Abstract
Circular RNA (circRNA) has been shown to participate in various tumors, including lung cancer. In the present study, we explored the expression and functional relevance of hsa_circ_0003288 in human non-small cell lung cancer (NSCLC). We verified that hsa_circ_0003288 expression was upregulated in lung cancer tissues and cell lines. Overexpression of hsa_circ_0003288 dramatically promoted lung cancer cell proliferation, colony formation, inhibited apoptosis, and increased cell migration and invasion in vitro. Xenograft experiments showed that hsa_circ_0003288 overexpression accelerated tumor growth in vivo. Mechanistically, hsa_circ_0003288 negatively regulated miR-145 to exert the oncogenic role in lung cancer. Overexpression of miR-145 decreased cell proliferation, induced apoptosis, and suppressed migration and invasion in lung cancer. Additionally, miR-145 co-transfection abolished the oncogenic role of hsa_circ_0003288. Collectively, these findings identified a novel regulatory role of hsa_circ_0003288/miR-145 axis in the progression of NSCLC.
Introduction
Non-small-cell lung cancer (NSCLC) accounts for approximately 80–85% of all lung cancers, the leading cause of cancer-related death worldwide. In 2015, the estimated number of new lung cancer cases in China and America is 733,300 and 221,200, respectively [1]. Although significant advances achieved in lung cancer diagnosis and treatment, the five-year overall survival rate of NSCLC remains unsatisfactory [2]. Therefore, it is urgent to elucidate the pathogenesis underlying tumorigenesis and to develop novel therapies against NSCLC.
Circular (circ) RNAs are a large class of endogenous non-coding RNAs that covalently link 5’ and 3’ ends of RNA to generate a circular structure [3]. A large number of studies have demonstrated that circRNAs play diverse roles in biological processes, including proliferation, differentiation, apoptosis, inflammation, and oxidative stress [4, 5, 6]. Recent achievements in high-throughput sequencing technology have allowed the identification of many circRNAs with unknown functions in human pathophysiological processes [7]. For instance, Li et al. have identified that hsa_circ_0000792 could serve as a prognostic biomarker for human lung adenocarcinoma [8]. Chen et al. demonstrated that hsa_circ_100395 inhibits lung cancer development via sponging miR-1228/TCF21 pathway [9]. Of note, hsa_circ_0003288, located on chromosome 2 (32703702–32718734) and generated by back-splicing of the BIRC6 transcript, has been reported to function as a positive regulator for hepatocellular carcinoma (HCC) progression [10]. Studies have demonstrated that circRNAs are predominantly present in the cytoplasm, where they interact with miRNAs to modulate the expression of miRNA targets, thereby regulating the malignant behaviors in human cancers [11]. Functionally, hsa_circ_0003288 has been to act as a miR-3918 sponge to release Bcl-2 in HCC [10]. In the present study, we aimed to investigate the biological roles of hsa_circ_0003288 in lung cancer and explore the underlying regulatory mechanisms of hsa_circ_0003288 in NSCLC.
Materials and methods
Ethical approval
The present study has been reviewed and approved by the Internal Review Board of Department of Geriatrics, Ruijin Hospital, Shanghai Jiaotong University. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Cell lines
NSCLC cell lines including NCI-H460, A549, NCI-H1650, and NCI-H1299 were purchased from ATCC, and cultured in DMEM with 10% FBS (Hyclone), penicillin (100 IU/ml) and Streptomycin (100
Clinical samples
A total of 30 patients with NSCLC, who underwent routine surgery at the Ruijin Hospital, School of Medicine, Shanghai Jiao Tong University, Shanghai, China from 2013 to 2017, were included. Their diagnosis was made by a pathological examination, and none of the patients received any preoperative treatment before admission. The patients were classified according to the most recent version of the staging system (8th edition). This study was approved by the Institutional Review Board and Ethics Committee of Ruijin Hospital. Written informed consent was obtained from all patients.
Quantitative real-time PCR (qPCR)
RNA was extracted using RNeasy Mini Kit (QIAGEN, Hilden, Germany) and quantitation of total RNA was conducted using the NanoDrop (Thermo Fisher Scientific, Waltham, MA, USA). cDNA was generated using PrimeScript RT reagent kit with gDNA Eraser (TaKaRa Biotech, Kyoto, Japan) for RT-PCR with mixture containing 12
Hsa_circ_0003288 is upregulated in NSCLC tissues and cell lines. (A) Relative expression of hsa_circ_0003288 in thirty pairs of NSCLC tissues and the adjacent normal tissue was determined by qRT-PCR. (B) Expression of hsa_circ_0003288 was detected in NCI-H460, A549, NCI-H1650, and NCI-H1299 and human bronchial epithelial cells. Results are expressed as mean 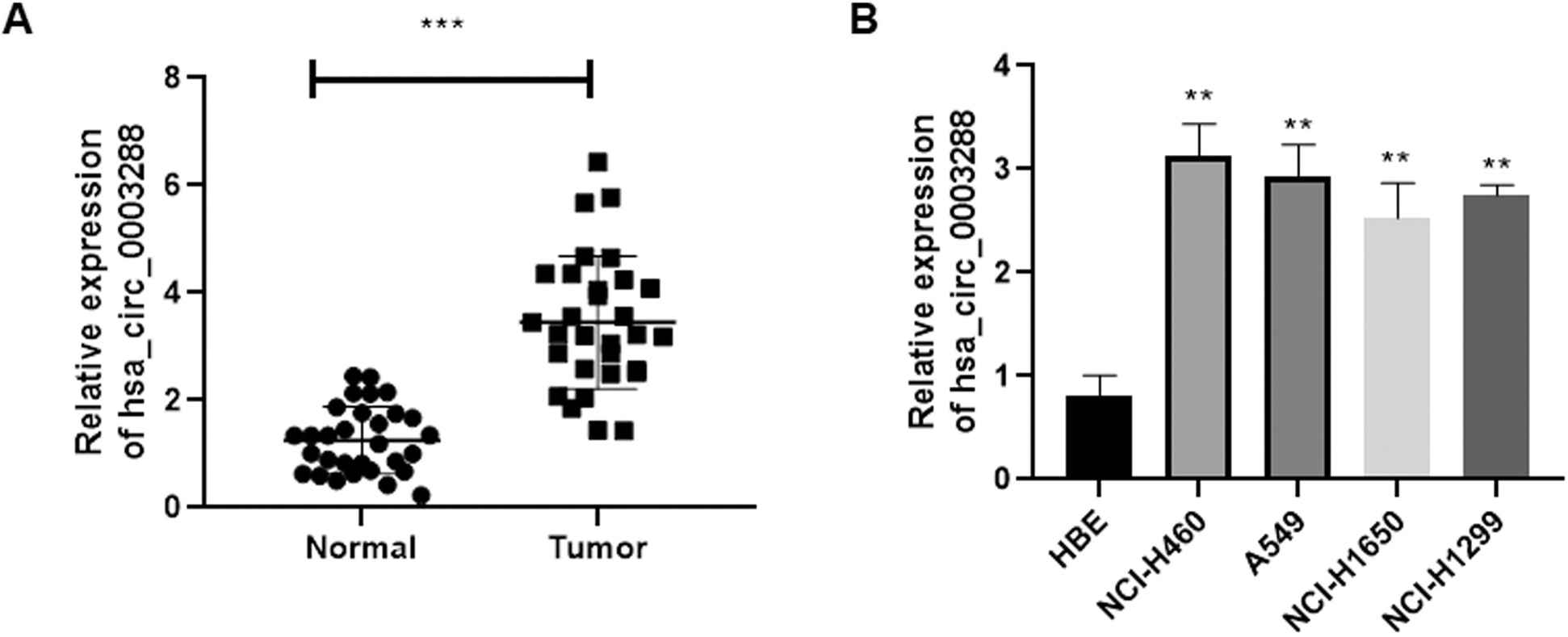
Cells at the density of 3
Colony formation assay
For clone formation assay, cells were seeded in the six-well plates (200 cells/well). After incubation for 2 weeks, cells were stained with crystal violet and viable cells were counted using an automated colony-counting system.
Terminal deoxynucleotidyl transferase d-UTP nick end labelling (TUNEL) staining
Cell apoptosis was analyzed by TUNEL assay using an in-situ cell death detection kit (Roche, Mannheim, Germany) according to the manufacturer’s instruction. In brief, cells were fixed in 4% paraformaldehyde, incubated in 3% H
Dual-luciferase reporter assay
A hsa_circ_0003288 was synthesized with either mutant or wild-type region and cloned into the psiCHECK-2 vector (Applied Biosystems, USA). All cell lines were transfected using Lipofectamine 2000 (Invitrogen, USA). Cells were co-transfected with luciferase reporter plasmids carrying hsa_circ_0003288 wt or hsa_circ_0003288 mut, and miR-145 mimics or miR-NC using Lipofectamine 2000. After forty-eight hours, cells were harvested and the luciferase activity was measured by the Dual-Luciferase Reporter Assay System Kit (Promega, Madison, USA).
Transwell assay
Cell migration and invasion was measured using transwell chamber (Costar, Massachusetts, USA) according to the manufacturer’s protocol. Transfected cells at a density of 2.0
Xenograft assay
The eight-week-old male BALB/c mice were purchased from Shanghai SLAC Laboratory Animal Co. Ltd. (Shanghai, China), and the animal experiments were approved by the Institutional Animal Care and Use Committee of Ruijin Hospital, Shanghai Jiaotong University. Cells carrying hsa_circ_0003288 or si-hsa_circ_0003288 were collected and 2
Statistical analysis
Data were expressed as mean
Results
Hsa_circ_0003288 is upregulated in NSCLC tissues and cell lines
Firstly, we detected the expression profile of hsa_circ _0003288 in thirty paired NSCLC clinical samples and the adjacent normal tissues using quantitative real time PCR (qRT-PCR). Our results found that hsa_circ_0003288 was dramatically upregulated in NSCLC tissues, when compared to the adjacent normal tissues (Fig. 1A). Consistently, a qRT-PCR analysis confirmed elevated expression of hsa_circ_0003288 in lung cancer cell lines – i.e., NCI-H460, A549, NCI-H1650, and NCI-H1299 – than in the normal bronchial epithelial cells (HBE) (Fig. 1B), suggesting that hsa_circ_0003288 may be involved in the tumorigenesis of NSCLC.
Effects of hsa_circ_0003288 on cell proliferation, colony formation, migration, and invasion in NSCLC cells. Hsa_circ_0003288 was overexpressed or downregulated in NCI-H460 and A549 cells. (A and B) Relative expression of hsa_circ_0003288 was determined in NCI-H460 and A549 cells using qRT-PCR. (C and D) Cell proliferation of tumor cells was examined using MTT assay. (E and F) Colony number of tumor cells were counted using the colony formation assay. (G and H) Cell migration and invasion was assessed using the Matrigel Transwell assay. Magnification 200 x. EV: empty vector; NC: negative control. Results are expressed as mean 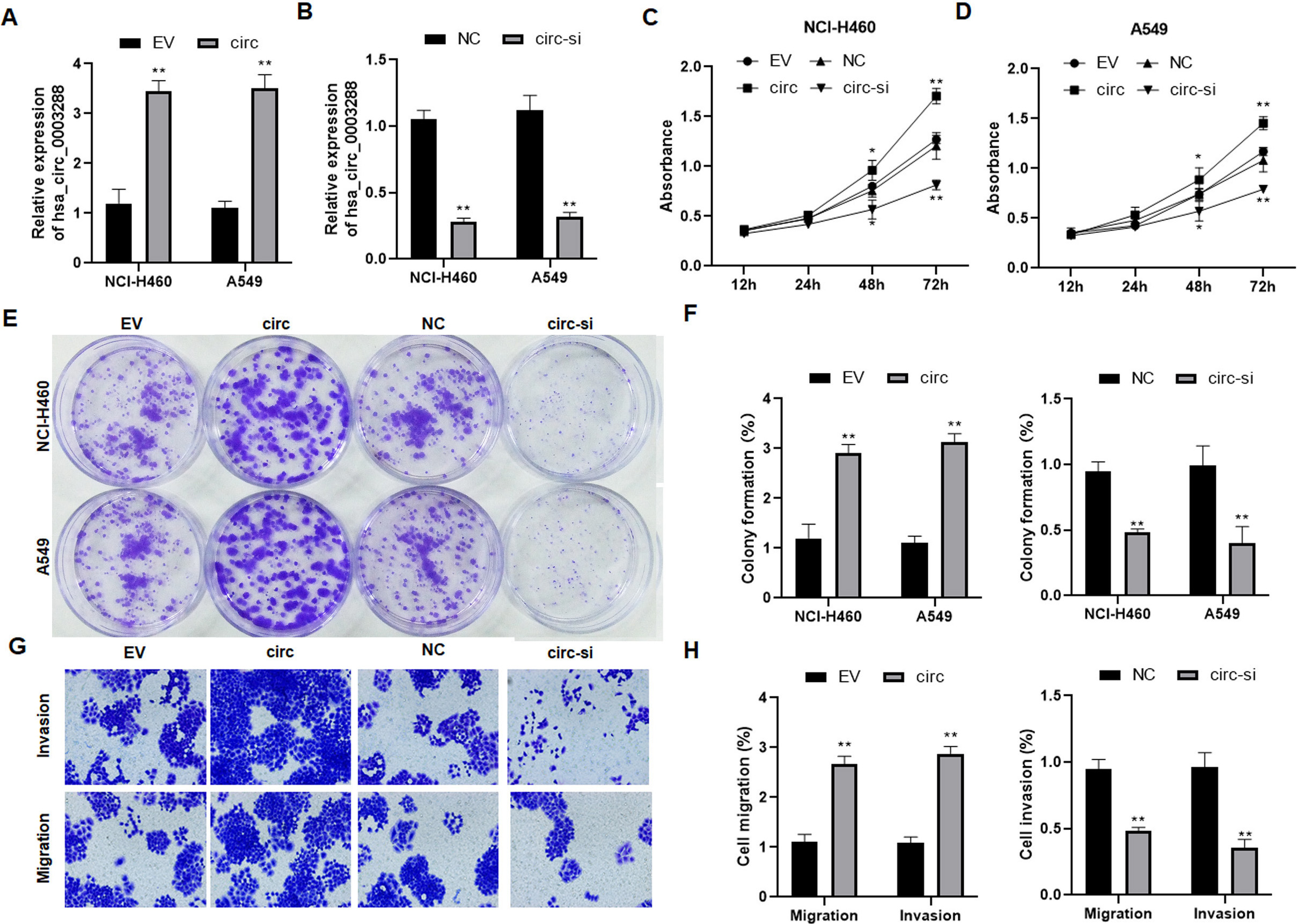
Effects of hsa_circ_0003288 on apoptosis in NSCLC cells. Hsa_circ_0003288 was overexpressed or downregulated in NCI-H460 and A549 cells. TUNEL assay was used to determine the levels of apoptosis rates in tumor cells. (A and B) Representative images of TUNEL positive cells. Magnification 200 x. (C and D) Quantification of apoptosis rates. EV: empty vector; NC: negative control. Results are expressed as mean 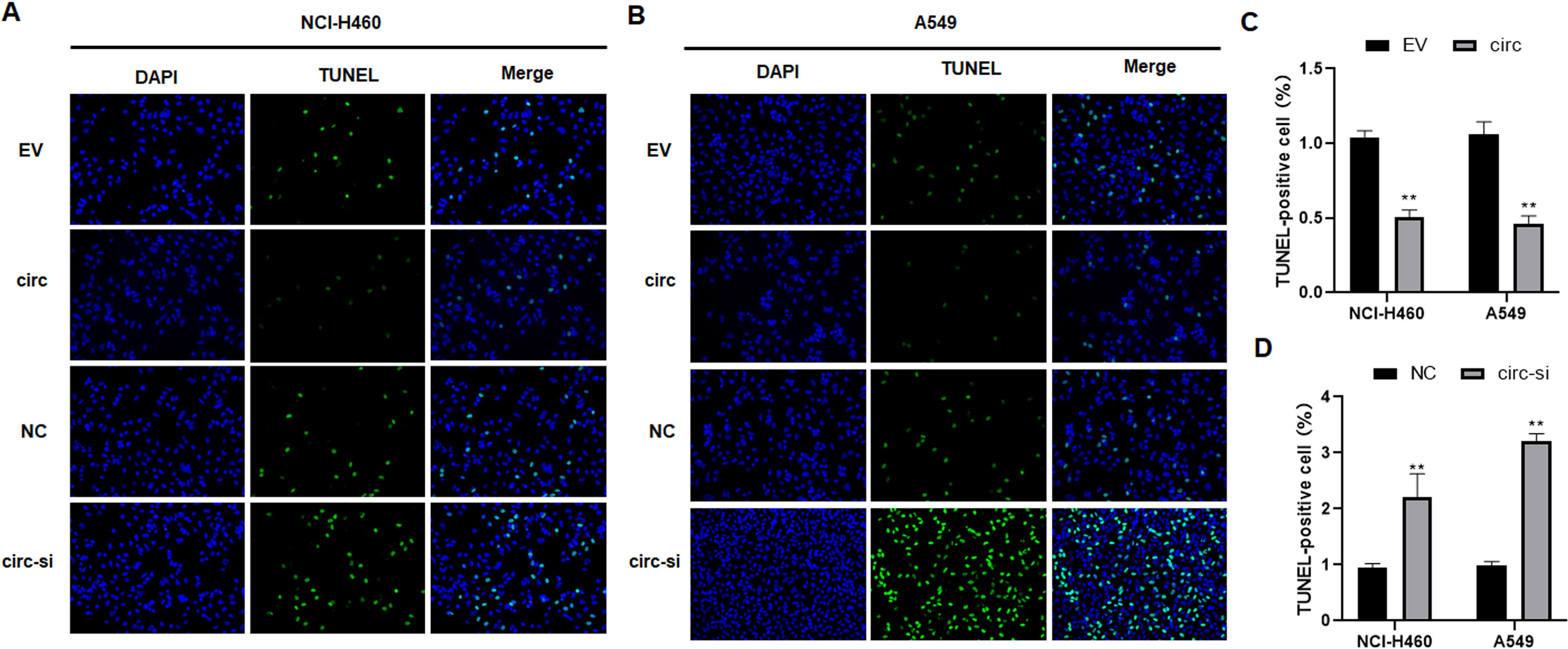
Hsa_circ_0003288 promotes tumor growth and suppresses apoptosis in vivo. NCI-H460 and A549 cells with upregulated or downregulated hsa_circ_0003288 were injected subcutaneously into the flank of nude mice. (A and B). Tumor volume in each group over five weeks. (C and D) A TUNEL assay was used to determine the apoptosis in each group. Magnification 200 x. EV: empty vector; NC: negative control. Results are expressed as mean 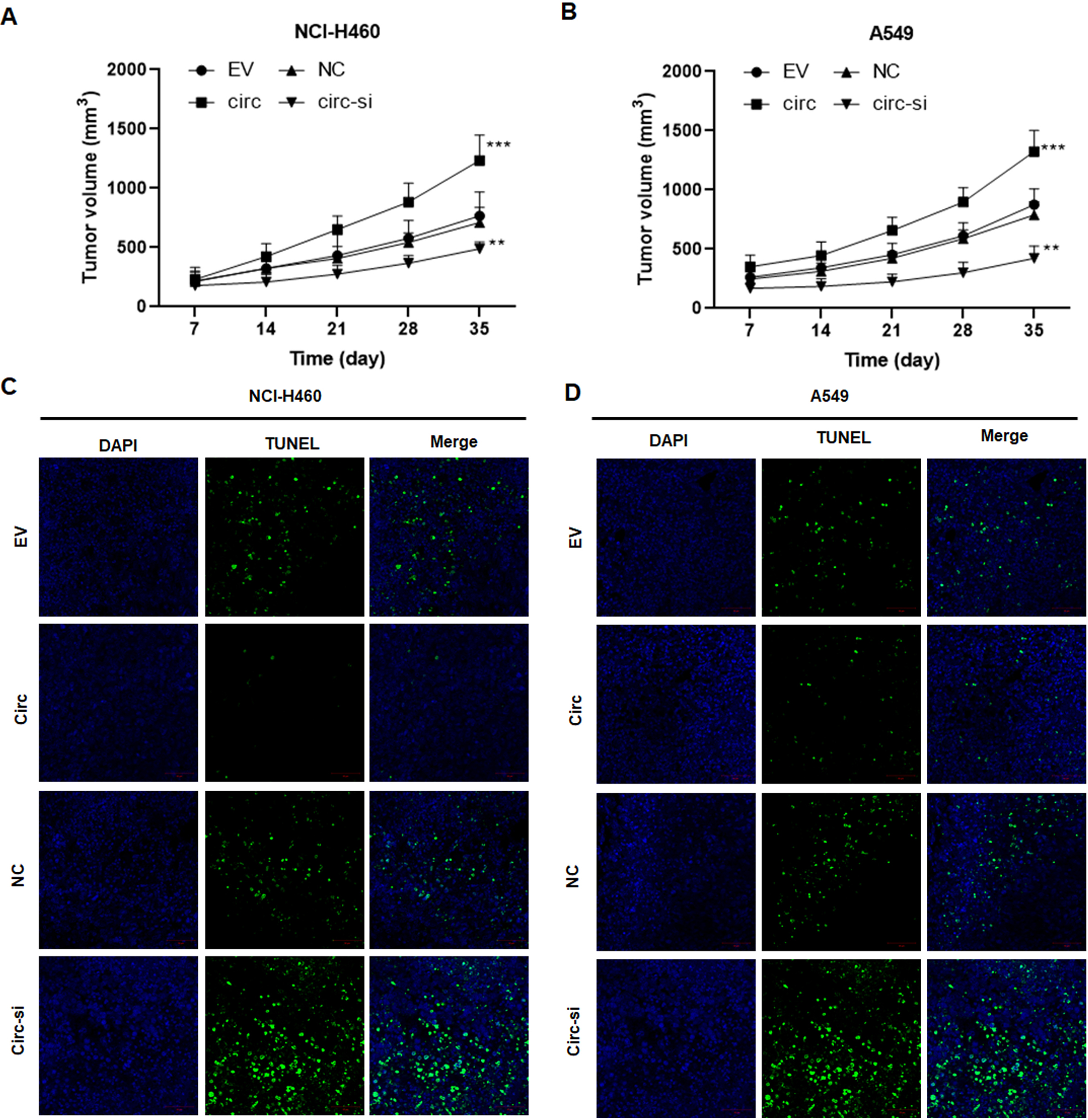
To explore the biological roles of hsa_circ_0003288 in NSCLC, NCI-H460 and A549 cells were transfected with hsa_circ_0003288 overexpressing plasmids or designed hsa_circ_0003288 siRNA, respectively (Fig. 2A and B). Consequently, overexpression of hsa_circ_0003288 significantly promoted tumor cell proliferation (Fig. 2C and D), colony formation (Fig. 2E and F), and invasion and migration (Fig. 2G and H). Conversely, the malignant phenotype was obviously reversed in NCI-H460 and A549 cells following knockdown of hsa_circ_0003288 (Fig. 2C–H). Additionally, TUNEL assay revealed overexpression of hsa_circ_0003288 dramatically suppressed apoptosis, whereas knockdown of hsa_circ_0003288 induced potent apoptosis in NSCLC cells (Fig. 3A–D). Consistent with the in vitro findings, the overexpression of hsa_circ_0003288 dramatically promoted tumor growth (Fig. 4A and B) and suppressed apoptosis (Fig. 4C and D), while knockdown of hsa_circ_0003288 inhibit the malignant phenotype of NSCLC in nude mice inoculated with NCI-H460 and A549 cells overexpressing or downregulating hsa_circ_0003288 (Fig. 4A–D). Collectively, these results indicate that hsa_circ_0003288 could exhibit an oncogenic role in NSCLC cells.
Hsa_circ_0003288 binds directly to miR-145. (A) Bioinformatics analysis of potential hsa_circ_0003288/miR-145 interactions. (B and C) A dual luciferase reporter plasmid containing hsa_circ_0003288-wt or hsa_circ_0003288-mut was co-transfected into NCI-H460 and A549 cells along with miR-145 mimics or miR-NC, and luciferase activities were determined. (D and E) Expression of miR-145 in NCI-H460 and A549 cells with upregulated or downregulated hsa_circ_0003288. EV: empty vector; NC: negative control. Results are expressed as mean 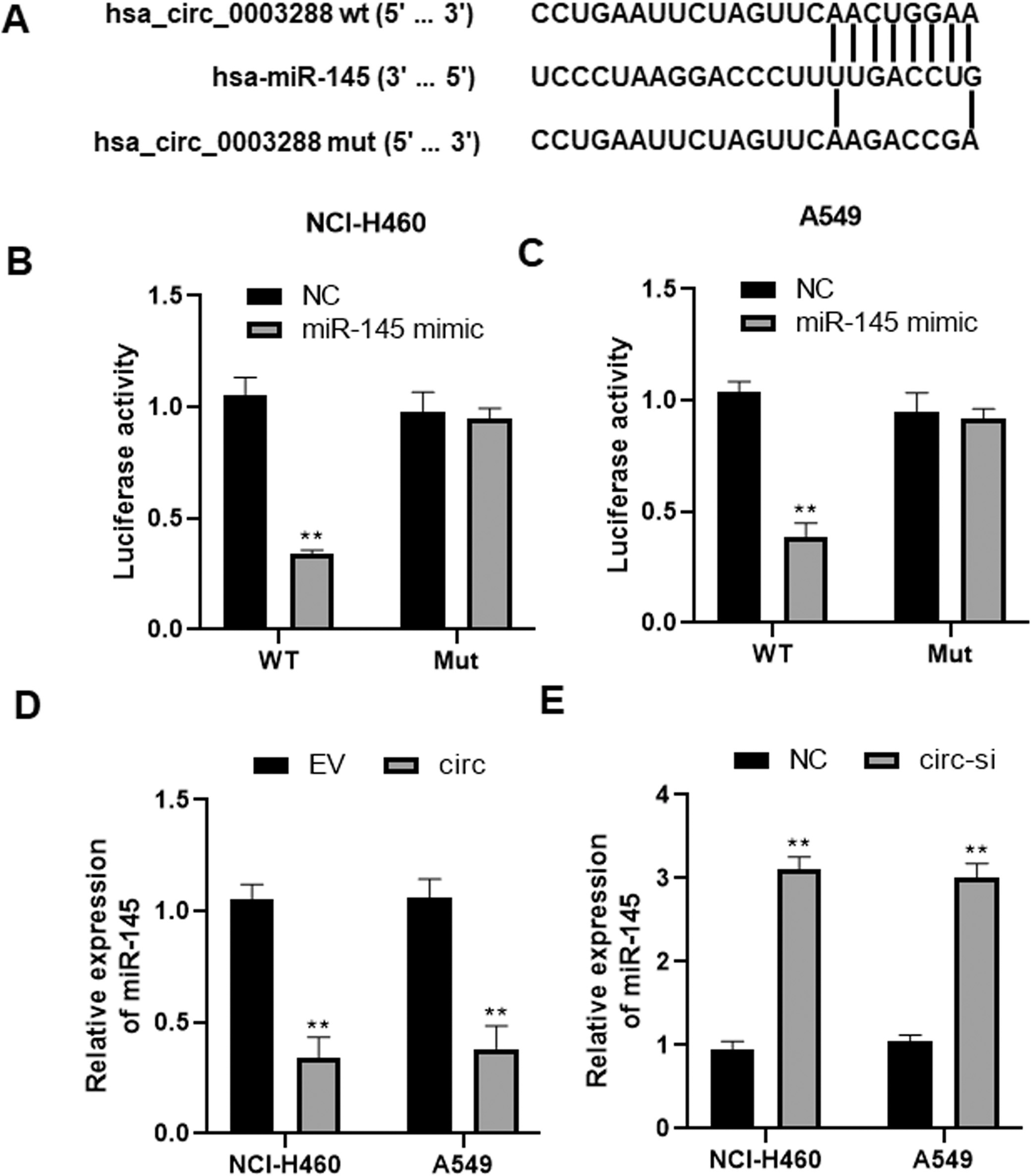
To elucidate the molecular mechanism by which hsa_circ_0003288 exhibit its oncogenic role in NSCLC, we searched potential targets of hsa_circ_0003288 using online software Circular RNA Interactome (
Hsa_circ_0003288 plays an oncogenic role via regulating miR-145 in NSCLC. NCI-H460 and A549 cells were transfected with hsa_circ_0003288 alone or combined with miR-145. (A and B) Cell proliferation of tumor cells was examined using MTT assay. Cell invasion (C and E) and migration (C and F) were detected using the Matrigel Transwell assay. Magnification 200 x. (D and G) TUNEL assay was used to determine the levels of apoptosis rates in tumor cells. Magnification 200 x. EV: empty vector; NC: negative control. Results are expressed as mean 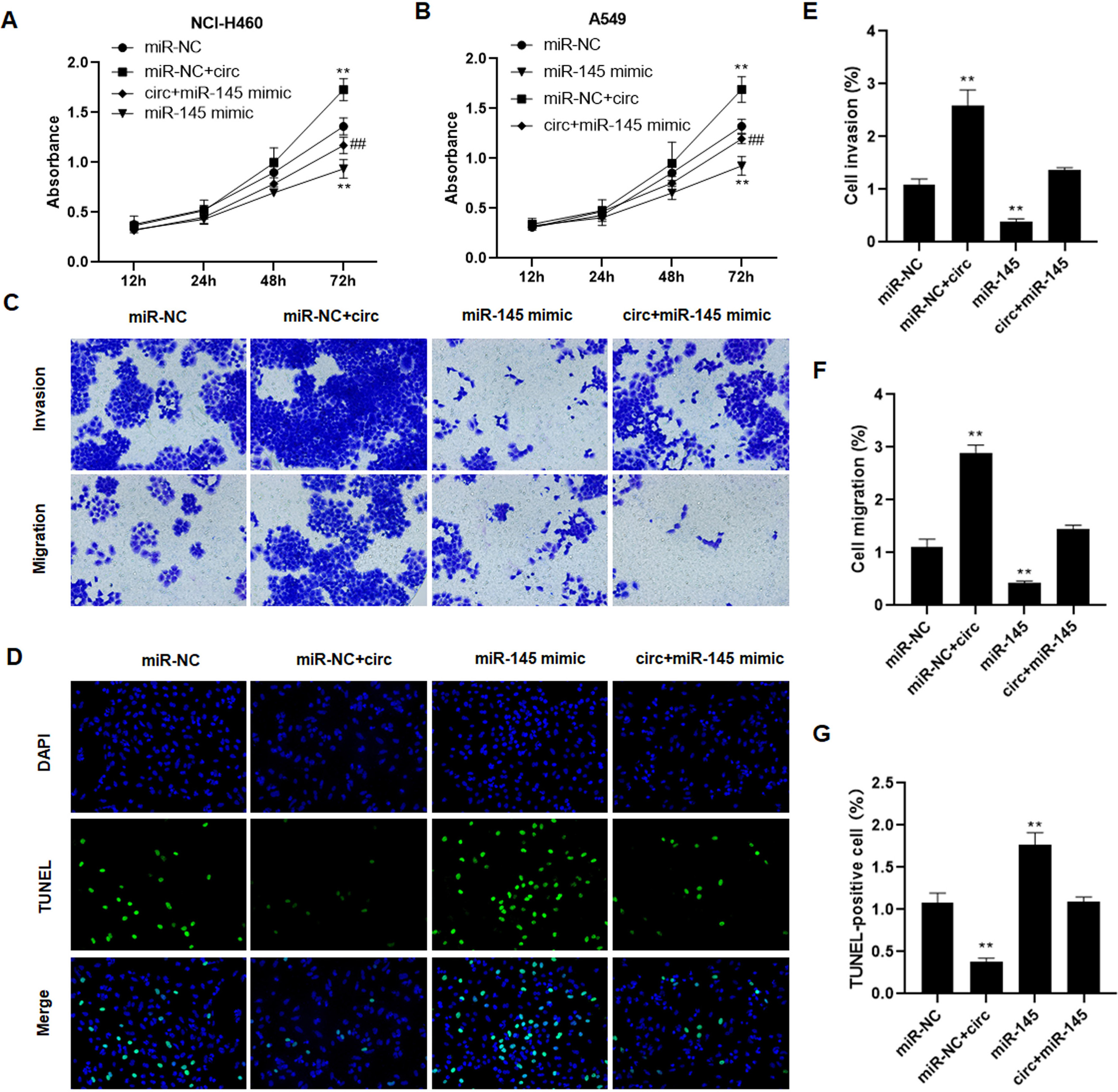
To ask whether hsa_circ_0003288 exerts its oncogenic role by regulating miR-145, we performed rescue assays by transfection with miR-145 mimic in NCI-H460 and A549 cells. Consequently, hsa_circ_0003288 increased NSCLC cell proliferation (Fig. 6A and B), potentiated migration and invasion (Fig. 6C, E and F), and suppressed apoptosis (Fig. 6D and G). Nevertheless, miR-145 transfection alone decreased cell proliferation (Fig. 6A and B), suppressed migration and invasion (Fig. 6C, E and F), and induced apoptosis (Fig. 6D and G) in tumor cells. Strikingly miR-145 co-transfection abrogated such oncogenic role of hsa_circ_0003288 in lung cancer cells (Fig. 6A–G). These findings demonstrated that hsa_circ_0003288 exerts its oncogenic role by regulating miR-145 in lung cancer.
Discussion
CircRNAs play a diverse role in human cancer physiological and pathological processes. A large amount of evidence demonstrates that circRNA is frequently deregulated and its aberrant expression may lead to malignant behaviors, such as excessive growth, resistance to apoptosis, and metastasis [12]. Hence, it is critical to reveal the relationship between circRNA and tumorigenesis. In the present report, we identified that hsa_circ_0003288 functions as a positive regulator in the progression of lung cancer.
Various studies have demonstrated that abnormal expression of circRNA is closely associated with the malignant phenotype in human cancer, such as hepatocellular carcinoma, breast cancer, bladder cancer, and lung cancer [13]. For instance, hsa_circ_100395 expression is remarkably downregulated in lung cancer tissues and correlates with TNM stage and lymphoid node metastases, representing a novel biomarker for prognosis of this cancer [9]. CircFGFR1 has been reported to accelerate progression and confer to anti-PD-1 resistance by sponging miR-381-3p in NSCLC cells [14]. In our study, we found that hsa_circ_0003288 was upregulated in lung cancer samples and cell lines. Consistently, by using the high-throughput sequencing technology, Yang et al. identified that hsa_circ_0003288 is aberrantly expressed in hepatocellular carcinoma and its knockdown suppresses the malignant behaviors, including proliferation and migration [10]. In the present study, our data showed that overexpression of hsa_circ_0003288 could promote cell proliferation, decrease apoptosis, and potentiate migration and invasion in vitro; these malignant behaviors were reversed in hsa_circ_0003288-silenced lung cancer cells. Additionally, we showed that hsa_circ_0003288 promoted tumor growth and suppressed apoptosis of lung cancer in vivo. These findings revealed hsa_circ_0003288 as an oncogene for NSCLC.
CircRNAs regulates pathophysiological processes via different mechanisms, e.g., RNA-binding protein (RBP) sponge, as miRNA sponge, or mRNA regulator [4, 5, 6]. Multiple studies have been conducted on the roles of miRNAs in cancer. Many aberrantly expressed miRNAs have been identified to be involved in the initiation and progression of NSCLC. Circular RNAs often functions as sponges of miRNAs. For example, circ_0074027 has been found promote cell proliferation, apoptosis, and invasion via sponging miR-185-3p in NSCLC [15]. Hsa_circ_0007059 could blunt proliferation and epithelial-mesenchymal transition in lung cancer cells via suppressing miR-378 [16]. Based on this feature of circRNA, we identified that miR-145 interacted directly in lung cancer cells. MiR-145 has been acknowledged to be a tumor suppressor in various human from different origins, such as lung, liver, kidney, prostate, colon, etc. [17, 18, 19]. For instance, miR-145 exhibits a negative role on TGF-
In summary, our study for the first time demonstrates that hsa_circ_0003288 is significantly elevated in NSCLC tissues. Functional and mechanical investigations reveal that hsa_circ_0003288 plays an oncogenic role via modulating miR-145, which has been identified as a tumor suppressor in several human cancers. These findings provide a novel therapeutic target for treatment of lung cancer.
Funding
This work was supported by Scientific Research Project of Shanghai Municipal Commission of Health and Family Planning (201840083); Weak discipline construction project of Shanghai Municipal Commission of Health and Family Planning (2015ZB0503) and Production, study and research project of Shanghai Colleges and Universities (RC20190079).
Author contributions
Conception: Li-Na Pan.
Interpretation or analysis of data: Yun-Fang Ma.
Preparation of the manuscript: Jia-An Hu.
Revision for important intellectual content: Zhi-Hong Xu.
Supervision: Li-Na Pan.
Footnotes
Acknowledgments
We thank all the patients who agreed to participate in this study.
Conflict of interest
The authors declare that they have no conflict of interest.
