Abstract
BACKGROUND:
The dysregulation of microRNA-802 (miR-802) has crucial roles in cancer progression. Nevertheless, the bio-function of miR-802 in cervical cancer remains unclear.
OBJECTIVE:
Hence, we illuminated the potential roles of miR-802 in cervical cancer cell growth, migration, and invasion.
METHODS:
The levels of miR-802 and myosin regulatory light chain interacting protein (MYLIP) were measured using qRT-PCR assay. The potential effects of miRNA-802 on cervical cancer cell proliferation and metastatic phenotypes were determined using CCK-8, colony formation, wound healing and Transwell invasion assays. MYLIP was validated as a downstream target gene of miRNA-802 using bioinformatics analysis tool and luciferase report gene assay. The impact of miR-802 on the growth of cervical cancer cell in vivo was analyzed using xenograft model. The expression of MYLIP was measured by western blotting and immunohistochemistry (IHC).
RESULTS:
MiRNA-802 was distinctly down-regulated in cervical cancer cells as well as clinical cervical cancer samples. Upregulation of miRNA-802 significantly inhibited the growth and aggressiveness of cervical cancer cell. Additional, MYLIP was a functional target of miR-802. MYLIP was ovrerexpressed in cervical cancer and MYLIP level was negatively associated with the level of miR-802. Overexpression of MYLIP eliminated the inhibitory effects of miR-802 on growth and metastatic-related traits of cervical cancer cell. In vivo, miR-802 also markedly reduced the tumor growth of cervical cancer cell and decreased the expression of MYLIP.
CONCLUSIONS:
MiR-802 inhibits the growth and metastatic-related phenotypes of cervical cancer cell through targeting MYLIP.
Introduction
Cervical cancer is the most common malignant tumor in gynaecology [1]. Although the surgical treatments and chemotherapy of cervical cancer have been improving constantly, the overall survival of patients with cervical cancer is still unsatisfactory, mainly owing to diffusion and metastasis rate [2]. Deep investigation of the metastasis of cervical cancer cell and exploration of novel treatment targets are urgently needed.
MicroRNAs (miRNAs), which are noncoding RNAs, post-transcriptionally regulate the expression of protein-coding genes by complementarily binding to the 3
This study mainly aims to illuminate the underlying mechanism of miRNA-802 in regulation of cervical cancer. We demonstrate that miR-802 is distinctly downregulated in human cervical cancer and miR-802 regulates cervical cancer cell growth, migration and invasion by modifying MYLIP expression.
Materials and methods
Cell culture and cervical cancer tissues
Cervical cancer cell lines (SiHa, CasKi and C33A) and the human cervical surface epithelial cell line, HcerEpic were obtained from Nanjing Keygen Biotech. Co., Ltd (Nanjing, Jiangsu, China). Cells were cultured in DMEM with 10% FBS supplemented with 1% penicillin/streptomycin. Cells were cultured in an atmosphere containing 5% CO
Association of miR-802 expression with clinicopathologic features in patients with cervical cancer
Association of miR-802 expression with clinicopathologic features in patients with cervical cancer
FIGO, International Federation of Gynaecology and Obstetrics.
MiR-NC (negative control), miR-802 mimics and miR-802 inhibitor were purchased from GenePharma (Shanghai, China). The sequence used was as follows: miR-802 sense, 5
Quantitative real-time PCR (qRT-PCR) assay
Total RNAs were abstracted from cells or tumor tissues using TRIzol reagent (Beyotime Biotechnology, Nanjing, Jiangsu, China). 1
Cell Counting Kit-8 (CCK-8) and colony formation assay
In cell proliferation assay, cervical cancer cells were cultured into 96-well plates. 10
Migration assay
Cervical cancer cells were seeded into six-well plates. After 24 hours, an artificial wound in each well was made using a 100
Invasion assay
Transwell chambers were coated with 30
Luciferase reporter gene assay
Fragments of MYLIP 3
Western blotting analysis
Proteins were extracted using RIPA Buffer (Beyotime Biotechnology, Nanjing, Jiangsu, China). Equal amounts of proteins were separated by 10% SDS-PAGE and transferred to PVDF membrane. The membrane was incubated with MYLIP (cat. ab74562, Abcam, Cambridge, UK) or GAPDH (cat. #5174, Cell Signaling Technology, Danvers, MA, USA) antibody (Cell Signaling Technology, Danvers, MA, USA). HRP-conjugated goat anti-rabbit IgG antibody (cat. #7074, Cell Signaling Technology) was used as secondary antibody. Target bands were visualized using ECL (Pierce).
Animal experimental
MiR-NC or miR-802 stable transfected SiHa cells (5
Levels of miR-802 expression. A. Expression levels of miR-802 in cervical cancer tissues and adjacent normal tissues. 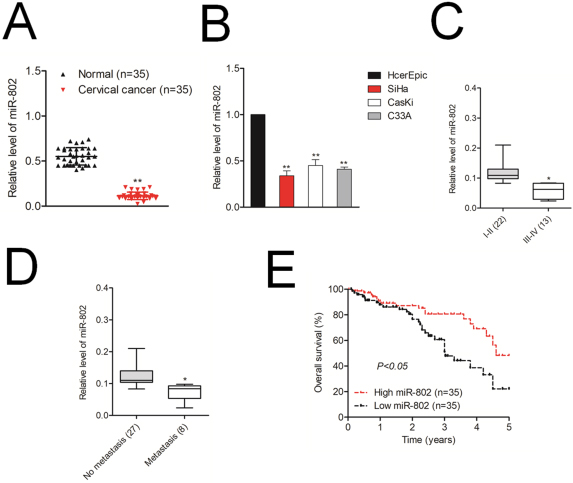
All data were analyzed using SPSS19.0 statistical software and were shown as Mean
Biological effects of miR-802 on cervical cancer cell. A. The levels of miR-802 in SiHa and CasKi cell was measured using qRT-PCR assay. B. Cell growth rate in SiHa and CasKi cell was determined by CCK-8 assay. C. Colony forming ability of SiHa and CasKi cells. D. Cell migration ability was measured in SiHa and CasKi cells. E. Cell invasion ability was measured in SiHa and CasKi cells. 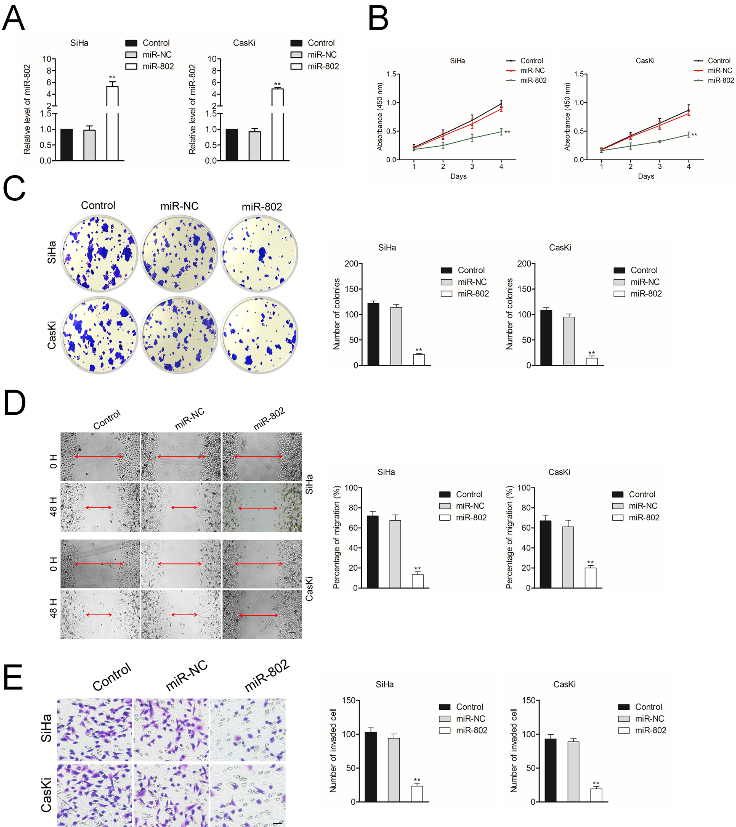
MiR-802 is downregulated in cervical cancer
To investigate to the level of miR-802 in cervical cancer tissues, 35 cases of patients with cervical cancer were collected and miR-802 level was analyzed using qRT-PCR analysis. As indicated in Fig. 1A, miR-802 was downregulated in cervical cancer samples when compared to that in corresponding normal samples. In addition, miR-802 was significantly downexpressed in three human cervical cancer cells (SiHa, CasKi and C33A) when compared to that in the human cervical surface epithelial cell line, HcerEpic (Fig. 1B). In addition, miR-802 level was inversely correlated with the progression of cervical cancer pathological grade and metastasis (Fig. 1C and D). Finally, the patients with cervical cancer harboring lower level of miR-802 showed significantly shorter overall survival than did the cervical cancer patients with higher level of miR-802 (Fig. 1E). All these results imply that miR-802 is downexpressed in cervical cancer and may be closely related to cervical cancer progression.
MYLIP is a target of miR-802. A. Bioinformatics analysis revealed predicted binding sites between miR-802 and MYLIP. B. The common targets of miR-802 identified using TargetScan and miRTarBase. C. Relative luciferase activity in SiHa and CasKi cell cotransfected with miR-802 mimic and MYLIP 3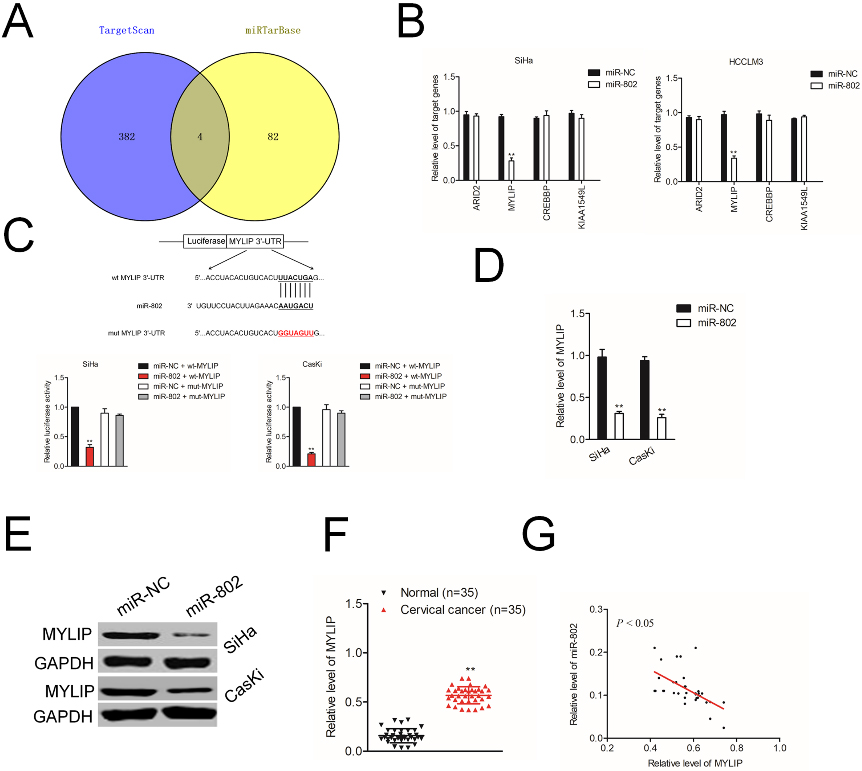
We next explored the effect of miR-802 on the aggressiveness of cervical cancer cell, SiHa and CasKi was transfected with miR-NC or miR-802. As shown in Fig. 2A, miR-802 level was increased after transfection of miR-802 mimics. Next, the results of CCK-8 test implied that the cell viabilities of SiHa and CasKi cell were significantly suppressed after miR-802 transfection (Fig. 2B). Consistently, colony formation assay indicated that in miR-802 overexpressing group, the colonies of SiHa and CasKi cell were distinctly reduced compared to that in miR-NC group (Fig. 2C). Wound healing assay and Transwell invasion was applied to uncover the influence of miR-802 on the migrate ability and invasion of cervical cancer cell in vitro. As shown in Fig. 2D and E, miR-802 overexpression remarkably inhibited the migration and invasion abilities of SiHa and CasKi. These results demonstrate that miR-802 exerts a cancer suppressive effect in the aggressive phenotype of cervical cancer cell.
Overexpression of MYLIP reverses the effects of miR-802 on cervical cancer cell. A. The levels of MYLIP in SiHa and CasKi cell that transfected with miR-802 mimic and pc-MYLIP were assessed using qRT-PCR assay. B. The levels of MYLIP in SiHa and CasKi cell that transfected with miR-802 mimic and pc-MYLIP were assessed using western blotting assay. Cell growth rate with miR-802 mimic and pcDNA3.1-MYLIP. C. CK-8 analysis of SiHa and CasKi cells cotransfected with miR-802 mimic and pc-MYLIP. D. Colony forming ability of H SiHa and CasKi cells cotransfected with miR-802 mimic and pc-MYLIP. E. miR-802 mimic and pc-MYLIP was cotransfected into SiHa and CasKi cell. The migration of cell was detected using wound healing assay. F. Invasion ability of SiHa and CasKi cell cotransfected with miR-802 mimic and pc-MYLIP was analyzed using Transwell invasion assay. 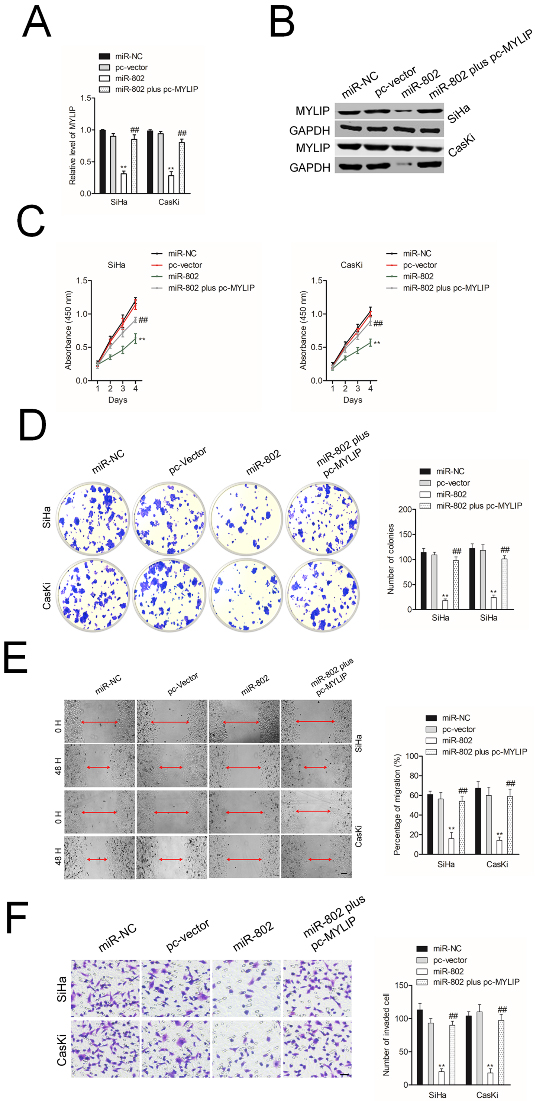
By using miRNAs bioinformatics tool (TargetScan and miRTarBase), four genes, including MYLIP were identified as the potential targets of miR-802 (Fig. 3A). Next, the results of qRT-PCR assay indicated that only the MYLIP gene was significantly inhibited in both miR-802 transfected SiHa and CasKi cell (Fig. 3B). The luciferase reporter assay was carried out and we observed that miR-802 transfection significantly inhibited the luciferase activity in cervical cancer cell transfected with wt-MYLIP whereas had no significant suppressive effect on luciferase activity in cervical cancer cell transfected with mut-MYLIP (Fig. 3C). Then, the levels of MYLIP in miR-802 transfected SiHa and CasKi were analyzed using qRT-PCR and western blotting assay. As shown in Fig. 3D and E, in SiHa and CasKi cell, the mRNA levels and protein expressions of MYLIP were both distinctly decreased in miR-802 overexpressing cell. Meanwhile, compared to normal tissues, the level of MYLIP was significantly higher in cervical cancer tissues (Fig. 3F). Additionally, an inversely relationship between miR-802 level and MYLIP level was observed in cervical cancer tissue as demonstrated by qRT-PCR analysis (Fig. 3G). All these data suggest that MYLIP is a target gene of miR-802.
Restoration of MYLIP abrogates the inhibitory impact of miR-802 on cervical cancer cell
To find the core role of MYLIP in miR-802 medicating cervical cancer cell aggressiveness, SiHa or CasKi cell was cotransfected with miR-802 and pc-MYLIP. As showed in Fig. 4A and B, the level of MYLIP was significantly reduced in miR-802 transfected cervical cancer cell as demonstrated by qRT-PCR and western botting assay. However, cotransfection of miR-802 mimics and pc-MYLIP impaired the suppressive impact of miR-802 on MYLIP expression. The CCK-8 results and colony formation assay suggested that in both SiHa and CasKi cell, miR-802 transfection repressed the cell proliferation and decreased colony formation ability (Fig. 4C and D). Additional, in SiHa and CasKi cell, the migration and invasion abilities were markedly decreased by miR-802 whereas cotransfection of miR-802 with pc-MYLIP abolished the inhibitory action of miR-802 on the migration and invasion (Fig. 4E and F). All these findings suggest that restoration of MYLIP abrogates the inhibitory impacts of miR-802 on cervical cancer cell.
MYLIP silencing abrogates the promoting impact of miR-802 inhibitor on cervical cancer cell. A. The levels of MYLIP in SiHa and CasKi cell that transfected with miR-802 inhibitor and siMYLIP were assessed using qRT-PCR assay. B. The levels of MYLIP in SiHa and CasKi cell that transfected with miR-802 inhibitor and siMYLIP were assessed using western blotting assay. Cell growth rate with miR-802 inhibitor and siMYLIP. C. CK-8 analysis of SiHa and CasKi cells cotransfected with miR-802 inhibitor and siMYLIP. D. Colony forming ability of SiHa and CasKi cells cotransfected with miR-802 inhibitor and siMYLIP. E. miR-802 inhibitor and siMYLIP was cotransfected into SiHa and CasKi cell. The migration of cell was detected using wound healing assay. F. Invasion ability of SiHa and CasKi cell cotransfected with miR-802 inhibitor and siMYLIP was analyzed using Transwell invasion assay.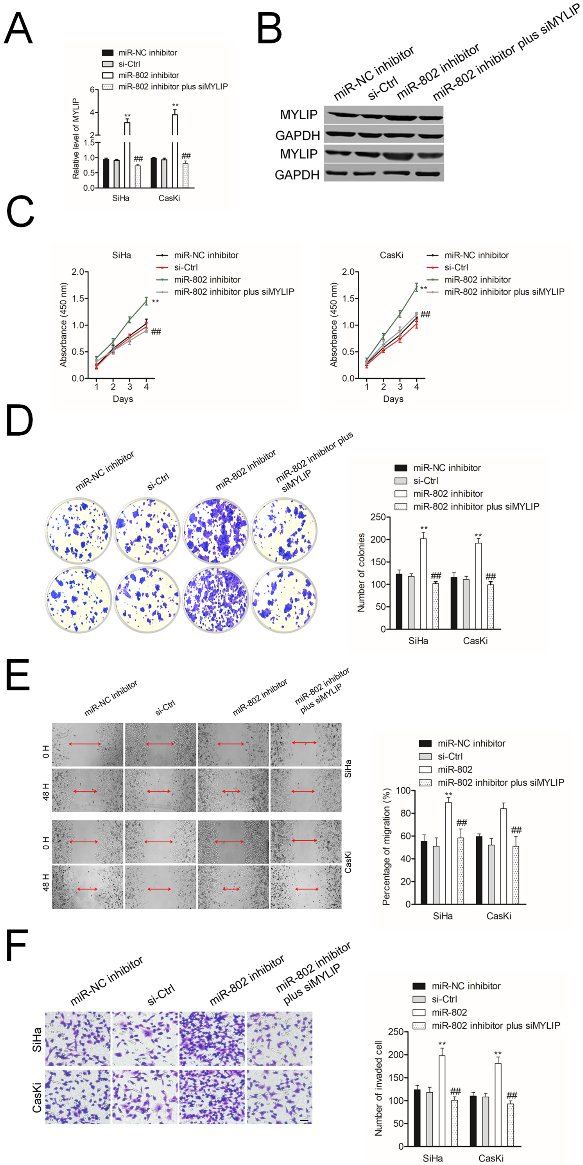
To confirm the core role of MYLIP in miR-802 medicating cervical cancer cell aggressiveness, SiHa or CasKi cell was cotransfected with miR-802 inhibitor and siMYLIP. As showed in Fig. 5A and B, the expression of MYLIP was raised in miR-802 inhibitor transfected cell. However, cotransfection of miR-802 inhibitor and siMYLIP impaired the promoting impact of miR-802 inhibitor on MYLIP expression. The CCK-8 results and colony formation assay suggested that in SiHa and CasKi cell, miR-802 inhibitor increased the proliferation and colony formation ability. However, cotransfection of miR-802 inhibitor and siMYLIP reversed the promoting effect of miR-802 inhibitor on SiHa and CasKi cell proliferation and colony formation (Fig. 5C and D). Additional, in SiHa and CasKi cell, the migration and invasion abilities were markedly increased by miR-802 inhibitor whereas cotransfection of miR-802 inhibitor with siMYLIP abolished the promoting impact of miR-802 inhibitor on the migration and invasion (Fig. 5E and F). All these findings suggest that MYLIP silencing abrogates the promoting impact of miR-802 inhibitor on cervical cancer cell.
MiR-802 inhibits SiHa cell growth in vivo. A. The tumor volumes of BALB/c nude mice injected by SiHa cells transfected with miR-802 or miR-NC. B. The tumor weights were measured for miR-802 and miR-NC groups. C. The body weights of BALB/c nude mice injected by SiHa cells transfected with miR-802 or miR-NC. D. The miR-802 level in miR-802 and miR-NC group. E. The level of MYLIP in miR-802 and miR-NC group was detected using qRT-PCR assay. F. The immunohistochemical (IHC) staining pictures for Ki67 in miR-802 and miR-NC group. G. MYLIP-stained sections of transplanted tumors in miR-NC or miR-802 group. 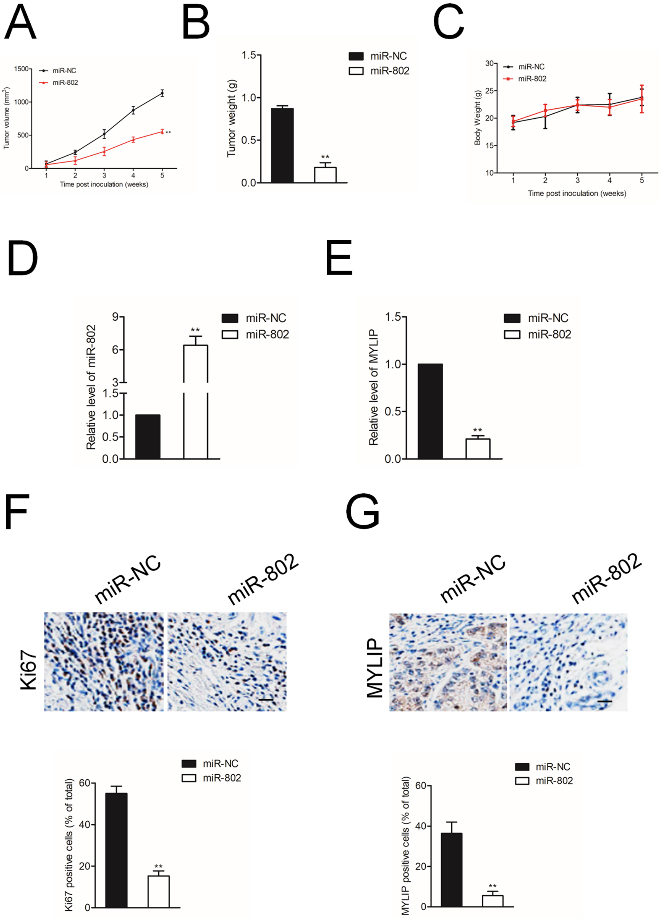
Finally, we validated the essential functions of miR-802 in cervical cancer cell growth in vivo. Xenograft model was constructed by injection miR-802 stable transfected SiHa cell into nude mice. The tumor volume and body weight of nude mice were recorded each week. As shown in Fig. 6A and B, the tumor volume and tumor weight in mice injected with miR-802 transfetced SiHa cell were significantly smaller than miR-NC group. Nevertheless, the body weight of two groups didn’t exhibit significant difference (Fig. 6C). 35 days after the inoculation, tumor tissues were separated and subjected for qRT-PCR test. As shown in Fig. 6D-6E, the level of miR-802 was markedly up-regulated in miR-802 transfected group, and the level of MYLIP was downregulated in miR-802 transfected group. The immunohistochemical (IHC) staining using Ki67 antibody indicated that miR-802 significantly decreased the proliferation of SiHa cell in vivo (Fig. 6F). Then, IHC staining was conducted using tumor tissue to detect the expression of MYLIP (Fig. 6G) and we observed that MYLIP was remarkably downregulated in miR-802 transfected group. All these findings indicate that miR-802 inhibits the growth of cervical cancer cell in vivo.
Discussion
Cervical cancer has the high mortality rate among malignancies. Its survival rate is still very low due to recurrence and metastasis [15, 16]. Hence, a better investigation of the precise mechanism of cervical cancer metastasis has important clinical value for the effective treatment of cervical cancer. Increasing evidences have suggested that miRNAs act as vital regulators in a variety of cancer cell biological processes, including growth, invasion, and metastasis. Therefore, miRNAs can be used as effective diagnostic markers and drug targets for therapy [17]. The crucial functions of miRNAs in the progression of cervical cancer have been identified in recent decades. In this study, miR-802 was down-regulated in cervical cancer tissues and cell lines. Decreased level of miR-802 was associated with the advanced progression and poor prognosis of patients with cervical cancer. These observations indicated miR-802 might be a promising diagnostic biomarker and target for the treatment of cervical cancer.
Previous investigations have revealed the essential actions of miR-802 in modulating the development of cancer [6, 12]. In glioblastoma multiforme (GM), miR-802 suppresses the cell proliferation, invasiveness, and epithelial-mesenchymal transition (EMT) of GM cell through modulating SIX4 [18]. Some reports have indicated that miRNA-802 is dysregulated in various cancers [6, 8, 19]. The expression level of microRNA-802 is down-regulated in ovarian cancer related cell lines and tissues [8]. MiR-802 is also significantly downregulated in colorectal cancer tissues and cell lines. Overexpression of miR-802 profoundly inhibited viability, migration and invasion of colorectal cancer cells [20]. All these observations have demonstrated that miR-802 plays as a tumor suppressor gene in cancers. In cervical cancer, we observed that miR-802 was remarkably downexpressed in clinical tissues. Consistently, miR-802 was also downregulated in cervical cancer cell lines when compared to that in HcerEpic cell. Meanwhile, the low level of miR-802 was closely related with the advanced stage and poor prognosis of patients with cervical cancer. Importantly, the lower level of miR-802 related to the poor overall survival of patients with cervical cancer. Enforced expression of miR-802 results in the reduction growth and metastasis in tongue squamous cell carcinoma and prostate cancer cells [7, 21]. We proved that upregulation of miR-802 suppressed the cell growth and colony formation of cervical cancer cell. In addition, transfection of miR-802 significantly reduced the migration and invasion abilities of cervical cancer cell in vitro.
To investigate the mechanism responsible for miR-802 mediated anti-cancer functions in cervical cancer, the target of miR-802 in cervical cancer was analyzed. Interestingly, myosin regulatory light chain interacting protein (MYLIP) was identified as a target of miR-802 using the bioinformatics databases. MYLIP, which belongs to the cytoskeletal protein clusters, is involved in the regulation of cell migration and movement [22]. Through interacting the cell membrane proteins with myosin cytoskeleton, MYLIP plays an important function in the maintenance of cellular morphology, the remodeling of cytoskeletal proteins, the modulation of cell motility and the adhesion of cells with extracellular matrix (ECM) [23]. In the current study, miR-802 bound the 3’-UTR of MYLIP as demonstrated by luciferase reporter gene assay. In addition, overexpression of miR-802 inhibited the expression of MYLIP in cervical cancer cell. Importantly, there was a negative relationship between miR-802 level and MYLIP level was observed.
Furthermore, to validate the crucial function of MYLIP and its relationship with miR-802 medicating cervical cancer cell metastasis, cervical cancer cell was transfected miR-802 and MYLIP overexpression plasmid. We proved that restoration of MYLIP significantly reversed the inhibitory actions of miR-802 on the aggressiveness of cervical cancer cell. As expected, in cells cotransfected with miR-802 inhibitor with siRNA targeting MYLIP, the promoting impacts of miR-802 on the growth, invasion and migration of cervical cancer cell were reversed by MYLIP siRNA. All these findings suggest that miR-802 restrains the aggressive phenotypes of cervical cancer cell by regulating MYLIP.
In conclusion, our data demonstrated the crucial functions of miR-802 in human cervical cancer. MiR-802 was down-expressed in cervical cancer and was associated with the poor clinical outcomes of patients with cervical cancer. Upregulation of miR-802 restrains the growth, migration, and invasion of cervical cancer cell through modulating MYLIP. We establish a potential cervical cancer-associated miR-802-MYLIP regulatory network and explore its underlying mechanisms involved into cervical cancer progression.
Footnotes
Conflict of interest
The authors declare no conflict of interest.
