Abstract
BACKGROUND:
The expression of neuropilin-1 (NRP-1) in Epstein-Barr virus (EBV)-associated lymphomas and its relationships with clinicopathological parameters was investigated.
METHODS:
The researchers compared 111 cases of patients with lymphoma to 20 cases of reactive lymphoid hyperplasia. In situ hybridization was applied to observe the expression of EBV-encoded RNA (EBER) in lymphomas, and immunohistochemistry was used to detect the NRP-1 expression in lymphoma tissues and lymph node tissues with reactive hyperplasia.
RESULTS:
In these 111 cases, the EBER of 62 cases (55.9%) appeared positive. NRP-1 was relatively highly expressed in lymphomas (
CONCLUSIONS:
NRP-1 exhibited higher expression in lymphomas, and it was positively expressed in EBV-positive lymphomas. Moreover, highly expressed NRP-1 can be used as an undesirable independent prognostic factor in NHL.
Introduction
Lymphoma is a type of malignancy that develops in the lymph nodes and/or extranodal lymphoid tissues. The two main types of lymphoma include Hodgkin’s lymphoma (HL) and non-Hodgkin’s lymphoma (NHL) [1]. It has been suggested that the morbidity rate of NHL ranked seventh among all malignancies, with its mortality rate ranking ninth [2]. Recently, researchers have focused their studies on the treatment of lymphomas [3, 4].
The Epstein-Barr virus (EBV) consists of a double helix of DNA and has a high infection rate (approximately 90%) in humans. Once activated, this type of virus (which often lies dormant) causes various cancers such as nasopharyngeal carcinoma, lymphoma, gastric cancer, etc. [5, 6, 7, 8, 9]. The viral proteins produced by EBV include latent membrane proteins (LMP-1, LMP-2A, and LMP-2B) and nuclear antigens (EBNA1, EBNA2, EBNA3a, EBNA3b, EBNA3c, and EBNA-LP). Moreover, EBV produces viral RNAs (EBER-1 and EBER-2). LMP-1 has been confirmed as a carcinogen involved in the initiation and progression of many tumors [10, 11, 12]. Since the mechanism by which EBV leads to lymphomas is thus far not very well understood, it is worth exploring [13, 14, 15].
Neuropilin-1 (NRP-1) is a transmembrane glycoprotein that is versatile and cell-surface expressed [16, 17]. NRP-1 has been found expressed in vascular endothelial cells [18, 19], and its overexpression has also been detected in neuroglioma, gastric cancer, breast cancer, and lung cancer tumors, which has been regarded as an unwanted prognosis [20, 21, 22, 23, 24]. Currently, there are only a limited number of studies on the associations between NRP-1 and lymphomas. Vadasz et al. employed the immunohistochemistry (IHC) method to detect the expression of NRP-1 in HL tissues [25]. Huang et al. applied gene expression profiling and microarray analysis to observe the highly expressed NRP-1 in EBV-positive NK/T-cell lymphoma (NKTCL) tissue microarray, and the high expression correlated with angiogenesis [26]. Wang et al. discovered that in nasopharyngeal carcinoma, NRP-1 could directly interact with glycoprotein B (gB
Only a few studies have explored NRP-1 in lymphomas, and no reports have been found on NRP-1 expression and its role in EBV-associated lymphoma. In this research, in situ hybridization was applied to observe the expression of EBV in NKTCL, HL, diffuse large B-cell lymphoma (DLBCL), and anaplastic large cell lymphoma (ALCL) tissues. Moreover, immunohistochemistry was used to detect the expression of NRP-1 in lymphoma tissues [28, 29]. We examined the different expressions of NRP-1 in lymphomas with positive and negative EBV, then we analyzed the relationships between NRP-1 and clinicopathological parameters and investigated the prognostic significance of NRP-1. In addition, we explored the possibility that NRP-1 could be used to indicate the clinical course and the prognosis of EBV-associated lymphoma.
Materials and methods
Tissue samples
A total of 111 cases of NKTCL, HL, DLBCL, and ALCL were collected from the Department of Pathology of the First Hospital Affiliated of Guangxi Medical University during the period of March 2012 to May 2016, and 20 cases of normal tissues of reactive lymphoid hyperplasia were selected as controls. None of the cases involved had undergone chemotherapy, radiotherapy, or other tumor treatments before operation and biopsy, and these cases had not been diagnosed with diseases of the immune system, tumors other than lymphoma, or connective tissue diseases. The present study was approved by the Research Ethics Committee of the First Affiliated Hospital of Guangxi Medical University (Nanning, China), and written informed consent was obtained from all patients.
Immunohistochemistry
The main reagents used for the immunohistochemistry included anti-NRP-1 antibody (sc-5307, mouse monoclonal antibody purchased from Santa Cruz Biotechnology), anti-VEGF165 antibody (ab46154, rabbit polyclonal antibody purchased from Abcam), VEGFR-2 antibody (55B11, rabbit monoclonal antibody purchased from Cell Signaling Technology), LMP-1 antibody (CS1-4, mouse monoclonal antibody), CD34 antibody (kit-0004, mouse monoclonal antibody purchased from Maxim Biotechnologies, Fuzhou), Ki67 antibody (ZA-0502, rabbit monoclonal antibody), 3,3’-diaminobenzidine kits (purchased from Zhong Shan Golden Bridge Biotechnology, Beijing), and rabbit/mouse horse radish peroxidase conjugated secondary antibody (purchased from Shanghai Long Island Biotech. Co., Ltd). The streptavidin-peroxidase method of immunohistochemistry was employed to detect the protein expressions of NRP-1, VEGF165, VEGFR-2, LMP-1, Ki67, and CD34. Paraffin sections 4
Hematoxylin-eosin staining of reactive hyperplasia of lymph node and lymphoma tissues. (A) Reactive hyperplasia of lymph node tissues. (B) NK/T-cell lymphoma. (C) Hodgkin’s lymphoma. (D) Diffuse large B-cell lymphoma. (E) Anaplastic large cell lymphoma.
Two pathologists who did not participate in the experiment observed the sections independently. Through in situ hybridization, when granules of brown-yellow or brown showed up in the nuclei of tumor cells, a tumor appeared positive, while a tumor was negative if blue or light purple cell nuclei were seen. IHC staining of NRP-1, VEGF165, and VEGFR-2 was scored based on the intensity of the staining and the percentage of positive cells. Based on the staining intensity, the colorless cells were graded 0, light yellow 1, brown-yellow 2, and brown 3. According to the percentage of positive cells, 0 was assigned for 0%, 1 for 1%–25%, 2 for 26%–50%, 3 for 51%–75%, and 4 for
Statistical analysis
SPSS 23.0 was used to carry out the statistical analysis. The median value of the CD34 staining microvessel count was mean
Results
Clinicopathological features
This research involved 111 cases of lymphoma, of which 40 were NKTCL, 29 were HL, 31 were DLBCL, and 11 were ALCL. Among these 111 cases, 71 patients were male (64%) and 40 were female (36%), with an average age of 40.7 years and a median age of 42 years (age range 3–89 years) (Table 1).
Expression of neuropilin-1 protein in lymphoma and its relationship with clinicopathological parameters
Expression of neuropilin-1 protein in lymphoma and its relationship with clinicopathological parameters
LDH, Lactate dehydrogenase; ECOG, Eastern Cooperative Oncology Group; NRP-1, Neuropilin-1; IPI, International prognostic index; NKTCL, NK/T-cell lymphoma; HL, Hodgkin’s lymphoma; DLBCL, Diffuse large B-cell lymphoma; ALCL, Anaplastic large cell lymphoma; EBER, EBV-encoded RNA; LMP-1, Latent membrane protein 1; VEGF165, Vascular endothelial growth factor 165; VEGFR-2, VEGF receptor 2;
Under the microscope, despite having a normal structure, the reactive lymphoid hyperplasia carried increasing numbers of lymphoid follicles, its size increased with an amplified germinal center or widened deep cortex, and the number of vessels increased; however, the NKTCL, HL, DLBCL, and ALCL structures were damaged (Fig. 1).
Results of in situ hybridization
We examined the expression of EBER in these 111 cases and found that 62 cases had positive expressions of EBER, comprising 55.9% (62/111). In terms of NKTCL, EBER was positively expressed in all 40 cases, with a positivity rate of 100%. In the HL cases, 75.9% of patients (22/29) showed a positive expression of EBER. In contrast, a negative expression of EBER was observed in the DLBCL and ALCL cases (Fig. 2).
In situ hybridization of EBV-encoded RNA of lymphoma tissues. (A) NK/T-cell lymphoma. (B) Hodgkin’s lymphoma. (C) Diffuse large B-cell lymphoma. (D) Anaplastic large cell lymphoma.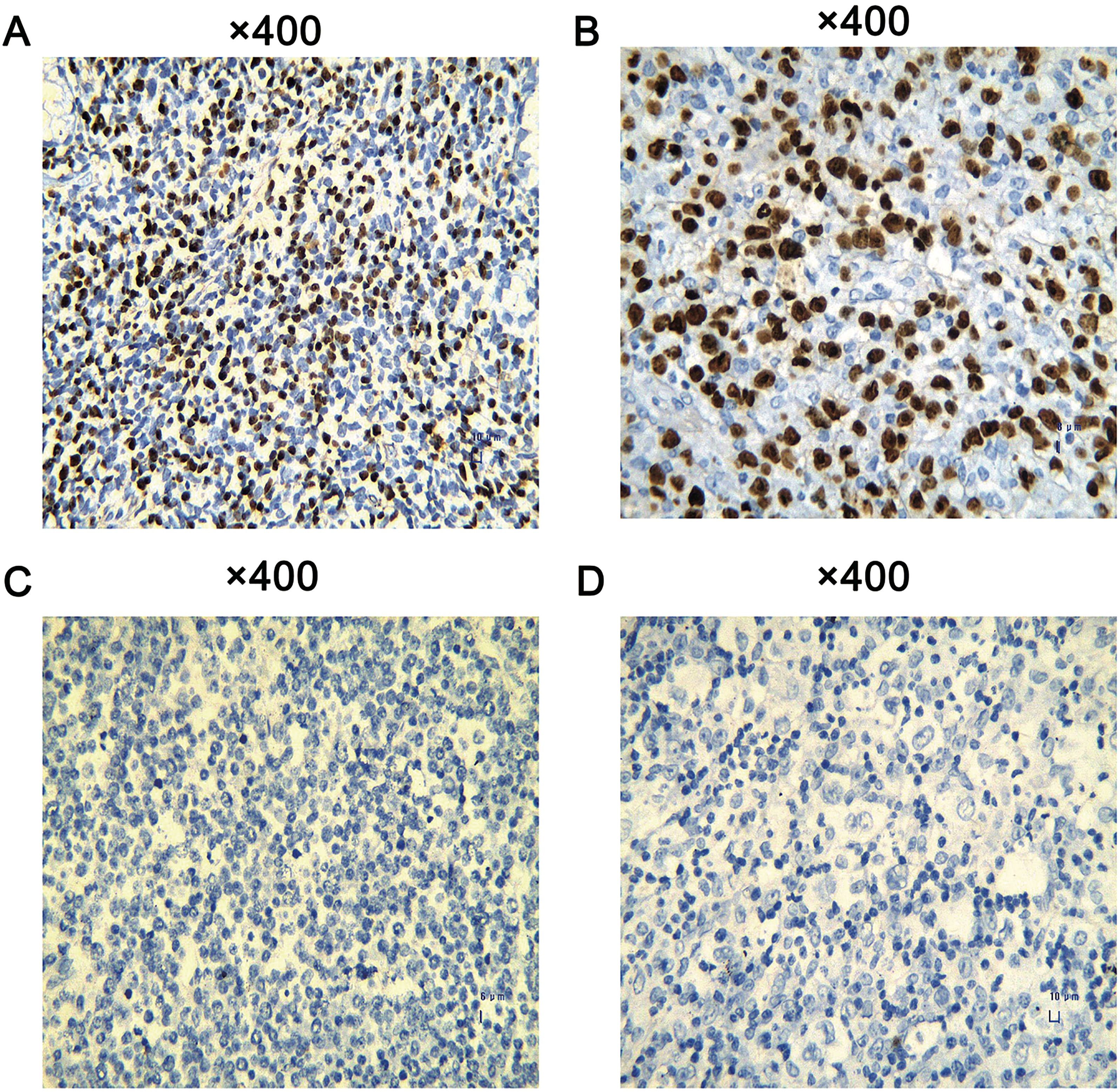
Immunohistochemistry of neuropilin-1 of reactive hyperplasia of lymph node and lymphoma tissues. (A) Reactive hyperplasia of lymph node. (B) NK/T-cell lymphoma. (C) Hodgkin’s lymphoma. (D) Diffuse large B-cell lymphoma. (E) Anaplastic large cell lymphoma.
Immunohistochemistry of VEGF165, VEGFR-2, LMP-1, Ki67, and CD34 of lymphoma tissues. (A) VEGF165. (B) VEGFR-2. (C) LMP-1. (D) Ki67. (E) CD34.
The IHC of NRP-1
The positivity rate of NRP-1 was relatively higher in the 111 cases (51.4%) than in the 20 control cases (20%), with statistical significance (
The IHC of VEGF165, VEGFR-2, LMP-1, Ki67, and CD34
Among the 111 cases, 76 (68.5%) had a positive expression of VEGF165; 63 (56.8%) had a positive expression of VEGFR-2; 50 (45%) had a positive expression of LMP-1; and 73 (65.8%) had a positive expression of Ki67. In 103 cases of lymphoma, 54 (52.4%) had a positive expression of CD34 (Fig. 4).
The relationships between NRP-1 and the clinicopathological parameters of 111 cases
It was discovered that NRP-1 was differently expressed in NKTCL, HL, DLBCL, and ALCL, with statistical significance (
Univariate analysis of clinical pathological parameters and prognosis in lymphoma
Univariate analysis of clinical pathological parameters and prognosis in lymphoma
OS, Overall survival; PFS, Progression-free survival; LDH, Lactate dehydrogenase; ECOG, Eastern Cooperative Oncology Group; NRP-1, Neuropilin-1; IPI, International prognostic index; NKTCL, NK/T-cell lymphoma; HL, Hodgkin’s lymphoma; DLBCL, Diffuse large B-cell lymphoma; ALCL, Anaplastic large cell lymphoma; EBER, EBV-encoded RNA; LMP-1, Latent membrane protein 1; VEGF165, Vascular endothelial growth factor 165; VEGFR-2, VEGF receptor 2;
The analysis indicated that the positive expression of EBER in lymphomas positively correlated with male patients (
The prognostic analysis of 111 cases
By the end of the follow-up period, 40 of the 111 patients (36%) had died. For the remaining 71 cases, the patients’ average survival period was 36.5 months, with the median survival period being 45 months (0.3–54.1 months). The univariate analysis of overall survival (OS) suggested that patients with the following characteristics were more likely to face a bad prognosis: advanced age (
The multivariate Cox regression analysis of OS revealed that the IPI score was considered an independent prognostic factor for lymphomas, with a risk ratio (RR)
Multivariate analysis of prognosis of lymphoma
Multivariate analysis of prognosis of lymphoma
OS, Overall survival; PFS, Progression-free survival; NRP-1, Neuropilin-1; IPI, International prognostic index; NKTCL, NK/T-cell lymphoma; HL, Hodgkin’s lymphoma; DLBCL, Diffuse large B-cell lymphoma; ALCL, Anaplastic large cell lymphoma; NHL, Non-Hodgkin’s lymphoma.
Univariate analysis of clinicopathological parameters and prognosis in Hodgkin’s lymphoma and non-Hodgkin’s lymphoma
OS, Overall survival; PFS, Progression-free survival; LDH, Lactate dehydrogenase; ECOG, Eastern Cooperative Oncology Group; NRP-1, Neuropilin-1; IPI, International prognostic index; NKTCL, NK/T-cell lymphoma; HL, Hodgkin’s lymphoma; NHL, Non-Hodgkin’s lymphoma; DLBCL, Diffuse large B-cell lymphoma; ALCL, Anaplastic large cell lymphoma; EBER, EBV-encoded RNA; LMP-1, Latent membrane protein 1; VEGF165, Vascular endothelial growth factor 165; VEGFR-2, VEGF receptor 2;
Univariate survival curves of the influences of neuropilin-1 protein expression on the overall survival (OS) and progression-free survival (PFS) of lymphoma patients. (A) OS of 111 cases of lymphoma patients. (B) PFS of 111 cases of lymphoma patients. (C) OS of Hodgkin’s lymphoma (HL) patients. (D) PFS of HL patients. (E) OS of non-Hodgkin’s lymphoma (NHL) patients. (F) PFS of NHL patients.
Analyses of lymphomas were conducted on the two main categories: HL and NHL (NKTCL, DLBCL, and ALCL). When the follow-up was completed, 9 of the 29 patients with HL had died. The remaining 20 patients had an average survival period of 35.6 months, with a median survival period of 45 months (3.5–45 months). The univariate analysis of OS demonstrated that patients who had a disappointing ECOG score were prone to a bad prognosis (
By the completion of follow-up, 31 of the 82 patients with NHL had died. The 51 remaining patients experienced an average survival period of 35.6 months (0.3–54.1). We conducted a univariate analysis of OS and found that patients with the following clinicopathological parameters were closely linked with a bad prognosis: senior age, two or more extranodal metastases, clinical stages III–IV, high levels of LDH, undesirable IPI scores, increased or decreased lymphocytes and leucocytes, and no radiotherapy or radio-chemotherapy (
Subsequently, a multivariate Cox regression analysis of OS was conducted to confirm that the independent prognostic factors for NHL included NRP-1 (RR
Discussion
EBV’s genome could encode multiple types of anti-apoptotic proteins and cytokines that lead to infection, activation, and proliferation. People with weak immune systems are more prone to suffer from lymphomas or EBV-related diseases, including nasopharynx cancer, gastric carcinoma, etc. [30, 31, 32, 33, 34]. Despite numerous studies on EBV, its specific roles and mechanism in lymphomas remain unclear.
It has been revealed that NRP-1 is overexpressed in neuroglioma, gastric, breast, and lung cancers, and thereby plays a boosting role in tumor cells [18, 20, 21, 23, 35, 36, 37, 38]. Vadasz et al. detected NRP-1 expression in 92% of HL tissues, while its expression was not observed in tissues of reactive lymphoid hyperplasia [25]. In addition, by gene expression profiling and microarray analysis, Huang et al. discovered overexpressed NRP-1 in NKTCL tissues with positive EBV expression, which was associated with angiogenesis [26]. Overall, the results indicated that aberrant expression of NRP-1 exists in lymphomas. Based on in situ hybridization, this research examined the expression of EBV in NKTCL, HL, DLBCL, and ALCL. Moreover, IHC aided our investigation of the expressions of NRP-1, VEGF165, VEGFR-2, LMP-1, CD34, and Ki67 in the four types of lymphoma. Further, we analyzed the relationships of EBV and NRP-1 with individual clinicopathological parameters and prognoses.
The in situ hybridization revealed that EBER was positively expressed in NKTCL, which was consistent with the results of Mo et al. [39] and Chen et al. [40], thereby suggesting that patients with NKTCL have higher EBV infection rates. In HL, the rate of EBER positivity reached 75.9%. The detection rate of EBV in HL appeared higher than that which has been found in previous studies [41], which was explained by the small sample size or false positives in the results. However, EBER was negatively expressed in DLBCL and ALCL, thereby indicating that DLBCL and ALCL have no relationship with EBV infection.
The expression of NRP-1 was obviously higher in lymphomas than in normal reactive lymphoid hyperplasia, with declining expression in EBV-positive lymphomas, EBV-negative lymphomas, and normal reactive lymphoid hyperplasia. Statistical significance was observed, which indicated that NPR-1 played a cancer-promoting role in lymphomas. We categorized the lymphomas into two types (HL and NHL) and conducted comprehensive analyses, finding that the overexpression of NRP-1 was positively associated with EBV-positive high expressions of VEGF165, VEGFR-2, and CD34, which suggests that NRP-1 correlates with EBV in lymphomas, and NRP-1 might accelerate the angiogenesis by interacting with VEGF165 and VEGFR-2. Nevertheless, its specific mechanism needs further confirmation. Numerous studies have demonstrated that in nasopharynx epithelial cells, NRP-1 can promote EBV infection, and the infection could be inhibited by downregulating NRP-1 [27]. Zhou et al. verified that in pancreatic cancer, the increased expression of NRP-1 could promote the expression of VEGFR-2, therefore inducing the growth of vascular endothelial cells; further, the suppression of interactions between NRP-1 and VEGF165 could deactivate the VEGFR signaling pathways [42]. Xu et al. found that in liver cancer, downregulating NRP-1 could reduce the expression of CD34 and then suppress the growth of cancer cells [43].
In this research, it was confirmed that EBER-positive was positively associated with a positive expression of LMP-1, male patients, and overexpressions of VEGFR-2 and CD34, thereby indicating that males were more likely to contract EBV-associated lymphomas, and EBV might promote the formation of tumor vessels. However, the specific mechanism required further studies for confirmation. In all these cases, EBER positivity was negatively associated with clinical stages III–IV, high ECOG scores, disappointing IPI scores (
The OS and PFS of patients with lymphomas were also analyzed in this research. The analysis of OS showed that patients with the following features were more likely to face a bad prognosis: senior age,
Conclusions
This study found that NRP-1 was overexpressed in lymphomas, particularly in EBV-positive lymphomas, which might be closely correlated with the infection of EBV and angiogenesis. EBV was much more easily detected in male patients and was positively related to the expression of VEGFR-2. EBV positivity could predict a better clinical course and improved prognosis for lymphoma patients. The analysis of prognoses revealed that senior age, clinical stages III–IV, and undesirable IPI scores could be utilized as independent factors of a poor prognosis. In addition, the high expression of NRP-1 could be used as an independent factor of a poor prognosis for NHL patients. In conclusion, NRP-1 has the potential to become the prognostic indicator for EBV-associated lymphomas.
Footnotes
Acknowledgments
This study was supported by the Open Fund of Guangxi Colleges and Universities Key Laboratory of Biological Molecular Medicine Research (GXBMR20 1601), the Guangxi Zhuang Autonomous Region Health, Family Planning Commission Self-Financed Scientific Research Project (Z20170556), the Natural Science Foundation of Guangxi in China (2017GXNS FAA198107, 2015GXNSFDA139028), and the Guang-xi Education Department Research Project (ZD20140 33).
Conflict of interest
The authors declare no conflict of interest.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-192437.
