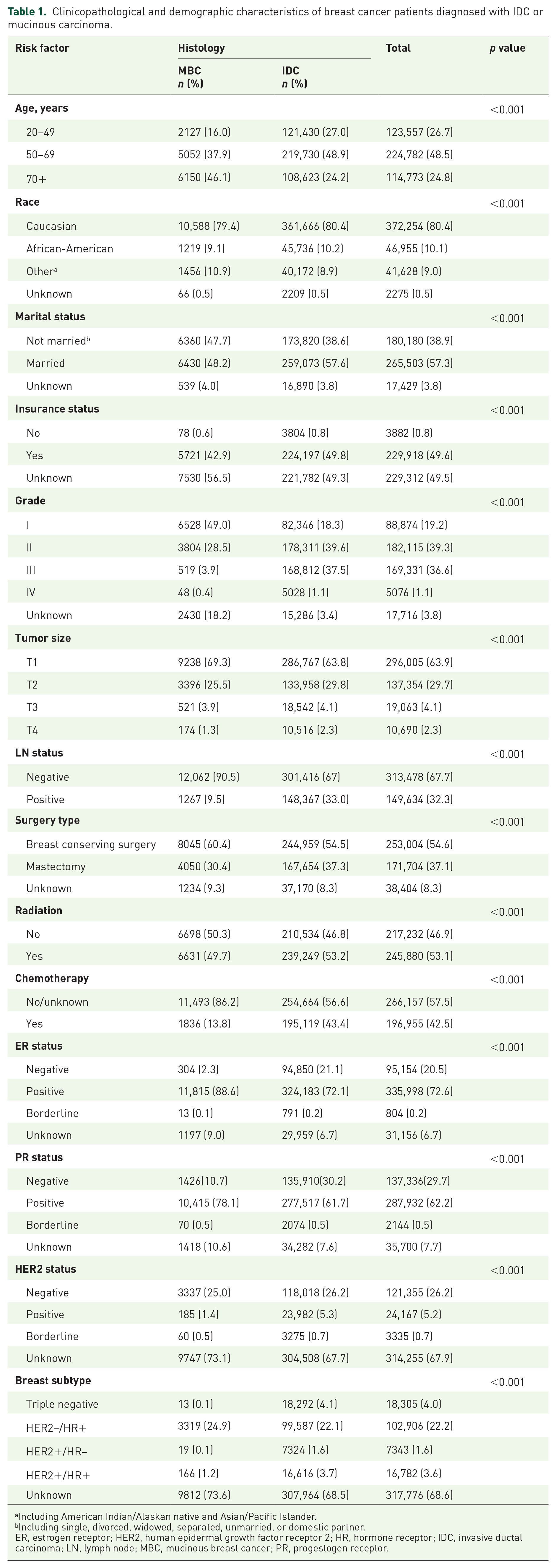
Including American Indian/Alaskan native and Asian/Pacific Islander.
Including single, divorced, widowed, separated, unmarried, or domestic partner.
ER, estrogen receptor; HER2, human epidermal growth factor receptor 2; HR, hormone receptor; IDC, invasive ductal carcinoma; LN, lymph node; MBC, mucinous breast cancer; PR, progestogen receptor.
Kaplan–Meier plots (Figure 1A) showed that patients with mucinous carcinoma had significantly better BCSS than IDC patients. To rule out the influence of demographic and clinical characteristic discrepancies between IDC and mucinous carcinoma, a 1:1 (mucinous carcinoma:IDC) matched case–control analysis was carried out (N = 24,578) and the results confirmed mucinous carcinoma to be a favorable histology (Figure 1B). Further univariate and multivariate analyses (Supplemental material Table S1 online) also indicated that patients with mucinous carcinoma had better prognoses than patients with IDC.

Comparison of breast cancer-specific survival (BCSS) between mucinous carcinoma and invasive ductal carcinoma (IDC) using the log-rank test based on all patients from the SEER database (A) and after 1:1 propensity score matching (B).
Regarding prognostic factors for patients with mucinous carcinoma, univariate and multivariate analysis results are shown in Table 2. Old age, African-American race, grade III and T2–T4, positive LN status, no radiation, receiving chemotherapy, and negative ER and PR status were significantly associated with worse prognosis. It is worth noting that receiving chemotherapy was predictive of poor outcomes (univariate, hazard ratio = 1.88, 95% CI: 1.532–2.307, p < 0.001; multivariate, hazard ratio = 1.348, 95% CI: 1.03–1.763, p = 0.029), which led us to consider the necessity of chemotherapy in patients with mucinous carcinoma.
Univariate and multivariate analysis of breast cancer-specific survival predictors in patients with mucinous carcinoma using Cox proportional hazard model.
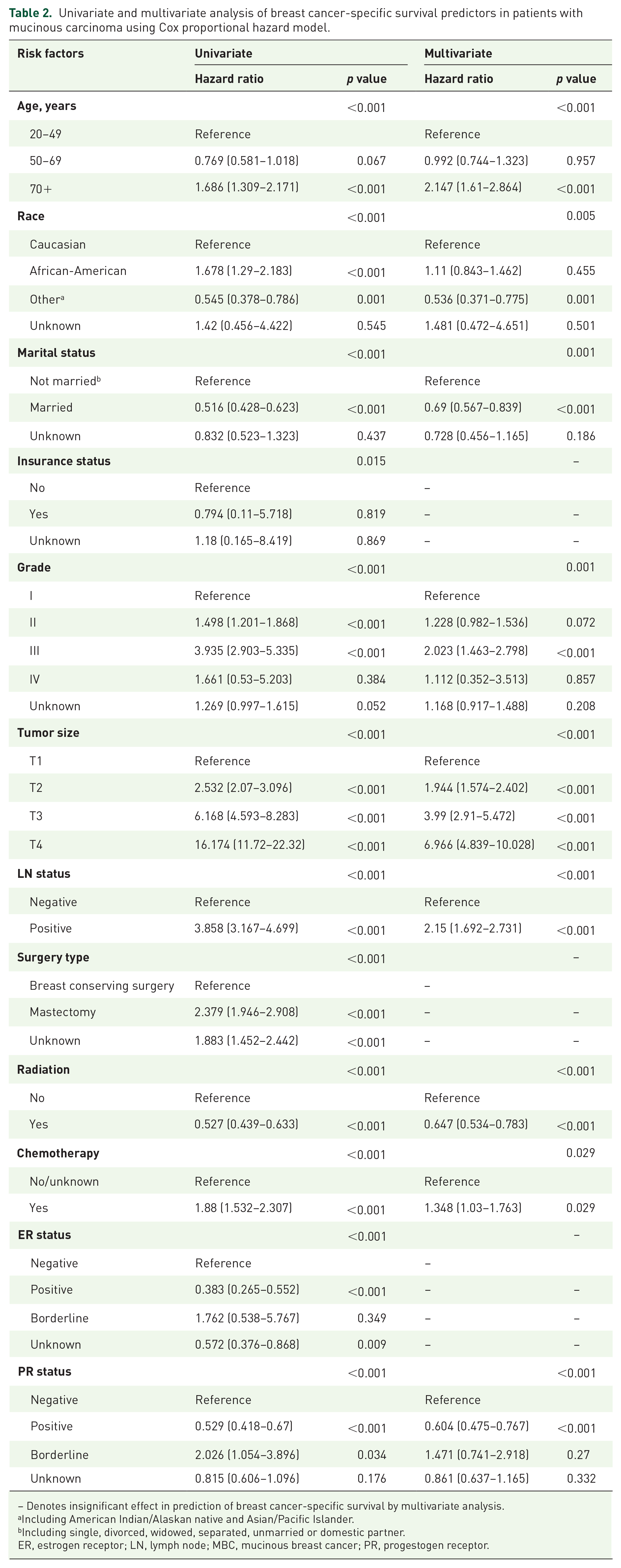
– Denotes insignificant effect in prediction of breast cancer-specific survival by multivariate analysis.
Including American Indian/Alaskan native and Asian/Pacific Islander.
Including single, divorced, widowed, separated, unmarried or domestic partner.
ER, estrogen receptor; LN, lymph node; MBC, mucinous breast cancer; PR, progestogen receptor.
Predictive factors for receiving chemotherapy among patients with mucinous carcinoma
Mucinous carcinoma has long been recognized as a histological subtype with a favorable prognosis. The effect of chemotherapy on patients with mucinous carcinoma and the optimal population of patients with mucinous carcinoma to receive chemotherapy remains under discussion. The differences in clinicopathological variables between the chemotherapy and no/unknown chemotherapy groups are shown in Table 3. Patients receiving chemotherapy tended to be younger, with larger, higher grade, HR−/HER2+ tumors with lymph node involvement. Regarding treatment patterns, patients receiving chemotherapy were more likely to undergo mastectomy and radiotherapy. Multivariate analysis revealed that younger age, African-American race, higher grade, larger tumor size, lymph node involvement, negative PR status, triple negative subtype, and receiving mastectomy and radiation were independently associated with chemotherapy administration (Supplemental Table S2).
The association between different chemotherapy administration status and patients’ demographics and clinicopathological characteristics.
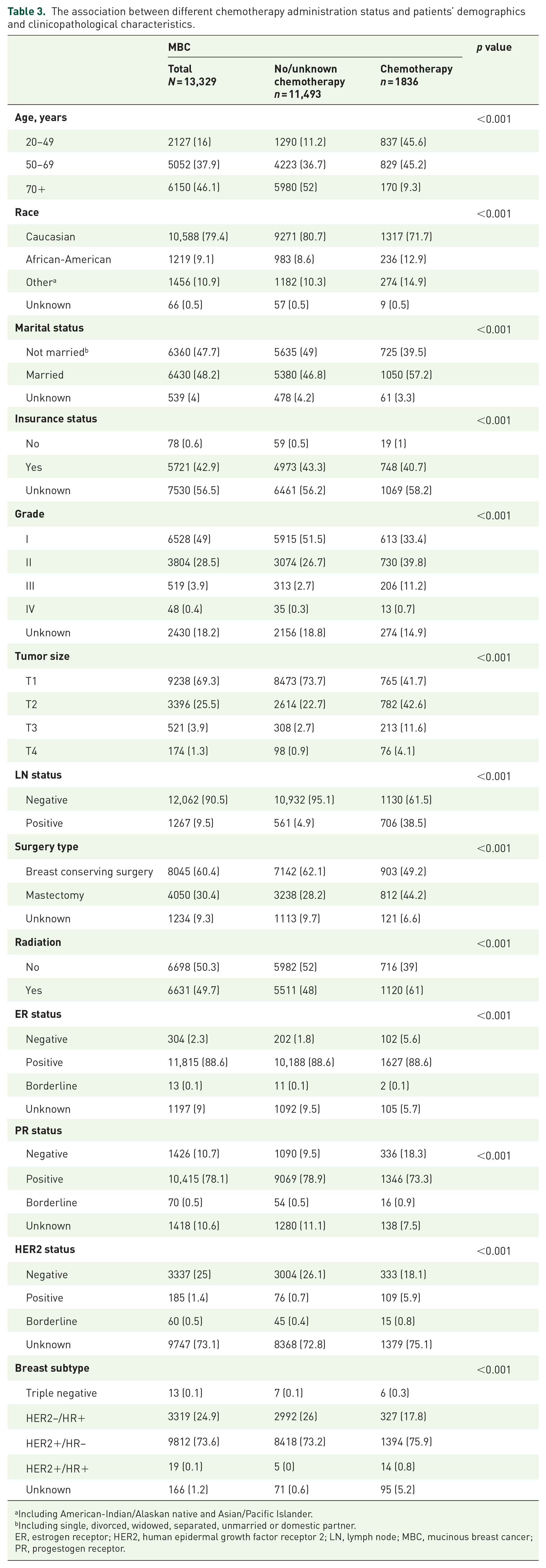
Including American-Indian/Alaskan native and Asian/Pacific Islander.
Including single, divorced, widowed, separated, unmarried or domestic partner.
ER, estrogen receptor; HER2, human epidermal growth factor receptor 2; LN, lymph node; MBC, mucinous breast cancer; PR, progestogen receptor.
Influence of chemotherapy on the prognoses of specific mucinous carcinoma subgroups
A detailed comparison of clinicopathological and demographic characteristics according to different chemotherapy administration status was conducted in the HR+/LN+, HR+/LN, HR–/LN+, and HR−/LN– subgroups of patients (Supplemental Table S3). According to the National Comprehensive Cancer Network (NCCN) guidelines (version 1.2018) for patients with mucinous carcinoma, those diagnosed with HR–/LN+ and HR−/LN− were supposed to receive chemotherapy, and HR+/LN+ to consider chemotherapy.
The median follow-up time for patients with mucinous carcinoma of chemotherapy and no/unknown chemotherapy groups were 94 and 78 months, respectively. The 5-year BCSS rate for patients with mucinous carcinoma who underwent adjuvant chemotherapy was 96.5%, compared with 97.9% for those who did not undergo adjuvant chemotherapy (p < 0.001) (Figure 2). Stratifying the population according to the above NCCN criteria, we found that the 5-year BCSS rates for HR+/LN+, HR−/LN+, and HR−/LN− patients with mucinous carcinoma who underwent adjuvant chemotherapy were 99.2%, 71.4%, and 88.6%, respectively, compared with 99.3%, 82.5%, and 93.9% for those who did not undergo adjuvant chemotherapy. Chemotherapy did not improve the prognosis of BCSS either in HR+/LN+ (p = 0.568; Figure 3A), HR−/LN+ (p = 0.141; Figure 3B), or HR−/LN− subgroup (p = 0.701; Figure 3C). However, for the HR−/LN+ and HR−/LN− subgroups, the results were probably not reliable due to the small sample size and need further studies to demonstrate. Subsequent analysis of the subgroup of HR+/LN− patients, who did not need chemotherapy according to the NCCN guidelines, showed that the 5-year survival rate was 99.3% in the chemotherapy group, compared with 98.4% in the non-chemotherapy group (p = 0.398). Kaplan–Meier analysis revealed that chemotherapy could not improve BCSS, but had the tendency to endanger survival (Figure 3D). Considering that some results we found were contradictory to the NCCN guidelines, we carried out a 1:1 matched case–control analysis. The 5-year survival rates were 95.8%, 88.9%, and 99.4% for patients undergoing chemotherapy, compared with 93.9%, 100%, and 98.8% for those who did not undergo chemotherapy. Chemotherapy did not improve the BCSS after PSM in the HR+/LN+, HR−/LN−, or HR+/LN− subgroups (Figure 4). However, for the HR−/LN+ subgroups, PSM was not practicable due to its small sample size. All the aforementioned results are summarized in Figure 5.

The survival outcomes of patients with mucinous carcinoma who underwent adjuvant chemotherapy and those who did not undergo adjuvant chemotherapy.

Impact of chemotherapy on patients with mucinous carcinoma with HR+/LN+ (A), HR−/LN+ (B), HR−/LN− (C), or HR+/LN− (D) status.

Impact of chemotherapy on patients with mucinous carcinoma with HR+/LN+ (A), HR−/LN− (B), or HR+/LN− (C) status after 1:1 matching.
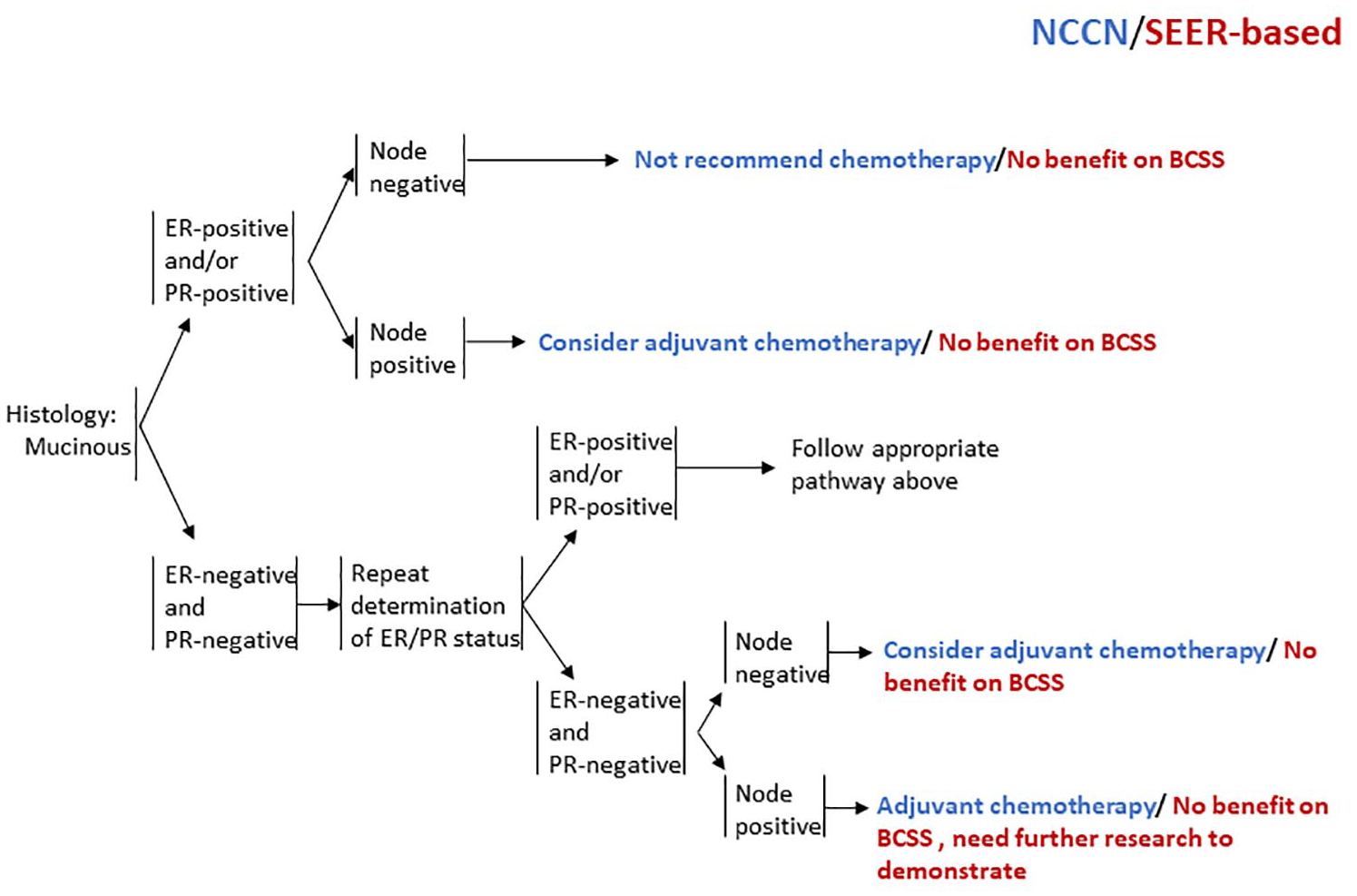
Summary of the effect of chemotherapy on patients with mucinous carcinoma classified by hormone receptor and lymph node status and presenting the difference compared with the NCCN guidelines for favorable histologies.
Discussion
Mucinous carcinoma is a unique type of neoplasm of the breast which is morphologically distinct from IDC. Previous studies have reported that mucinous carcinoma accounts for only 1–6% of all primary breast cancers; therefore, it is difficult to identify a sufficient number of patients, considering the rarity of mucinous carcinoma in the general population. In this study, we retrospectively investigated the demographic, clinicopathological, and prognostic features of mucinous carcinoma based on the SEER database. Consistent with previous studies,23–25 our findings indicated that mucinous carcinoma had more favorable clinicopathological characteristics than IDC, such as well-differentiated histological grade, smaller tumor size, less LN involvement, higher proportion of HR positivity, less HER2 overexpression, and the HR+/HER2– subtype was more common. Both uni- and multivariate analysis confirmed that patients with mucinous carcinoma had better BCSS than those with IDC. Cao et al. 21 performed a relatively large retrospective analysis in Chinese women and confirmed that pure mucinous carcinoma showed less aggressive behavior, was rarely associated with LN involvement, and had better prognosis than IDC. Fu et al. 26 also revealed that early stage patients with mucinous carcinoma had a better prognosis than patients with IDC, even after adjusting for demographic and clinicopathological characteristics. We conducted a 1:1 mucinous carcinoma:IDC PSM analysis to ensure that the outcome discrepancies did not result from an incongruous demographic or clinicopathological baseline, and the results remained the same. This further validated that the mucinous carcinoma subtype has a favorable prognosis.
As discussed above and as previous studies indicate, the majority of mucinous carcinoma tumors had higher expression of ER and PR, and lower expression of HER2, which are characteristics of luminal A and luminal B molecular subtypes. The currently recommended treatment for patients with luminal A breast cancer is endocrine therapy, 27 and whether to add cytotoxic chemotherapy to endocrine therapy remains under debate. In the decision-making regarding the addition of chemotherapy, clinical factors, including tumor size, grade, age, Ki67, and nodal status, are most influential, apart from genomic gene expression. In the case of mucinous carcinoma, our findings indicate that patients receiving chemotherapy tended to be younger; African-American; with larger, higher grade, and triple negative tumors; nodal involvement; and treated with mastectomy and radiation. Tumor size 28 has been questioned by several studies because the size measurement generally included the extracellular mucinous component, which may overestimate the actual size of the tumor. However, Elledge et al. demonstrated that tumor size remained an independent prognostic factor of disease-free survival (DFS) after multivariate analysis. 5 LN involvement5,6,29 was recognized as an important prognostic factor when making clinical decisions21,30,31 and as an indicator for chemotherapy. For luminal A and luminal B breast cancer, a significant reduction in the use of chemotherapy was observed in nodal negative patients from 2001 to 2008. This reduction might be associated with the consideration that the concomitant risk of life-threatening toxicities and physical, mental, and financial stress of chemotherapy outweighed the potential gain in overall survival or DFS. Several studies, including BRENDA (breast cancer care under evidence-based guidelines), 27 IBCSG (International Breast Cancer Study Group) IX Trial, 32 NSABP B20, 33 and several meta-analyses, 34 revealed strong evidence that there were no additional benefits from chemotherapy over endocrine therapy alone. However, for node-positive patients, the recommendations diverged. The BRENDA study found no clinical benefits for chemotherapy in addition to endocrine therapy. In contrast, Albain et al. 35 showed in clinical trial SWOG 8814 for postmenopausal, node-positive luminal A breast cancer that adjuvant chemotherapy plus tamoxifen improved DFS over that with tamoxifen treatment alone. Specifically, for patients with mucinous carcinoma, the NCCN guidelines recommend that patients with HR−/LN+ receive chemotherapy and with HR+/LN+ consider chemotherapy, while our results revealed that regardless of whether HR+/LN+ or HR−/LN+, patients did not benefit from chemotherapy. It is also worth noting that receiving chemotherapy would worsen the prognosis of patients with mucinous carcinoma in multivariate analysis, although previous results only proved that the prognostic value of adjuvant chemotherapy no longer existed after multivariate Cox analysis. 5 Some studies claimed that adjuvant systematic therapy could not provide the expected benefit for patients with mucinous carcinoma because the favorable outcomes of mucinous carcinoma were mainly related to its intrinsic biological characteristics. 5 Moreover, the St. Gallen guidelines did not recommend chemotherapy administration in ER+ patients, especially when the tumor is highly responsive to endocrine therapy and has a low probability of recurrence. 36 Tseng et al. reported that adjuvant endocrine therapy was adequate for patients with a positive HR status.36,37 Our findings also showed that chemotherapy is not beneficial for HR+ subgroups with respect to BCSS. For HR− patients, Fu et al. 26 indicated that it is not feasible to guide clinical practice according to ER and PR status because the sample size of HR− patients was too small.
The 21-gene and/or 70-gene scores were demonstrated to be effective in clearly identifying premenopausal patients with luminal tumors who might be exempt from chemotherapy. Since the gene expression assays’ results were not available in the SEER database, we currently cannot evaluate the effect of gene expression analyses in the decision to administer chemotherapy for patients with mucinous carcinoma in this study. However, before the 21-gene and/or 70-gene signatures, many genomic and transcriptomic profiling approaches have been applied to identify signatures associated with prognosis, and they have demonstrated that some special histological types of breast cancer, including mucinous carcinomas, are more than mere architectural patterns, but are also distinct entities at the molecular level.9,38–43 Furthermore, the morphological difference between endocrine and mucinous B carcinomas cannot be explained by the patterns of the transcriptomic profiles.9,40 On the other hand, several studies have raised the concern that genomic assessment might overestimate the risk of recurrence. In the NSABP B20 trial, 33 of patients with node-negative and luminal A breast cancer who were retrospectively analyzed using Oncotype Dx and defined as being at a “genomic” high risk of relapse but only received tamoxifen therapy alone, 60.5% remained 40 recurrence-free at 10 years. The SWOG S8814 trial 35 reported analogous results in node-positive breast cancer patients. Taken together, gene expression analysis and ensuing enhancement in hazard assessment provided advanced tools for identifying active micro-metastatic disease. However, more developed tools and large, direct randomized comparisons of chemoendocrine therapy versus endocrine therapy alone were desperately needed to help identify the optimal population to receive chemotherapy and make more self-tailored treatment plans.
Our study had several limitations. First, due to the uncertainties regarding data completeness of the SEER program, the sensitivity of SEER data to identify chemotherapy receipt specifically for breast cancer was moderate (68.6%) in comparison with SEER-Medicare data. 44 Therefore, further analysis augmenting SEER data with other data sources (e.g. SEER-Medicare, National Cancer Database) may add credibility to the results and is needed in the future. Second, the rarity of mucinous carcinoma renders this study a retrospective study, which may cause the potential ensuing bias. Third, we failed to differentiate the subtypes of mucinous carcinoma, such as pure and mixed mucinous carcinoma. However, pure and mixed mucinous carcinoma displayed similar patterns of genetic aberrations in unsupervised clustering analysis, 45 which may not exert a significant effect on our results. Last, SEER database lacks information on Ki-67 status, targeted therapy and hormonotherapy, which was therefore not included in our analysis. The incorporation of these variables would help determine their effects in multivariate regression analysis, and derive more precise effect of chemotherapy on outcomes, which are needed in the future. Despite these limitations, our study further advanced the knowledge of demographics, prognosis, and therapeutic patterns of patients with mucinous carcinoma. To the best of our knowledge, this study is the first to investigate the effect of chemotherapy on survival outcomes in specific subgroups of patients with mucinous carcinoma.
As far as we know, this study was the first to investigate the effect of chemotherapy on patients with mucinous carcinoma according to the classification of NCCN guidelines based on a large-scale retrospective analysis of the SEER database. Our results support the NCCN recommendations that HR+/LN− patients do not benefit from chemotherapy. We also found that HR+/LN+, HR−/LN+, and HR−/LN− patients did not benefit from chemotherapy, which needs to be examined in a larger sample size to confirm in the future.
Supplemental Material
sj-pdf-1-tam-10.1177_1758835920975603 – Supplemental material for Evaluation of efficacy of chemotherapy for mucinous carcinoma: a surveillance, epidemiology, and end results cohort study
Supplemental material, sj-pdf-1-tam-10.1177_1758835920975603 for Evaluation of efficacy of chemotherapy for mucinous carcinoma: a surveillance, epidemiology, and end results cohort study by Hanwen Zhang, Ning Zhang, Yaming Li, Yiran Liang and Qifeng Yang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-2-tam-10.1177_1758835920975603 – Supplemental material for Evaluation of efficacy of chemotherapy for mucinous carcinoma: a surveillance, epidemiology, and end results cohort study
Supplemental material, sj-pdf-2-tam-10.1177_1758835920975603 for Evaluation of efficacy of chemotherapy for mucinous carcinoma: a surveillance, epidemiology, and end results cohort study by Hanwen Zhang, Ning Zhang, Yaming Li, Yiran Liang and Qifeng Yang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-3-tam-10.1177_1758835920975603 – Supplemental material for Evaluation of efficacy of chemotherapy for mucinous carcinoma: a surveillance, epidemiology, and end results cohort study
Supplemental material, sj-pdf-3-tam-10.1177_1758835920975603 for Evaluation of efficacy of chemotherapy for mucinous carcinoma: a surveillance, epidemiology, and end results cohort study by Hanwen Zhang, Ning Zhang, Yaming Li, Yiran Liang and Qifeng Yang in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The manuscript has been proofread by SAGE language editing services.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No. 81672613; No. 81874119), Special Foundation for Taishan Scholars (No. ts20190971), Special Support Plan for National High Level Talents (Ten Thousand Talents Program W01020103), National Key Research and Development Program (No. 2018YFC0114705), and Qilu Hospital Clinical New Technology Developing Foundation (No. 2018-7; No. 2019-9).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
