Abstract
BACKGROUND:
Breast cancer (BC) is a common malignancy in women. Some molecules, including endocan, are still under investigation as potential prognostic factors in BC.
OBJECTIVE:
In the present study, we aimed to determine the relationship between endocan expression and clinicopathological prognostic parameters in BC.
METHODS:
Two hundred and fifty-five patients diagnosed with BC were included in the present study. The immuno-reactivity scoring (IRS) system was used to reveal the tissue endocan expression levels.
RESULTS:
We found that endocan expression is associated with tumor necrosis, tumor size, and the presence of lobular carcinoma in situ (LCIS) in BC. There was no relationship between endocan expression and survival as well as other clinicopathological prognostic parameters.
CONCLUSION:
Endocan overexpression in BC is associated with some prognostic parameters, including tumor necrosis, tumor size, and the presence of LCIS. Further studies with larger series are needed to reveal the clinical and therapeutic implications of endocan in BC.
Introduction
Breast cancer (BC) is the most common malignancy in women worldwide [1, 2]. The incidence of cancer, particularly BC, and its mortality rate, has been increasing for the last 50 years. BC is the leading cause of death among women, accounting for 15% of all cancer deaths [2, 3]. In BC, well-known prognostic factors are the patient’s age, tumor grade, tumor size, axillary lymph node status, and vascular invasion [2, 4]. Given the long latency and relatively young age of onset of BC, the ability to predict prognosis would be of great value when selecting the optimal therapy for each patient and avoiding overtreatment [5].
Some molecules involved in breast carcinogenesis are potential prognostic factors, and hormone receptors, markers of proliferation (e.g., Ki67), and EGFR-2 (Her-2) are some of these prognostic factors. Other markers, such as cyclin D1, cyclin E, and plasminogen activators and inhibitors are under investigation but are not yet used routinely [2, 4].
Endocan, also called endothelial cell-specific-molecule-1 (ESM-1), is a dermatan sulfate proteoglycan and a product of endothelial cells. It is highly regulated by vascular endothelial growth factor (VEGF) and some cytokines, such as tumor necrosis factor alpha (TNF alpha), interleukin-1 (IL-1) and fibroblast growth factor-2 (FGF-2) [1, 6]. Endocan may be secreted by tumor endothelium as well as normal tissue endothelium, such as kidney, colon, and lung endothelia [1]. There are studies that focus on serum endocan levels or tissue expressions as a marker of prognosis in different cancer types, such as colorectal, hepatocellular, ovarian, renal cancers, and pancreatic neuroendocrine tumors [7, 8, 9, 10, 11]. In BC, the relation between serum endocan levels and prognosis has been investigated in a few studies [1, 12]. Based on our literature research, there are no studies that employ endocan immunohistochemistry in BC.
In the present study performed using immunohistochemistry, we aimed to determine the association between traditional prognostic factors, such as tumor grade, tumor stage, histological and molecular subtype, hormone receptor status, lymph node metastasis, distant metastasis and survival, and endocan expression in BC.
Material and methods
After approval from the local ethics committee was obtained, 255 consecutive patients with BC who had undergone modified radical mastectomy between January 2009 and December 2018, were included in the study. Relevant data, such as age, tumor diameter, multifocality, tumor stage, lymph node metastasis, distant metastasis, survival, and hormone receptor status, were obtained from patients’ records. Hematoxylin-eosin stained pathology slides were examined by two pathologists (E.C and F.D) in order to assess pathological prognostic parameters, such as histologic subtype, tumor grade, and the presence of tumor necrosis and to determine the appropriate tissue block for immunohistochemical staining.
The clinical and pathological staging of the cases were reevaluated based on the American Joint Committee on Cancer (AJCC) 2018 TNM BC staging system.
The molecular subtypes of the tumors were determined based on the ki-67 proliferation index, Her-2, and the hormone receptor status of the tumors. Accordingly, five different subtypes were obtained. The luminal subtypes A and B indicate hormone receptor positive breast cancer. The most common type is luminal A, which has a low grade, low Ki 67 score, and good prognosis. The luminal B subtype expresses more proliferation and her-2 genes and fewer ER-related genes. This subtype can be divided into two categories according to her-2 positivity or negativity. Her-2 and triple negative BC subtypes are high grade and more aggressive types displaying high systemic and local risks [1].
Tumors were divided into two categories based on IRS (low IRS group [IRS
Immunohistochemical staining procedure
Paraffin-embedded tissues of the chosen slides were collected and 4
The sections were deparaffinized at 60
Evaluation and scoring of immunohistochemically stained slides
The immunohistochemical slides stained with endocan, were evaluated and scored by a pathologist (EC) who was blinded to patients’ data. The entire slide was viewed at 100X magnification using a light microscope. The IRS system was used to determine endocan expression levels. This system, previously used by Wang et al. [13], depends on multiplication of staining intensity and the percentage of positive cells. The IRS system is explained in Table 1.
Immunorectivity scoring (IRS) system of Endocan
Immunorectivity scoring (IRS) system of Endocan
IR score
SPSS v25.0 was used for statistical analysis. The Kolmogorov-Smirnov Z test was used to test the normality of the distribution. Parametric analysis methods were employed for normal distributions, and for abnormal distributions, nonparametrical tests were preferred. When two groups were compared, independent t tests were used to determine statistical significance. Analysis of variance (ANOVA) and nonparametric Kruskall-Wallis tests were used to compare the mean scores of more than two groups. Statistical significance was considered as
Results
Two hundred and fifty-five patients diagnosed with BC were evaluated in this study. All patients were female, and their mean age was 55 years (24–88).
We observed cytoplasmic staining in tumor cells with endocan (Figs 1 and 2). Endocan expression was reduced in normal acini compared to tumor cells (Fig. 2).
Strong and diffuse cytoplasmic Endocan staining in a grade 2 tumor (Endocan, Original Magnification, X200).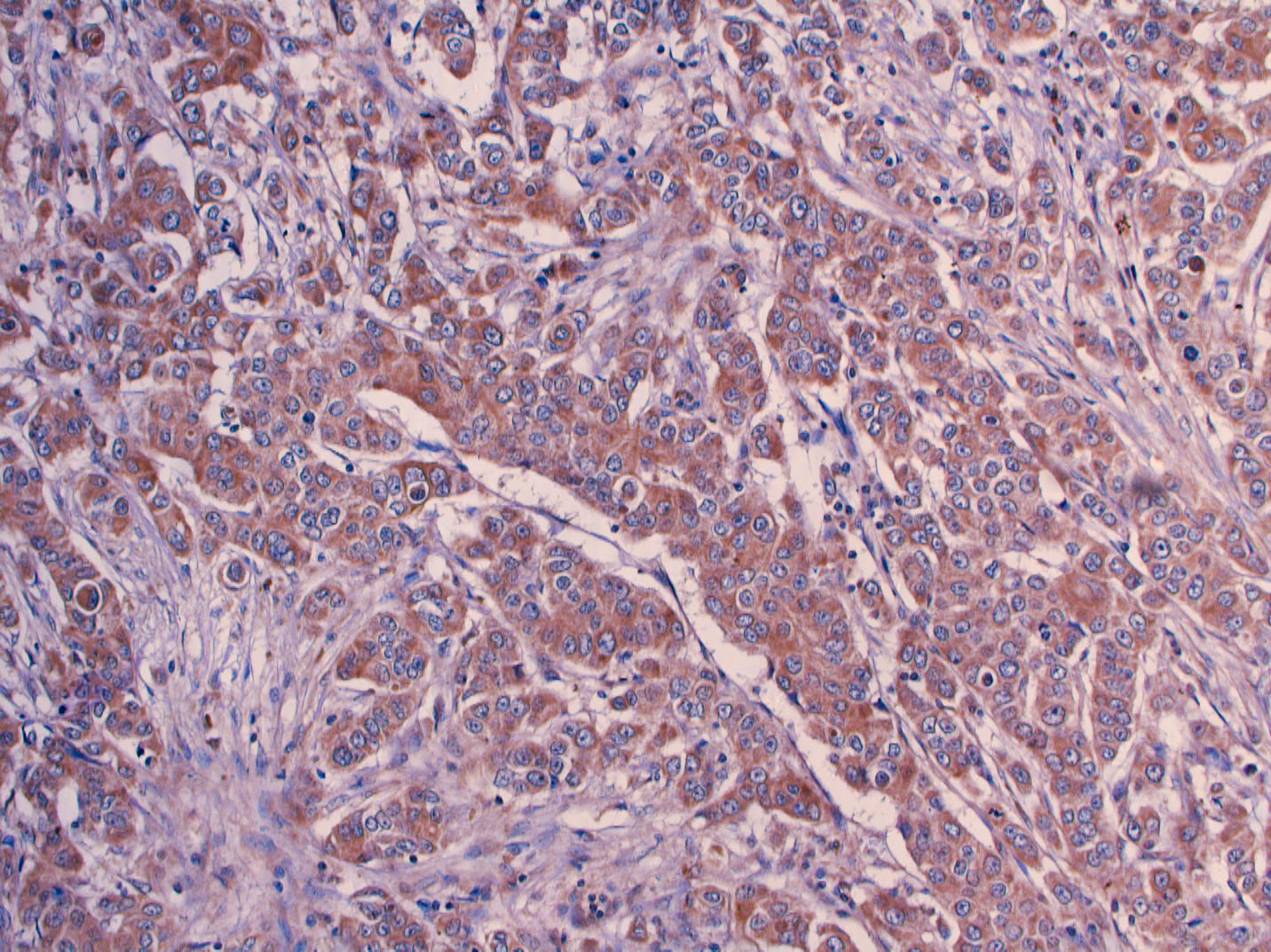
Endocan staining was mild in normal acini compared to adjacent tumor tissue (Endocan, Original Magnification, X200).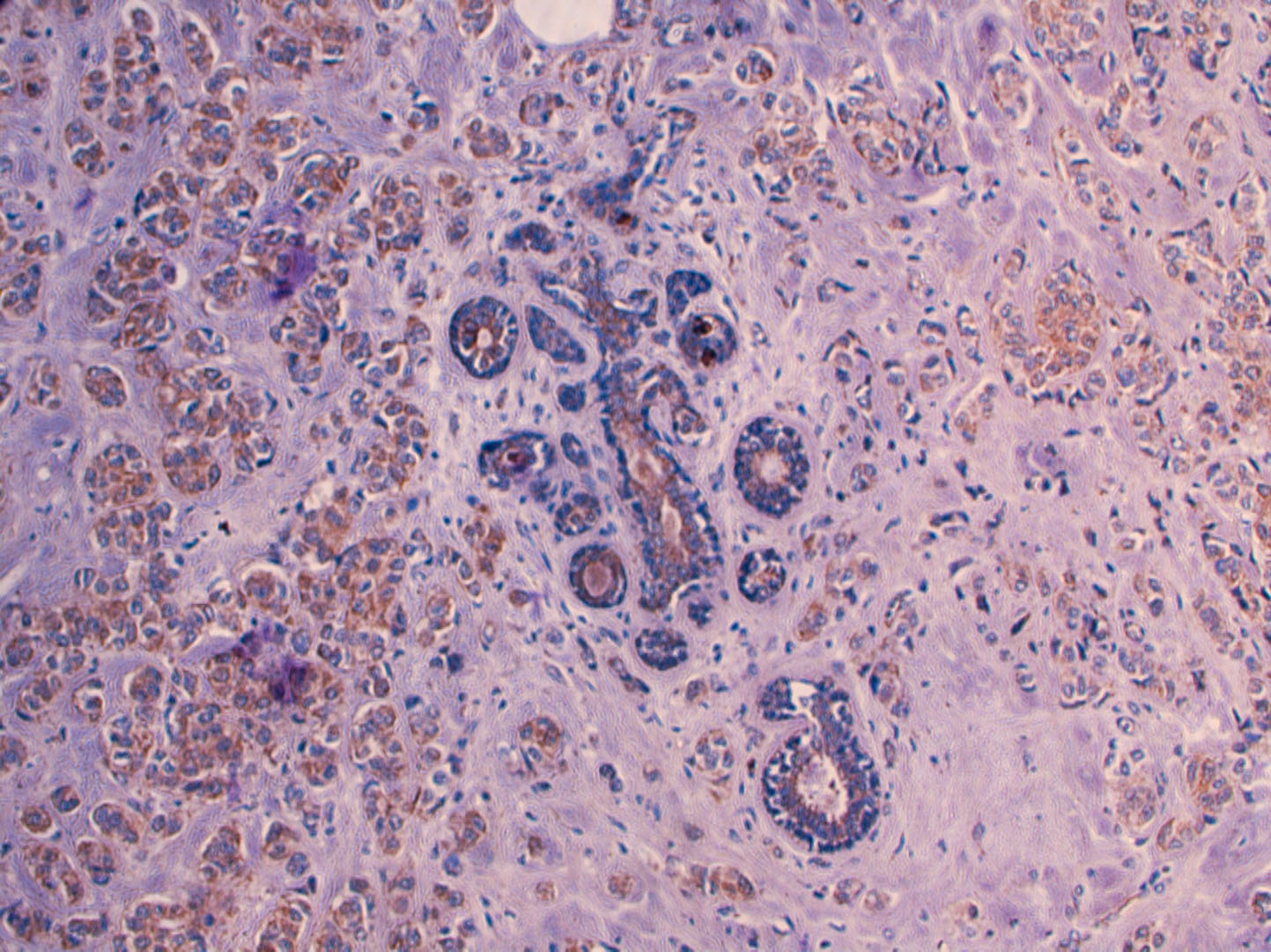
The clinicopathological characteristics of the patients are summarized in Table 2. Two hundred and twenty-five patients were diagnosed with invasive carcinoma, not otherwise specified (NOS) and its variants. Of the variants, five cases (1.9%) were carcinoma with apocrine differentiation, four (1.6%) cases were invasive carcinoma (NOS), medullary variant, one (0.4%) case was invasive carcinoma (NOS) with neuroendocrine differentiation, and one (0.4%) case was invasive carcinoma (NOS), pleomorphic variant. Other invasive carcinomas included 16 (6.4%) cases of invasive lobular carcinoma, three (1.2%) cases of invasive papillary carcinoma, three (1.2%) cases of cribriform carcinoma, one (0.4%) case of metaplastic carcinoma, four (1.6%) cases of mucinous carcinoma, one (0.4%) case of secretory carcinoma, and two (0.8%) cases of tubular carcinoma. No statistically significant difference was detected between histologic subtypes of BC in terms of endocan expression (
Relation of endocan expression with clinicopathological characteristics
Of the 16 tumors diagnosed as invasive lobular carcinomas, nine had accompanying lobular carcinoma in situ (LCIS). In these cases, the endocan expression was higher compared to those with ductal carcinoma in situ (DCIS) or without DCIS or LCIS (
In our study, the smallest tumor was 0.2 cm, while the largest was 14 cm. The mean tumor diameter was 3.1 cm. No statistically significant difference was observed between T stages in terms of endocan expression. However, when we divided the cases into two categories according to the size of the tumors (
Thirty-one tumors had necrosis. Tumors without necrosis showed higher endocan expression than tumors with necrosis (
Table 3 summarizes the relationship between endocan expression and progression, relapse, and site of metastasis. There was no statistically significant relationship between endocan expression and progression, relapse status, or site of metastasis (
Relation of progression, relapse and site of metastasis with endocan expression
Comparison of OS of the patients in the different IRS groups
Comparison of PFS of the patients in the different IRS groups
The median follow-up time was 54 (1–120) months. We compared the tumors with IRS system scores
Kaplan Meier curves for progression free survival (PFS) of Endocan expressions.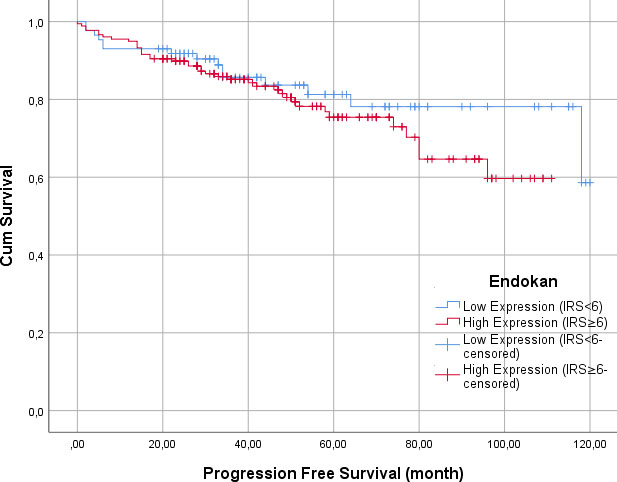
Kaplan Meier curves for overall survival (OS) of Endocan expressions.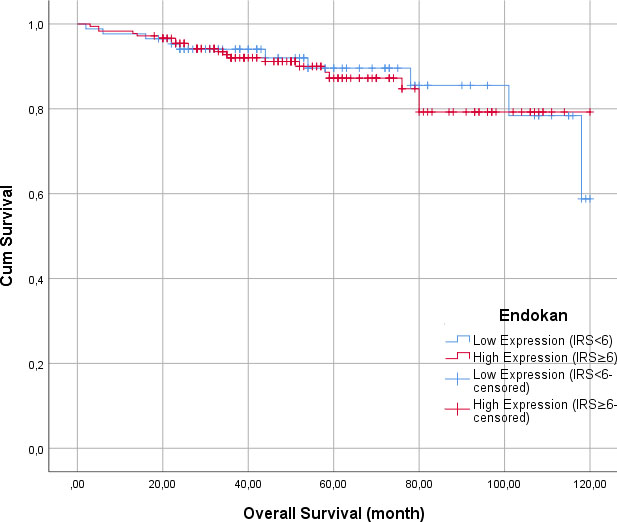
In the present study, we analyzed immunohistochemical endocan expression in BC patients and found that endocan expression is associated with tumor necrosis, tumor size, and presence of LCIS in BC.
Angiogenesis is an active process that is regulated by many pro-angiogenic and antiangiogenic molecules and has a key role in many tumors including BC [1, 4]. It is important in tumor development and progression and is related to tumor aggression [1].
Angiogenic growth factors, such as FGF and VEGF, are involved in the regulation of endocan mRNA expression in vitro as well as endocan secretion [14, 16]. Endocan, which is a circulating proteoglycan, can be detected at sites of inflammation and tumors. Structurally and functionally, endocan has recently been recognized as a potential key player in cancer and inflammatory diseases [14].
Endocan plays a role in cell proliferation and migration and has proangiogenic properties [15]. It promotes the secretion of growth factors and the proliferation of blood vessels. Therefore, plasma levels of endocan are associated with tumor hypoxia [1]. In our study, we found higher endocan expression in tumors without necrosis than in tumors with necrosis. Our finding supports the association of endocan with tumor angiogenesis and hypoxia.
Endocan stimulates tumor cell proliferation and is overexpressed in tumor cells and promotes tumor growth [16, 17, 18]. Kim et al. [19] revealed that endocan immunoreactivity significantly correlated with the tumor size as well as depth of invasion, nodal status, distant metastasis, and Dukes’ stage and was an independent prognostic factor for disease recurrence and worse survival outcome. In another study on colorectal carcinoma, Zuo et al. [7] suggested that the expression of endocan was associated with the development and differentiation of colorectal cancer. However, in their study, no correlation was found between endocan expression and tumor size or tumor stage. In our study on BC, we found a negative correlation between tumor size and endocan expression. In tumors smaller than 2 cm (T1), higher endocan expression was observed. This is inconsistent with the effect of endocan on tumor growth suggested in the literature.
LCIS is accepted as an indolent precursor for invasive lobular BC rather than a pure risk factor [20]. There is still no standard care approach for patients with LCIS [21]. The present study revealed a higher endocan expression in patients with LCIS than in those without LCIS. Endocan overexpression may be associated with the presence of LCIS and therefore, constitutes a risk for the development of invasive lobular BC.
Endocan is also a biomarker of tumor progression [14]. In many cancer types, endocan was associated with bad prognosis [22, 23, 24, 25]. Huang et al. [8] suggested that endocan expression was correlated with poor survival in human hepatocellular carcinoma. Lin et al. [10] found that endocan expression was correlated with poor progression-free survival in patients with pancreatic neuroendocrine tumors. Triple negative BC patients with high endocan expression had worse relapse-free survival rates than those with low endocan expression [12]. Ates et al. [1] found that plasma endocan levels were higher in metastatic BC than in nonmetastatic BC. Additionally, patients with high plasma endocan levels had shorter event-free survival rates. In our study, there was no statistically significant difference between high and low endocan expression groups in terms of PFS and OS. Hence, we could not propose an association between endocan expression and survival.
This study is unique in that it is the first in which endocan expression in BC is evaluated using the immunohistochemical method. Endocan immunohistochemical staining is a simple, inexpensive, and reliable assay [9].
There are also some limitations to our study. The study was performed in a single center with patients who did not have uniform or randomized treatment or follow-up. To establish a causal relationship presenting endocan as a prognostic biomarker in BC, further prospective studies with larger patient samples have to be conducted.
Conclusions
In conclusion, Endocan overexpression in BC is associated with some prognostic parameters including tumor necrosis, tumor size, and the presence of LCIS. Further studies with larger patient series are needed to reveal the clinical and therapeutic implications of Endocan in BC.
Footnotes
Acknowledgments
This study was supported by grants from the Selcuk University Coordinatorship of Scientific Research Projects with the project number 20401002.
Author contributions
Conception: Zeliha Esin Celik
Interpretation or analysis of data: Fatih Demir, Harun Yonar
Preparation of the manuscript: Zeliha Esin Celik
Revision for important intellectual content: Serdar Ugras
Supervision: Serdar Ugras
