Abstract
OBJECTIVE:
Bone mesenchymal stem cells (BMSCs) have been widely researched in cancer treatment, including hepatocellular carcinoma (HCC). This study intended to discuss the mechanism of miR-20a-3p in BMSCs-extracellular vesicles (EVs) in HCC apoptosis.
METHODS:
BMSCs were isolated and identified. EVs derived from BMSCs were extracted and identified. After overexpressing or inhibiting miR-20a-3p expression in BMSCs, EVs were extracted and acted on HCC cells and transplanted tumors. HCC cell apoptosis in the treatment of BMSCs-conditioned medium, BMSCs-EVs and/or miR-20a-3p mimic/inhibitor was evaluated, with the detection of levels of TRAIL and TRAIL-related proteins. A functional rescue experiment about c-FLIP was carried out in HCC cells. The target binding relationship between miR-20a-3p and c-FLIP was detected. The subcutaneous tumorigenesis model of mice was established and injected with BMSCs-EVs to estimate the effect of BMSCs-EVs-miR-20a-3p on HCC growth.
RESULTS:
EVs isolated from BMSCs conditioned medium promoted the apoptosis of HCC cells. After BMSCs-EVs treatment, TRAIL levels, downstream proteins and miR-20a-3p were increased significantly, but the expression of c-FLIP was decreased. miR-20a-3p could target c-FLIP. BMSCs-EVs inhibited the growth of HCC cells, decreased c-FLIP expression, increased TRAIL levels, and promote the of HCC cell apoptosis. BMSCs-EVs with overexpressing miR-20a-3p further enhanced the apoptotic effect of HCC cells in vitro and in vivo.
CONCLUSION:
BMSCs-EVs-carried miR-20a-3p targets c-FLIP and increases TRAIL levels in HCC cells, thus promoting TRAIL-related apoptosis.
Keywords
Introduction
Hepatocellular carcinoma (HCC) ranks the sixth most diagnosed primary cancer in the world and the third leading cause for cancer related mortality [1]. It is estimated that the global incidence and related deaths of HCC are rising over the last decades partially due to a growing number of obesity [2, 3]. The major risk factors for HCC are chronic infections with hepatitis B virus and hepatitis C virus, together with alcohol consumption, metabolic syndromes, obesity, diabetes and frequent exposure to aflatoxin [4]. The surgical intervention (excision or transplantation), radio-frequency catheter ablation and percutaneous ethanol injection are potentially effective for about 30
Mesenchymal stem cells (MSCs) are the main components of tumor microenvironment, and play an important role in the progress and metastasis of liver cancer [7]. A previous study has suggested that the communication between MSCs and cancer cells may be a potential therapeutic target to inhibit the growth of cancer cells [7]. Bone marrow MSCs (BMSCs) play a regulatory role in a variety of diseases and tumors [8]. However, the research on BMSC mechanism in HCC is limited, so we choose to explore the role of BMSCs in HCC. The expression of miR-20a-3p in human umbilical cord MSCs is high [9], while the expression of miR-20a-3p in HCC is low [10]. Therefore, we chose miR-20a-3p to study. MSCs could secrete several kinds of extracellular vesicles (EVs) to maintain tissue homeostasis for optimal tissue function [11]. EVs have profound impacts on normal physiology and pathological processes, and EVs contain a broad range of nucleic acids such as mRNA and small non-coding RNAs [12]. EVs derived from BMSCs have great promise to treat neurological and neurodegenerative conditions due to their anti-inflammatory and neuroprotective properties [13]. In addition, as an important member of tumor necrosis factor (TNF) family, TNF-related apoptosis-inducing ligand (TRAIL) is an encouraging anticancer protein, which can specifically kill diverse cancerous cells without damaging the normal ones [14]. HCC cells exhibit major resistance to TRAIL-induced apoptosis [15]. TRAIL is an antitumor protein and is considered as a potential target in HCC due to its promotion of apoptosis in HCC cells [16]. In the combination with chemotherapeutic drugs, like doxorubicin and 5-fluorouracil, TRAIL serves an effective approach for HCC treatment [17]. From all above, we make a hypothesis that there may be underlying mechanisms of TRAIL, miR-20a-3p and BMSCs-EVs in HCC progression. In this regards, we aimed to explore the therapeutic efficiency of BMSCs-EVs in HCC.
Materials and methods
Ethics statement
This study was performed with the approval of the Clinical Ethical Committee of Chengdu First People’s Hospital. Informed consent was received from all subjects. All procedures were strictly conducted in accordance with the code of ethics. Significant efforts were made to minimize the animals used and their suffering.
Isolation and identification of BMSCs
As previously described [18], the primary BMSCs were obtained from the bone marrow of healthy donors. Then cells were cultured in Dulbecco’s modified Eagle medium (DMEM) supplemented with antibiotics (100 U/mL penicillin and 100 U/mL streptomycin) and 10% fetal bovine serum (FBS) (Thermo Fisher Scientific Inc., Waltham, MA, USA) at 37
BMSCs of the 3
BMSCs of the third generation were cultured in DMEM with 10% FBS to 80% fused monolayer cells. BMSCs were cultivated with 1
Extraction and identification of BMSCs-EVs
BMSCs were seeded into 6-well plates and were adhered for 24 hours. BMSCs were washed with serum-free
BMSCs-EVs were isolated from CM by differential ultracentrifugation (according to the size and density of particles, the centrifugation force is increased by stages). Then, BMSCs-EVs were identified through nano particle tracking analysis, transmission electron microscope and western blotting to detected the expression of CD63 (1: 1000, ab59479, Abcam), CD81 (1: 1000, ab79559, Abcam), tumor susceptibility gene 101 (TSG101) (1: 1000, ab30871, Abcam), with CM added with EV-targeting phospholipase inhibitor GW4869 (CM-GW, MedChemExpress, LLC, NJ, USA) as the control [19]. The protein content of EVs was determined and standardized by the bicinchoninic acid (BCA) kit (Thermo Fisher).
miR-20a-3p mimic, miR-negative control (NC) and miR-20a-3p inhibitor synthesized by Shanghai Genechem Co., Ltd. (Thermo Fisher) were transfected into BMSCs according to Lipofectamine
HCC cell culture and treatment
HCC cell lines Huh7 (from human male liver tumor), HLcz01 (from liver tumor of a human male with chronic hepatitis C), SMMC-7721 (from human male liver tumor) and LH-86 (from human male liver tumor) were all purchased from American type culture collection (ATCC, Manassas, Virginia, USA), and cultured in DMEM with 1% penicillin and streptomycin and 10% FBS at 37
HCC cells were assigned into blank group (without any treatment), CM group (cultured with CM), CM-GW group (cultured with CM-GW), BMSCs-EVs group (cultured with 5
3-(4,5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) assay
Cells were seeded into 96-well plates (5
Flow cytometry
Cells were seeded into a 12-well plate (8
Western blot analysis
The total proteins were extracted from cell and tissue homogenates and protein concentration was determined based on the instructions of BCA kit (Thermo Fisher). After that, the proteins were added with loading buffer and boiled at 95
Reverse transcription quantitative polymerase chain reaction (RT-qPCR)
Total RNA in cells was extracted using one-step method of TRIzol (Invitrogen). Then, the extracted high-quality RNA was verified by ultraviolet analysis and formaldehyde deformation electrophoresis. Fluorescent qPCR was performed on the instructions of RT-qPCR kit (Thermo Fisher). All primers of RT-qPCR (Table 1) were designed and synthesized by Sangon Biotech Co., Ltd. (Shanghai, China), with glyceraldehyde-3-phosphate dehydrogenase or U6 as the internal reference. After the reaction, curves of amplification and dissolution were identified, and the 2
Primer sequences of RT-qPCR
Primer sequences of RT-qPCR
Note: RT-qPCR, reverse transcription quantitative polymerase chain reaction; TRAIL, tumor necrosis factor-related apoptosis-inducing ligand; miR, microRNA; GAPDH, glyceraldehyde-3-phosphate dehydrogenase; F, forward; R, reverse.
The c-FLIP fragments containing binding sites with miR-20a-3p were cloned to pmirGLO bisoligosaccharide enzyme vector (Promega Corp., Madison, Wisconsin, USA) to construct pmirGLO-c-FLIP-wild type (WT) plasmids. Identical c-FLIP fragments except the mutant ones of miR-20a-3p binding sites were used to construct pmirGLO-c-FLIP-mutant type (MUT). These two kinds of plasmids were respectively transfected with 293T cells (ATCC) and then with miR-20a-3p mimic or miR-NC. Consequently, luciferase activity was determined via dual luciferase reporter assay system (Promega) after 48 hours, with the ratio of relative activity as luciferase activity of firefly to that of renilla luciferase measured.
Tumor xenograft in nude mice
LH-86 cells collected by centrifugation were re-suspended. Each nude mouse (Nanjing University Institute of model animal research, Nanjing, China) was subcutaneously injected with 20
TUNEL staining
Sections were treated and stained in TUNEL reaction buffer. Then, each section was stained using 4’,6-diamidino-2-phenylindole, and added with fluorescence quencher. The pictures were captured by a fluorescence microscope (BX51, Olympus) within 24 h. Apoptotic cells were calculated and analyzed using Image J software.
Statistical analysis
Statistical analysis was conducted by SPSS 21.0 (IBM Corp. Armonk, NY, USA). All the data were in normality distribution checked by the Kolmogorov-Smirnov test. Measurement data were expressed as mean
Identification of BMSCs. A, BMSCs isolated from the bone marrow of healthy volunteers, left: BMSCs cultured for 1 day, right: BMSCs cultured for 5–6 days; B, flow cytometry screening of cell surface antigens in the third generation of BMSCs showed that CD29 and CD90 were positive, while CD34 was negative; C, Alizarin red staining showed BMSCs had osteogenic differentiation ability; D, oil red O staining showed BMSCs had lipogenic differentiation ability. Replicates 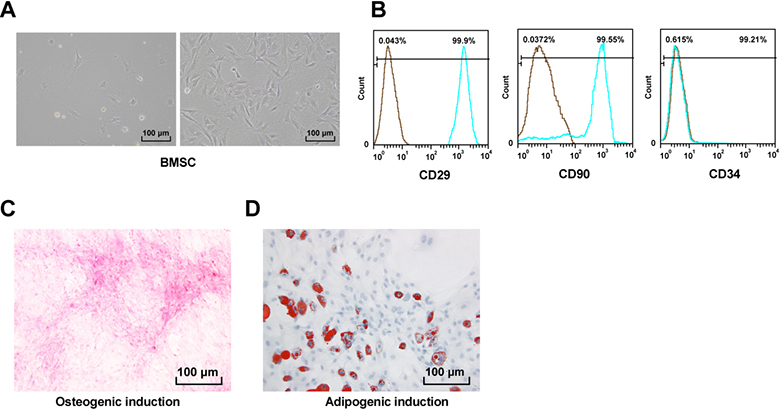
Identification of BMSCs
The primary BMSCs were in spindle-shaped adherence. After 5–6 days, BMSCs grew rapidly and reached 80% confluence in a whirlpool (Fig. 1A). Then primary cells were cultured around 14 days, and remained relatively stable after long-term passage to 20
BMSCs-EVs promote HCC cell apoptosis
To explore the role of BMSCs in the apoptosis of HCC cells, we first applied the CM of BMSCs to Huh7, HLCZ01, SMMC-7721 and LH-86 cells, and through flow cytometry we found the apoptosis rate was significantly increased after culture in CM (
BMSCs-EVs promote HCC cell apoptosis. A/G, flow cytometry showed that the apoptosis rate of HCC cells was increased significantly after BMSCs-CM treatment, while the CM with GW4869 (CM-GW) had no significant effect on promoting apoptosis (A); B, western blot analysis detected the protein levels of Bcl-2 and Bax; C, the morphology of BMSCs-EVs was observed by transmission electron microscope; D, the concentration and size of BMSCs-EVs were analyzed by nanoparticles; E, western blot analysis detected the expression of BMSCs-EVs specific markers, with CM-GW as the control; F, MTT detected the proliferation of LH-86 and Huh7 cells treated with EVs of different concentrations; The apoptosis rate of LH-86 and Huh7 cells treated with high concentration BMSCs-EVs (50 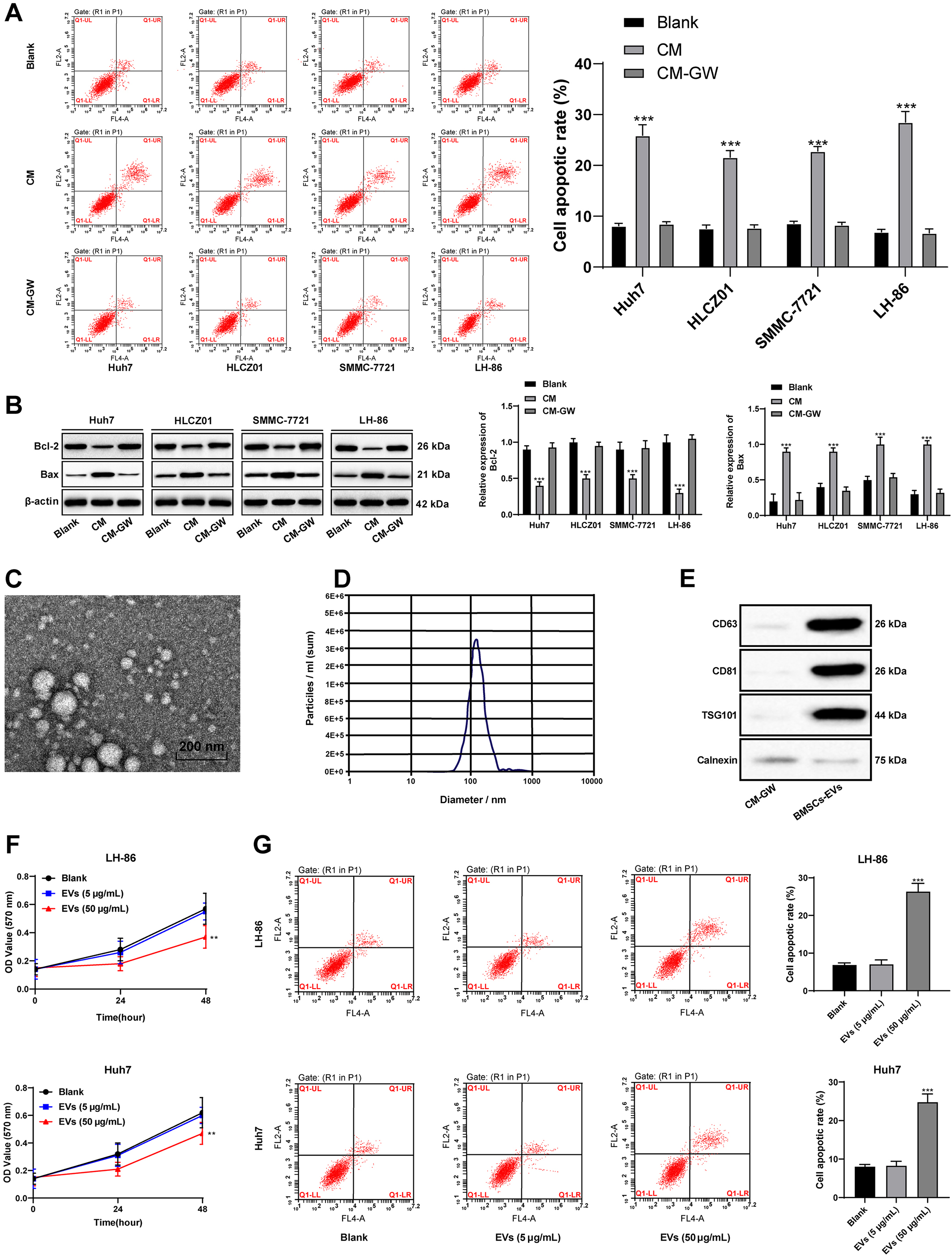
BMSCs-EVs promote HCC cell apoptosis by increasing the sensitivity of HCC cells to TRAIL. A, RT-qPCR detected the mRNA expression of TRAIL in LH-86 and Huh7 cells treated with 50 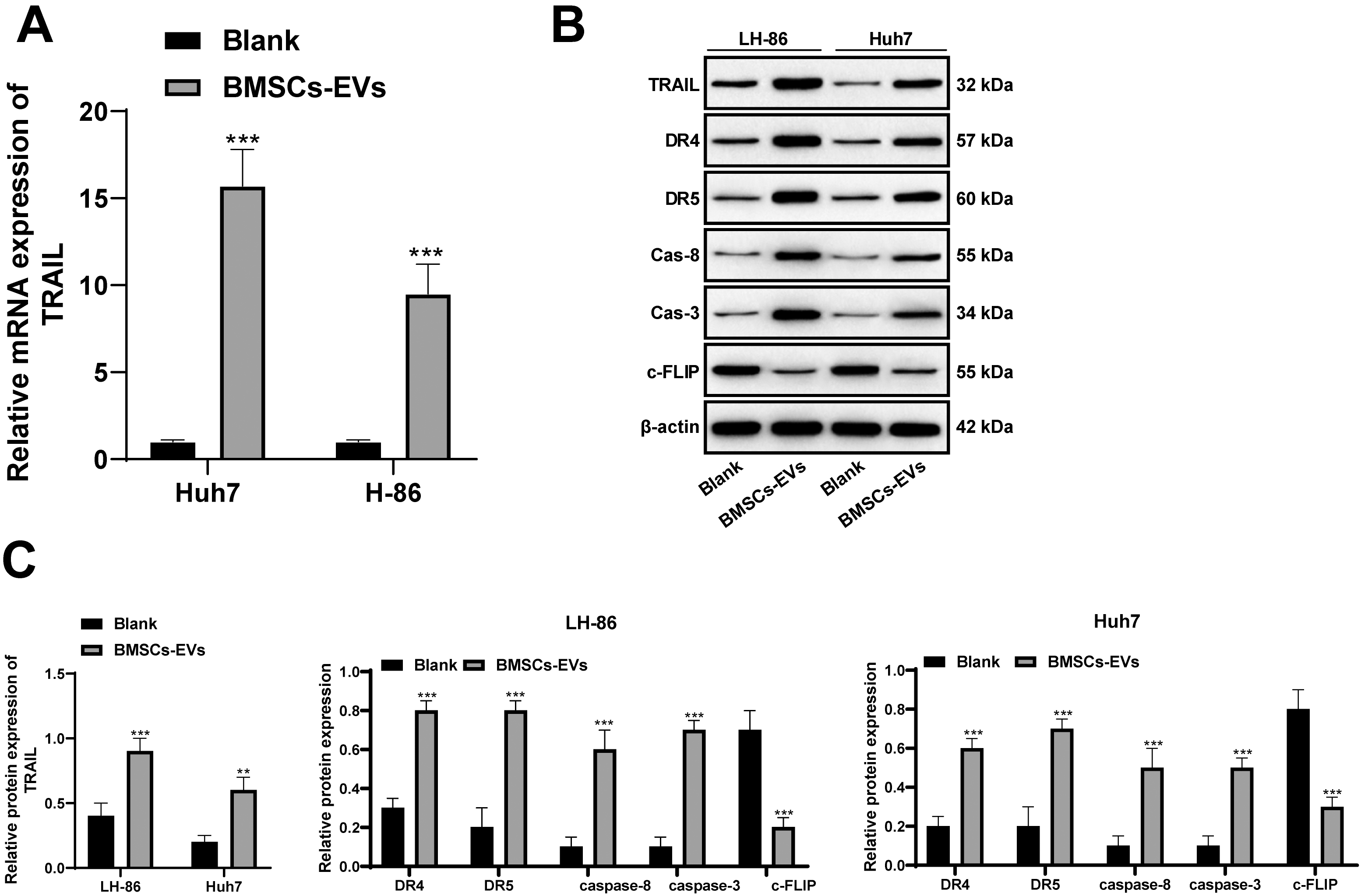
We speculated that the EVs secreted by BMSCs might play a role in this process, so we added the EV inhibitor GW4869 to BMSCs, and then extracted its CM (CM-GW) to treat HCC cells. It was found that BMSCs-CM lost its effect on HCC cells after inhibiting the formation of EVs (Fig. 2A and B).
To further confirm that BMSCs promote HCC cell apoptosis through the EVs, EVs was isolated from the CM of BMSCs by differential ultracentrifugation. Under the transmission electron microscope, EVs were oval in shape and about 100 nm in size (Fig. 2C). The peak particle size was about 100 nm and the concentration was about 3.5
Next, the separated BMSCs-EVs were applied to LH-86 and Huh7 cells. MTT results showed no significant difference in the effect of low concentration (5
HCC, like other tumors, has inherent resistance to TRAIL induced apoptosis [16]. To explore the relationship between BMSCs-EVs and TRAIL-induced apoptosis, we found TRAIL levels in HCC cells (LH-86 and Huh7) were significantly increased, and the expression of DR4 and DR5, Caspase-8 and Caspase-3 related to TRAIL-induced apoptosis were also upregulated (all
BMSCs-EVs carry miR-20a-3p into HCC cells and miR-20a-3p targets c-FLIP
As we mentioned in the introduction the role of EVs and miR, we speculated that BMSCs-EVs might enhance the sensitivity of TRAIL in HCC cells by transferring miR, and thus promote apoptosis. The EV can play its role in regulating disease by transferring miRs [22, 23]. miR-20a is highly expressed in the BMSC-EVs [9]. Through literature review, we found that miR-20a was downregulated in HCC patients [10], and in online analysis of database (
BMSCs-EVs carry miR-20a-3p into HCC cells and miR-20a-3p targets c-FLIP. A, online database (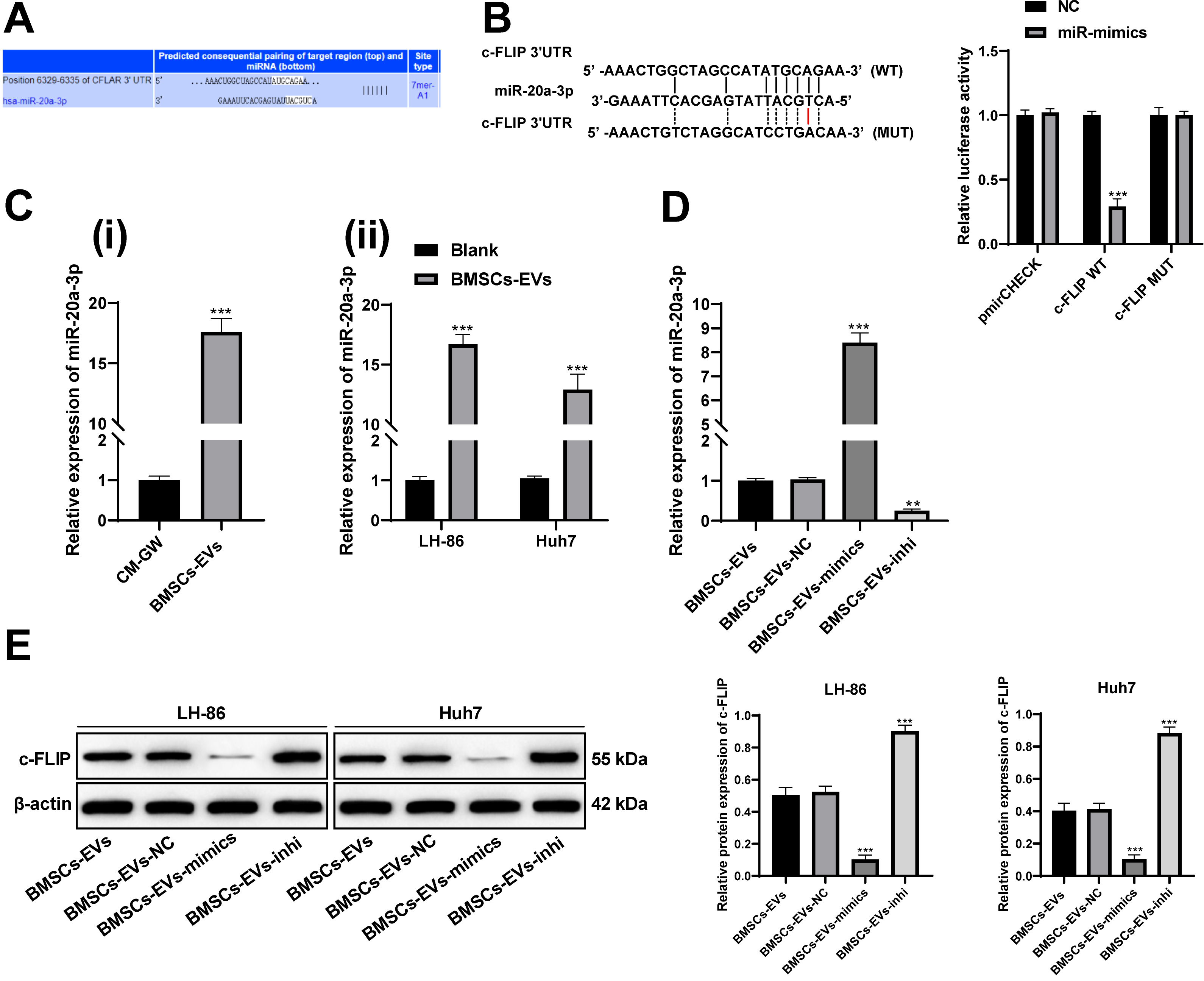
Overexpression of c-FLIP reverses the apoptosis promoting effect of BMSCs-EVs on HCC cells. A, western blot analysis detected the protein levels of c-FLIP after transfection; B, flow cytometry detected HCC cell apoptosis; the concentration of BMSCs-EVs was 50 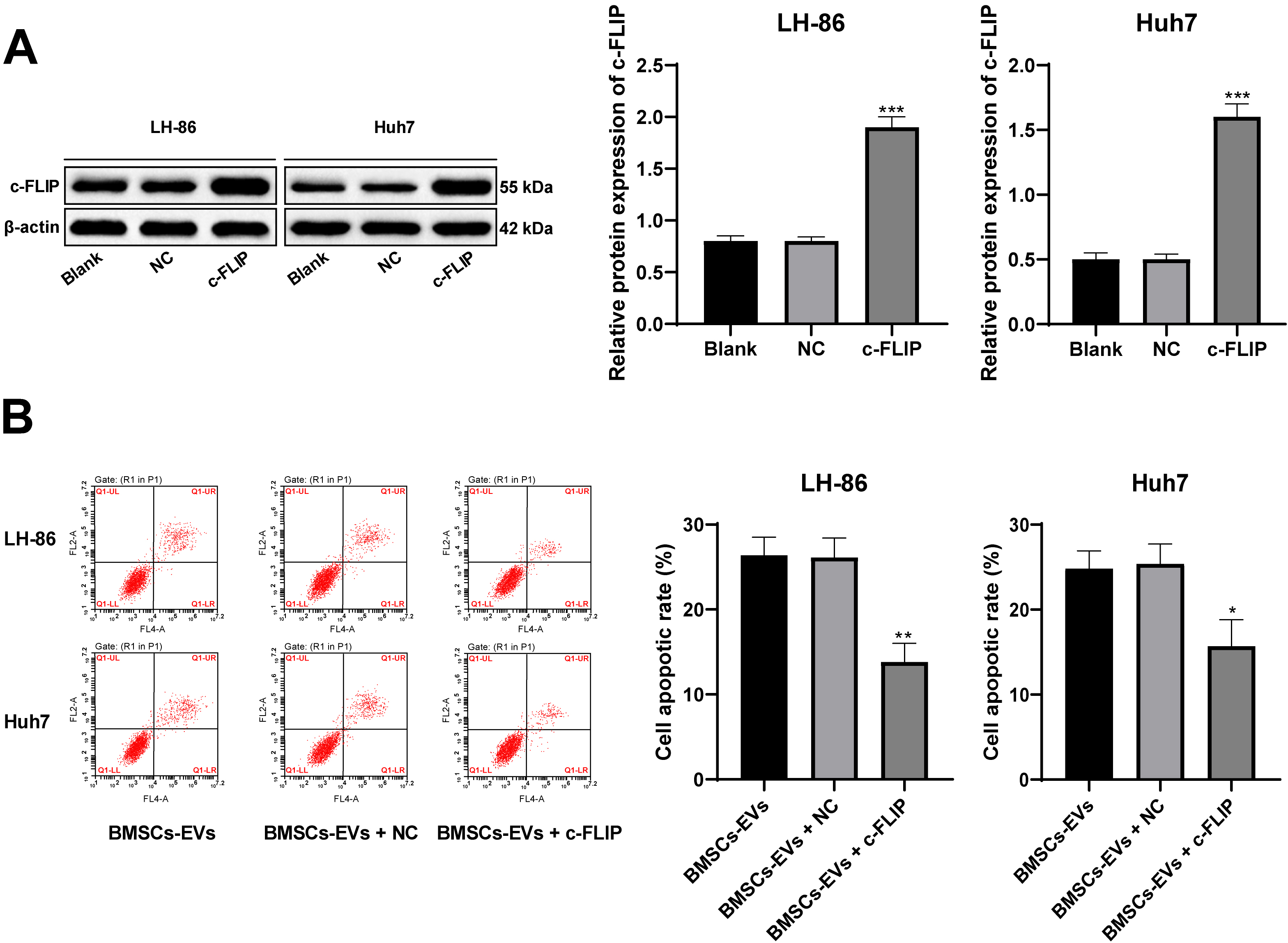
Then we transfected miR-20a-3p mimic/inhibitor into BMSCs and extracted the EVs (BMSCs-EVs-mimic and BMSCs-EVs-inhi). miR-20a-3p expression in BMSCs-EVs-mimic and BMSCs-EVs-inhi increased or decreased significantly (
In the functional rescue experiment, we first constructed a vector expressing c-FLIP and transfected it into LH-86 and Huh7 cells. We found c-FLIP expression was increased significantly, indicating the transfection was successful (Fig. 5A). After BMSCs-EVs were used to treat LH-86 and Huh7 cells overexpressing c-FLIP, the apoptotic rate was significantly lower than those only treated with BMSCs-EVs (
BMSCs-EVs inhibit the growth of HCC tumors in mice
After 7 days, BMSCs-EVs, BMSCs-EVs-mimic or BMSCs-EVs-inhi were injected into the tail vein every other day. The tumor volume and weight of mouse in each group were compared. Compared with the control, BMSCs-EVs effectively inhibited the growth of HCC tumor (Fig. 6A and B), increased TUNEL-positive rate in tumors (Fig. 6C), reduced c-FLIP expression and increased TRAIL expression (all
BMSCs-EVs inhibit the growth of HCC tumors in mice. A, the growth curves of tumor volume in mice in each group; B, representative images of tumors and histogram of tumor weight; C, TUNEL staining detected the apoptosis; D, western blot analysis detected c-FLIP and TRAIL levels in tumor homogenate. 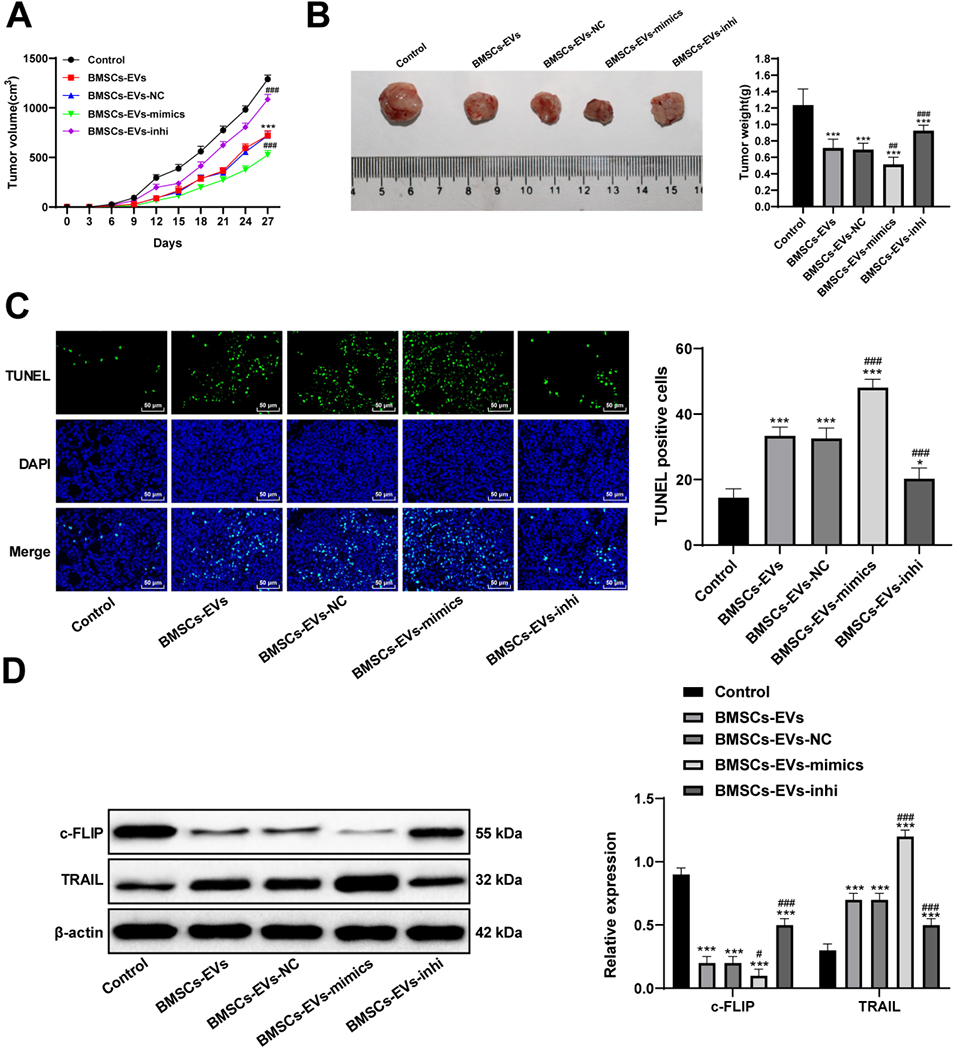
HCC almost ran an acute fulminating course and held an extremely terrible prognosis with low rates of resectability and high rates of recurrence after surgery, especially responding awfully to anticancer agents and radiotherapies [26]. A recent research has indicated that ADSCs conditioned medium could suppress HCC cell growth by repressing cell proliferation and migration, and facilitating cell apoptosis [27]. In this study, we evaluated the underlying mechanism of TRAIL and BMSCs-EVs in HCC apoptosis using both in vivo and in vitro experiments to confirm our hypothesis. Consequently, the observations in this study supported that miR-20a-3p carried by BMSCs-EVs targets c-FLIP and increases TRAIL levels in HCC cells, thus promoting TRAIL-related apoptosis.
Study in animal models has indicated that HCC may originate from genetically mutated BMSCs, BMSCs can modulate their microenvironment via secreted cytokines and BMSCs have been recognized as a putative target of molecular therapies to treat and prevent HCC [28]. The first result of our study was that CM of BMSCs promoted HCC cell apoptosis. Then we added the EV inhibitor GW4869 to BMSCs, and then extracted its CM to treat HCC cells. It was found that BMSCs-CM lost its effect on HCC cell apoptosis after inhibiting the formation of EVs. MTT results further showed 50
In addition, TRAIL levels in LH-86 and Huh7 cells were increased, and the expression of DR4 and DR5, Caspase-8 and Caspase-3 related to TRAIL-induced apoptosis were also upregulated after BMSCs-EVs treatment. It is highly noteworthy that mice administered soluble recombinant TRAIL exhibited smaller size of breast adenocarcinoma xenograft tumors [31]. TRAIL alone is able to inhibit HCC cell viability and promote cell apoptosis [32]. Additionally, MSC-delivered TRAIL displays an anti-proliferative effect and a tumor-supportive function, showing greater capability to induce apoptosis in primary tumor cells [33]. TRAIL can trigger apoptosis upon binding to cell death receptors (DR4 and DR5) to activate cytoplasmic death domain and recruit FADD assembled in the death inducing signaling complex, which then interacts with caspase-8 and caspase-10 and activates proapoptotic signaling cascades [16]. The use of TRAIL alone in treating HCC may lead to chemotherapeutic resistance or reduced drug efficacy, because chemotherapeutics or biological agents upregulate DR4 and DR5 and enhance TRAIL-induced apoptosis in HCC, simultaneously upregulating decoy receptors [17]. The mice lack of TRAIL are more likely to develop tumors after administration of carcinogens and show faster development of malignant tumors in the blood system, and TRAIL is further confirmed to inhibit solid tumor growth and xenograft tumors [34], which supported the results in the transplanted tumors in nude mice in our study. HCC cells are commonly TRAIL-resistant, while adjuvant agents that can increase the sensitivity of cancer cells to TRAIL may improve the curative effect of TRAIL-based combined therapy [35].
As a gene delivery system in cancer therapy, EVs have attracted more and more attention. They can transfer nucleic acids, proteins and lipids to receptor cells naturally [22]. miR is a key regulatory molecule of biochemical pathway, and many miRs are abnormally expressed in HCC [36]. In this study we verified that BMSCs-EVs carry miR-20a-3p into HCC cells and miR-20a-3p targets c-FLIP. Patients with decreased miR-20a expression have notably shorter relapse free survival and overall survival [37]. Similarly, a previous study provides a new strategy to improve the chemosensitivity of HCC via adipose tissue derived MSCs-EVs mediated miR-199a-3p [38]. After treatment of BMSCs-EVs-mimic, c-FLIP was reduced, indicating that BMSCs-EVs promote HCC cell apoptosis by increasing the sensitivity of HCC cells to TRAIL. c-FLIP is always expressed in human HCC, and its overexpression suggests a shorter period of recurrence-free survival, while silencing c-FLIP upregulates drug-induced apoptosis, and may be a therapeutic potential for HCC treatment [39]. Interestingly, miR-20a targets CFLAR, facilitates activation of caspase-8 and caspase-3, and enhances sensitivity of HepG2 cells towards TRAIL-induced apoptosis, and subsequently reduced cell proliferation [10]. To sum up, BMSCs-EVs could inhibit c-FLIP expression and promote apoptosis induced by TRAIL pathway by carrying miR-20a-3p into HCC cells.
Together these results offer insights into the mechanism of TRAIL and BMSCs-EVs in HCC development, whereby BMSCs-EVs inhibit c-FLIP expression and promote TRAIL-induced apoptosis by carrying miR-20a-3p into HCC cells. The theoretical basis of anticancer therapy should be carried forward with improved understanding of BMSCs-EVs and their interaction with anticancer genes, which can help to develop personalized strategies for optimizing HCC diagnosis and prognosis. Further study should be conducted to find out the exact application approach for HCC based on results obtained from this study. We selected four kinds of cells to explore the role of BMSCs in the apoptosis of HCC cells. To further study whether BMSCs play a role by secreting EVs and the mechanism of EVs, considering the limitation of experimental funds, we only selected two cells with better therapeutic effect of BMSCs for following experiments according to the results in Fig. 2A.
Footnotes
Conflict of interest
The authors declared that they have no competing interests.
