Abstract
BACKGROUND:
It is essential in the management of ovarian cancers to identify the patients who will benefit from primary complete cytoreductive surgery and those who will rather benefit from neoadjuvant chemotherapy.
OBJECTIVE:
To evaluate the predictive value of preoperative inflammatory markers, i.e. platelet to lymphocyte ratio (PLR), neutrophil to lymphocyte ratio (NLR), monocyte to lymphocyte ratio (MLR), red cell distribution width (RDW), and serum CA125 level for surgical outcome in epithelial ovarian cancer.
METHODS:
A retrospective study was carried out in Sanglah Hospital, Denpasar, Bali. A total of 54 patients with epithelial ovarian cancer who underwent primary exploratory laparotomy from January 2018 to November 2019 was recruited. Data about clinical characteristics, preoperative inflammatory markers, serum CA125 level, and surgical outcome (optimal vs. suboptimal) was collected from the medical records. Predictive value of the markers were evaluated using ROC curve to determine their accuracy (area under the curve, sensitivity, specificity, positive and negative predictive value).
RESULTS:
Mean age, parity, and tumor size did not differ between the study groups (
CONCLUSION:
Preoperative serum inflammatory markers (PLR, MLR, and RDW) may serve as useful markers to predict the surgical outcome with fair accuracy in patients with epithelial ovarian cancer.
Introduction
Ovarian cancer still has the highest mortality rate among malignant tumors of the female reproductive system [4]. Its mortality rate among Indonesian women even ranks the highest in Asia (6.1 per 100.000 population) [20]. One of Indonesian survey organization, Riskesdas 2007, reported that around 20% (927 people from 4,647 cancer respondence) diagnosed as ovarian or cervical cancer and 795 people of them in reproductive age (20–59 years-old) with low socioeconomic background [7]. From 1989–1992, there was 1,726 cases of gynecological cancer in Dr. Cipto Mangunkusumo, Jakarta, Indonesia and 13.6% was ovarian cancer. Patients proned to visit medical healthcare when stage II-IV ovarian cancer. It was reported that ovarian cancer costed 22.6% death from 327 death of gynecological cancer. Data from 1996–2001, Dr. Cipto Mangunkusumo Hospital obtained 214 patients with ovarian cancer. The 5-year survival rate on stage I-IV about 76.39%, 66.67%, 24.64%, and 8.13%, respectively, with median survival approximately 1,294 days [22]. Another study from Prof Dr. RD Kandou Manado, Indonesia included 95 patients with ovarian cancer from 2013–2015 showed the majority cases were women aged
The prognosis is poor for advance stage, in which the five year survival rate is only 25% [17]. The major prognostic factor that determines the survival of patients with ovarian cancer is the residual tumor after primary cytoreductive surgery [8]. One meta-analysis suggested that for each 10% increase in the proportion of patients undergoing complete cytoreduction to no gross residual disease (R0) was associated with a significant and independent 2.3-month increase in cohort median survival compared to a 1.8-month increase in cohort median survival for optimal cytoreduction (residual disease
The ability to identify a subset of patients who are most likely to achieve optimal cytoreduction/R0 is of paramount importance. Poor candidate of surgery who are less likely to achieve that ideal target may have delayed chemotherapy after unnecessary laparotomy and their prognosis can be adversely affected. If optimal cytoreduction/R0 is difficult to attain at primary cytoreduction, the use of neoadjuvant chemotherapy followed by interval debulking to allow for R0 is recommended [12]. Currently, there are no general consensus on the preoperative method for establishment of cytoreducibility. Several studies have suggested the use of preoperative imaging modalities (laparoscopy, CT, PET scan) or serum biomarkers (CA 125, HE-4) but those modalities are sometimes expensive and inaccessible for low-resource setting that commonly present in developed country such as Indonesia. Identification of low-cost biological indicators that can help to improve prediction of the surgical outcome will be crucial.
Cancer is a pro-inflammatory state. On the other hand, it was suggested that inflammatory reaction and immune status have played a pivotal role in tumorigenesis and cancer progression, i.e. they generate a tumor-related inflammatory microenvironment which exerted an important role in the pathogenesis and development of tumors [6, 16, 10]. Inflammation often manifests as neutrophilia, thrombocytosis and relative lymphopenia in the peripheral blood [11]. Several inflammatory markers which are simply obtained from peripheral blood count such as absolute platelet count (PLT), platelet to lymphocyte ratio (PLR), monocyte to lymphocyte ratio (MLR), neutrophil to lymphocyte ratio (NLR), and the red cell distribution width (RDW) are gaining much attention for its role in predicting cancer progression and survival. In a study by Qin et al. [18], it was reported that the RDW, absolute neutrophil count (N), NLR, PLR, and CA-125 level were significantly higher in the ovarian cancer than benign ovarian tumor. Thus, serum inflammatory markers obtained from peripheral blood count may serve as potential predictors for surgical outcome in patients with epithelial ovarian cancer undergoing primary cytoreductive surgery.
This study is aimed to evaluate the predictive value of preoperative inflammatory markers obtained from the peripheral blood count (PLR, NLR, MLR, and RDW) and serum CA125 level for tumor resectability in epithelial ovarian cancer. These parameters are convenient, inexpensive, and simple to measure for the low resource setting.
Methods
Design
This is a retrospective study that was carried out in Gynecologic Oncology Division, Obstetric and Gynecology Department, Sanglah Hospital/Faculty of Medicine Udayana University, Bali, Indonesia, during January 2018 to November 2019. This study is approved by the Ethical Committee Faculty of Medicine Udayana University (No. 1921/UN14.2.2.VII.14/LT/2020).
Characteristics of study population
Characteristics of study population
*Normal distribution.
Patients with ovarian cancer who underwent primary exploratomy laparotomy from January 2018 to November 2019 and had given their written consent to participate in this study were recruited into the study population Patients with non epithelial type of ovarian cancer, those who underwent conservative staging, incomplete data about inflammatory markers, those with systemic diseases (diabetes mellitus, cardiovascular disease, kidney disease, blood disease, acute inflammation, anemia), recent iron therapy, venous thrombosis for period of
Variables and measurement
Data about the clinical characteristics (i.e., age parity tumor size, FIGO stage, histologic type), preoperative inflammatory markers, and serum CA 125 level were all obtained from the medical records of the patients. We used the 2014 FIGO ovarian cancer staging system to determine the stage of our patients. In our hospital, preoperative venous blood samples (3 mL) were obtained from each patient from their first visit to the Gynecologic Oncology Outpatient Clinic and placed in EDTA-K2 anticoagulation tubes Whole blood cell parameters were determined with a Cell Dyn Ruby
Serum inflammatory markers and CA125 level across the study population
Serum inflammatory markers and CA125 level across the study population
*Normal distribution.
Optimized cut off value and AUC for each serum inflammatory markers and CA125
Surgical outcome was classified as optimal or suboptimal and determined using Gynecologic Oncology Group criteria (i.e., optimal cytoreduction: a post-operative surgical residuum of
Data were analyzed using SPSS version 24.0. Univariate analysis was used to generate frequencies and percentages of categorical variables. Continuous variables were presented as mean
Results
The characteristics of the study population were summarized in Table 1. Mean age, parity, and tumor size did not differ between the study groups (
The group with suboptimal outcome had significantly higher PLR, NLR, MLR, and RDW value (
Using the ROC curve (Fig. 1), a cut off value and AUC was optimized for each predictor (Table 3). Sensitivity, specificity, positive and negative predictive value, and accuracy for predicting suboptimal outcome were summarized in Table 4. The association between the inflammatory marker and surgical outcome were summarized in Table 5. The strongest association were observed for PLR, followed by RDW and MLR, consecutively.
The ROC curves of each inflammatory markers and CA-125.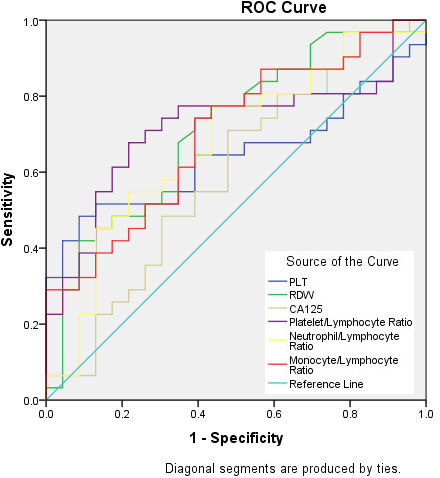
Sensitivity, specificity, positive and negative predictive value, and accuracy for each predictor in predicting suboptimal outcome
The association between the inflammatory marker and surgical outcome
In this study, we found that the level of serum inflammatory markers were significantly higher in the group of patients with suboptimal surgical outcome. Three inflammatory markers, i.e. PLR, MLR, and RDW, can predict the surgical outcome with fair accuracy. A similar result was reported by Ashrafganjoei et al. [2]. They reported that a statistically significant relation was found between surgical outcomes and PLR (
Platelets are involved in key steps of malignancy progression [3]. The role for platelets has been demonstrated in tumor metastasis, tumor growth and angiogenesis. Bambace and Holmes [3] proposed several roles of platelets in tumorigenesis, including: i) tumor cell-induced platelet aggregation following tumor cell intravasation into the vasculature, thereby the platelets “protect” or “cloak” the circulating tumor cells from physical clearance and immune surveillance, (ii) platelets facilitate tumor cell arrest within the vasculature, endothelial cell retraction and subsequent tissue invasion, (iii) platelets induce endothelial cell proliferation and angiogenesis, and (iv) platelet-tumor and platelet stromal interactions in the tumor microenvironment depend, in part, on platelet activation and platelet protein release, which contribute to the inflammatory response. Furthermore, platelet-related proteins and metabolites that facilitate proteolysis and tissue remodeling also enhance tumor growth and metastasis. Thrombocytosis found in ovarian cancer can be explained through a mechanism in which ovarian cancer produce the inflammatory cytokine interleukin-6 (IL-6) and induce platelet-production regulating hormone thrombopoietin (TPO) in the liver, which in turn, stimulate thrombopoiesis [19].
PLR and NLR are two of the inflammatory response markers, which have been reported to relate with cancer prognosis in various cancers [24, 25]. In a recent meta-analysis by Zhao et al. [28], it was suggested that PLR and NLR could be effective prognostic predictors of ovarian cancer. High PLR adversely affected OS (pooled HR 2.05, 95% CI 1.70–2.48) and PFS (pooled HR 1.85, 95% CI 1.53–2.25) in ovarian cancer. Similarly, high NLR had a poor prognostic impact on OS (pooled HR 1.70, 95% CI 1.35–2.15) and PFS (pooled HR 1.77, 95% CI 1.48–2.12) in ovarian cancer. Another meta-analysis by Zhu et al. [29] also reported similar result. PLR and NLR are simple and easily accessible markers that can be used to predict the prognosis of ovarian cancer patients.
The role of RDW in malignant tumors has also attracted much attention. The RDW is a measure of the range of variation in the size of erythrocyte. The RDW reflects erythrocyte cell volume heterogeneity and is a part of the whole blood cell count. A recent meta-analysis by Wang et al. [26] that involved 49 studies with 19,790 individuals reported that high RDW level adversely affected both OS and DFS/PFS/RFS. Colorectal cancer (CRC) had the strongest relationship with poor OS, followed by hepatic cancer (HCC). Negative OS outcomes were also observed in hematological malignancies [26].
The association between RDW and ovarian cancer has been reported in a recent study by Qin et al. [18]. The RDW was negatively correlated with the hemoglobin concentration and positively correlated with the cancer stage, NLR, PLR, and CA 125 level. The AUC of the RDW was 0.876 (95% confidence interval 0.829–0.923). In our study, RDW had the second best performance in predicting the surgical outcome, the AUC was 0.712 (95% CI: 0.572–0.852) and it had the same sensitivity level as PLR with a cut off value of 13.19%. Wang et al. [26] reported the cut off value of RDW between 13% and 14% was associated with worse HRs for OS and DFS/PFS/RFS than others. A study by Li et al. [15] reported that RDW with cutoff 14.5% had independent prognostic significance for OS in patients with epithelial ovarian cancer. The mechanism on how RDW is increased in ovarian cancer is still unclear. In our study, we did not collect other relevant laboratory data regarding inflammatory factors (e.g., inflammatory mediators, the erythrocyte sedimentation rate and C-reactive protein), erithropoietin, vitamin B6 and B12 concentrations, and serum ferritin concentration due to costly issues. These indicators can help to clarify more about elevated RDW’s mechanism in patients with ovarian cancer. To our knowledge, our study is the first study to report the role of RDW in predicting the surgical outcome in patients with epithelial ovarian cancer. Thus, we propose that RDW is a potential marker of ovarian cancer prognosis.
In a study by Xiang et al. [27], preoperative MLR was reported to be a significant predictor of advanced stages, advanced pathologic grades, and positive lymphatic metastasis in ovarian cancer patients. The optimal cut off value of MLR was reported to be 0.23. In this study, MLR was able to predict the surgical outcome of patients with epithelial ovarian cancer, the AUC was 0.700 (95% CI: 0.560–0.839), sensitivity 74.2%, specificity 59.1%, and optimal cut off value was set to be 0.24.
In this study, the performance of CA 125 in predicting the surgical outcome was poor. With a cut off value of 300.85, the AUC was 0.593 (95% CI: 0.436–0.750), sensitivity was 54.8%, and specificity was 60.9% However, in a recent study by Arab et al. [1], they suggested the best cut-off point for the serum CA125 level to distinguish suboptimal debulking was 420 U/ml with 73.0% (95%CI: 0.5590.862) sensitivity and 78.3% (95% CI 0.684–0.862) specificity. When we adopted this cut off value, the sensitivity and specificity of CA 125 in predicting the suboptimal outcome was 45.2% and 69.6%, respectively. A higher cut off value was proposed in a meta-analysis by Skang et al. A cut-off value of 500 U/ml gave a sensitivity of 68.9% (95% CI 62.0–75.1%) and a specificity of 63.2% (95% CI 53.7–71.7%). However, the optimal cut off value for CA 125 that best correlate with the surgical outcome still need to be determined in further study.
Our study is subjected to several limitations. First, our sample is relatively small. Second, we included patients with epithelial ovarian cancer in all stages. Finally, the follow-up studies should be require to make these results stronger. The patients who are most likely to benefit from optimal cytoreduction are those with advance stage (i.e. FIGO stage III to IV), thus a study selectively involving patients in advance stage is still needed. However, our study is the first study that investigate multiple inflammatory markers in predicting the surgical outcome in patients with epithelial ovarian cancer in Indonesia, from which the data are rarely reported. To date, there is no specific study to explain about hormone-related factors from patients.
Conclusion
Preoperative serum inflammatory markers such as PLR, MLR, and RDW may serve as useful markers to predict the surgical outcome with fair accuracy in patients with epithelial ovarian cancer.
