Abstract
BACKGROUND:
Long non-coding RNA (lncNRA) forkhead box D3 antisense RNA 1 (FOXD3-AS1) has been proved to promote or suppress the occurrence and development of multiple types of human tumors. However, the function and mechanism of FOXD3-AS1 in non-small cell lung cancer (NSCLC) are scarcely understood.
METHODS:
qRT-PCR was used for detecting FOXD3-AS1, miR-150 and SRC kinase signaling inhibitor 1 (SRCIN1) mRNA expression in NSCLC tissues, and the relationship between pathological characteristics of NSCLC patients and FOXD3-AS1 expression level was analyzed. With human NSCLC cell lines H1299 and A549 as cell models, CCK-8 and BrdU assays were employed for detecting cancer cell proliferation, and Transwell assay was employed for detecting cell invasion ability. Dual luciferase reporter gene assay and RNA immunoprecipitation (RIP) assay were used for the verification of the targeting relationshipe between FOXD3-AS1 and miR-150, and Western blot was employed for detecting SRCIN1 protein expression.
RESULTS:
FOXD3-AS1 expression was significantly reduced in NSCLC tissues and cell lines, and low expression of FOXD3-AS1 was closely related to positive lymph node metastasis and relatively high tumor grade. FOXD3-AS1 over-expression inhibited the proliferation and invasion of H1299 cell lines, while its knockdown promoted the proliferation and invasion of A549 cells. Additionally, it was confirmed that FOXD3-AS1 suppressed the expression of miR-150 by targeting it, and up-regulated the expression of SRCIN1.
CONCLUSIONS:
FOXD3-AS1 indirectly enhances the expression of SRCIN1 by targeting miR-150, thereby inhibiting NSCLC progression.
Introduction
Lung cancer (LC) is a tumor with the highest morbidity and mortality in the world [1, 2]. Non-small cell lung cancer (NSCLC) is the leading pathological sub-type of LC [3]. Since there is a lack of apparent clinical manifestations in the early stage, many NSCLC patients are in advanced stages when they are diagnosed, and the prognosis of these patients is unsatisfactory [4]. In this context, finding out effective biomarkers and therapy targets is of great significance.
Long non-coding RNA (lncRNA)’s transcripts are larger than 200 nt, without open reading frames and protein coding function [5]. LncRNA has many biological functions and contributes greatly to gene expression regulation [6, 7]. According to existing research data, abnormally expressed lncRNAs have been found in various tumors, affecting tumor cell proliferation, differentiation, apoptosis and metastasis [8, 9]. LncRNA forkhead box D3 antisense RNA 1 (FOXD3-AS1) has tumor-promoting or -suppressing effects in different cancers. For example, it, as a carcinogenic lncRNA, promotes the progression of glioma via regulating FOXD3 transcription [10]. In breast cancer, knockdown of FOXD3-AS1 inhibits cancer cell proliferation, migration and invasion [11]. Conversely, as a tumor-suppressing lncRNA, FOXD3-AS1 inhibits neuroblastoma progression by repressing PARP1-mediated CTCF activation [12]. Nevertheless, FOXD3-AS1’s function and mechanism in NSCLC have not yet been clearly elucidated.
MicroRNAs (miRNAs) can pair with the 3’ untranslated region (3’-UTR) of the messenger RNA (mRNA), thus causing mRNA degradation or translation inhibition, and regulate target gene expression [13]. They take part in the development of various diseases including cancers [14, 15, 16, 17, 18]. Specifically, miR-148a suppresses NSCLC cell migration and invasion by targeting Wnt1 [15]; miR-1254 facilitates NSCLC cell proliferation via targeting SFRP1 [16]. In addition, recent studies show that miR-150 expression is increased in various tumors including NSCLC, indicating that miR-150 is a carcinogenic miRNA [17, 18]. However, the upstream mechanism that causes miR-150 dysregulation in NSCLC needs to be further explored.
The study aimed to explore FOXD3-AS1’s expression characteristics, clinical significance, biological functions and mechanism in NSCLC. It was demonstrated that FOXD3-AS1 expression was reduced in NSCLC tissues and cells, and it significantly suppressed NSCLC cell proliferation and invasion. In terms of mechanism, it was confirmed that FOXD3-AS1 targeted miR-150, and up-regulated tumor suppressor SRC kinase signaling inhibitor 1 (SRCIN1).
Materials and methods
Tissue sample
This study got the approval of the Ethics Committee of Shenzhen University General Hospital and was performed according to the Declaration of Helsinki. 50 NSCLC patients who underwent surgery from June 2016 till June 2018 in Shenzhen University General Hospital were randomly selected. The cancer tissue samples and adjacent normal tissues were collected. All samples were collected during surgery and immediately stored in
Cell culture and transfection
Normal bronchial epithelial cell line (16HBE cells) and NSCLC cell lines (H1299, NCI-H460, A549 and L9981 cells) were bought from China Center for Type Culture Collection (CCTCC, Wuhan, China). Cells were cultured in RPMI 1640 medium (Gibco, Carlsbad, CA, USA) supplemented with 100 U/mL penicillin, 100
Control vector, FOXD3-AS1 over-expression plasmid, siRNA negative control (si-NC), siRNA against FOXD3-AS1 (si-FOXD3-AS1), mimics control (miR-NC), miR-150 mimic, inhibitors control (MiR-in) and miR-150 inhibitors were bought from GenePharma Co., Ltd. (Shanghai, China). H1299 and A549 cells in logarithmic growth were inoculated into 6-well plates, cultured in 5% CO
qRT-PCR
TRIzol reagent (Invitrogen, Carlsbad, CA, USA) was used to extract total RNA of NSCLC tissues or cells. PrimeScript RT kit (TaKaRa, Dalian, China) was used for synthesizing cDNA. SYBR Premix Ex Taq II (TaKaRa, Dalian, China) was used for qRT-PCR, and Bio-Rad CFX96TM Manager (Bio-Rad, Hercules, USA) software was used for data analysis. Primers were designed and synthesized from BGI (Shenzhen, China), and the specific primer sequences were shown in Table 1. GAPDH and U6 functioned as the internal references.
Primer sequences of qRT-PCR
Primer sequences of qRT-PCR
Based on the manufacturer’s instructions, cell proliferative ability was measured by CCK-8 assay. In short, transfected cells were inoculated into 96-well plate (5
BrdU experiment
BrdU cell proliferation detection kit (Solarbio, Beijing, China) was used to evaluate cell proliferation. H1299 and A549 cells in logarithmic growth phase were inoculated into 96-well plate (5
Transwell assay
Transwell system (8
Luciferase reporter assay
Luciferase reporter vectors (FOXD3-AS1-MUT and FOXD3-AS1-WT) were constructed by Promega Corporation (Promega, Madison, WI, USA). H1299 and A549 cells were inoculated into 48-well plate, and they were cultured until the confluence reached 70%. Furthermore, with Lipofectamine
RNA immunoprecipitation (RIP)
EZMagna RIP kit (Millipore, Billerica, MA, USA) was used for RIP assay according to the manufacturer’s instructions. In brief, H1299 and A549 cells were lysed in RIP lysis buffer. Subsequently, the cell extract was incubated in RIP buffer with magnetic beads coupled with anti-Ago2 antibody (Millipore, Billerica, MA, USA). The negative control was mouse IgG (Millipore, Billerica, MA, USA). Subsequently, the collected samples were processed with proteinase K. At last, qRT-PCR was performed.
Correlation of FOXD3-AS1 expression with multiple clinicopathological characteristics in patients with lung cancer
Correlation of FOXD3-AS1 expression with multiple clinicopathological characteristics in patients with lung cancer
RIPA lysis buffer (Beyotime Biotcchnology, Shanghai, China) buffer solution was used for extracting total protein. The protein samples were separated by 8% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and then transferred to PVDF membranes (Millipore, Billerica, MA, USA). Subsequently, the membranes were incubated primary antibodies for 12 h at 4
Statistical analysis
Statistical analysis was carried out by SPSS17.0 statistical software (SPSS Inc., Chicago, IL, USA). Measurement data were shown as mean
Results
FOXD3-AS1 expression in NSCLC and its clinical significance
We firstly used qRT-PCR to detect FOXD3-AS1 expression in cancer tissues and adjacent normal tissues of 50 NSCLC patients, and found that FOXD3-AS1 expression in NSCLC tissues was greatly down-regulated in comparison with adjacent normal tissues (Fig. 1A). In addition, qRT-PCR was used to detect FOXD3-AS1 expression level in NSCLC cell lines (H1299, L9981, NCI-H460 and A549 cells) and normal bronchial epithelial cell line (16HBE cells), and it was demonstrated that FOXD3-AS1 expression in NSCLC cells was also greatly reduced compared with 16HBE cells (Fig. 1B). Subsequently, the relationship between FOXD3-AS1 expression level and cliniopathological characteristics was analyzed by Chi-square test, and the results suggested that low FOXD3-AS1 expression was significantly related to positive lymph node metastasis and high tumor grade (Table 2,
FOXD3-AS1 expression in NSCLC tissues and cells. A. qRT-PCR was used for detecting FOXD3-AS1 expression in NSCLC tissues and normal lung tissues of 50 patients. B. qRT-PCR was used for detecting FOXD3-AS1 expression in lung cancer cell lines (NCI-H460, L9981, A549 and H1299 cells) and normal bronchial epithelial cell line (16HBE cells). 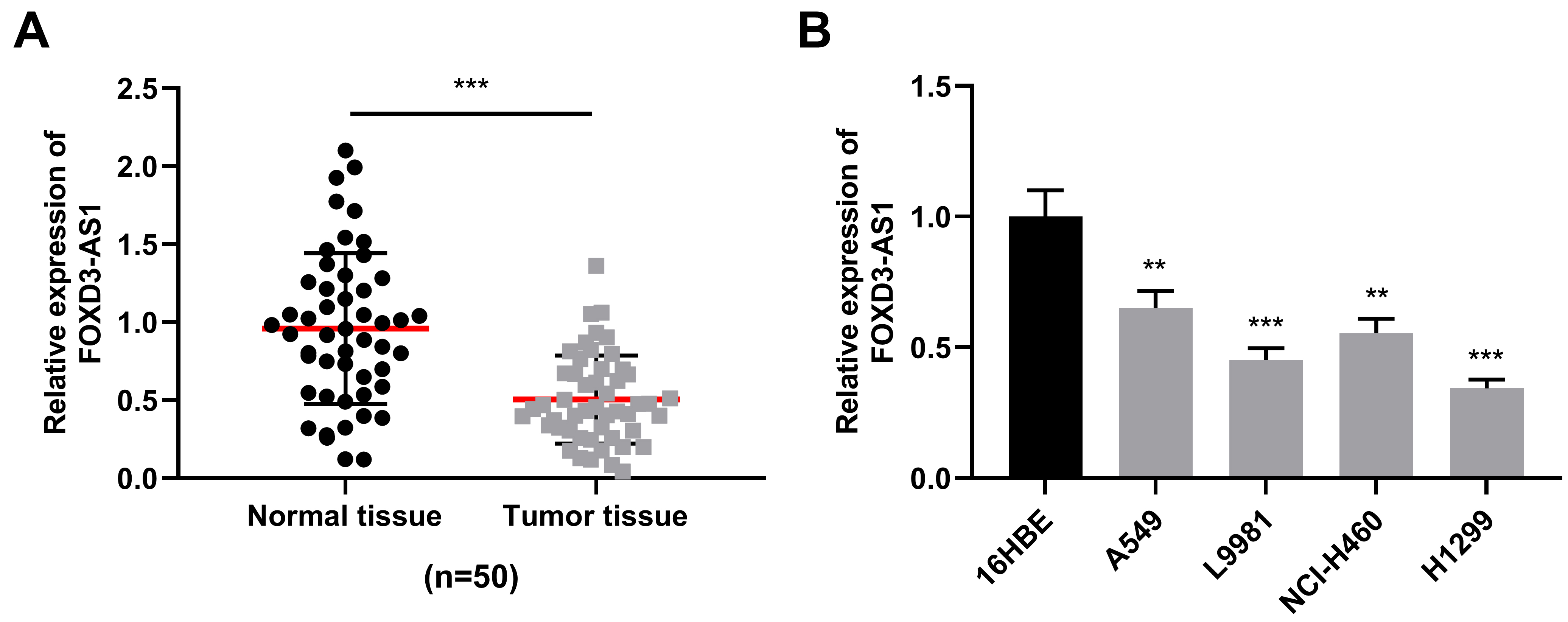
FOXD3-AS1 inhibited lung cancer cell proliferation and invasion. A. qRT-PCR was used to detect the expression of FOXD3-AS1 in H1299 cells transfected with FOXD3-AS1 over-expression plasmid and A549 cells transfected with FOXD3-AS1 siRNAs. B and C. BrdU and CCK-8 experiments were used for detecting NSCLC cell proliferation. D. Transwell assay was used for detecting NSCLC cell invasion. 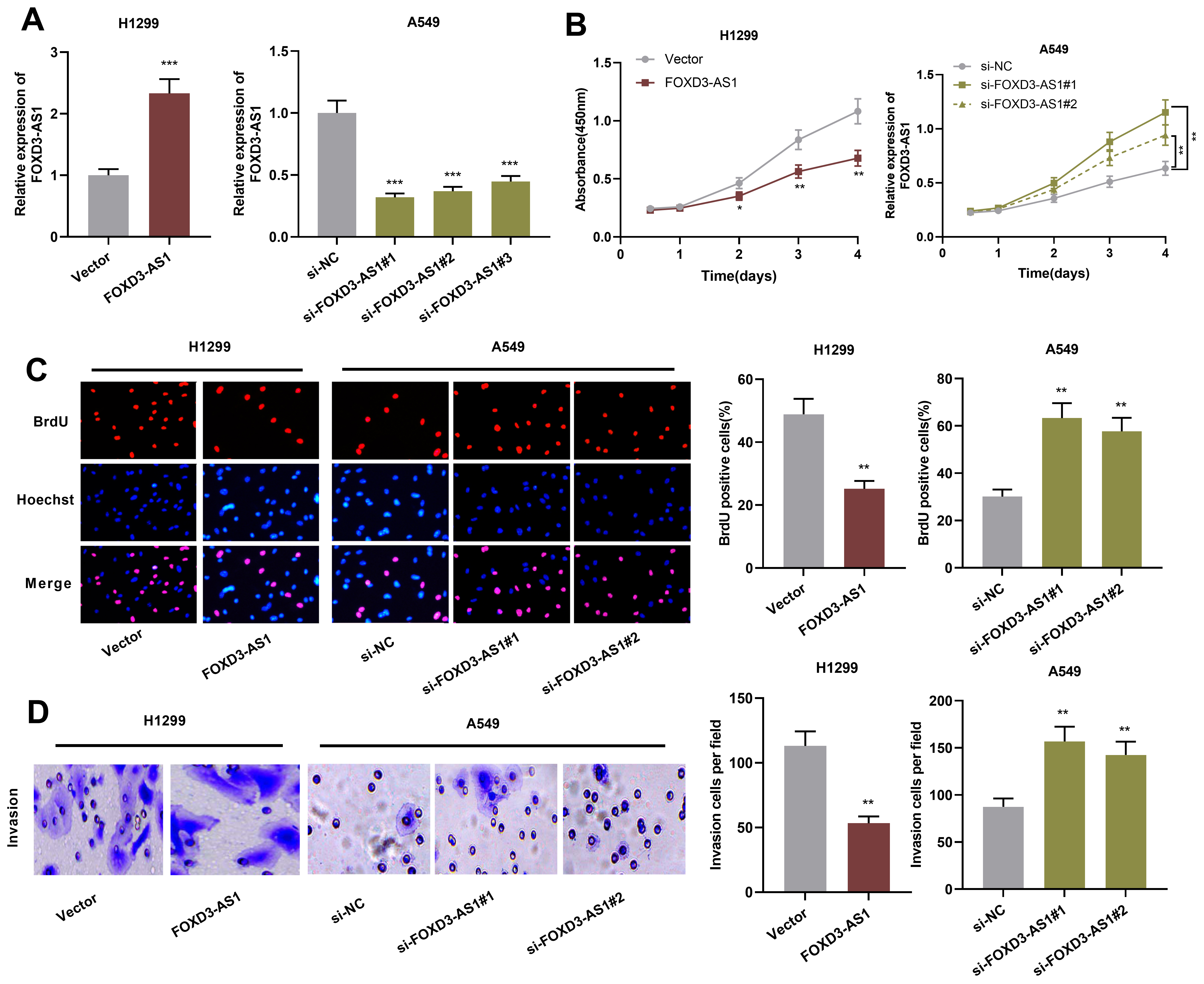
FOXD3-AS1 inhibited EMT of NSCLC cells. A and B. qRT-PCR (A) and Western blot (B) were used for detecting EMT-related markers N-cadherin, E-cadherin and Snail mRNA expressions in NSCLC cells after over-expression or knockdown of FOXD3-AS1. 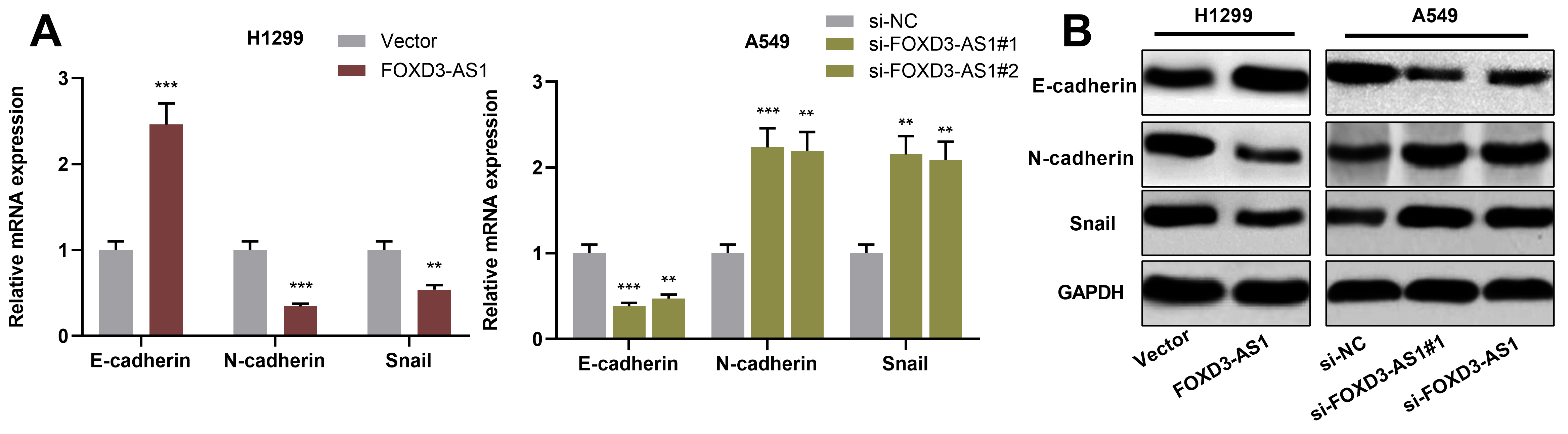
MiR-150 was a target of FOXD3-AS1 in NSCLC. A. A binding site between miR-150 and FOXD3-AS1 was predicted by bioinformatics analysis. B. qRT-PCR was used for detecting the correlation between FOXD3-AS1 expression and miR-150 expression in NSCLC samples. C. qRT-PCR was used for detecting miR-150 expression in NSCLC cells with over-expression or knockdown of FOXD3-AS1. D. Dual luciferase reporter gene assay displayed that miR-150 mimics could reduce the luciferase activity of FOXD3-AS1-WT reporter. E. Direct interaction of miR-150 and FOXD3-AS1 in NSCLC cells was identified by RIP analysis. 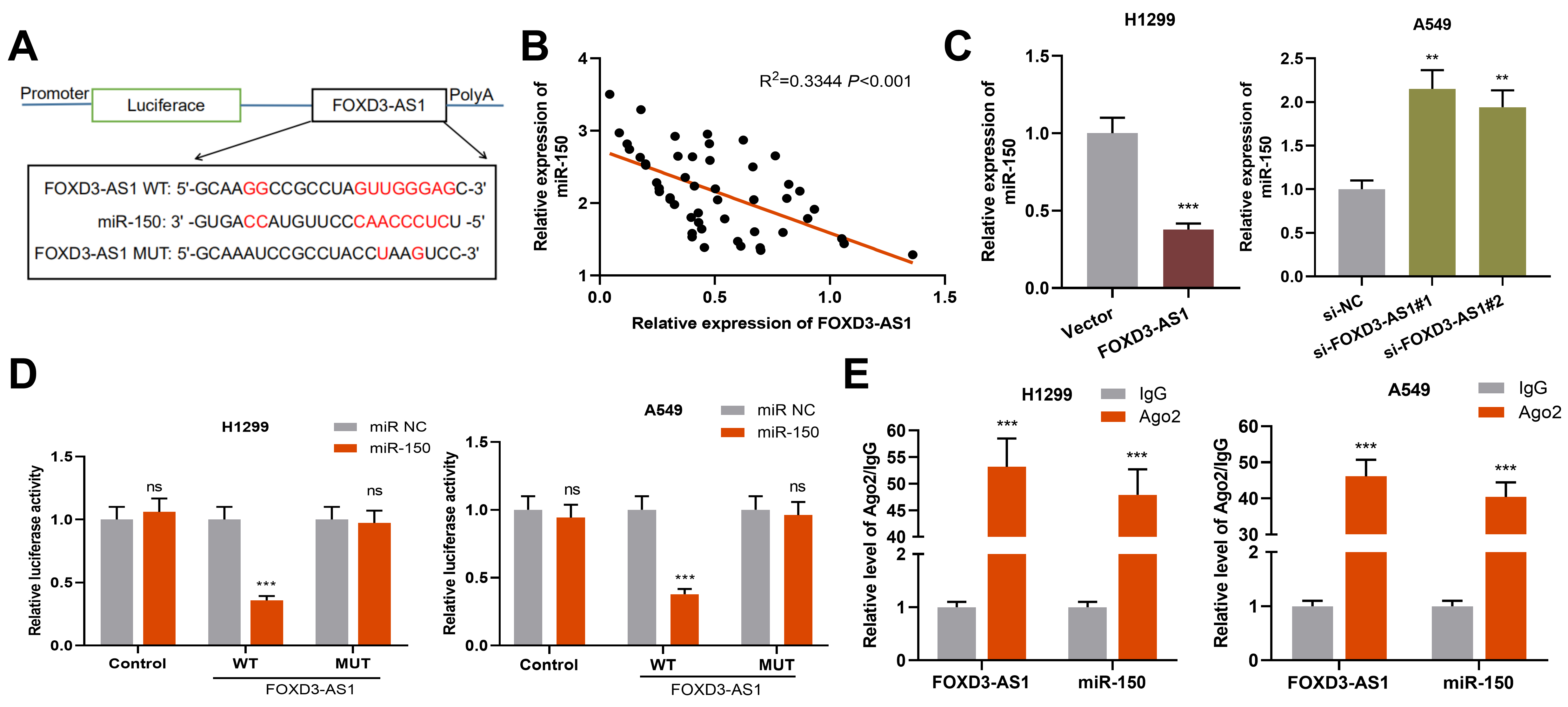
MiR-150 partly reversed the inhibition of lung cancer cells by FOXD3-AS1. A. MiR-150 mimics were used to transfect into H1299 cells with FOXD3-AS1 over-expression, and miR-150 inhibitors were used for transfecting A549 cells with FOXD3-AS1 knockdown, and then miR-150 expression in NSCLC was detected via qRT-PCR. B and C. BrdU and CCK-8 experiments were used for detecting NSCLC cell proliferation. D. Transwell assay was used for detecting NSCLC cell invasion. 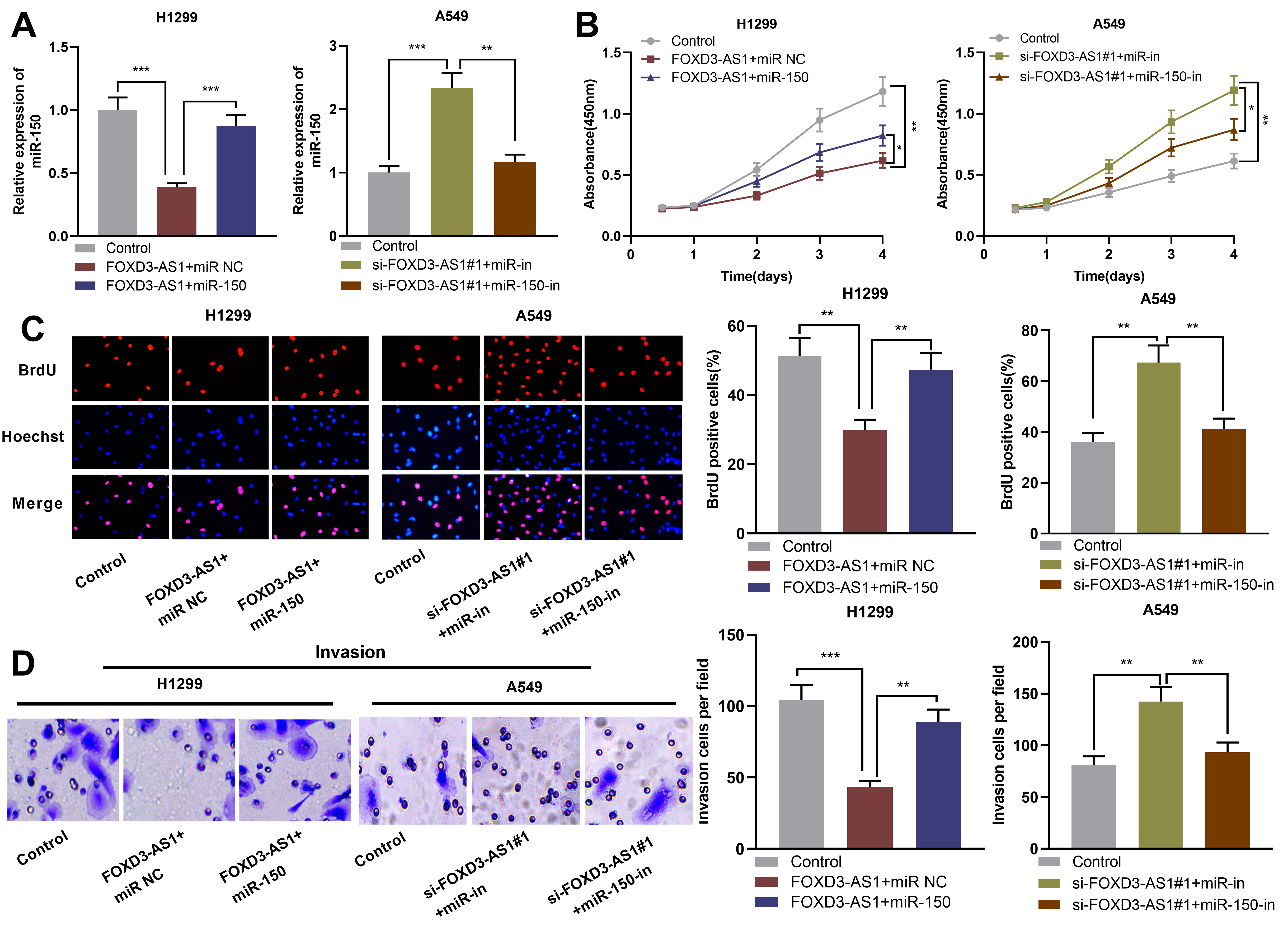
FOXD3-AS1/miR-150 axis regulated SRCIN1 expression. A. qRT-PCR was used for detecting the regulation of FOXD3-AS1 or miR-150 on SRCIN1 mRNA expression. B. Western blot was used for detecting the regulatory function of FOXD3-AS1 or miR-150 on SRCIN1 protein expression. C. qRT-PCR was used for detecting the correlation of SRCIN1 mRNA with the expression of miR-150 or FOXD3-AS1 in NSCLC samples. 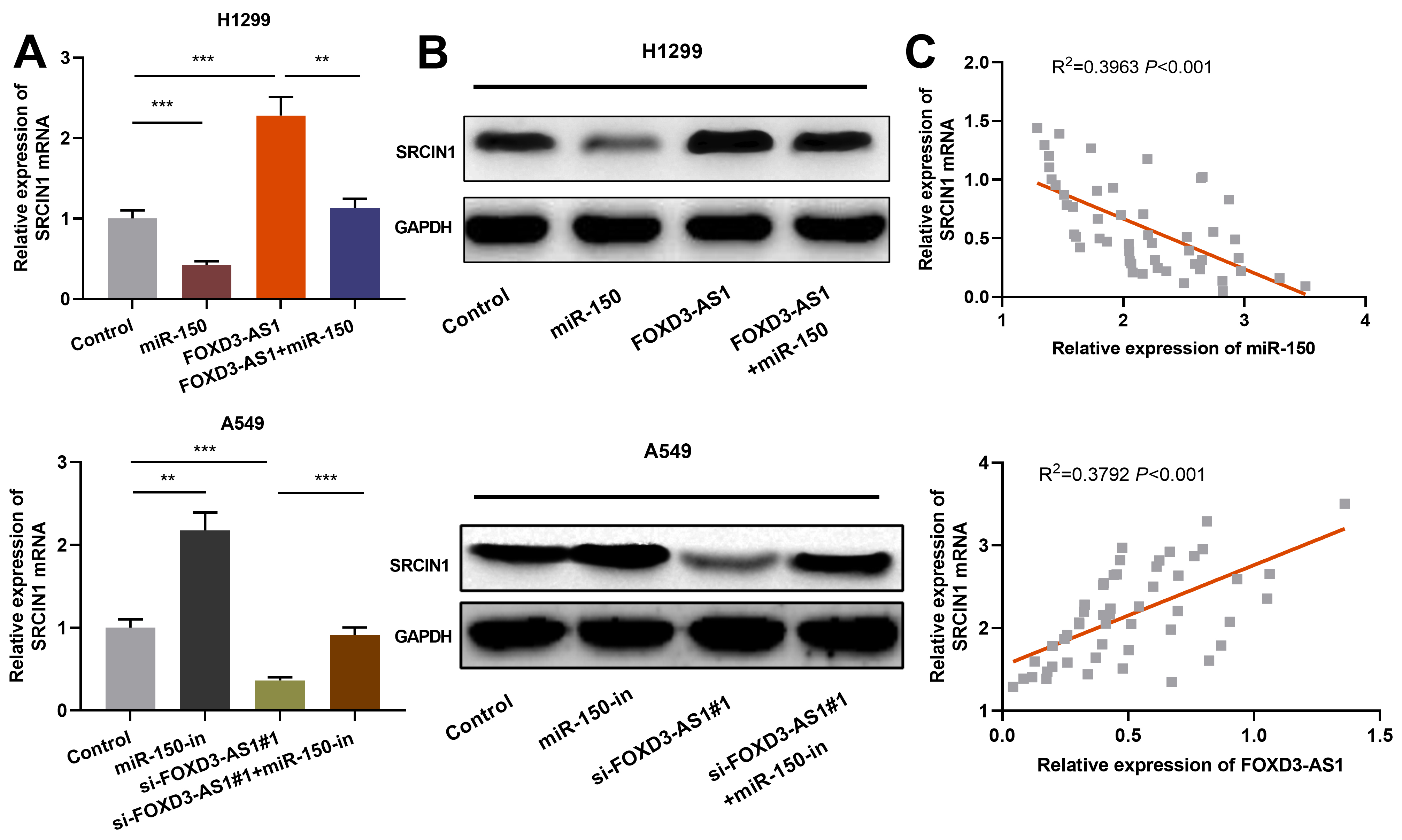
Next, FOXD3-AS1 over-expression plasmids were transfected into H1299 cells, and FOXD3-AS1 siRNAs were employed to knock down FOXD3-AS1 in A549 cells. Following that, cell transfection efficiency was detected by qRT-PCR (Fig. 2A). Subsequently, CCK-8 assay and BrdU assay were then used for detecting cell proliferation. As shown, FOXD3-AS1 over-expression inhibited H1299 cell proliferation, while knockdown of FOXD3-AS1 facilitated A549 cell proliferation (Fig. 2B and C). In addition, Transwell assay was used for detecting FOXD3-AS1’s effect on NSCLC cell invasion, and the results suggested that FOXD3-AS1 over-expression inhibited H1299 cell invasion and knockdown of FOXD3-AS1 facilitated A549 cell invasion (Fig. 2D). Additionally, qRT-PCR and Western blot were used to detect the expression of EMT-related markers N-cadherin, Snail and E-cadherin in H1299 and A549 cells. It was found that FOXD3-AS1 over-expression enhanced the expression of E-cadherin, and decreased the expressions of N-cadherin and Snail, while knockdown of FOXD3-AS1 had adverse effects (Fig. 3A and B). These data indicate that FOXD3-AS1 takes part in inhibiting NSCLC cell proliferation, invasion and EMT.
FOXD3-AS1 targeted miR-150 in NSCLC
Next, using StarBase database, we searched for the potential downstream miRNAs which could be sponged by FOXD3-AS1, and miR-150 was predicted (Fig. 4A). Subsequently, we analyzed the correlation between FOXD3-AS1 expression and miR-150 expression in NSCLC samples, and it was found that their expressions had a negative correlation (Fig. 4B). In addition, we found that FOXD3-AS1 over-expression in H1299 cells greatly inhibited miR-150 expression, while knockdown of FOXD3-AS1 in A549 cells caused an increase in miR-150 expression (Fig. 4C). To further verify the targeted relationship between FOXD3-AS1 and miR-150, we performed dual luciferase reporter gene assay. As shown, miR-150 significantly inhibited the luciferase activity of FOXD3-AS1-WT reporter and exerted no obvious effect on FOXD3-AS1-MUT reporter (Fig. 4D). Furthermore, RIP assay confirmed that miR-150 and FOXD3-AS1 were enriched in Ago2-containing microribonucleoproteins in comparison with control IgG (Fig. 4E). In a word, these findings indicate that in NSCLC, miR-150 is a downstream target of FOXD3-AS1.
MiR-150 reversed the inhibiting effect of FOXD3-AS1 in NSCLC
Next, miR-150 mimics was transfected into H1299 cells with FOXD3-AS1 over-expression, and miR-150 inhibitors into A549 cells with FOXD3-AS1 knockdown, so as to further investigate the function of FOXD3-AS1/miR-150 axis in NSCLC. qRT-PCR results indicated that transfection with miR-150 mimics reversed the down-regulation of miR-150 caused by the over-expression of FOXD3-AS1, while transfection with miR-150 inhibitors attenuated miR-150 up-regulation caused by knockdown of FOXD3-AS1 (Fig. 5A). CCK-8, BrdU and Transwell experiments showed that miR-150 attenuated the inhibiting effect of FOXD3-AS1 over-expression on the malignant biological behaviors of H1299 cells, and the promotion of knockdown of FOXD3-AS1 on the malignant biological behaviors of A549 cells was partly reversed by miR-150 inhibitors (Fig. 5B–D). In addition,Western blot results showed that the inhibitory effect of overexpression of FOXD3-AS1 on EMT was greatly reversed by miR-150 mimics, and knockdown of miR-150 reduced the promoting effect of down-regulation of FOXD3-AS1 on EMT (Supplementary Fig. 1). These results indicated that FOXD3-AS1 took part in inhibiting NSCLC cell proliferation, invasion and EMT by targeting miR-150.
Regulation of FOXD3-AS1/miR-150 on the expression of SRCIN1
The aforementioned data have confirmed that FOXD3-AS1 can target miR-150 in NSCLC. In addition, SRCIN1 has an inhibiting effect on NSCLC progression and is targeted by miR-150 [19]. Therefore, we then explored the relationship between FOXD3-AS1 and SRCIN1 in NSCLC. Through qRT-PCR and Western blot, we found that FOXD3-AS1 over-expression could increase SRCIN1 mRNA and protein expression, while knockdown of FOXD3-AS1 had opposite effects. Moreover, miR-150 over-expression reduced SRCIN1 expression in NSCLC cells and attenuated the up-regulation of SRCIN1induced by over-expression of FOXD3-AS1, while inhibition of miR-150 increased SRCIN1 expression and reversed the down-regulation of SRCIN1 expression by knockdown of FOXD3-AS1 (Fig. 6A and B). Besides, SRCIN1 mRNA was negatively correlated with the expression of miR-150 and positively correlated with the expression of FOXD3-AS1 in NSCLC tissues (Fig. 6C). The above results show that FOXD3-AS1 regulates SRCIN1 expression in NSCLC cells by targeting miR-150.
Discussion
LncRNA is of great importance in tumorigenesis and cancer progression, and it affects the biological behaviors of cancer cell by transcriptional, epigenetic and post-transcriptional regulation [20, 21, 22, 23]. There is more and more evidence showing that lncRNA is involved in NSCLC progression. For example, over-expressed lncRNA LCAL1 induces NSCLC cells’ aerobic glycolysis via regulating AMPK/HIF1
MiRNAs contribute to the regulation of multiple biological processes [28, 29]. Abnormal expression of miR-150 is commonly found in multiple tumors and it promotes cancer progression. For example, miR-150 over-expression reduces apoptosis, and facilitates the growth and colony formation of breast cancer cells [30]. MiR-150 facilitates cervical cancer cell proliferation, migration and invasion by targeting PDCD4 [31]. Studies have demonstrated that lncRNA can function as an endogenous competitive RNA (ceRNA) to adsorb miRNA, and this mechanism is involved in the cancer progression. For example, lncRNA BLACAT1 regulates the expression of CCR2 by targeting miR-150-5p to promote breast cancer cell proliferation and metastasis [32]. Down-regulated lncRNA ZFAS1 inhibits the development of melanoma via regulating miR-150-5p/RAB9A axis [33]. In the present work, it was also found that there was a least one binding site between miR-150 and FOXD3-AS1, and RIP assay and dual luciferase reporter gene experiments confirmed that FOXD3-AS1 could adsorb miR-150. Importantly, FOXD3-AS1 expression in NSCLC tissues had a negative correlation with miR-150 expression, and FOXD3-AS1 could regulate the expression of miR-150 expression in NSCLC cells. Rescue experiments also proved that the tumor-suppressive role of FOXD3-AS1 was partly dependent on its regulatory function on miR-150. Collectively, these data suggested that miR-150 was an oncogenic miRNA in NSCLC, and FOXD3-AS1 took part in inhibiting NSCLC progression by adsorbing miR-150.
SRCIN1, is also known as p140CAP, whose gene is located in chromosome 17q21.1, and its main function is to inhibit Src kinase activity. Existing studies show that SRCIN1 expression in various cancers is decreased, and it mainly functions as a tumor suppressor. For example, SRCIN1 negatively regulates STAT3 and Src signal transduction, inhibits anchorage-independent growth and migration, as well as functions as an independent biomarker for the prognosis of neuroblastoma patients [34]. It is reported that miR-181a facilitates Src/VEGF signal pathway by targeting SRCIN1 to promote angiogenesis of colorectal cancer [35]. And in pancreatic cancer, miR-374a facilitates cancer cell proliferation, migration and EMT via targeting SRCIN1 [36]. Interestingly, miR-150 is reported to facilitate NSCLC progression via targeting SRCIN1 [19]. In the work, consistently, it was discovered that miR-150 was able to negatively regulate SRCIN1 in NSCLC cells, and that miR-150 and SRCIN1 mRNA expressions were negatively correlated in NSCLC tissues. Besides, over-expression of FOXD3-AS1 significantly enhanced SRCIN1 mRNA and protein expression, while knockdown of FOXD3-AS1 caused a decrease in SRCIN1 mRNA and protein expression, and FOXD3-AS1 expression had a positive correlation with SRCIN1 expression in NSCLC tissues. The above results suggest that FOXD3-AS1 up-regulated SRCIN1 expression by targeting miR-150.
To sum up, this research for the first time reveals that FOXD3-AS1 expression is greatly decreased in NSCLC tissues and cells, and low expression of FOXD3-AS1 is strongly associated with adverse pathological parameters of patients. This work also confirms that FOXD3-AS1 participates in inhibiting NSCLC cell proliferation and invasion via regulation of miR-150/SRCIN1 axis. However, this study is limited to in vitro experiments, and it is necessary to further validate this point of view in animal models in the future.
Footnotes
Acknowledgments
This work is supported by Natural Science Foundation of China (Grant No. NSFC30872543).
Conflict of interest
The authors declare that they have no competing interests.
Supplementary data
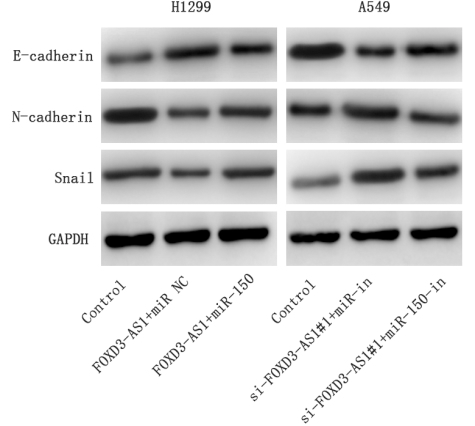
Western blot was used to detect the expression of E-cadherin, N-cadherin and Snail expressions in NSCLC cells after transfection.