Abstract
BACKGROUND:
Several molecules are highly expressed in the serum of cancer patients, and can be used as serological markers. This approach has become one of the important auxiliary diagnostic methods for cancer.
AIM:
To investigate the correlation between the serum levels of EphA2 and VEGF-A and the pathogenesis of colorectal cancer (CRC) as well as the potential value of these molecules in the diagnosis of CRC.
METHODS:
ELISA was used to detect the levels of EphA2 and VEGF-A in the peripheral venous serum of 106 newly diagnosed patients with CRC and 69 normal controls. The relationship between the serum EphA2 and VEGF-A levels and the clinicopathological characteristics of CRC patients was analyzed. ROC analysis was used to investigate the diagnostic value of the serum EphA2 and VEGF-A levels in CRC, and the optimal cutoff value was calculated.
RESULTS:
The serum levels of EphA2 and VEGF-A in the CRC group were higher than those in the control as well as CEA, the serum level of EphA2 was positively correlated with the VEGF-A levels, but neither was significantly associated with the clinicopathological parameters of CRC. The ROC curve showed that the single index AUC was
CONCLUSION:
The serum levels of EphA2 and VEGF-A are high in CRC patients, and the combine detection of CEA, EphA2 and VEGF-A can significantly improve the diagnostic accuracy of CRC.
Introduction
Among malignant tumors, colorectal cancer (CRC) ranks third in incidence and second in mortality worldwide [1]. In 2020, there were approximately 147,950 patients who were newly diagnosed with colorectal cancer in the United States [2], and 12% were under the age of 50; in addition, approximately 52,980 patients will die from colorectal cancer, and 7% will be under the age of 50 [2, 3]. According to the American Cancer Society (ACS) CRC statistics, from 2000 to 2014, the incidence of CRC increased by 22% and the mortality rate increased by 13% [4], and from 2008 to 2017, the mortality rate of colorectal cancer among people under the age of 50 increased at an average annual rate of 1.3% [2]. The above data are cause for concern; the whole population of patients with colorectal cancer is rapidly becoming younger. Therefore, finding new methods to screen for early CRC is particularly important for its prevention and treatment.
Erythropoietin producing hepatocellular receptor (Eph receptor) is the largest family of receptor tyrosine kinases (RTK), and these receptors can be classified based on their structure and receptor ligand specificity into EphA (EphA1–8, 10), consisting of nine A-type Eph receptors, and EphB (EphB1–4, 6), consisting of five B-type Eph receptors [5]. The RKT family is widely involved in many pathophysiological processes, such as embryonic development, inflammatory responses and angiogenesis; furthermore, most Eph receptors and ephrin are dysfunctional in various types of cancer, and these changes are also associated with tumor progression, tumor malignancy, and prognosis [6, 7]. Therein, EphA2 has been shown to over-expressed in various cancers, such as non-small cell lung cancer (NSCLC), prostate cancer (PC), breast cancer (BC), esophageal cancer, melanoma, and CRC [8, 9, 10, 11, 12, 13]. Previous studies have shown that high expressed level of EphA2 is closely related to the apoptosis of tumor cells. Moreover, inhibiting the expression of the EphA2 gene could effectively induce apoptosis, reduce melanoma cell viability, and abolish tumorigenic growth. Therefore, EphA2 is considered a promising target for tumor therapy [11].
Vascular endothelial growth factor (VEGF) is an important proangiogenic factor, and the VEGF family comprises five structurally related members: VEGFA, VEGF-A, VEGF-B, VEGF-C, VEGF-D and placental growth factor (PLGF) [14]. VEGF-A has received the most attention, and it acts through three structurally related VEGF receptor tyrosine kinases (RTKs), VEGFR1, VEGFR2 and VEGFR3, to promote vascular endothelial cell proliferation and migration, prevent vascular endothelial cell apoptosis and luminal network degeneration, and increase vascular permeability [15]. VEGF is highly expressed in tumors, can promote tumor angiogenesis, plays an important role in maintaining the continuous growth of tumors, and is also essential for tumor metastasis. VEGF has been implicated in the processes of development, invasion and metastasis in different cancer types including CRC [16]. At present, bevacizumab, – an antibody against VEGF –, combined with chemotherapy is being used in the treatment of CRC in clinical practice and has achieved certain positive effects [17]. The detection of VEGF expression may be of great practical value in predicting the metastatic potential and guiding the treatment of CRC.
To date, the expression of EphA2 and VEGF and their association with the clinical features of CRC are not fully understood, and previous studies have mainly focused on tissue samples of primary tumor rather than human serum samples or animal experiments [18, 19]. Based on this, we collected serum samples from patients with CRC and investigated the relationship between the serum levels of EphA2 and VEGF-A and the clinicopathological characteristics of the patients to further clarify whether circulating EphA2 and VEGF-A levels can be used as effective diagnostic markers for CRC.
Materials and methods
This study was conducted in the Department of Gastrointestinal Surgery, Department of General Surgery, the First Affiliated Hospital of Anhui Medical University. From March 2018 to October 2018, 131 consecutive Han Chinese patients with CRC were enrolled in this study. These patients had not previously received surgery or any chemotherapy or radiotherapy, and all of them were diagnosed with colon cancer or rectal cancer by colonoscopy and tissue biopsy before the operation. These patients had complete general information, no other malignancies, no uncontrolled acute or chronic diseases (nephropathy, cardiovascular disease, rheumatism, etc.), and the female patients were not pregnant or breastfeeding. Finally, 25 patients were excluded from the study, and a total of 106 were included as the experimental group (CRC group).
Sixty-nine healthy Han Chinese subjects who volunteered to undergo total colonoscopy in our outpatient clinic during the same period and were found to be without polyps or inflammatory bowel disease were included as a control group.
General data, such as gender, age, and body mass index (BMI), were recorded for both groups, and the subjects completed a general questionnaire that asked about their smoking habits (those who smoked more than five cigarettes per day on average in any one year were defined as smokers, and all the female subjects in both groups were nonsmokers), alcohol consumption (those who consumed an average of more than 25 g of alcohol per day were defined as drinkers), medication history (e.g., the use of antihypertensive drugs, opsonizing lipid drugs, aspirin, thyroid hormones), family history, and any history of hypertension.
The preoperative laboratory serological test results, such as blood glucose, blood lipids, CEA, CA125, and CA19-9, were recorded in the CRC group. The patients in the CRC group underwent radical resection of the tumor by Professor Yongxiang Li’s team. Pathological features such as tumor stage, histopathological type, lymph node metastasis status, vascular tumor thrombus, and nerve invasion were recorded according to the postoperative pathology. The 8th edition of colorectal cancer staging criteria (NCCN, 2017) was used in this study. Tumor sites were divided into the right colon, left colon, and rectum. The right colon included the cecum, ascending colon, and the proximal two-thirds of the transverse colon; the left colon included the distal one-third of the transverse colon, descending colon, and sigmoid colon; and the rectum included areas below the sigmoid colon and above the dentate line. The Ethics Committee of the First Affiliated Hospital of Anhui Medical University approved the study, and all the subjects gave informed consent.
Blood sample collection
After fasting and water deprivation for one night, 5 ml of peripheral venous blood was collected from the antecubital vein of all 175 subjects (106 CRC patients and 69 healthy subjects) in the two groups between 6:00 and 7:00 the next morning. The blood samples were centrifuged within 30 min (4000 g, 10 min), the upper layer of serum was transferred into a sterilized 1.5 ml EP tube, and the serum samples were stored in a
Measurement of EphA2 and VEGF-A
The serum levels of EphA2 and VEGF-A were measured by human EphA2 enzyme-linked immunosorbent assay kits based on the double-antibody sandwich method (Wuhan Elabscience Biotechnology Co., Ltd., model E-EL-H0861c) and human VEGF-A enzyme-linked immunosorbent assay kits (Wuhan Elabscience Biotechnology Co., Ltd., model E-EL-H0111c) in strict accordance with the kit instructions. The ELISA kits showed no cross-reactivity with any cytokines. The EphA2 levels are expressed in ng/ml, the VEGF-A levels are expressed in pg/ml, and the intraplate and interplate variation coefficients were less than 10%. Standard, control, and sample optical density (OD) values were assessed at a wavelength of 450 nm. The concentration was calculated and determined by the OD values of the sample and standard curve.
Other variables
Blood glucose and blood lipid levels were measured using an automatic biochemical analyzer (MODULEP800, Roche, Switzerland), and the analysis was performed by the laboratory department of the hospital. Blood glucose was measured by the glucose oxidase method, TG and TC were measured by the enzymatic method, and HDL-C and LDL-C were measured by the direct method. Serum levels of CEA, CA19-9, and CA125 were measured by electrochemiluminescence (Roche, cobas601). The reference ranges were CEA: 0–5 ng/mL; CA19-9: 0–34 U/mL; and CA125: 0–35 U/mL.
Statistical analysis
Excel 2013 was used for data summarization and collation. Statistical analysis was conducted with Windows vers.16.0 SPSS statistical software (SPSS, Chicago, IL, USA), MedCalc19.0 software (
Comparison of clinical data between CRC group and control group
Comparison of clinical data between CRC group and control group
BMI, Body mass index; FPG, fasting plasma glucose; TC, total cholesterol; TG, triglyceride; HDL-C, high density lipoprotein cholesterol; EphA2, Erythropoietin producing hepatocellular receptor A2; VEGF-A, vascular endothelial cell growth factor A; M, Median; SD, Standard deviation; P25, 25th percentile; P75, 75th percentile.
A total of 106 patients with CRC (CRC group) and 69 healthy individuals (control group) were included in the study. Descriptive characteristics of the subjects in both groups are summarized in Table 1. The age of the CRC group was significantly higher than that of the control group, and the level of serum TC in the CRC group was lower than that in the control group, but there was no significant difference. The serum levels of HDL-C and BMI of the CRC group were lower than those of the control group (
The preoperative serum levels of EphA2 and VEGF-A in the CRC group were higher than those in the control group: the serum levels of EphA2 in the two groups were 285.48 (241.17, 349.10) ng/mL vs 258.02 (210.67, 294.04) ng/mL,
Serum CEA, EphA2 and VEGF-A levels in CRC group and control group. A. Serum CEA levels in CRC group and control group (The results of Fig. 1A and Table 3 showed that the serum CEA level of the CRC group was 4.49 (2.20, 8.82) ng/mL, which was significantly higher than that of the control group (2.07, 3.59) ng/mL, 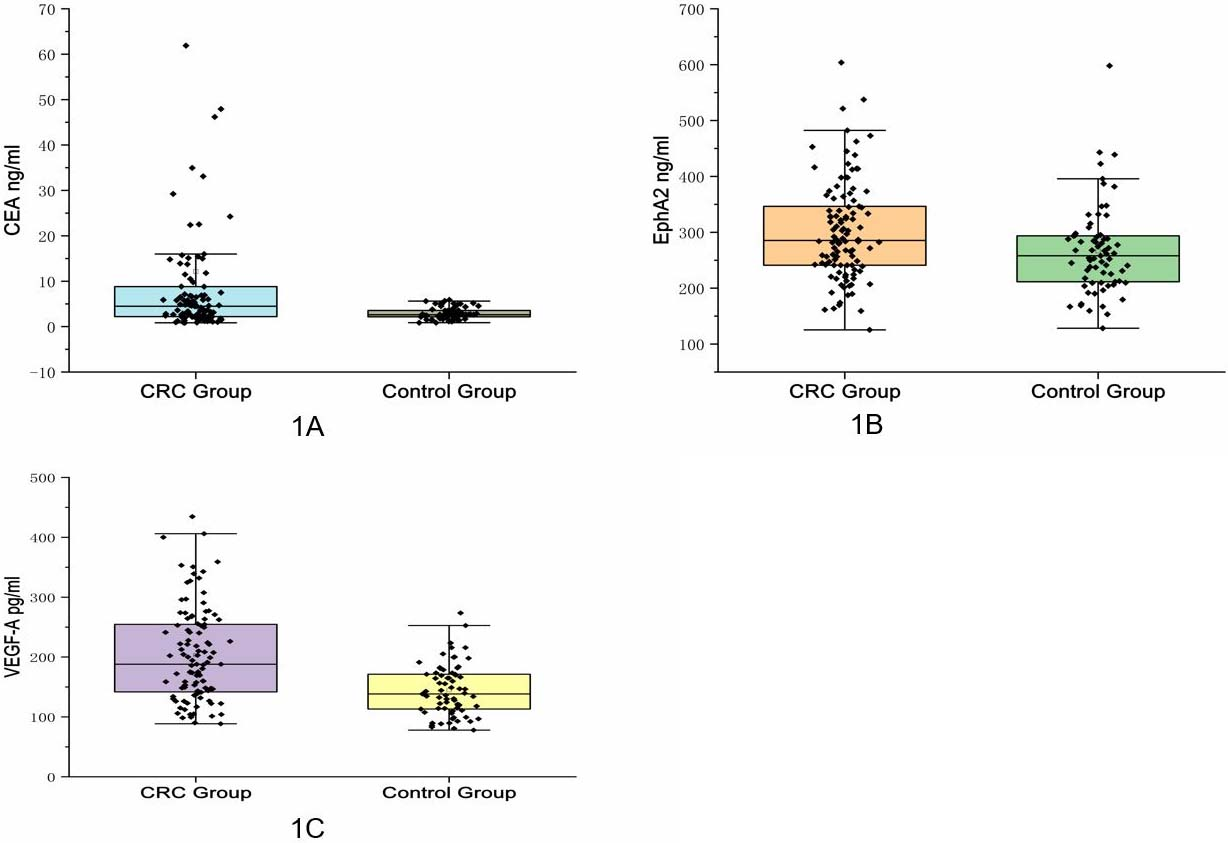
Receiver operating characteristics (ROC) curve for serum EphA2, VEGF-A and CEA for the diagnosis of colorectal cancer. A. ROC curve of single index for CRC diagnosis. B. ROC curve of combined indicators for CRC diagnosis.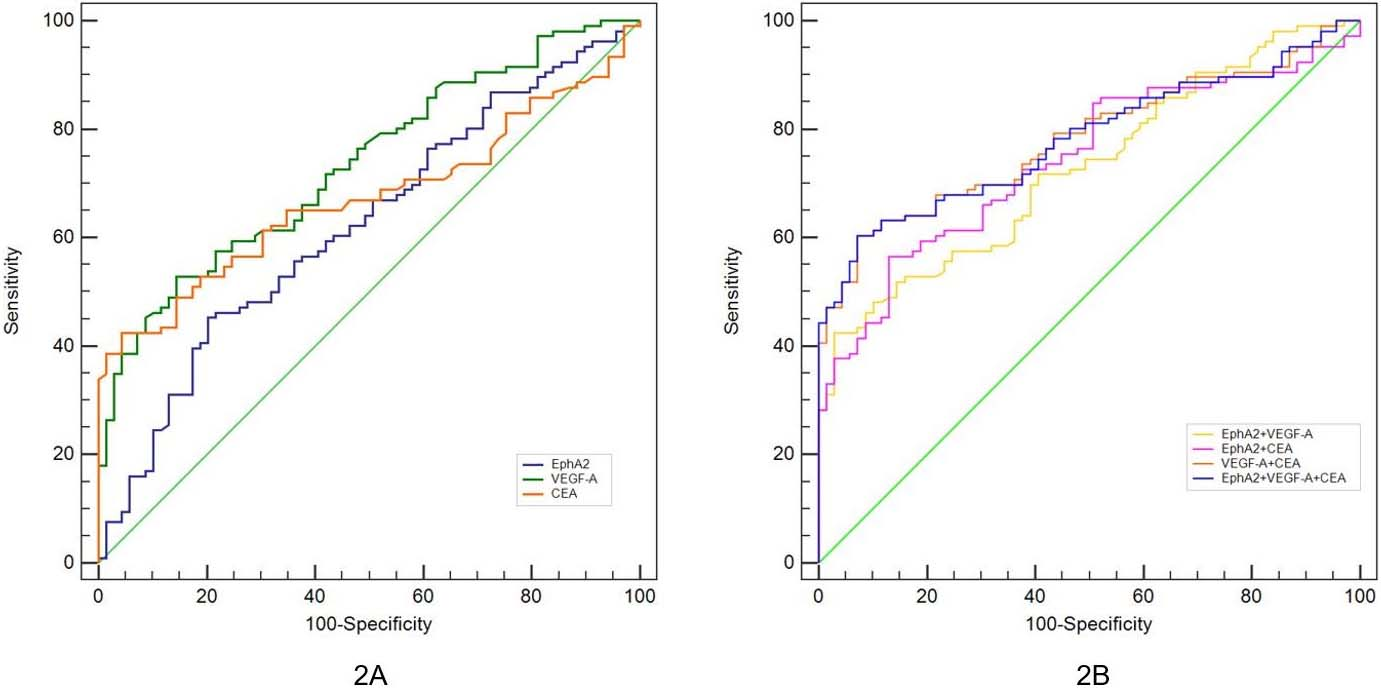
The patients in the CRC group were divided into different subgroups according to their age, sex, tumor size, location, stage, differentiation, presence of lymph node metastasis, presence of vascular tumor thrombus, presence of neural invasion, and serum levels of CEA, CA19-9, and CA125. Multiple comparisons were performed to determine whether there were differences in the serum levels of EphA2 and VEGF-A between the subgroups (Table 2). The expression levels of EphA2 and VEGF-A were not significantly associated with age, sex, or other clinicopathological parameters of the patients with CRC, including tumor size, location, lymph node metastasis, TNM stage, vascular tumor thrombus, and neurological invasion. The serum level of EphA2 was positively correlated with the serum level of VEGF-A (
Comparison of serum levels of EphA2 and VEGF-A with clinicopathological features in patients with CRC
SD, standard deviation; RC, Right hemicolon; LC, Left hemicolon; LNM, lymph node metastasis; CEA, carcinoembryonic antigen; CA19-9, carbohydrate antigen 19-9; CA125, carbohydrate antigen 125.
Pearson correlation analysis showed that the level of EphA2 in the serum was mildly positively correlated with medication intake (
Correlation between serum levels of EphA2 and VEGF-A and other parameters in patients with CRC
BMI, Body mass index; CEA, carcinoembryonic antigen; CA19-9, carbohydrate antigen 19-9; CA125, carbohydrate antigen 125; EphA2, Erythropoietin producing hepatocellular receptor A2; VEGF-A, vascular endothelial cell growth factor A;
To verify the ability of each indicator to diagnose CRC, we determined the expression levels of EphA2, VEGF-A, and CEA in 176 serum samples from patients with CRC and normal controls, generated ROC curves with MedCalc 19.0 software, and calculated the cut-off values of the optimal concentrations of the three serum indicators, as shown in Fig. 2 and Table 4. The ROC curve analysis of a single indicator showed that the AUC of EphA2 was 0.622 (95% CI: 0.545–0.694), VEGF-A was 0.734 (95% CI: 0.662–0.798) and CEA was 0.673 (95% CI: 0.598–0.742) (Fig. 2A). We can see that serum VEGF-A, whose accuracy (Jordan index) is only 38.3%, has the best efficacy when tested as a single indicator. EphA2 is not superior to CEA and VEGF-A for the diagnosis of CRC. The analysis of the ROC curve by the combined detection of three or two markers showed that the VEGF-A+CEA scheme had the highest diagnostic accuracy (53.1%), and its AUC was 0.782 (95% CI: 0.598–0.742) (Fig. 2B). The combination of the three was not superior to the combination of VEGF-A and CEA in the diagnosis of CRC, and there was no significant difference in their AUC (
In addition, we also tried to identify the optimal cut-off values of the three indicators for CRC diagnosis, and the AUC for EphA2 to detect CRC was determined to be
ROC curve characteristic parameters of EphA2, VEGF-A and CEA in the diagnosis of CRC
EphA2, Erythropoietin producing hepatocellular receptor A2; VEGF-A, vascular endothelial cell growth factor A; CEA, carcinoembryonic antigen; CI, Confidence Interval;
CRC is one of the most common malignancies and is a leading cause of cancer-related death worldwide [20]. There are data indicating that people born after approximately 1990 in the United States have a doubled risk of colon cancer and a four-fold increased risk of rectal cancer than adults born in 1950; in addition, currently, nearly one-third of patients with rectal cancer are younger than 55 years of age [21]. This strongly suggests that CRC has a trend of affecting increasingly younger people and an increasing incidence. Early diagnosis can improve CRC survival, and the American Cancer Society (ACS) recommends starting CRC screening at age 45 for regular screening targeting adults with average risk, especially those over 50 years of age [22]. However, colonoscopy cannot be universally used for CRC screening because it is invasive and expensive, so it is of great importance to find new screening tools.
EphA2, a member of the Eph receptor family class A, is overexpressed in a variety of human tumors, including CRC, and is, considered one of the most promising cell membrane-associated tumor antigens [23]. EphA2 is overexpressed in esophageal squamous cell carcinoma (ESCC) and also positively associated with tumor differentiation and lymph node metastasis, suggesting that patients with EphA2 overexpression have a worse prognosis [8].
Not only ESCC, EphA2 expression has also been shown to be positively correlated with breast cancer (BC) progression [24], and recent studies have shown that reduced expression of EphA2 in BC can inhibit the clonal growth of BC cells [25]. During tumor progression, EphA2 is also one of the key regulatory signals of angiogenic mimicry [26] (VM). VM describes the formation of an epithelial-independent tumor microcirculatory system that is different from traditional angiogenesis and is another important vascular recruitment route.
In osteosarcoma, the inhibition gene of EphA2 expression of in cell experiments in vitro reduced the VM caused by osteosarcoma cells, decreased the proliferation rate, and increased the apoptosis rate [27]. Furthermore, EphA2 is overexpressed in hepatocellular carcinoma (HCC), and EphA2 is a direct target of microRNA-520d-3p (miR-520d-3p), which has a tumor suppressive effect in human tumors. It indicated that miR-520d-3p may exert its an anti-cancer effect by inhibiting the expression of EphA2 [28]. These are good indications that EphA2 plays an important role in tumorigenesis, and it may become a molecular marker for tumor treatment and prognosis of tumor prognostication.
Angiogenesis plays a crucial role in the development of tumors [29], a process involving many angiogenesis and inhibitory factors [30]. During angiogenesis, VEGF can specifically act on vascular endothelial cells to promote cell proliferation, migration and angiogenesis [31]. VEGF is also involved in tumor angiogenesis mimicry [26]. In a CRC mouse model, inhibiting VEGF expression causes a significant reduction in tumor numbers, and inhibition of VM was also observed [32]. In addition to VEGF, EphA2 and its major ligand, ephrin-A1, have become increasingly recognized as playing important roles in tumorigenesis and neovascularization in recent years [33]. Recent studies have shown that EphA2 is required for the VEGF-mediated induction of angiogenesis, but the mechanism is still unclear [34]. EphA2 may help regulate the endothelial cell response to VEGF-mediated tumor angiogenesis through the angiogenic factor Slit2 [35]. VEGF upregulates the expression of EphA2 in human brain microvascular endothelial cells (HBMECs) by binding to VEGFR2 and subsequently activating the intracellular PI3K/Akt and ERK1/2 signaling pathways, which contribute to an increase in paracellular permeability [34]; these phenomena may contribute to the metastasis of the tumor. This indicates some type of correlation between EphA2 and VEGF in both tumor-bearing mice and clinical cancer patients. However, the relationship between the serum levels of EphA2 and VEGF and CRC is not clear.
Based on these studies, we explored the relationship between the clinical characteristics of CRC patients and serum EphA2 and VEGF levels to provide a basis for the diagnosis and treatment of CRC. In this study, we compared the levels of EphA2 and VEGF-A in the serum from 106 patients with CRC and 69 normal healthy subjects, and the results showed that serum EphA2 and VEGF-A levels were significantly higher in CRC patients than in controls (
We further analyzed the relationship between EphA2 and VEGF-A expression and clinicopathological features, and the results showed that the expression levels of EphA2 and VEGF-A were not significantly associated with age, sex or other clinicopathological parameters of the patients with CRC, including tumor size, location, lymph node metastasis, TNM stage, vascular tumor thrombus or neurological invasion. In addition, this study observed a correlation between the serum VEGF-A and CEA levels, which is consistent with the study by Bestas et al. [39], but EphA2 was not observed to be associated with CEA.
As mentioned above, many studies have confirmed that the expression of EphA2 and/or VEGF-A is increased in CRC [40, 41], which is consistent with the results of this study. In addition, some studies have found that EphA2 exhibits an upward trend with increased CRC staging, and the expression level of EphA2 in cancer tissues is also significantly correlated with clinical staging [18, 24], but we did not find any such correlations in this study. In contrast, this study found that the expression level of EphA2 in the serum of patients with advanced CRC exhibited a downward trend compared with that in the serum of patients with early CRC, although there was no significant difference. De Robertis et al. validated the tissue samples of 1,663 patients with colorectal cancer, and the findings suggested that the expression level of EphA2 has no correlation with tumor staging progression [42].
The somewhat conflicting results regarding the prognostic or predictive value of EphA2 overexpression in solid tumors might be associated with the complex nature and diverse roles of the EphA2 molecule (both the receptor and its ligand). For example, EphA2 receptors are regulated through ligand-dependent pathways, which can inhibit tumor cell proliferation in most cases, and promote tumor progression through ligand-independent regulatory pathways. In addition, the source of the samples in different studies may be different (the expression level of EphA2 in serum and tissue is not parallel, and humans have more influencing factors than mice). Finally, differences may arise due to the relatively small sample size of this study and the shortcomings of the retrospective study design.
Furthermore, we also provided information on the diagnostic use of EphA2 and VEGF-A for CRC and calculated the optimal cutoff values. For a single indicator, the area under the ROC curve (AUC) of VEGF-A for the diagnosis of CRC was 0.734, with higher accuracy than EphA2 and CEA. In comparison, EphA2 did not show a particular advantage. When used in combination for diagnosis, whether two or three of these factors were used together, the AUCs were
However, our research also has shortcomings, and the sample size was relatively small. Due to insufficient patient follow-up time, the relationship between EphA2 and VEGF-A for CRC prognosis cannot be determined, but the patient’s survival status will be further followed up to investigate that in the future.
In short, the various functions of the EphA2 molecule make it attractive for research, and our results could constitute a reference for further stimulating such research.
