Abstract
Claudins are indispensible in modulating the permeability of epithelial and endothelial cells and in the maintenance of cell polarity. In order to verify the function of claudin-6 in the development of gastric cancer, we investigated claudin-6 expression in different gastric disease tissues. Moreover, we further explored whether overexpression of claudin-6 altered proliferation, apoptosis, migration, invasiveness, differentiation in BGC-823 cells and the potential mechanism. Immunohistochemistry was performed to detect the in situ expression of claudin-6 in different gastric disease tissues; moreover, cell culture, real-time PCR and western blot were used to evaluate the effect of overexpression of claudin-6 in vitro and the related mechanism. The results of immunohistochemical staining showed that the positivity of claudin-6 was significantly higher in superficial gastritis than that in gastric cancer. Overexpression of claudin-6 induced differentiation of BGC-823 cells by inhibiting the JNK pathway. However, it had no effect on proliferation, apoptosis, migration or invasiveness in vitro. The expression of claudin-6 was decreased in gastric cancer. Overexpression of claudin-6 induced differentiation of gastric cancer cells by inhibiting the JNK pathway.
Introduction
The gastric epithelial barrier is a vital defensive mechanism that helps to maintain epithelial integrity and protect us from gastric diseases. When the integrity of gastric epithelial barrier is impaired, abnormal lateral diffusion of intracellular molecules and the invasion of bacteria and virulence factors from gastric mucosa into the organic body can induce many kinds of gastric diseases and even gastric cancer [1]. The tight junction, a multicomplex of membrane proteins, plays a critical role in keeping epithelial barrier function. As an important ingredient in tight junction, claudin family which consists of 27 proteins is indispensable in the maintenance of cell polarity, barrier function and in the permeability of epithelial cells [2, 3, 4]. The abnormal expression and dislocation of claudins may play pivotal roles in tumorigenesis. For instance, claudin-1, which is highly expressed in the intestinal type of gastric cancer, is correlated with tumor invasion and migration [5, 6]. Besides, our previous work found that claudin-11 and -23 were down-regulated in gastric cancer [7], which suggested that abnormal expression of claudins may be closely related to the initiation of gastric cancer.
Claudin-6 is an important member of the claudin family. Some researchers reported that claudin-6 played an important role in the formation of mouse embryonic epithelial as well as the development of mouse endoderm [8, 9]. Moreover, claudin-6 has been found to be a crucial marker of mouse multipotential stem cells. Overexpression of claudin-6 in mouse multipotential stem cells can induce epithelial morphological changes [10, 11]. Gastric tissue is derived from endoderm, it is therefore desirable to specify whether claudin-6 is abnormally expressed or not during the progression of gastric cancer.
Although the role of claudin-6 in the development and progression of diseases has attracted more and more attention, the exact expression status of claudin-6 in gastric cancer is still unclear, and its potential biological effect on gastric cancer cells has never been reported before. In the current study, we aimed to compare the expression profile of claudin-6 in different gastric diseases, and to explore whether claudin-6 over-expression can regulate biological behavior of gastric cancer cells and its potential mechanism.
Materials and methods
Research subjects and ethics statement
One hundred and nine patients with superficial gastritis and 109 patients with atrophic gastritis were selected from a health check-up program involving gastroscopy for gastric cancer screening in Zhuanghe, Liaoning Province, from 2008 to 2011. In addition, 109 patients with gastric cancer who underwent surgical treatment without chemoradiotherapy or other therapy before surgery between 2012 and 2015 were enrolled from the anorectal department of the First Hospital of China Medical University (Supplementary Table 1). In the above 109 cases of gastric cancer, we also obtained paracancerous tissues in 58 of them, which were stored in the refrigerator at minus 80 degrees for PCR and western blot. In addition, about 5 percent of the follow-up data were incomplete.
Clinicopathological parameters and survival in GC
Clinicopathological parameters and survival in GC
GC, gastric cancer; MST median survival time.
This study was approved by the Ethics Committee of the First Hospital of China Medical University Shenyang, China, and written informed consent was obtained from all participants.
Slides were deparaffinized in xylene, rehydrated in a graded alcohol series, and washed in tap water. The tissue sections were incubated in boiling sodium citrate buffer in a steam pressure cooker for antigen retrieval. Next, endogenous peroxidase was blocked using 3% hydrogen peroxide for 30 min, and the sections were washed with phosphate-buffered saline (PBS), pH 7.4. Tissue collagen was blocked by the addition of 10% normal goat serum at 37
Cell culture
Gastric cancer cell line AGS was purchased from the American Type Culture Collection (ATCC; Manassas, VA, USA); BGC-823, MGC-803, SGC-7901, MKN-45 and HGC-27 were purchased from the cell bank of Chinese Academy of Sciences (Shanghai, China). Real-time PCR and western blot was performed to detect the expression of claudin-6 in different gastric cancer cell and the results indicated that BGC-823 could be selected as tool cell for further investigation. Claudin-6 overexpression BGC-823 cell (823OE) and negative control BGC-823 (823NC) were purchased from Genechem (Shanghai, China). AGS was cultured with DMEM-F12 medium while other cell lines were cultured with RPMI 1640 medium supplemented with 10% fetal bovine serum (FBS) (Hyclon, USA) in a 95% humidified incubator at 37
Lentivirus production and infection
The human CLDN6 coding region was cloned into pGV303GFP vector (Shanghai GeneChem). The lentiviral constructs were packaged in plasmids, and transfected to BGC-823 cell. Stable cell line was verified by real-time PCR and western blot.
RNA extraction and real-time PCR
Total RNA was extracted with Trizol buffer (Thermo Fisher Scientific, Massachusetts, USA). RNA was reverse transcribed to cDNA using PrimeScript RT Master Mix (Takara). Real-time PCR was carried out on Eppendorf equipment using SYBR Premix Ex Taq (Takara, Liaoning, China) in accordance with the manufacturer’s protocols.
Protein extraction and western blot analysis
Total proteins were extracted with RIPA buffer (Aidlab Biotechnologies, Beijing, China). Tissue lysates were centrifuged at 14,000 rpm for 20 min at 4
Cell proliferation assay
5
Transwell migration assay
Transwell assay was performed to evaluate cell migration ability (Corning Life Sciences USA). The lower chamber was filled with 600 ul RPMI 1640 medium containing 10% FBS. Cells (5
Claudin-6 expression in super gastritis, atrophic gastritis, gastric cancer tissues from different individuals
Claudin-6 expression in super gastritis, atrophic gastritis, gastric cancer tissues from different individuals
The mRNA (A, 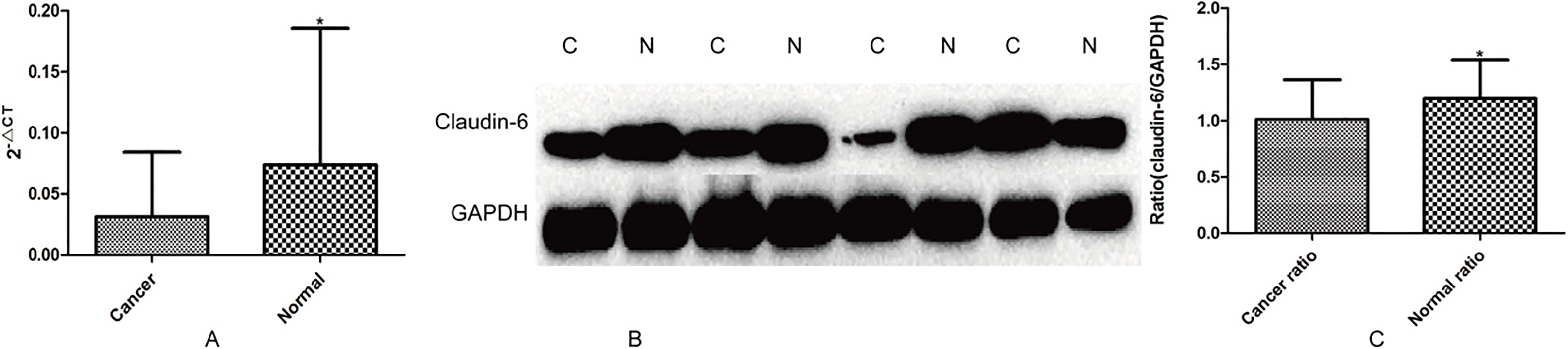
Total RNA (
Statistical analysis
All statistical analyses were performed using SPSS18.0 software (Chicago, IL, USA). Chi-square was used to analyze differences in expression of claudin-6 between different gastric diseases from different individuals. Comparison of gene differential expression between 823NC and 823OE was performed by student’s
Results
Differential expression of claudin-6 in different gastric tissues
Real-time PCR and Western blot were used to detect the general trends of claudin-6 expression in 58 cases of gastric cancer tissues and adjacent normal tissues. The result showed that claudin-6 expression was significantly higher in adjacent normal tissues than that in gastric cancer tissues (Fig. 1). We further observed claudin-6 expression in different gastric diseases tissues from different individuals by immunohistochemical staining, and the result indicated that claudin-6 expression was significantly higher in superficial gastritis than that in atrophic gastritis and gastric cancer (
Positive expression of claudin-6 (A and B) and negative expression of claudin-6 (C) in superfical gastritis; Positive expression of claudin-6 (F) and negative expression of claudin-6 (D and E) in atrophic gastritis; Positive expression of claudin-6 (I) and negative expression of claudin-6 (G and H) in gastric cancer.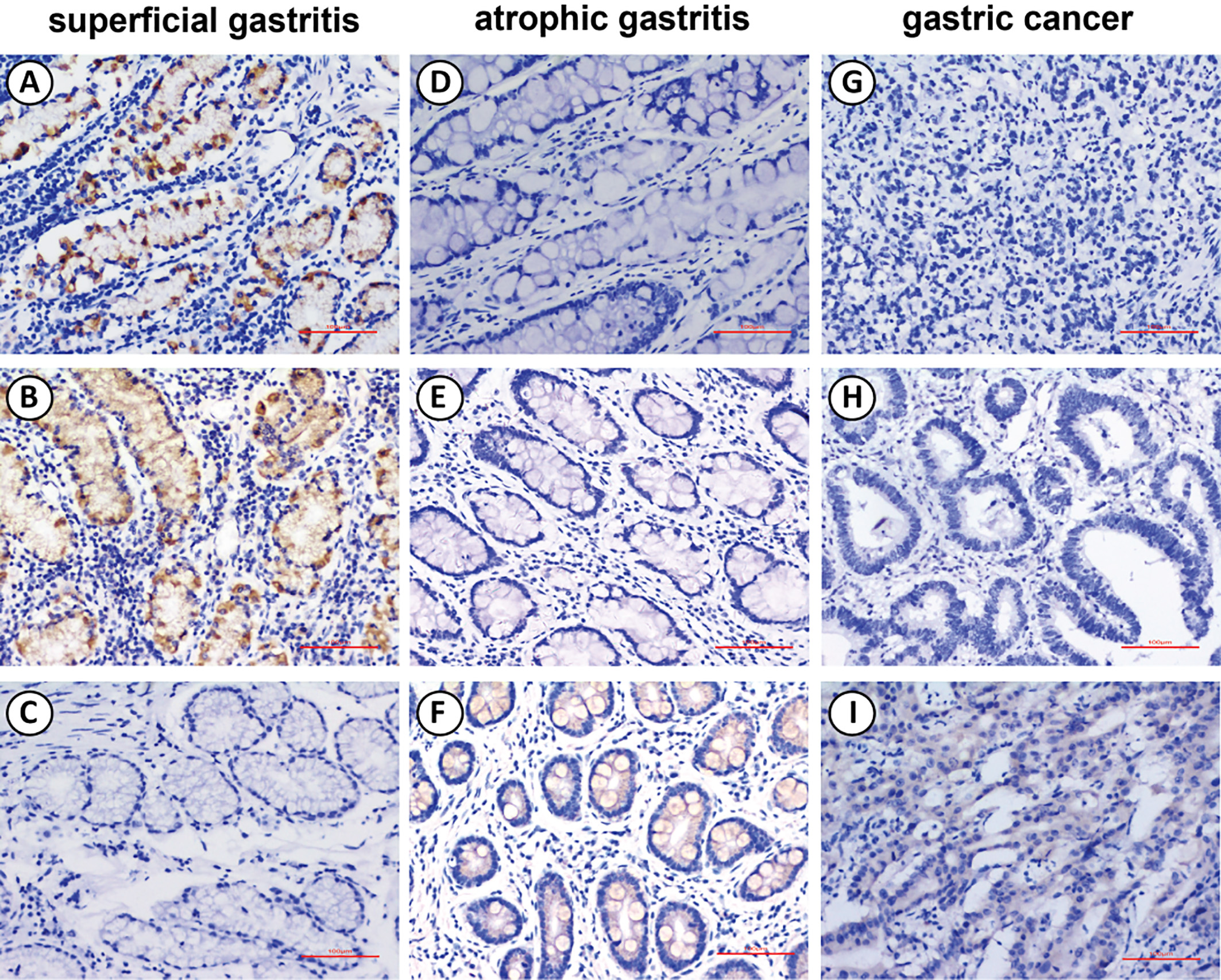
The results showed that claudin-6 was high expressed in AGS at both mRNA and protein level, low expressed in other cells, especially in BGC823 (Fig. 3). We further chose BGC823 to establish cell line stably expressed claudin-6. Whether the 823NC and 823OE were constructed successfully was verified by real-time PCR and Western blot (Fig. 4).
The mRNA (A) and protein (B) expression of Claudin-6 in different gastric cancer cell lines.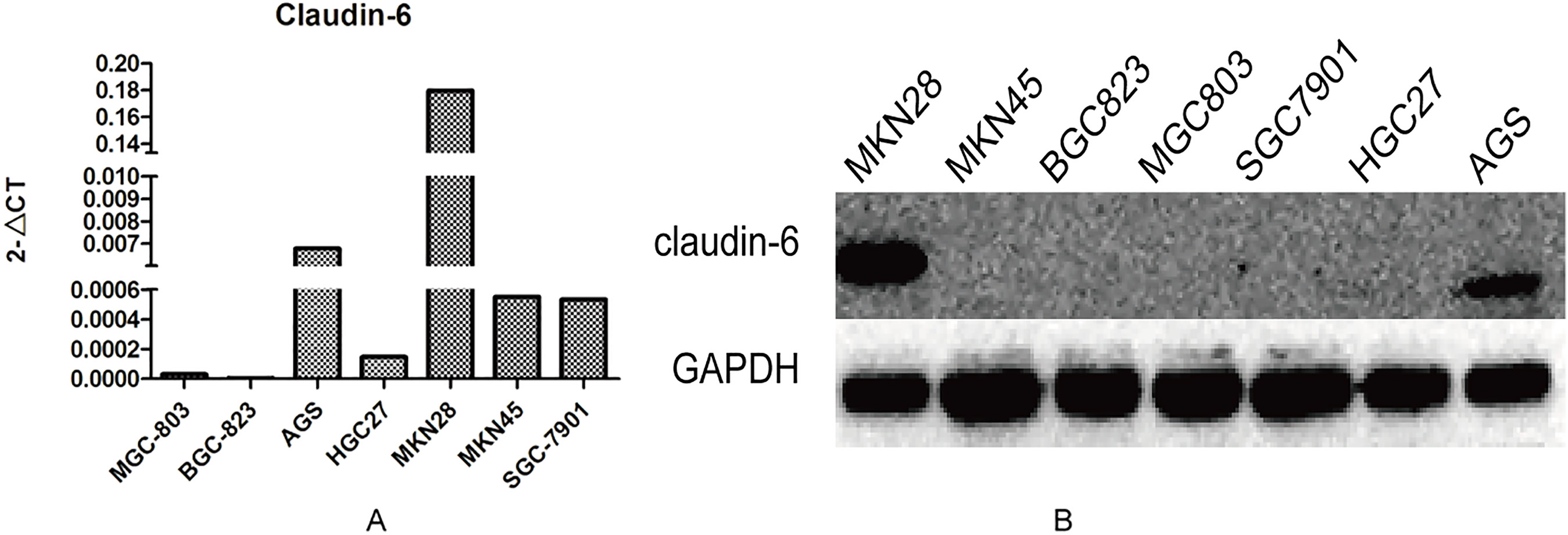
The mRNA (A, 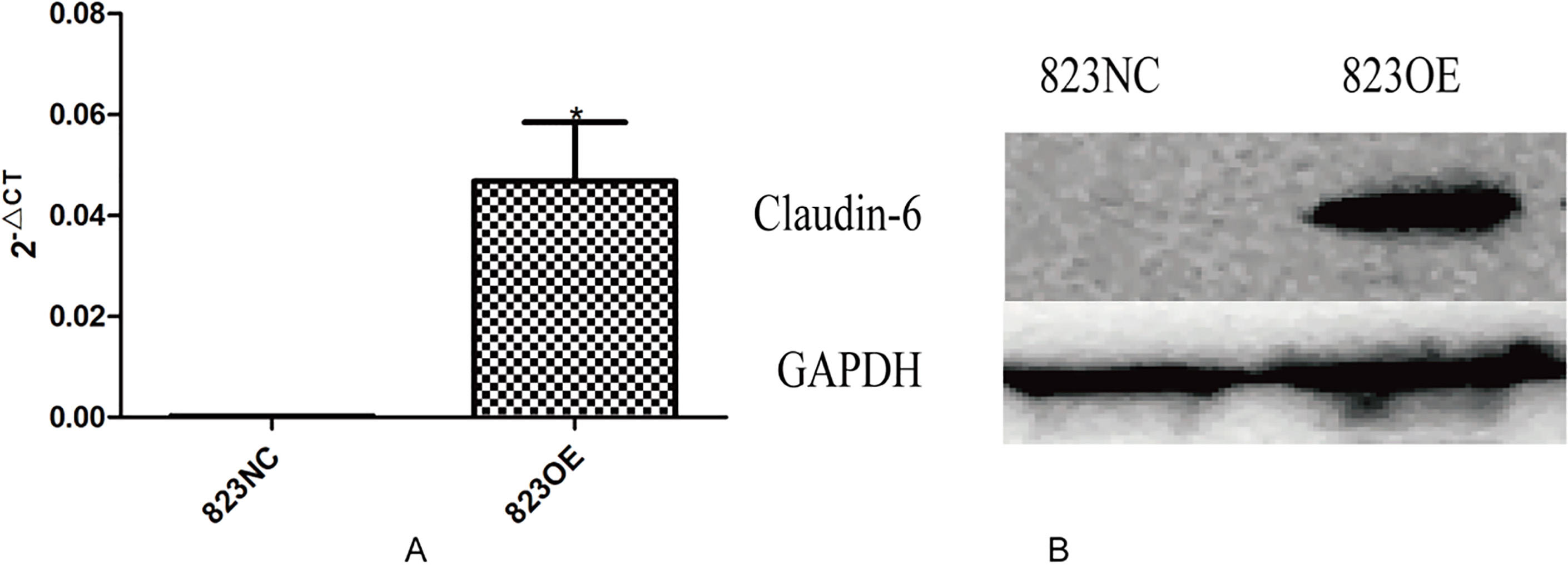
The results of CCK-8 test (A, 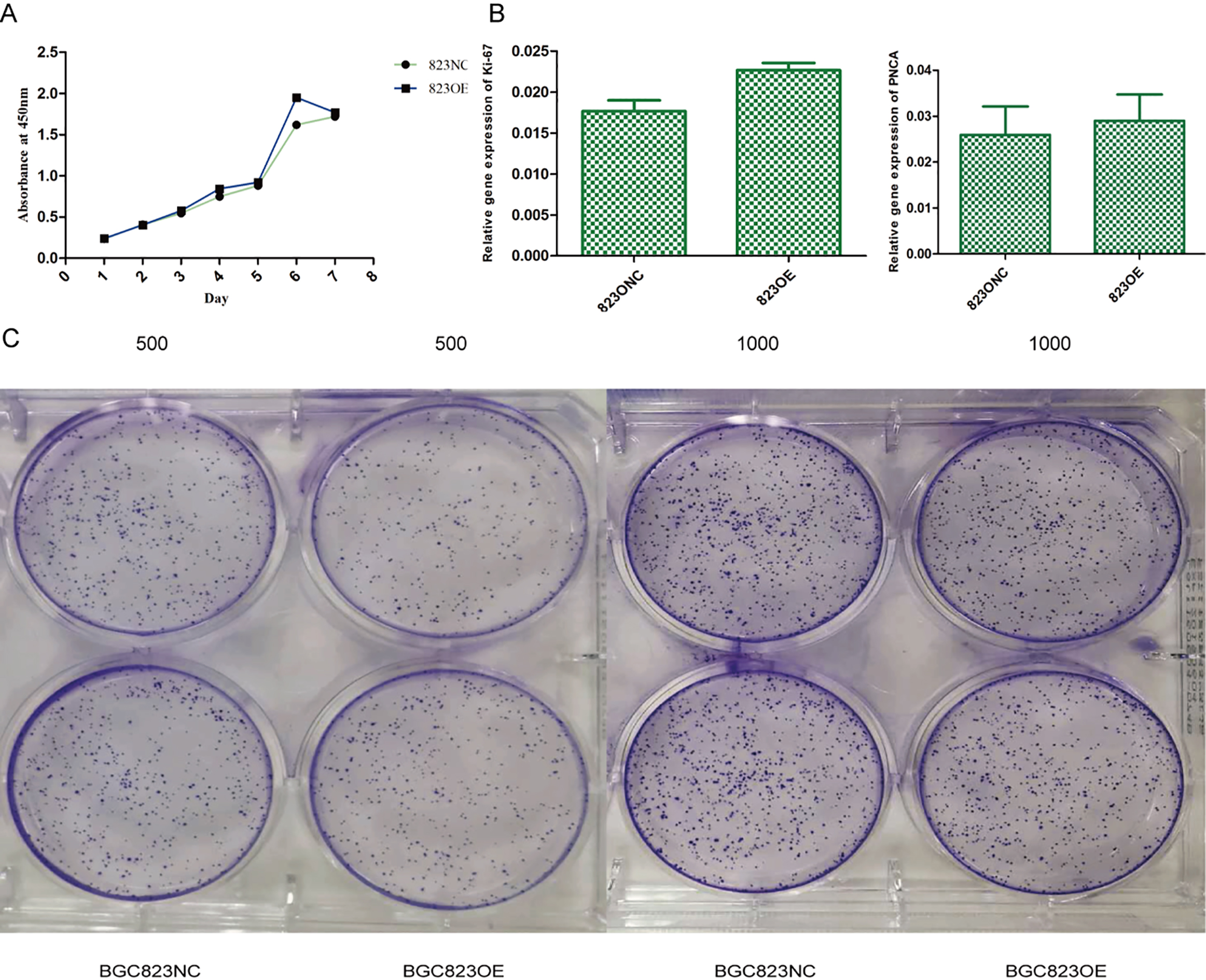
The mRNA expression of caspase-3 and TFAR-19 in 823NC cells and 823OE cells (A, 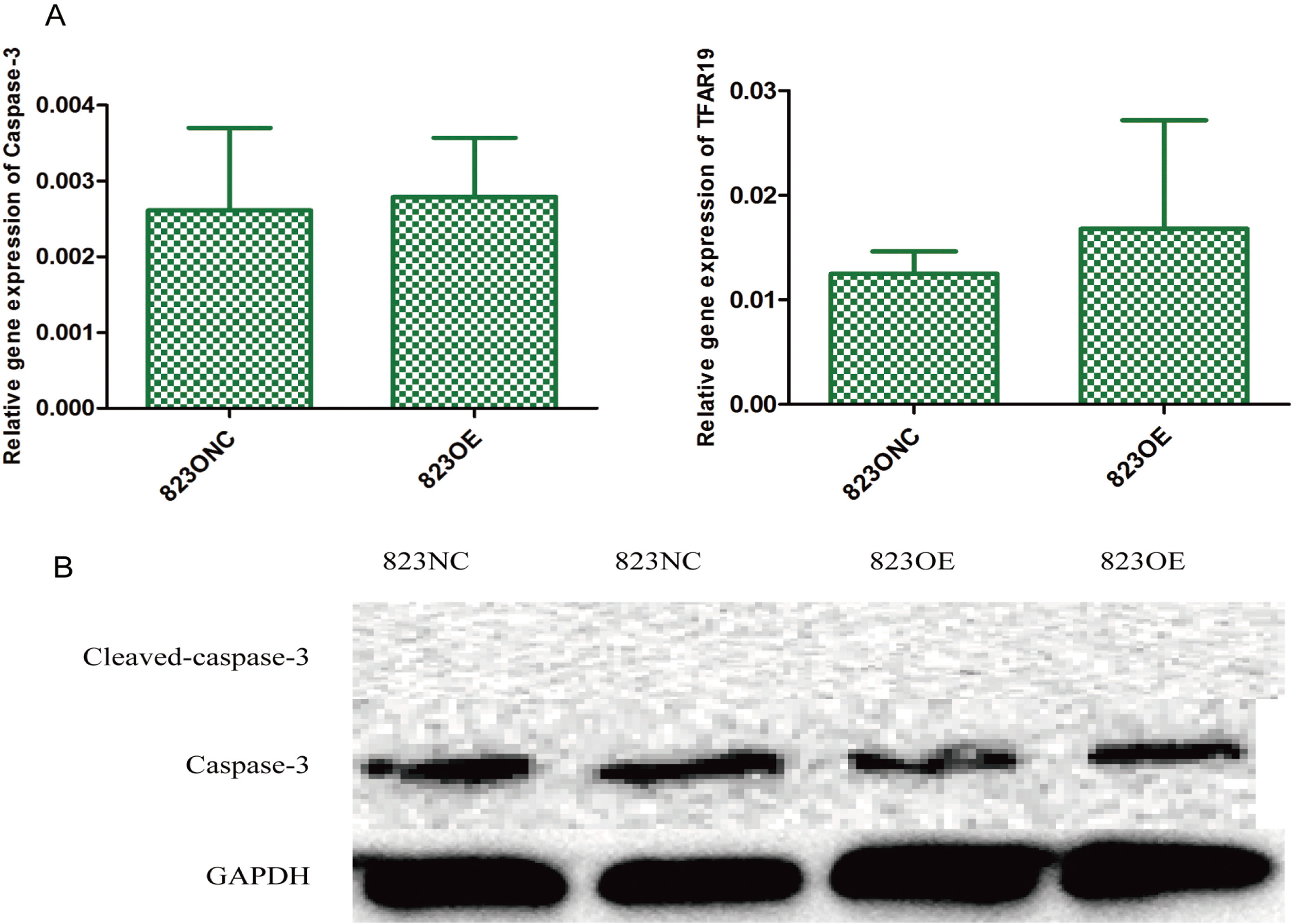
The result of transwell assay (A, 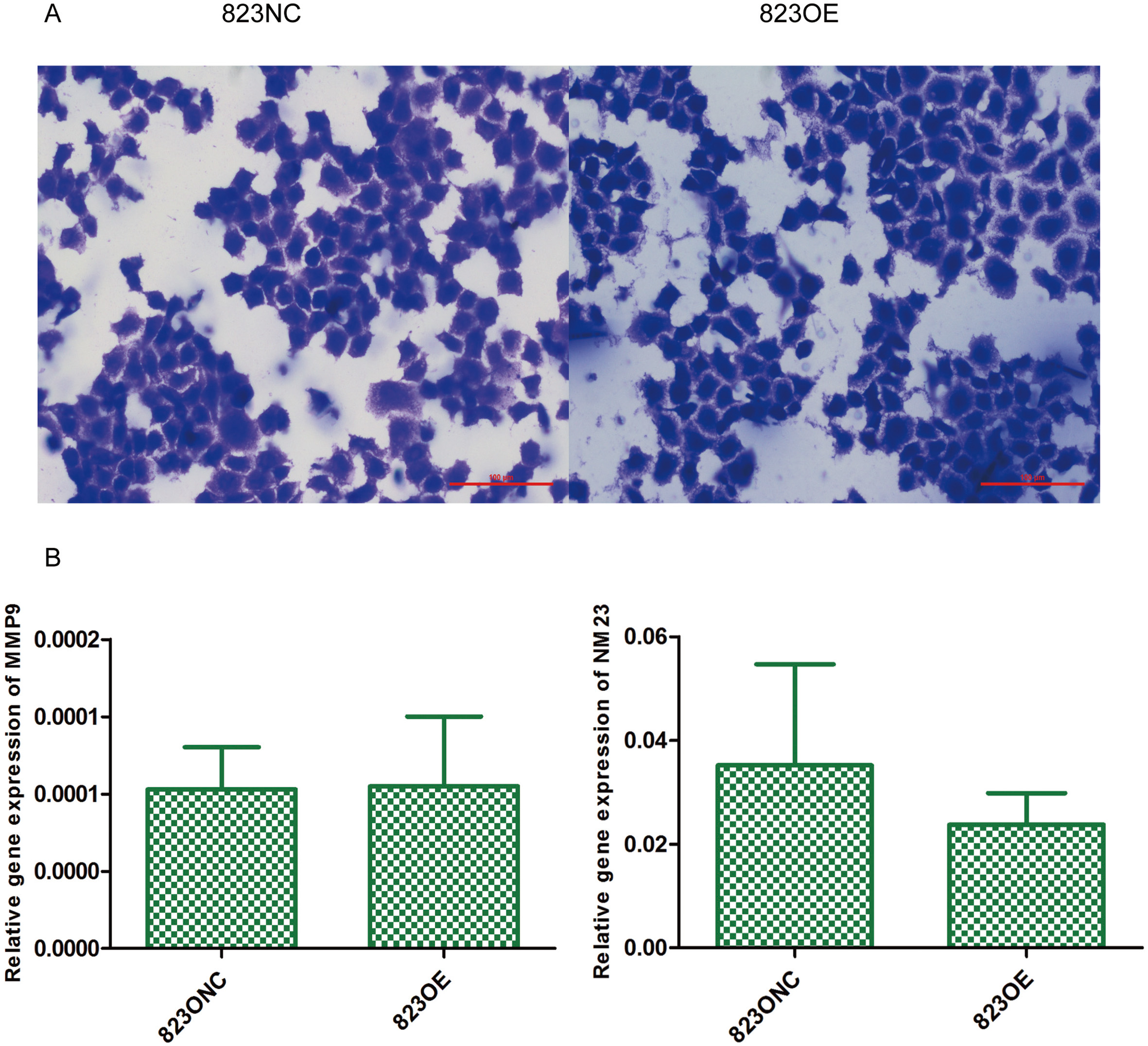
The mRNA expression of SOX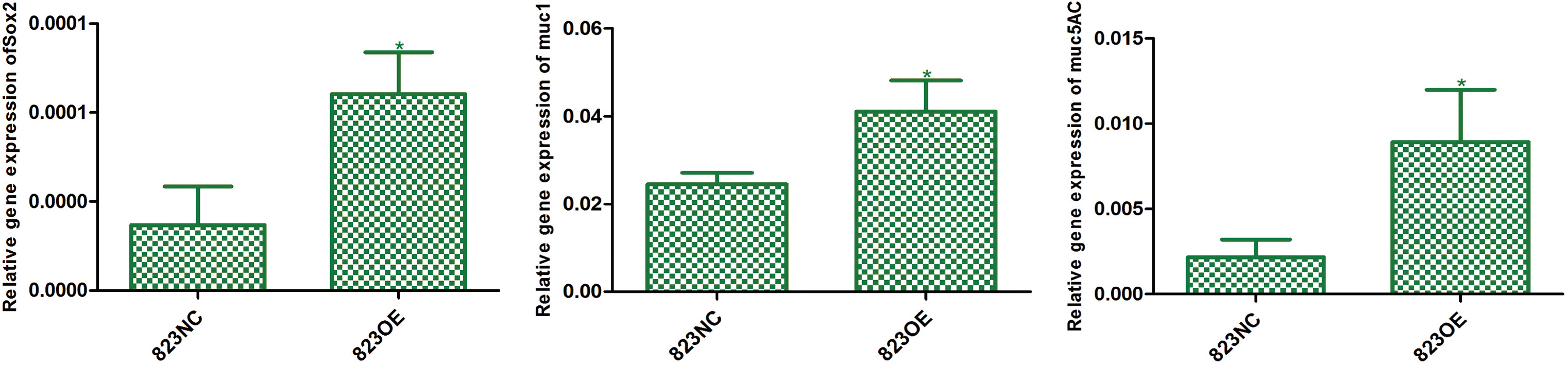
The expression of FN1 (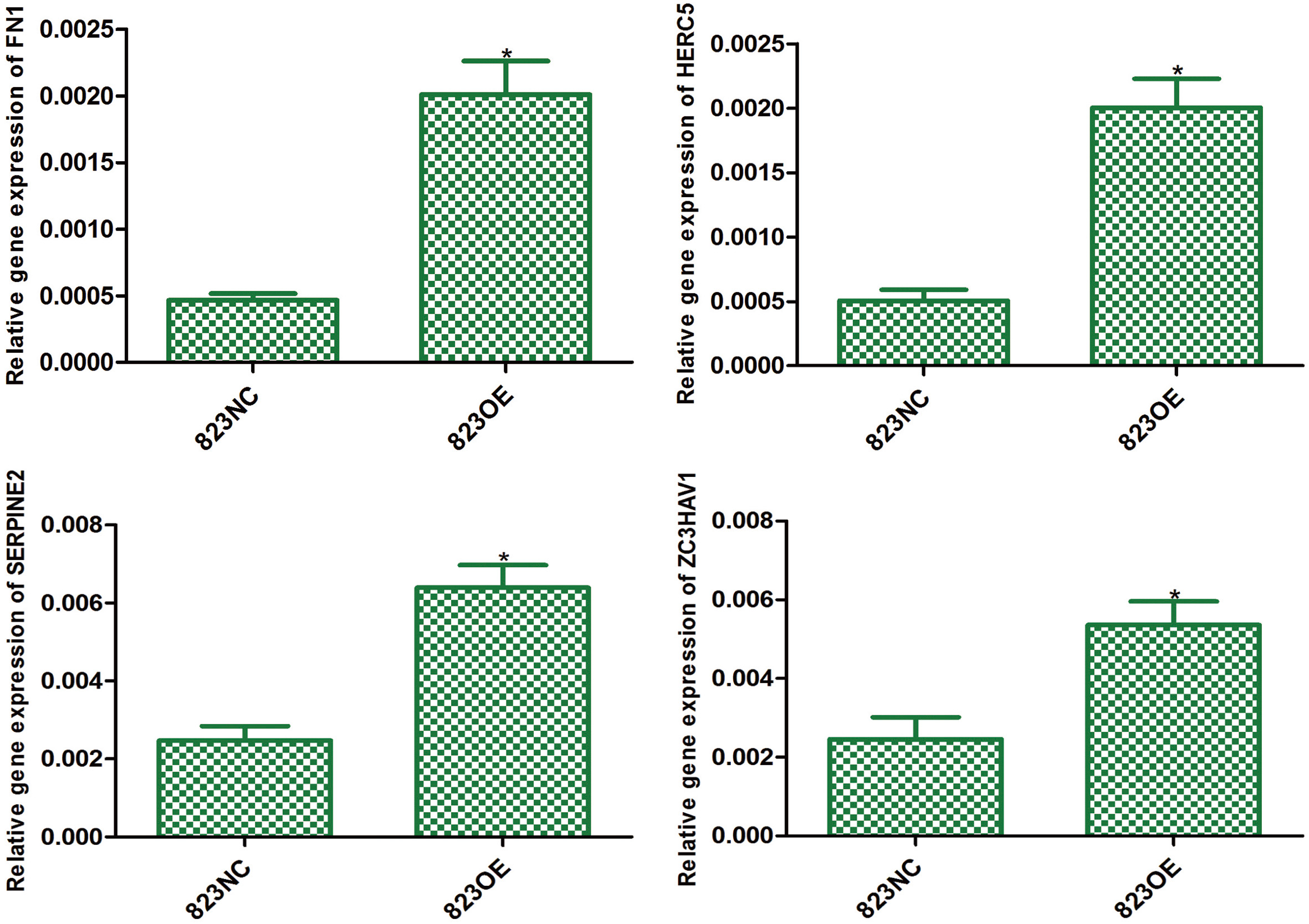
We used CCK-8 test and plate clone formation assay to detect cell proliferation rate and found that claudin-6 overexpression have no significant influence on cell proliferation (Fig. 5A and C). Besides, we also used real-time PCR to detect the expression of Ki-67 and PNCA which can indirectly reflect the proliferation rate and also got a negative result (Fig. 5B). In order to observe cell apoptosis ability, real-time PCR was performed to detect the expression of caspase-3 and TFAR-19 and western blot was performed to detect the expression of caspase-3 and cleaved caspase-3. However, no significant result was found (Fig. 6). MMP-9 and NM23 were detected by real-time PCR to observe cell apoptosis ability. Transwell assay was also performed. Nevertheless, no positive result was found (Fig. 7).
Claudin-6 overexpression promotes differentiation in BGC-823 cells
Indexes which can reflect cell differentiation ability were detected by real-time PCR. The result showed that claudin-6 overexpression can significantly up-regulate the expression of SOX
The expression of P38 (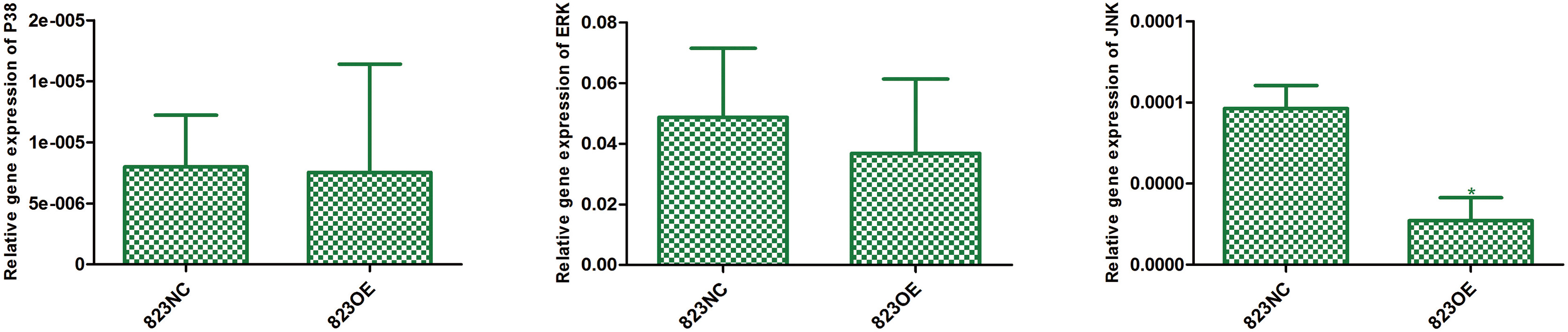
The result of gene expression profile showed that 95 genes were significantly up-regulated and 2 were down-regulated in 823OE compared to 823NC. According to KEGG pathway information, enrichment analysis of differentially expressed genes showed that MAPK pathway was the most significantly up-regulated one. We chose genes with more than 2 fold variations for further conformation and the result revealed that FN1, HERC5, SERPINE2 and ZC3HAV1 increased 4.3, 3.95, 2.59 and 2.18 fold respectively (Fig. 9). Moreover, we compared the differential expression of P38, JNK and ERK between 823OE and 823NC, and found that the expression of JNK was significantly decreased in 823OE (Fig. 10).
Discussion
In this study, we found that the expression of claudin-6 was significantly higher in adjacent normal tissues than that in gastric cancer tissues by real-time PCR and western blot. Besides, we also revealed that claudin-6 expression in superficial gastritis and atrophic gastritis was significantly higher than that in gastric cancer. We further explored the biological effect and potential pathway by constructing gastric cancer cells with claudin-6 stably expressed. The result showed that claudin-6 overexpression had no impact on cell proliferation, apoptosis, invasion and migration but can promote cell differentiation by inhibiting the activity of JNK pathway.
Some researchers have reported that claudin-6 was abnormally expressed in several malignant tumors. For example, claudin-6 expression was lower in non-small cell lung cancer (NSCLC) tissue than in normal adjacent tissue samples, and low claudin-6 expression was an independent indicator of prognosis in NSCLC patients [12]. Besides, Zhang et al. found that claudin-6 expression was significantly lower in cervical cancer than in adjacent normal tissues [13]. All of these results indicated that claudin-6 may play a defensive role in the occurrence and development of cancer. In this study, we used immunohistochemical methods to detect the expression of claudin-6 in different gastric diseases. Similarly, our results showed that the expression of claudin-6 was lower in gastric cancer than that in superficial gastritis, which is in accordance with the results of Lin et al. [14]. We speculated that decreased expression of claudin-6 may destroy epithelial integrity and thus increasing para-cellular leakage which not only played an essential role in increasing tumor cell migration ability but also increased nutrients’ supply for tumor cells.
Some researchers revealed that claudin-6 overexpression could play an anti-cancer role. For instance, the absence of claudin-6 expression in breast cancer tissue and breast cancer cell MCF-7 was associated with promoter methylation. 5-Aza treatment can increase claudin-6 expression, thus inhibit MCF-7 cell migration and invasion ability [15]. In addition, claudin-6 overexpression can induce cervical cancer cell apoptosis as well as inhibiting cell proliferation ability [16]. However, there was only one research about the effect of claudin-6 on gastric cancer and got a controversial result that claudin-6 overexpression could promote AGS cell proliferation, invasion and migration ability [17]. In this study, we found that claudin-6 overexpression had no influence on gastric cancer cell proliferation, apoptosis and migration ability, but promoted cell differentiation. This result was consistent with previous studies that claudin-6 played an anti-cancer role in cancer. Claudin protein is a four-time transmembrane protein, containing two extracellular rings, amino-terminal and carboxyl-terminal, and the difference in carboxyl-terminal structure leads to differences in the physiological functions of claudin family members. We hypothesized that claudin-6 may influence the signal pathway of cell differentiation through its carboxyl-terminal interaction with intracellular signal proteins, and ultimately promote cell differentiation.
In our study, claudin-6 overexpression can up-regulate molecular markers related to gastric mucosal differentiation, such as MUC1 and MUC5AC and transcriptional factor SOX
In order to explore the potential pathway, gene expression profile was performed. According to gene expression profile and KEGG pathway information, enrichment analysis of differentially expressed genes showed that MAPK pathway was the most significantly up-regulated one. We chose genes with more than 2 fold variations and those included in MAPK pathway for further conformation and the result revealed that FN1, HERC5, DUSP4, SERPINE2 and ZC3HAV1 increased 4.3, 3.95, 2.64, 2.59 and 2.18 fold respectively.
FN1, a member of the fibronectin family, is essential to the development of cancer [27]. For instance, Gorogh et al. discovered that fibronectin expression in laryngeal squamous cell cancer tissue was significantly lower than that in normal tissue [28]. Fibronectin can induce disease progression by affecting the activity of the MAPK signaling pathway [29]. Therefore, we speculated that claudin-6 overexpression can increase FN1 expression and then regulating the differentiation of gastric cancer cells by MAPK signal pathway.
HERC5 is a kind of E3 ubiquitin ligase that contains a HECT domain and an RCC-like domain [30, 31]. HERC5 can regulate cell proliferation and differentiation by interacting with NME2 gene and further affecting the development of tumors [32, 33]. Wrage et al. found that the decreased expression of HERC5 in lung cancer was associated with hypermethylation and the high incidence of brain metastases leading to a worse prognosis [34]. The results of this study showed that claudin-6 overexpression can significantly up-regulate the expression of HERC5, suggesting that claudin-6 may change the methylation status of certain genes in gastric cancer cells by increasing the expression of HERC5, thus affecting the biological behavior of gastric cancer cells.
DUSP4 is known as mitogen-activated protein kinase phosphatase 2 (MKP2), which plays an important role in cell proliferation, apoptosis and differentiation by deactivating MAPKs (including ERK, p38 and JNK) through dephosphorylation [35, 36]. Zhang et al. found that the expression of DUSP4 was significantly down-regulated in metastatic gastric cancers with deeper infiltration, and DUSP4 overexpression could inhibit the proliferation and invasion ability of gastric cancer cells [37]. The results of this study show that claudin-6 can significantly up-regulate the expression of DUSP4, suggesting that claudin-6 may promote the differentiation of gastric cancer cells by up-regulating the expression of DUSP4.
JNK pathway belongs to MAPK classical pathway. The abnormal activation or inactivation of JNK plays an important role in the nervous system diseases, immune diseases, and cancers. JNK is a kind of serine/threonine kinase. Many external stimuli play an important role in cell proliferation, differentiation and apoptosis by changing the activity of JNK pathway. JNK has been found to be activated in many human squamous cell carcinomas, and TNFR1/MKK7/JNK/AP-1 cascade can promote the formation of human epithelial tumors [38, 39, 40]. In addition, the expression of JNK was also reported to be up-regulated in the thyroid cancer, breast cancer and colon cancer [41, 42, 43]. In this study, we found that claudin-6 overexpression can significantly down-regulated the expression of JNK.
In summary, our results showed that claudin-6 was closely related to the occurrence of gastric cancer, and claudin-6 overexpression may inhibit the activity of JNK/MAPK signaling pathway by up-regulating the expression of DUSP4, FN1 and HERC5 genes, and then promoting differentiation.
Footnotes
Conflict of interest
All authors do not have a commercial or other association that might pose a conflict of interest.
