Abstract
BACKGROUND:
The highest risk factor for mortality among malignant tumors is metastasis. Endocrine gland-derived vascular endothelial growth factor (EG-VEGF) is an angiogenic factor which biological activity is mediated via two G protein-coupled receptors, prokineticin receptor1 (PROKR1) and PROKR2. Recent studies suggested that EG-VEGF expression is deregulated in multiple cancers including colorectal cancer (CRC).
METHODS:
Using distinctive CRC and peritoneal carcinomatosis (PC) cohorts and a corresponding control cohort, we determined the circulating levels of EG-VEGF and its in situ expression, and that of its related receptors.
RESULTS:
Circulating EG-VEGF levels were significantly increased in patients with metastatic PC compared to CRC and control patients (
CONCLUSION:
Altogether these findings suggest that EG-VEGF/receptors system might be an important actor in the CRC progression into PC and might be involved in the ability of tumor cells to invade other organs. Circulating EG-VEGF could be proposed as a prognostic marker in human CRC and its progression into PC.
Introduction
Associated metastasis to gastrointestinal cancer, especially colorectal cancer (CRC) still constitutes a serious health problem, and is the major cause of death in CRC patients, with approximately 50% of survival in patients with multiple metastatic sites [1, 2]. Among processes that contribute to the course of metastasis in CRC is angiogenesis. This process is one of the most critical steps in tumorigenesis [3, 4]. Recently, attention has been directed towards anti-angiogenesis therapies, in which tumor proliferation is inhibited by blocking the formation of new tumor vessels, using angiogenesis inhibitors and drugs [5, 6].
Endocrine gland-derived vascular endothelial growth factor (EG-VEGF), also known as Prokineticin-1 (PROK-1) is the canonical member of the prokineticin family. This family accounts two members PROK-1 and PROK-2, also called Bv8 [7]. EG-VEGF was reported to selectively act on the endothelium of endocrine gland cells [8, 9]. Although EG-VEGF exhibits the same actions on the vascular system as the ones described for VEGF, these factors do not have any homologies, they constitute distinct proteins from two different families, with multiple regulatory functions [7]. Prokineticin receptor (PROKR) 1 and 2 were identified as EG-VEGF receptors, they belong to G-protein coupled receptors and mediate multiple functions upon their activation by EG-VEGF or its analog, the prokineticin 2 also called Bv8 [10, 11]. Human EG-VEGF cDNA comprises 1378-bp, encoding for a protein of 105 amino acids, including a classical signal sequence of 19 residues and a mature protein of 86 amino acids with a molecular weight of 8.6 kDa. Its biological effects have been associated with neuronal survival, pain sensation, contraction of gastrointestinal smooth muscle, placental development and hematopoiesis [12, 13].
Among the different types of cancer, CRC is characterized by its susceptibility to trigger hematogeneous metastasis [14]. It is one of the third most common malignancies worldwide, and the fourth leading cause of cancer-related mortality that affects both genders equally [15]. The incidence and survival rates of CRC are significantly different between different countries around the world [18]. Once a gastrointestinal malignancy has reached the stage of peritoneal carcinomatosis (PC), this is usually considered as the final stage of the metastatic cancer.
To date, 15 to 20% of patients with metastatic CRC show isolated PC with significant clinical relevance. For a long time, PC has been considered a consequence of systemic dissemination of the cancer via the bloodstream [19]. It is now well established that angiogenic processes contribute to CRC progression. These processes are orchestrated by multiple growth factors that control the angiogenesis balance.
PC arising from digestive cancers is mostly associated with a poor prognosis. Although its symptoms are non-specific, indirect signs such as bowel obstruction ascites can be indicative but are not disease specific [19]. For CRC, peritoneal seeding of cancer cells leading to PC is rather common [20]. At the time of the first diagnosis, it is estimated that the peritoneal surface is already effected in 8–15% of CRC cases [21, 22]. Importantly, in approximately 25% of patients with recurrent CRC, the peritoneal cavity seems to be the only metastatic site of the disease [23, 24]. To date, much is known about CRC development; nevertheless, mechanisms that are associated with its progression are still to be investigated.
Recent studies reported positive correlations between the prokineticin family of proteins and CRC development. PROK-2 has recently been qualified as an angiogenic factor of colorectal cancer [25]. The canonical member of the prokineticin family, EG-VEGF has been reported to be associated with CRC development and metastasis [26, 27]. Importantly, EG-VEGF mRNA expression, measured within CRC tumor specimens was correlated to the severity of the CRC disease [28]. Nevertheless, no studies have investigated EG-VEGF circulating levels and expression in CRC and PC patients. Also, few studies have investigated the status of the expression of EG-VEGF receptors, PROKR1 and PROKR2 in CRC.
The present study was conducted to determineEG-VEGF, PROKR1 and PROKR2 expression in normal and tumor colorectal tissues. Both circulating and local levels were compared between control, CRC and PC patients.
Materials and methods
Blood collection
Collection and processing of human samples was approved by the university ethics committee, and informed consent was obtained from each patient. The consent authorizes the use of the blood and tissues for research purposes.
The study was restricted to patients, with histologically confirmed CRC and with PC as the only site of metastasis, with the ability to perform informed consent, for whom enough clinical documentation is available, with sampling at the primary CRC excision and with no previous chemotherapy for metastatic or advanced CRC.
Blood was collected into EDTA vacutainers and centrifuged at 5000 rpm for 10 minutes. Plasma was removed from the erythrocytes and stored at
Tumor biopsies collection
Colorectal specimens were obtained from 10 patients undergoing surgical resection of human advanced CRC. In addition 10 colorectal tissues from normal patients were used as positive control. The control specimens were obtained from patients who undergone regular colonoscopy and/or resection for benign colorectal diseases, such as hemorrhoidectomy. Shortly after collection, a fragment of each colorectal tissue, was fixed in 4% (vol/vol) paraformaldehyde for 24 h at room temperature, embedded in paraffin and cut in 5
EG-VEGF ELISA
EG-VEGF was measured by ELISA (Peprotech, Neuilly-sur-Seine, France) in the collected plasma. A total of 0.5
Demographic characteristics of all controls, colorectal (CRC) and peritoneal carcinomatosis (PC) patients
Demographic characteristics of all controls, colorectal (CRC) and peritoneal carcinomatosis (PC) patients
CRC, colorectal cancer; PC, peritoneal carcinomatosis; OR, Odds Ratio; CI, confidence interval; SD, standard deviation.
Clinicopathologic characteristics of colorectal cancer patients
Immunohistochemical staining was performed using the avidin-biotin-peroxidase method. Paraffin-embedded tissue Sections 5
Statistical analysis
Quantitative and qualitative variables were summarized in terms of median (range), mean (standard deviation), and frequency (percentage). Statistical comparisons between groups were made using chi-square or one-way ANOVA test. Calculations were performed using SPSS statistics version 21 (SPSS Inc., Chicago, IL, USA) and SigmaPlot (Jandel scientific software, San Rafael, CA). The differences were considered significant at
Results
Patient characteristics
The CRC was located in the colon in 32 patients (64%), and in 18 patients (36%) in the rectum. The mean ages (
The experimental groups consisted of 58 patients of both genders (CRC
Distribution of all control colorectal cancer (CRC) and peritoneal carcinomatosis (PC) patients by age groups.
EG-VEGF serum levels measured by ELISA in control (CTL), colorectal cancer (CRC) and peritoneal carcinomatosis (PC) patients. A total of 99 serum samples were analyzed (*
Considering the serum tumor Carcinoembryonic antigen (CEA) marker of our patients, we analyzed the relationship of its levels in correlation with the TNM stage of CRC. Positive correlation (
Of the 50 patients with CRC, 8 (16%) had liver metastasis as site of tumor dissemination. In the PC patients, no clinical or radiological evidence of other systemic spreading associated to PC was observed.
A) CEA circulating levels in relation to TNM stage of CRC. (*
EG-VEGF protein expression in healthy human colorectal mucosa and human primary colorectal cancer tissues. Expression was assessed by immunohistochemistry. Representative photographs show EG-VEGF immunoreactivity in normal human colorectal mucosa (A), in human primary colorectal cancer (B). Photograph in panel C reports a negative control. All sections were counterstained with heamatoxylin. TC: tumor cell, CL: crypts of lieberkùhn, MM: muscularis mucosae, LP: lamina propria, L: Lumen, GC: goblet cells, AE: absorptive erythrocytes. Magnification: A: x400, B and C: x200.
Immunohistochemical staining of PROKR1 in human colorectal tissues. Photographs show representative PROKR1 immunoreactivity in normal human colorectal mucosa (A), in human primary colorectal cancer (B) and a negative control (C). All sections were counterstained with Heamatoxylin. TC: tumor cell, CL: crypts of lieberkùhn, LP: lamina propria, GC: goblet cells, AE: absorptive erythrocytes, MM: muscularis mucosae, L: Lumen. Magnification: A: x400, B and C: x200.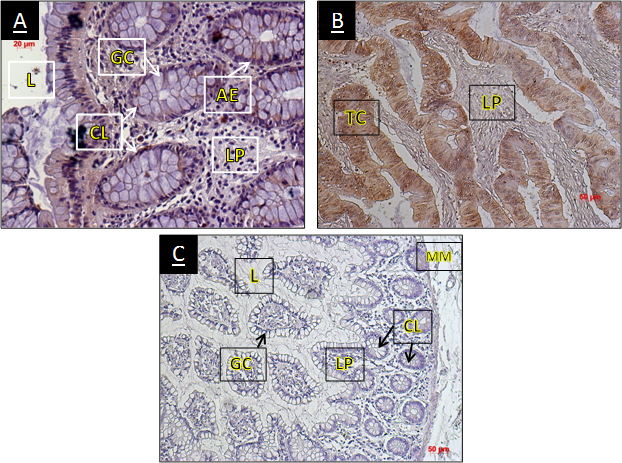
First, we compared EG-VEGF protein circulating levels in the control group and in the patient groups with CRC and PC, using ELISA test. Circulating EG-VEGF was detectable in all tested sera. There was a significant difference between control cases, CRC patients and PC patients (
EG-VEGF protein expression and clinicopathologic factors in CRC patients
Analysis of the relationship between EG-VEGF circulating levels in CRC patients and the clinicopathologic characteristics reported interesting correlations when considering the TNM classification of CRC. The expression of EG-VEGF was positively correlated with the tumor size (pT3, pT4) and nodal (N2) stage of the disease (Fig. 3B). These correlations were statistically significant (
Immunolocalization of EG-VEGF protein and its receptors PROKR1 and PROKR2 in colorectal cancer
Comparison of EG-VEGF protein expression in normal and colorectal cancer tissues
The EG-VEGF protein was strongly expressed in the primary lesion of CRC in comparison with normal colorectal tissue (Fig. 4A and B). Its expression in healthy human colorectal tissue was weak, heterogeneous and limited to the cytoplasm of tubular epithelial cells (crypts of lieberkùhn). The staining was detected in the cytoplasm of both the goblet and the absorptive cells. In the primary lesion of CRC, strong staining was observed in both the cell membrane and the cytoplasm, also we observed an expression in the nucleus of the lamina propria cells. The negative control did not show any staining. Representative photographs are reported in Fig. 4C.
Comparison of PROKR1 and PROKR2 protein expression in normal and colorectal cancer tissues
The expression of PROKR1 and PROKR2 receptors in normal and CRC tissues showed heterogeneous localizations in CRC cases compared to control tissues. PROKR1 was localized both in the nucleus and in the cytoplasm of tumor cells with weak expression in the lamina propria, in CRC cases (Fig. 5B). Overall, in positive controls, widespread cytoplasmic distribution of PROKR1 expression was observed in absorptive cells along with a weak expression in the cells of the lamina propria (Fig. 5A). Overall, the intensity and patterns of this distribution varied. No significant difference could be observed when comparing control and CRC sections (Fig. 5). In contrast, PROKR2 protein was localized predominantly, both at the cell membrane and in the cytoplasm of tumor cells in CRC lesions, and the intensity was stronger at the cell membrane compared to the cytoplasm (Fig. 6B). In positive control cases, PROKR2 expression was especially localized in the nuclei and the cytoplasm of lamina propria cells and erythrocytes (Fig. 6A). No staining was observed in all negative controls. PROKR2 immunoreactivity was significantly higher in the colorectal primary lesions, compared to the positive control sections (Fig. 6).
Comparison of PROKR2 immunohistochemical staining in normal and pathological colorectal mucosa. PROKR2 immunoreactivity was detected in normal human colorectal mucosa (A) and in human primary colorectal cancer (B). (C) Represent a negative control of the staining. All sections were counterstained with Hematoxylin. TC: tumor cell, CL: crypts of lieberkùhn, LP: lamina propria, GC: goblet cells, AE: absorptive erythrocytes. Magnification: A and C: x400, B: x200.
Prokineticins are a family of secreted peptides with diverse regulatory roles [29]. EG-VEGF, the canonical member of this family exhibits the same effects on endothelial cells as those described for VEGF, the classical angiogenic factor. Nevertheless, EG-VEGF is structurally different from VEGF and uses different types of receptors. According to previous studies, the expression of EG-VEGF in normal human tissue is limited to steroidogenic tissues hormone-producing cells; including, ovary, testis and placenta [30], and does not occur in normal gastrointestinal membranes such as the stomach and colon [7, 31]. This factor is strongly involved in normal and pathological angiogenesis [32] and plays an important role in the regulation of tumor cell growth and survival [4, 13, 33, 34].
In the present study, we searched for a potential new marker for the growth of CRC and its progression to PC. Recent studies suggested that EG-VEGF might be deregulated in this type of cancer without investigating its circulating levels in patient’s vs normal individuals. Our results confirmed EG-VEGF and PROKR2 deregulations in situ in the CRC and bring strong evidences of its increase in the blood stream of CRC and PC patients, suggesting its potential use as prognostic biomarker of the disease.
CRC is a major cause of cancer-related morbidity and mortality worldwide [31, 35]. Our results show that CRC typically occurs in middle-age to elderly patients. In addition, most affected individuals are aged between 40–60 years. Hence, we propose that circulatingEG-VEGF and its levels of expression and localization in CRC could be informative on its progression into PC.
In our study, EG-VEGF protein levels increased in CRC patients compared to the control group and were even higher in PC; this strongly supports a potential role of this factor in the processes that contributes to the progression and metastasis of CRC into PC. This results are concordant to previous finding of Nagano et al. who demonstrated that in situ EG-VEGF mRNA levels are correlated to the severity of CRC [28]. Importantly, EG-VEGF was also shown to increase the invasion of tumor cells, which is an indicator of cell malignancy and an important element for the development of hematogenous, lymph node and peritoneal metastasis [36].
We observed that the mean value of circulatingEG-VEGF was higher in patient’s age group of 41–50 years old, however among PC patients, the higher mean value of circulating EG-VEGF was observed in patients with age group 51–60 years. Tumor staging was by far the most important prognostic predictor of clinical outcome for patients with CRC. We found that the expression of EG-VEGF was increased in patients with high tumor size (pT3, pT4) and nodal (N2) stage of CRC, as well as in those with high CEA levels, which correlate with advanced tumor stage. In a recent study, the CEA levels were reported to correlate with the number of circulating tumor cells (CTCs) in CRC patients [37]. This relationship between the increase in the levels of EG-VEGF expression and the progression of the tumor, like the traditional tumor biomarker CEA, confirms and supports the idea that EG-VEGF could be considered to act as prognosis-determining factor in the progression of human CRC. In accordance with a previous study, tumor (T) stage, nodal stage and liver metastasis were considered independent predictors of metachronous colorectal PC [38]. Moreover, Nakazawa et al. [26], suggest that the recurrence of CRC was more frequent and the prognosis was worse for the stage T3 patients with positive lesions expressing EG-VEGF compared to stage T3 negative lesions. This indicates that EG-VEGF expression could be an important factor for the infiltration and metastasis of human CRC. In the same study, clinicopathologic examination confirmed that EG-VEGF expression frequently occurred in cases with vascular invasion and hematogenous metastasis, including liver metastasis [26].
Goi et al. defined EG-VEGF and its receptorPROKR2 as prognostic markers in colorectal carcinoma [39, 40]. Importantly, this group also demonstrated that synergistic anti-tumoral effects were obtained when both EG-VEGF and VEGF signaling pathways were targeted. These findings strongly suggest complementary actions between VEGF andEG-VEGF in CRC development and progression [41].
It was estimated that the peritoneal cavity was the main site of metastases in approximately 25% of patients with CRC [23]. Furthermore, multiple studies support the association between angiogenic factors and tumor malignancy [42]. Kim et al. [43] showed increased angiogenesis in tumors with rapid proliferation, whereas Folkman [3] reported an increase in the tumor volume due to the active angiogensis in tumor tissue, confirming that tumor growth is angiogenesis dependent.
Multiple studies reported the association ofEG-VEGF with malignant tumors. These studies reported that, i) increased EG-VEGF expression is associated with increased malignancy of prostate cancer, neuroblastoma, thyroid cancer and pancreatic duct cancer, ii) EG-VEGF mRNA expression in the resected primary lesion of CRC patients is associated with a worse prognosis, and that iii) increasedEG-VEGF expression is associated with angiogenesis involving hepatic metastasis [14, 36, 44]. Hence, these studies strongly suggest that EG-VEGF must be considered as a new central actor of cancer development and progression.
PC from CRC was considered as a terminal condition resulting from tumor with median survival not exceeding six months [38]. Due to the low sensitivity of preoperative imaging techniques, the exact incidence of PC is unknown [45, 46]. Jayne et al. [38] reported that 8% from a total of 3019 CRC patients presented a synchronous PC and 5% with metachronous PC. In addition, a recent study in Sweden by Segelman et al. [47] confirmed that 4.3% of 11124 CRC patients presented with synchronous PC, while 4% with metachronous PC.
The immunolocalization experiments performed in this study revealed interesting patterns of distribution of the EG-VEGF and its two receptors PROKR1 and PROKR2 in CRC sections. This particular aspect strongly suggests the involvement of EG-VEGF as a potential prognostic factor in CRC. EG-VEGF, PROKR1 and PROKR2 were slightly expressed in normal colorectal mucosa, and highly expressed in CRC cells, indicating that EG-VEGF/receptors systems could be considered as new targets to treat CRC. Previous study evaluating the expression of PROKR2, reported that the prevalence of PROKR2 expression increased as tumor stage advances. This study suggested that PROKR2 could be used as a prognostic factor in human CRC.
Metastasis is the strongest risk factor for mortality among tumors malignancies. Understanding the mechanism of such processes may lead to new therapies of cancer. Many discoveries suggest that prokineticins, expressed at higher levels in cancers compared to normal tissues, may be cancer specific and, possibly, tissue specific targets. These aspects could open new frames for cancer-specific therapies.
Conclusion
Our results support the use of EG-VEGF and its rec- eptor PROKR2 as predictor and prognosis-determining factors of the progression of the CRC into PC. These proteins could also, represent therapeutic targets in the pathogenesis of this cancer.
Limits of the study
Because no previous study has addressed the comparison of EG-VEGF circulating levels in colorectal cancer, the present study was conducted with the main objective to measure and compare circulating EG-VEGF levels in control vs CRC and PC patients. In situ comparison of the levels of EG-VEGF, PROKR1 and PROKR2 expression was also investigated. While, EG-VEGF measurement at the circulating level were conclusive and brought strong informative new message, in situ analyses will need more sampling to better quantify the local levels of expression of the prokineticin system. A large cohort including, sera, mRNA and protein specimens collected in CRC and PC patients is under construction to validate these results at larger scale.
Footnotes
Acknowledgments
We acknowledge the following sources of funding: Institut National de la Santé et de la Recherche Médicale (U1036), University Grenoble-Alpes, Commissariat à l’Energie Atomique (DSV/iRTSV/BCI), University Hassan II of Casablanca, Faculty of Medicine and Pharmacy of Casablanca, and the National Center for Scientific and Technical Research (CNRST) of Rabat.
Conflict of interest
The authors declare no potential conflicts of interest.
Abbreviations
EG-VEGF: Endocrine Gland-Derived Vascular Endothelial Growth Factor. PROK: Prokineticin. CRC: Colorectal Cancer. PC: Peritoneal Carcinomatosis.CEA: Carcinoembryonic Antigen.
