Abstract
BACKGROUND:
Long non-coding RNA testis-specific transcript, Y-linked 15 (TTTY15) is oncogenic in prostate cancer, however its expression and function in colorectal cancer remain largely unknown.
METHODS:
Paired colorectal cancer samples/normal tissues were collected, and the expression levels of TTTY15, miR-29a-3p and disheveled segment polarity protein 3 (DVL3) were examined by quantitative real-time polymerase chain reaction (qRT-PCR); TTTY15 shRNA and overexpression plasmids were transfected into HT29 and HCT-116 cell lines using lipofectamine reagent, respectively; the proliferation and colony formation were detected by CCK-8 assay and plate colony formation assay; qRT-PCR and Western blot were used to analyze the changes of miR-29a-3p and DVL3; dual-luciferase reporter gene assay was used to determine the regulatory relationships between miR-29a-3p and TTTY15, miR-29a-3p and DVL3.
RESULTS:
TTTY15 was significantly up-regulated in cancerous tissues of colorectal cancer samples, positively correlated with the expression of DVL3, while negatively correlated with the expression of miR-29a-3p. After TTTY15 shRNAs were transfected into colorectal cancer cells, the proliferation and metastasis of cancer cells were significantly inhibited, while TTTY15 overexpression had opposite biological effects. TTTY15 shRNA could reduce the expression of DVL3 on both mRNA and protein levels, and the luciferase activity of TTTY15 sequence was also inhibited by miR-29a-3p. DVL3 was also validated as a target gene of miR-29a-3p, and it could be repressed by miR-29a-3p mimics or TTTY15 shRNA.
CONCLUSION:
TTTY15 is abnormally upregulated in colorectal cancer tissues, and it can modulate the proliferation and metastasis of colorectal cancer cells. It functions as the ceRNA to regulate the expression of DVL3 by sponging miR-29a-3p.
Introduction
Colorectal cancer (CRC) is a common malignancy of digestive tract. The morbidity of CRC ranks third among cancers, and the mortality of CRC ranks fifth [1, 2]. At present, the treatment approaches of CRC are limited. The main methods are surgery and chemotherapy, but postoperative recurrence and metastasis occurs in 40%–50% cases [3, 4, 5]. The survival rate and quality of life of patients with CRC are needed to be further improved. Exploring the mechanisms of the carcinogenesis and progression of CRC is important for improving the prognosis of CRC patients.
Long non-coding RNA (lncRNA) is a class of RNA molecules that are more than 200 nucleotides in length but cannot encode proteins. It regulates gene expression at the transcriptional, post-transcriptional, and epigenetic levels, thereby affecting cell differentiation, metabolism, proliferation and apoptosis [6]. In the development of some human cancers, the expression of genes on the Y chromosome plays an important role [7, 8, 9, 10]. LncRNA testis-specific transcript, Y-linked 15 (TTTY15) is located in the AZFa region of the Y chromosome, and a recent study shows that the expression of TTTY15 is up-regulated in prostate cancer tissues, and it can drive the progression of prostate cancer tissues by sponging let-7 [11]. Nevertheless, the expression and function of TTTY15 in CRC are much less explored.
MicroRNA (miRNA) is a kind of non-coding RNA (ncRNA) of about 22 nucleotides in length [12]. Reportedly, the dysregulation of miR-29a-3p is related to the carcinogenesis and progression of diverse cancers. It is down-regulated in liver cancer, and its low expression is associated with the lower survival rate of patients; moreover, overexpression of miR-29a-3p can inhibit the proliferation and migration of liver cancer cells by targeting IGF1R [13]. It is also significantly down-regulated in gastric cancer and its low expression facilitates the proliferation, migration and invasion of cancer cells [14]. There are also some studies reporting the expression, biological function and mechanism of miR-29a-3p in CRC [15, 16], but its upstream and downstream molecular mechanisms still need to be further studied.
Disheveled (DVL), a positive regulator of the Wnt signaling pathway, is located upstream of
This study mainly explored the expression and function of TTTY15 in CRC, and its regulatory function on miR-29a-3p/DVL3 axis in CRC. This work provides useful clues for the diagnosis and treatment of CRC.
Materials and methods
Tissue specimens
The tumor tissues and normal tissues of 51 male patients who underwent radical resection of CRC at Qinghai University Affiliated Hospital from April 2017 to April 2018 were randomly collected. None of patients received chemotherapy or radiotherapy before surgery. The control group specimens were resected from normal tissues of the same patient (at least 2 cm from the surgical margin), and no cancer cells were found during pathological examination. The collection and use of patient tissue samples were approved by the Qinghai University Affiliated Hospital Ethics Committee. All patients involved gave informed consent. Tissue samples were surgically removed and immediately stored at
Cell culture
Human CRC cell line (HCT-116, HT29, SW480, SW620, DLD-1 cells) and normal colonic epithelial cell line CCD841 were purchased from the Institute of Biochemistry and Cell Biology of the Chinese Academy of Sciences (Shanghai, China). The cells were cultured in RPMI-1640 medium (Gibco, Carlsbad, CA, USA) containing 10% fetal bovine serum (FBS; Gibco, Carlsbad, CA, USA) and 100 U/ml penicillin, 100
Cell transfection
The cells in the logarithmic growth phase were inoculated into a 6-well plate with 2.5
RNA isolation and qRT-PCR
Total RNA was extracted from tissues and cell lines according to the instructions of TRIzol reagent (Invitrogen, Carlsbad, USA). RNA purity and concentration were measured using the NanoDrop ND-2000 spectrophotometer. With M-MLV Reverse Transcriptase (Thermo Fisher Scientific, Inc., Rockford, IL, USA), 5
Primers used in this study
Primers used in this study
The cell proliferation curve was determined using CCK-8 assay. HCT-116 and SW480 cells (3000 cells per well) were seeded in 96-well plates and incubated for 1, 2, 3, and 4 d, respectively. Subsequently, 10
Plate colony formation assay
Cells in the logarithmic growth phase were trypsini-zed with 0.25% trypsin and dispersed into single cells and counted. The cell suspension was inoculated separately into dishes containing 3 mL culture solution (1000 cells per dish). Subsequently, the cells were cultured in an environment of 37
Transwell assay
Transwell assay was used to detect the migration and invasion, with Transwell chambers (Corning, NY, USA). Matrigel (BD Biosciences, Franklin Lakes, NJ, USA) was used in invasion assay, but not in migration assay. The cells in each group transfected for 48 h were taken, trypsinized, resuspended with serum-free medium, counted and seeded into the upper chamber. RPMI-1640 medium containing 10% FBS was added to the lower chamber. The cells were then cultured for 24 h. After that, the cells that failed to migrate or invade were removed. After the migrated or invaded cells being fixed for 4 minutes with 4% paraformaldehyde and stained with 0.5% crystal violet, the cells were rinsed by the tap water, and the cells were observed under an inverted microscope. Cell counting was performed using five fields of view, and the mean value was taken.
Flow cytometry
Cellular apoptosis was detected by flow cytometry. Cells in the logarithmic growth phase were washed twice with PBS, fixed in 70% ethanol, and stored at 4
Expression of TTTY15 in CRC tissues and cell lines. A. The qRT-PCR assay was employed to detect the expression of TTTY15 in 51 pairs of CRC tissues and paracancerous tissues. B. qRT-PCR assay was employed to detect the expression of TTTY15 in CRC cell lines (HCT-116, HT29, SW480, SW620, DLD-1 cells) and normal colorectal epithelial cell line CCD841. (Compared with Normal group or CCD841, * indicates 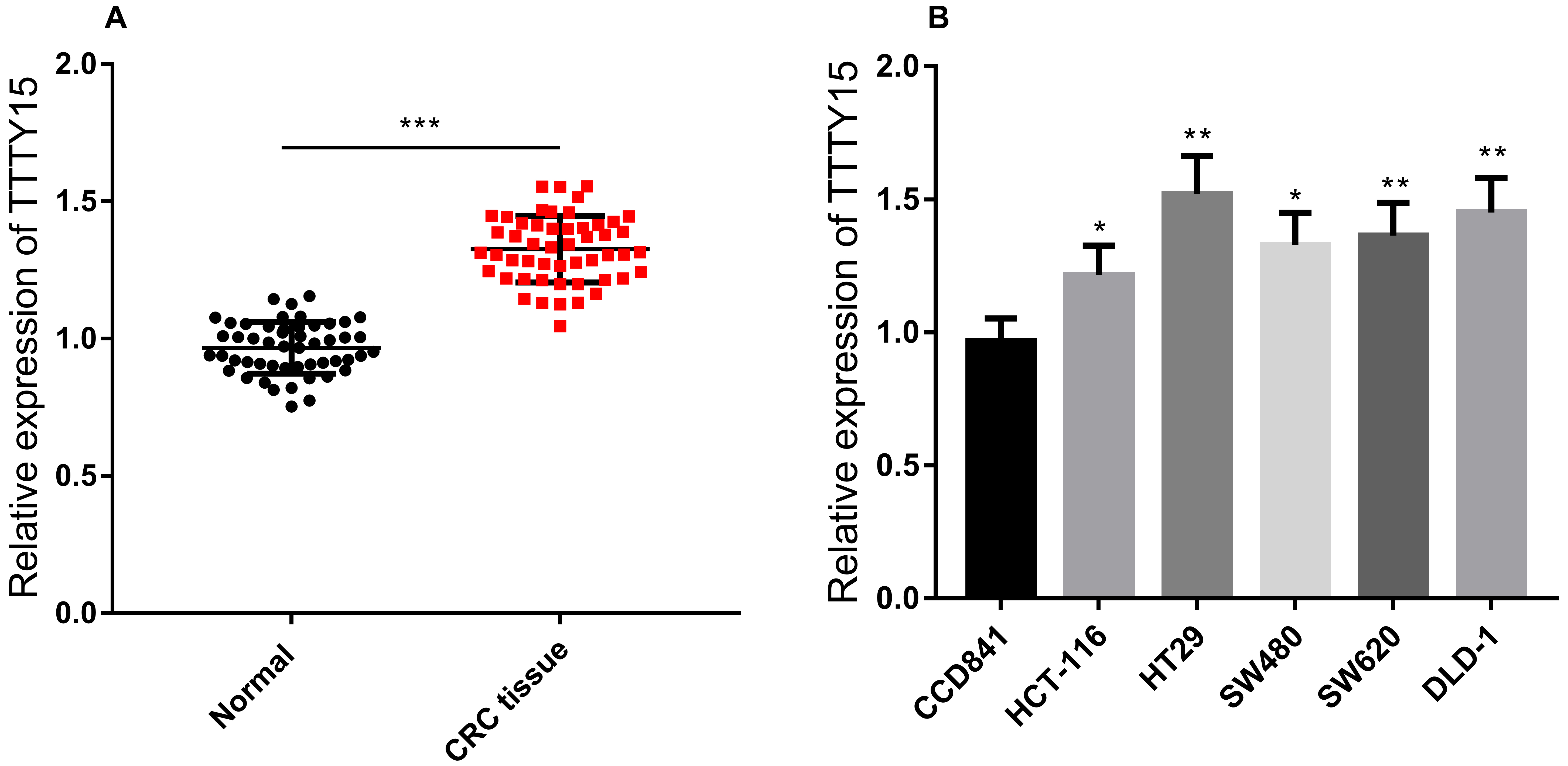
Luciferase reporter gene assay was performed using the dual-luciferase reporter assay system (Promega, Madison, WI, USA). The target fragments of wild type TTTY15 and mutant TTTY15 were constructed and integrated into pGL3 vector (Promega, Madison, WI, USA) to construct pGL3-TTTY15-wild type (TTTY15-wt) and pGL3-TTTY15-mutant (TTTY15-mut) reporter vectors. Then TTTY15-wt or TTTY15-mut reporter vector was co-transfected into HT29 cells with miR-29a-3p mimics or control miRNA mimics. After 48 h of transfection, luciferase activity was determined in accordance with the manufacturer’s instructions. The same method was used to construct DVL3-wt and DVL3-mut, and to detect the targeted binding relationship between miR-29a-3p and the 3’UTR of DVL3.
Western blot
RIPA lysis buffer (Beyotime Biotechnology, Beijing, China) containing PMSF was used to extract the protein from CRC cells. Protein samples were subjected for SDS-PAGE and transferred to the nitrocellulose (NC) membrane (Millipore, Billerica, MA, USA). After being blocked with 5% fat-free milk, the membrane was incubated with primary antibodies anti-DVL3, anti-
Statistical analysis
All experiments were performed in triplicate and repeated three times. Data analysis was performed using SPSS18.0 statistical software (SPSS Inc., Chicago, IL, USA). All data were expressed as mean
Result
TTTY15 was highly expressed in CRC tissues and cell lines
In order to explore the expression of TTTY15 in CRC, qRT-PCR assay was employed to detect TTTY15 expression in CRC tissues, normal tissues and CRC cell lines (HCT-116, HT29, SW480, SW620, DLD-1 cells) and normal colorectal epithelial cell line (CCD841 cells). The results revealed that TTTY15 was significantly up-regulated in CRC tissues and cell lines compared non-cancerous tissues and cells (Fig. 1A and B). To explore the significance of high expression of TTTY15 in CRC, we divided the patients into high-expression and low-expression groups according to the expression level of TTTY15. The results implied that the high expression of TTTY15 was significantly associated with lymph node metastasis in CRC (Table 2).
The correlations between TTTY15 expression and Clinicopathologic factors
The correlations between TTTY15 expression and Clinicopathologic factors
Next, we overexpressed TTTY15 in HCT-116 cell line and knocked down TTTY15 in HT29 cell line (Fig. 2A). The cell proliferation was examined by CCK-8 assay and plate colony formation assay. The results showed that the proliferation of HCT-116 cells was significantly increased after TTTY15 overexpression, and the proliferation of HT29 cells was decreased after TTTY15 knockdown (Fig. 2B–D). In addition, the migration and invasion were detected by Transwell assay, and the results showed that the migration and invasion of CRC cells were increased after overexpression of TTTY15; consistently, there was also a decrease in the invasion and migration of HT29 cells after knockdown of TTTY15 (Fig. 2E and F). After that, flow cytometry was employed to detect cell apoptosis. The results indicated that the apoptosis of HCT-116 cells was significantly decreased after TTTY15 overexpression, while the apoptosis of HT29 cells was increased after TTTY15 knockdown (Fig. 2G).
TTTY15 promotes the proliferation, invasion and migration of CRC cells and inhibits the apoptosis. A. The expression levels of TTTY15 in HCT-116 and HT29 cells after overexpression and knockdown of TTTY15 were detected by qRT-PCR. B–D. CCK-8 assay (B) and plate colony formation assay (C, D) were employed to detect the proliferation of TTTY15 overexpression and knockdown cells. E and F. The Transwell assay was employed to detect the migration and invasion of CRC cells after overexpression and knockdown of TTTY15. Flow cytometry was used to detect CRC cell apoptosis after TTTY15 overexpression and knockdown. (Compared with NC 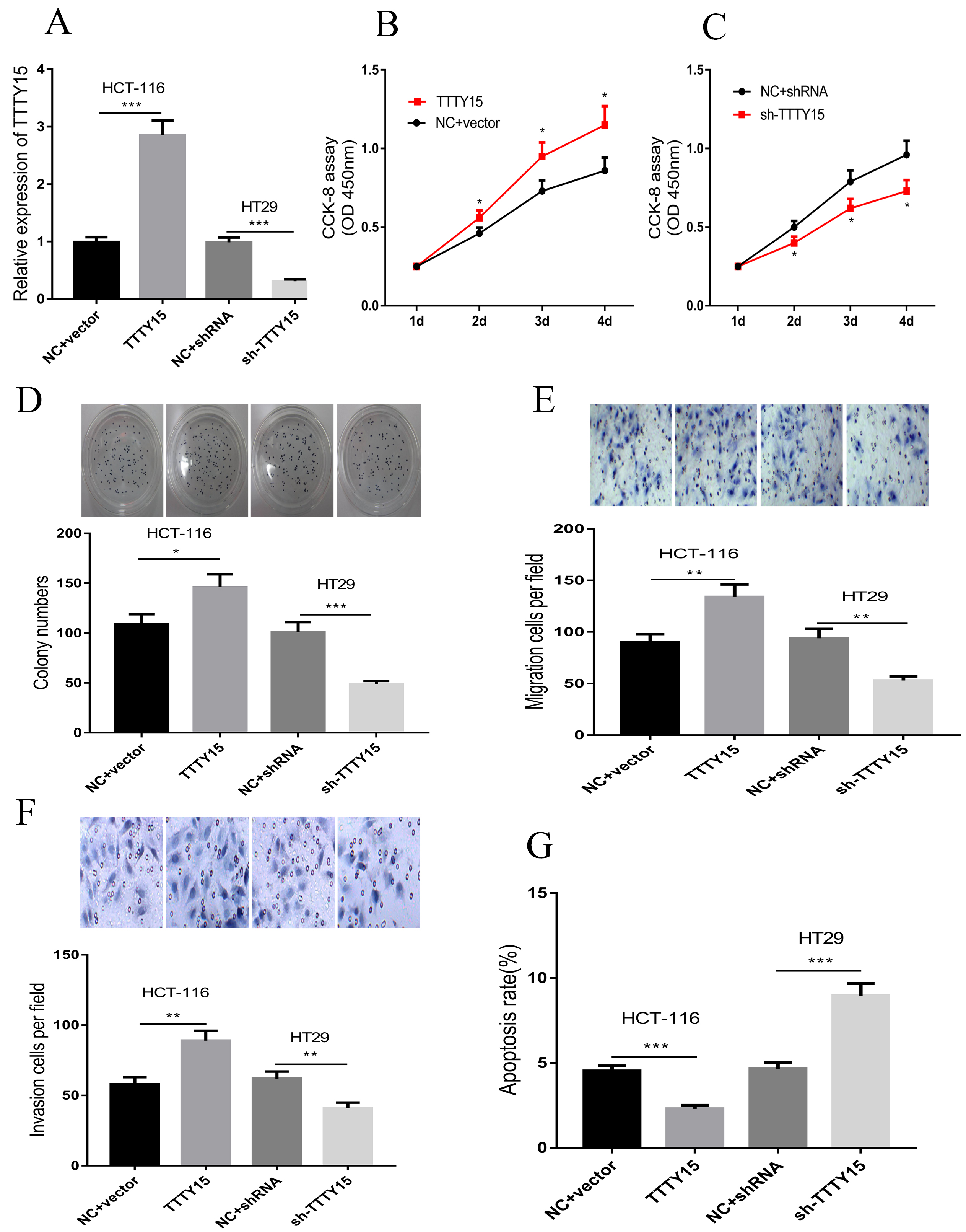
To further explore the downstream molecular mechanisms of TTTY15, we conducted a bioinformatics analysis by querying the LncBase Predicted v.2 database (
TTTY15 can directly bind to miR-29a-3p to promote the progression of CRC. A. It was predicted that there were two potential binding sites between TTTY15 and miR-29a-3p. B. The binding relationship between TTTY15 and miR-29a-3p mimics was verified by luciferase gene reporter assay. C. The expression of TTTY15 and miR-29a-3p in 51 groups of CRC tissues was detected by qRT-PCR, and the results showed that their expressions were negatively correlated. D. qRT-PCR assay was employed to detect the effect of overexpression and knockdown of TTTY15 on miR-29a-3p expression. E. The effect of overexpression and knock-down of miR-29a-3p on the expression of TTTY15 was detected by qRT-PCR assay. (Compared with control group/NC 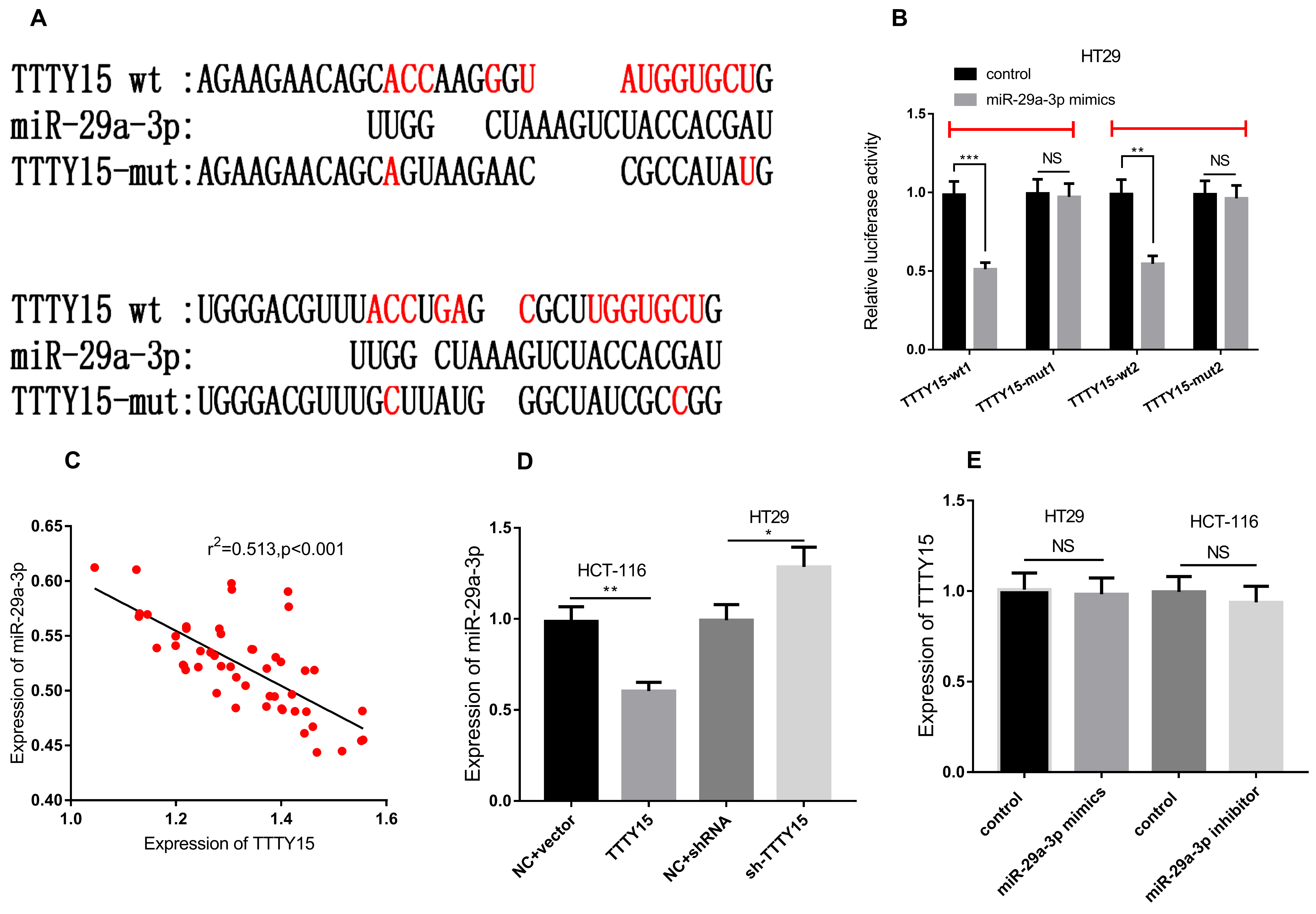
miR-29a-3p targeted and downregulated DVL3 in CRC. A. By querying the Starbase database, it was predicted that there was a potential binding site between miR-29a-3p and the 3’UTR of DVL3. B. The binding relationship between miR-29a-3P and DVL3 was verified by luciferase gene reporter assay. C–D. The expressions of TTTY15, miR-29a-3p and DVL3 in 51 cases of CRC tissues were detected by qRT-PCR, and the correlations between miR-29a-3p and DVL3, TTTY15 and DVL3 were analyzed. E–L. The effects of miR-29a-3p and TTTY15 expression on expression level of DVL3, 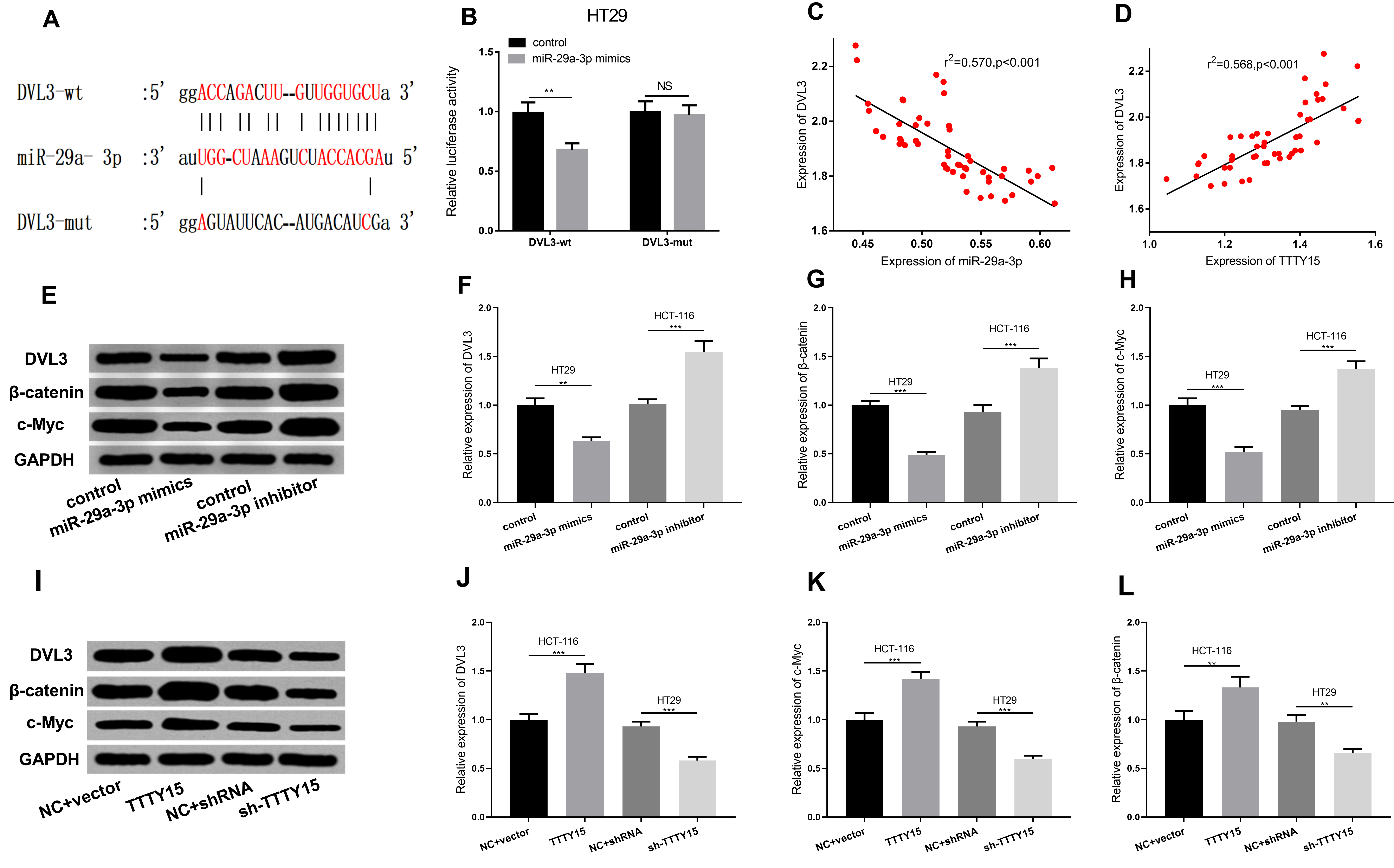
Recently, DVL3 was a direct target of the miR-29 family [22]. By querying StarBase database (
TTTY15 knockdown repressed CRC cell viability, migration, and invasion but induced cell apoptosis via regulating miR-29a-3p
Based on the above findings, we made a hypothesis that there could be a TTTY15/miR-29a-3p/DVL3 axis during CRC progression, so we then constructed TTTY15 knockdown cells and TTTY15 shRNA/miR-29a-3p inhibitor co-transfection cells with HT29 cells. The expression of TTTY15, miR-29a-3p, DVL3,
TTTY15 knockdown repressed CRC cell viability, migration, and invasion but induced cell apoptosis via regulating miR-29a-3p. A–F. The expressions of TTTY15, miR-29a-3p, DVL3, 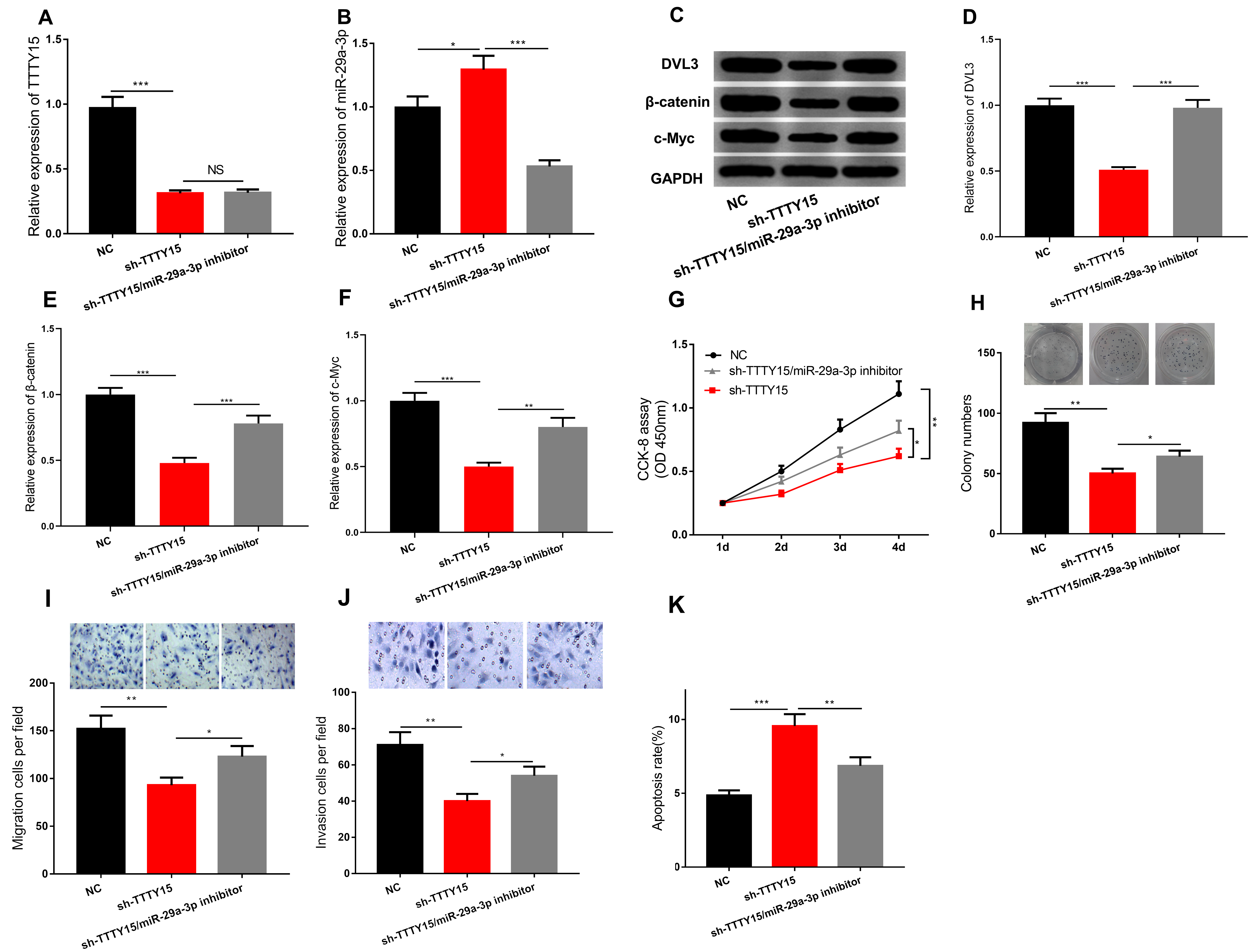
CRC is a common malignancy. Because early symptoms are not obvious, a large number of patients have metastatic disease when they are diagnosed and lose the opportunity for surgical treatment. The main therapeutic methods for patients with metastatic CRC are chemotherapy, and molecular targeting therapy, but the therapeutic effect is still not satisfactory [23, 24]. Exploring the molecular mechanism of CRC and finding feasible molecular therapeutic targets are important for improving patient survival.
Male and female have different susceptibility to different cancers, and their clinical outcomes and prognosis are different. Y chromosome has a specific effect on male’s development, physiology and disease [25]. It has been reported that the morbidity of CRC in males is significantly higher than that of females, which is associated with higher levels of estrogen in females [26]. Nonetheless, the impact of changes in gene expression on the Y chromosome on the development of CRC remains largely unknown. TTTY15 is located on the Y chromosome. It has been reported that the expression of TTTY15 is up-regulated in myocardial infarction; down-regulation of TTTY15 expression can inhibit hypoxia-induced cardiomyocyte apoptosis [27]. It has also been reported that the expression of TTTY15 is closely related to the carcinogenesis of prostate cancer: it can be fused with USP9Y, and TTTY15-USP9Y fusion is confirmed as a biomarker for early detection of prostate cancer [28]; TTTY15 is up-regulated in PC tissues, and down-regulation of its expression can inhibit the development of tumors [11]. Our study, for the first time, explored the expression and role of TTTY15 in CRC, and confirmed its oncogenic effect. To some extent, the gender differences in CRC are explained, providing a new therapeutic target for the treatment of this disease.
Accumulating reports imply that multiple miRNAs are dysregulated in CRC and function as a tumor suppressors or oncogenic factors. For instance, miR-296 is down-regulated in CRC tissues and cells, and its low expression can enhance the invasion and migration of CRC cells, and miR-296 can target S100A4 to inhibit epithelial-mesenchymal transition (EMT) of CRC cells [29]; miR-18b expression is remarkably increased in CRC tissues, and its overexpression promotes cancer cell proliferation by facilitating cell cycle progression by targeting CDKN2B [30]. In recent years, several studies have found that lncRNAs can regulate miRNA expression levels by sponging it and thus affect tumor progression [31, 32]. To explore the downstream molecular mechanism of TTTY15 in CRC, we identified the binding relationship between TTTY15 and miR-29a-3p. It should be noted that the role of miR-29a-3p in CRC has also been reported. Down-regulation of miR-29a-3p can promote the invasion and migration of CRC cells by regulating the expression of CDC42BPA [16]. We observed that in CRC tissues, the expression of TTTY15 and miR-29a-3p was notably negatively correlated, and it was found that the expression of miR-29a-3p was significantly increased after knocking down TTTY15; the expression of miR-29a-3p was significantly decreased after overexpression of TTTY15. These data not only explained the mechanism by which TTTY15 participated in CRC progression, but also helped clarify the reason of dysregulation of miR-29a-3p in CRC.
Additionally, previous studies have reported that in the osteoarthritis, the miR-29 family can negatively regulate the signaling pathways of Wnt, Smad, and NF-
Conclusion
In summary, TTTY15 expression is significantly up-regulated in CRC, and can enhance DVL3 expression via regulating miR-29a-3p and facilitate the development of CRC.The results of this study further deepen the understanding of the molecular mechanisms of CRC and provide new clues for its treatment.
Footnotes
Conflict of interest
The authors declare that they have no competing interest.
