Abstract
Background:
Hemophagocytic lymphohistiocytosis (HLH) is a severe hyperinflammatory condition often triggered by malignancies, especially T/NK-cell lymphoma-associated HLH (T/NK-LAHLH). Epstein-Barr virus (EBV) infection is strongly linked to T/NK-LAHLH and worsens prognosis. However, the prognostic value of whole-blood EBV DNA levels in T/NK-LAHLH remains unclear, necessitating further investigation to improve risk assessment and treatment strategies.
Objective:
To investigate the clinical characteristics and prognostic significance of whole-blood EBV DNA status in patients with T/NK-LAHLH.
Design:
A single-center, retrospective study was conducted, including 85 patients diagnosed with T/NK-LAHLH between January 2017 and August 2022. Patients were categorized based on EBV DNA status, and clinical outcomes were compared.
Methods:
EBV DNA levels were quantified using polymerase chain reaction (PCR) assays. Kaplan-Meier survival and Cox regression models to assess overall survival (OS) and identify independent prognostic factors.
Results:
A total of 85 T/NK-LAHLH patients were included, with a median age of 52 years (range: 18–81 years) and 60% male. The OS rates at 1, 3, 6, and 12 months were 66.6%, 49.8%, 33.8%, and 28.4%, respectively. Among these patients, 67 (78.8%) were EBV DNA-positive, while 18 (21.2%) were EBV DNA negative. EBV DNA-positive patients exhibited significantly lower platelet and globulin levels, higher IL-10 levels, and prolonged activated partial thromboplastin time compared to EBV DNA-negative patients (p < 0.05). The 6-month OS rate was significantly lower in EBV DNA-positive patients compared to EBV DNA-negative patients (22.5% vs 75.1%, p < 0.001). Multivariate analysis identified EBV DNA positivity as an independent risk factor for shorter 6-month OS (hazard ratio (HR): 4.715; 95% CI: 1.662–13.377; p = 0.004). Among the four patients who underwent allogeneic hematopoietic stem cell transplantation, all achieved complete remission and remained alive at the last follow-up.
Conclusion:
Whole-blood EBV DNA positivity is a significant prognostic factor for poor outcomes in T/NK-LAHLH patients. These findings highlight the need for incorporating EBV DNA monitoring into clinical management and further research to refine therapeutic strategies.
Plain language summary
Hemophagocytic lymphohistiocytosis (HLH) is a rare but severe illness caused by an overactive immune system. It can occur in people with certain cancers, especially T/NK-cell lymphoma. When this condition is linked to infection with the Epstein-Barr virus (EBV), the outcomes are often worse. This study examined how EBV levels in the blood relate to survival in people with HLH linked to T/NK-cell lymphoma. We analyzed data from 85 patients treated between 2017 and 2022. Most patients (78.8%) had EBV detected in their blood. These patients were sicker overall, with lower platelet levels, longer blood clotting times, and higher inflammation markers. Survival was significantly worse for patients with EBV in their blood. After six months, only 23% of these patients were alive, compared to 75% of patients without EBV. Our findings showed that having EBV in the blood was an independent factor linked to poor survival, along with being older than 50, high inflammation levels, and blood clotting problems. However, patients who received a bone marrow transplant did better, with all such patients in this study surviving. This research shows that testing for EBV levels in the blood can help doctors predict outcomes for patients with HLH and T/NK-cell lymphoma. Monitoring EBV levels could guide treatments and improve care for these high-risk patients. Bone marrow transplant may offer hope for long-term survival in some cases.
Keywords
Introduction
Hemophagocytic lymphohistiocytosis (HLH) is a life-threatening hyperinflammatory syndrome resulting from either genetic predisposition or acquired immune dysregulation. 1 Among secondary HLH cases, lymphoma-associated HLH (LAHLH) is the most common cause, accounting for approximately 58% of cases. 2 Patients with lymphoma complicated by Epstein-Barr virus (EBV) infection are at a significantly higher risk of developing HLH compared to those without EBV infection. 3 Previous studies, such as Yao et al., have estimated the prevalence of EBV infection in LAHLH at 54.8%. 4 Within this context, T-cell or natural killer cell lymphoma-associated HLH (T/NK-LAHLH) is notably more frequently associated with EBV infection compared to HLH associated with B-cell non-Hodgkin lymphomas. Furthermore, patients with T/NK-LAHLH tend to exhibit higher EBV DNA loads. 5 Despite these findings, the precise prognostic role of EBV DNA status in T/NK-LAHLH remains unclear, warranting further investigation.
Whole-blood EBV DNA detected by polymerase chain reaction (PCR) assays has emerged as a promising biomarker in this setting. 6 It is both easily accessible through peripheral blood samples and quantitative, making it highly suitable for clinical use. This biomarker provides valuable information not only about the disease burden at baseline but also about the dynamic response to therapy over time, enabling more informed clinical decisions.
Given these advantages, we conducted a retrospective study to explore the clinical features and prognostic significance of EBV DNA status in patients with HLH secondary to T/NK-cell lymphoma. Specifically, we aimed to evaluate the relationship between EBV DNA status and patient outcomes, analyze treatment responses, and identify key prognostic risk factors. Our findings contribute to the understanding of EBV’s role in T/NK-LAHLH and provide a foundation for future research and clinical management strategies.
Methods
Patients
Between January 2017 and August 2022, a total of 104 patients were diagnosed with T/NK-LAHLH at the First Affiliated Hospital of Zhejiang University. Patients meeting both the inclusion and exclusion criteria were enrolled in this study.
Inclusion criteria included:
A histopathological diagnosis of T/NK-cell lymphoma in accordance with the revised 2008 WHO classification.
Fulfillment of the 2004 diagnostic guidelines for HLH.
Exclusion criteria included:
HLH onset occurring during relapse or refractory stages of lymphoma.
Cases with more than 90% of clinical data missing.
Follow-up was conducted through December 2022, continuing until either the last follow-up visit or the patient’s death. The sample size for this retrospective study was determined by the number of eligible patients diagnosed during the study period. Of the 104 patients initially diagnosed, 85 patients were retained for analysis after applying inclusion and exclusion criteria (Figure 1). As this is a retrospective study, no formal statistical sample size calculation was performed a priori. However, the final sample size of 85 patients was considered sufficient to achieve meaningful subgroup analysis (e.g. EBV-positive vs EBV-negative groups) and assess significant differences in survival outcomes and clinical characteristics using multivariable models. The study was approved by the Institutional Review Board of the First Affiliated Hospital of Zhejiang University.

Flow chart of patients’ enrollment.
Quantitative PCR assay for EBV DNA
A commercial EBV-PCR fluorescent quantitative diagnostic kit (Shen Xiang Gene Co., Changsha, China) was used to detect EBV DNA extracted from peripheral whole-blood samples. Amplification data were analyzed using the ABI Applied Biosystems 7300 system (Waltham, Massachusetts, USA), with a sequence detection software specifically developed by ABI Applied Biosystems. Positive and negative controls were included in each quantitative PCR procedure to ensure reliability.
The viral loads were quantified and expressed in IU/mL, with a detection sensitivity threshold of 500 copies per milliliter. EBV DNA levels below 500 copies/mL were classified as negative, while levels of 500 copies/mL or higher were classified as positive. Accuracy was rigorously evaluated through internal quality control measures, with the coefficient of variation maintained within acceptable limits.
Clinical staging, treatment, and response evaluation
Lymphoma staging was performed using computed tomography (CT) or positron emission tomography (PET/CT) following the Ann Arbor system. Anti-HLH treatment protocols were categorized into the HLH-94 regimen, the ruxolitinib combined with corticosteroid (Ru-D) regimen, the doxorubicin-etoposide-prednisolone (DEP) regimen, and the corticosteroid with or without intravenous immunoglobulins (GC ± IVIG) regimen. Anti-lymphoma treatments were based on the specific lymphoma subtypes and included chemotherapy regimens such as cyclophosphamide-liposomal doxorubicin-vincristine-prednisolone (CHOP), cyclophosphamide-liposomal doxorubicin-vincristine-prednisolone-etoposide (CHOPE), asparaginase-gemcitabine-oxaliplatin (PGEMOX), and methotrexate-etoposide-dexamethasone-asparaginase (MESA). The evaluation of HLH treatment efficacy was based on the 2-week response criteria outlined by Marsh et al. 7
Statistical analysis
Statistical analyses were conducted using IBM SPSS Statistics 26.0 (IBM Corporation, Armonk, NY, USA) and R software (version 4.3.3, Vienna, Austria). Data were summarized as medians with ranges where applicable. The Mann-Whitney U test was used for comparisons of continuous variables, while categorical variables were analyzed using the Chi-square test.
Overall survival (OS) was defined as the time from HLH diagnosis to either death or the last follow-up for surviving patients. Survival rates were estimated using the Kaplan-Meier method, and group comparisons were conducted using log-rank tests. Receiver operating characteristic (ROC) curve analysis was performed to identify optimal thresholds for key prognostic indicators. Cox regression analysis was applied to assess potential prognostic factors. Statistical significance was defined as p < 0.05.
Sensitivity analysis
To evaluate the potential impact of including cases with undefined pathological subtypes of T-cell lymphomas, a sensitivity analysis was performed. This analysis excluded these cases (n = 25) and re-assessed the association between whole-blood EBV DNA status and OS using Kaplan-Meier survival analysis and Cox proportional hazard models. The sensitivity analysis aimed to ensure that the inclusion of these cases did not introduce significant bias in the study findings.
Results
Baseline characteristics
Among the 85 patients included in this study, 51 were males (60%) and 34 were females (40%), with a median age at HLH diagnosis of 52 years (range: 18–81 years). The baseline clinical characteristics and laboratory indicators of all patients are detailed in Table 1. The cohort included 35 cases (41.1%) of NK/T-cell lymphoma (NKTL), which consisted of 33 cases of extranodal NKTL, nasal type (38.7%), and 2 cases of aggressive natural killer cell leukemia (ANKL, 2.4%). In addition, the cohort included 2 cases of angioimmunoblastic T-cell lymphoma (AITL, 2.4%), 19 cases of peripheral T-cell lymphoma-not otherwise specified (PTCL-NOS, 22.3%), 2 cases of anaplastic large cell lymphoma (ALCL, 2.4%), and 2 cases of monomorphic epitheliotropic intestinal T-cell lymphoma (MEIT, 2.4%). The remaining 25 cases (29.4%) were T-cell lymphomas without confirmed pathological subtypes.
Baseline characteristics of patients with T/NK-LAHLH.
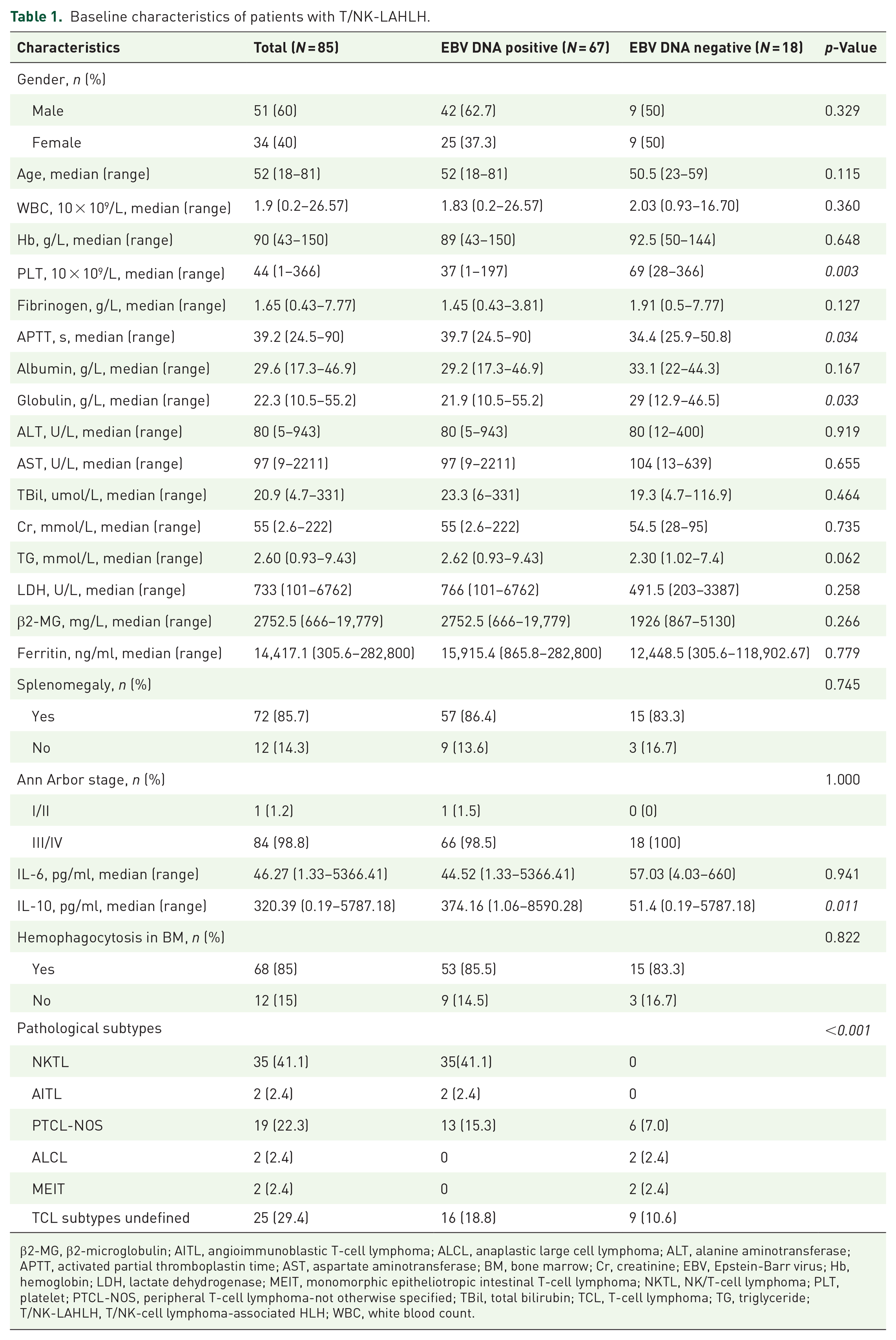
β2-MG, β2-microglobulin; AITL, angioimmunoblastic T-cell lymphoma; ALCL, anaplastic large cell lymphoma; ALT, alanine aminotransferase; APTT, activated partial thromboplastin time; AST, aspartate aminotransferase; BM, bone marrow; Cr, creatinine; EBV, Epstein-Barr virus; Hb, hemoglobin; LDH, lactate dehydrogenase; MEIT, monomorphic epitheliotropic intestinal T-cell lymphoma; NKTL, NK/T-cell lymphoma; PLT, platelet; PTCL-NOS, peripheral T-cell lymphoma-not otherwise specified; TBil, total bilirubin; TCL, T-cell lymphoma; TG, triglyceride; T/NK-LAHLH, T/NK-cell lymphoma-associated HLH; WBC, white blood count.
Most HLH patients (84 cases, 98.8%) were at advanced disease stages (Ann Arbor stage III–IV). Regarding clinical manifestations, fever was observed in all patients.
Treatment and response assessment
The 2-week overall response rate (ORR) for the entire cohort was 45.9% (39/85). Of the 85 patients, 62 received anti-HLH therapy alone, while 23 transitioned to anti-lymphoma chemotherapy after anti-HLH therapy. Among those treated exclusively with anti-HLH therapy within the first 2 weeks, the majority received GC ± IVIG (19/62, 30.6%), followed by the DEP regimen (16 cases, 5 of whom also received ruxolitinib, and 1 received asparaginase), the Ru-D regimen (14 cases), and the HLH-94 protocol (13 patients). Overall, most patients showed no response to anti-HLH therapy within this timeframe. Complete remission (CR) was achieved in 12 patients, and partial remission (PR) in 9 patients, resulting in an ORR of only 33.9%.
Patients treated with the DEP and Ru-D regimens demonstrated relatively better outcomes, with ORRs of 56.3% (CR in 6, PR in 3) and 64.3% (CR in 4, PR in 5), respectively. In contrast, the HLH-94 protocol had an ORR of 23.1% (CR in 2, PR in 1), while the GC ± IVIG regimen was ineffective, with no patients achieving a response.
For patients who transitioned to anti-lymphoma chemotherapy after a median of 5 days of anti-HLH therapy (range: 3–10 days), the initial anti-HLH regimens included HLH-94 (n = 2), GC ± IVIG (n = 13), and Ru-D (n = 8). Subsequent anti-lymphoma treatments included CHOPE (n = 13, 1 of whom also received asparaginase), PGEMOX (n = 3), CHOP (n = 4), and MESA (n = 3). Among these patients, nine achieved CR, nine achieved PR, and five showed no response, yielding an ORR of 78.3%.
Comparison of variables between patients in EBV DNA positive and EBV DNA negative group
We compared the clinical features of 67 patients in the EBV DNA-positive T/NK-LAHLH group and 18 patients in the EBV DNA-negative T/NK-LAHLH group (Table 1). The EBV DNA-positive group showed significantly lower platelet counts (PLT, p = 0.003), lower globulin levels (p = 0.033), longer APTT (APTT, p = 0.034), and higher IL-10 levels (p = 0.011) at the time of HLH diagnosis compared to the EBV DNA-negative group. Notably, all patients with NKTL and AITL had detectable EBV DNA in whole blood, whereas all patients with ALCL and MEIT were EBV DNA-negative, demonstrating a significant difference based on pathological subtype (p < 0.001).
No significant differences were observed between the two groups in other parameters, including white blood count, hemoglobin, fibrinogen, prothrombin time, alanine aminotransferase, aspartate aminotransferase, albumin, total bilirubin, creatinine, TG, β2-microglobulin, lactate dehydrogenase, ferritin, IL-6, splenomegaly, Ann Arbor stage, or evidence of hemophagocytosis in the bone marrow.
Survival outcome
The median follow-up period for patients with T/NK-LAHLH in our cohort was 12.6 months (95% CI: 7.404–17.796). The median OS was 82 days, and the OS rates at 1, 3, 6, and 12 months were 66.6%, 49.8%, 33.8%, and 28.4%, respectively (Figure 2(a)). EBV DNA-positive patients had significantly worse OS compared to EBV DNA-negative patients, with a median OS of 52 days versus not reached (p < 0.001). In addition, the 6-month OS rate was markedly lower in EBV DNA-positive patients compared to EBV DNA-negative patients (22.5% vs 75.1%, p < 0.001) (Figure 2(b)). Of the 85 patients, only 4 underwent allogeneic hematopoietic stem cell transplantation (allo-HSCT), while the remaining 81 did not. All patients who received allo-HSCT were alive at the last follow-up, with survival times of 270, 242, 200, and 188 days post-transplantation.

Overall survival in patients with T/NK-LAHLH. (a) All patients. (b) Stratified by whole-blood EBV DNA status.
ROC curve analysis revealed an area under the curve of 0.720 for age (95% CI: 0.612–0.829, p < 0.001), 0.603 for ferritin (95% CI: 0.478–0.728, p = 0.11), and 0.722 for APTT (95% CI: 0.613–0.830, p < 0.001). The optimal cut-off values were determined to be 50 years for age (sensitivity = 69%, specificity = 67%, Youden index = 0.359), 15,000 ng/mL for ferritin (sensitivity = 60%, specificity = 67%, Youden index = 0.263), and 40 s for APTT (sensitivity = 58%, specificity = 85%, Youden index = 0.425). Cox proportional hazard model for potential factors was conducted as shown in Table 2. Univariate analysis identified age, fibrinogen, APTT, EBV DNA status, and ferritin levels as significant factors associated with 6-month OS (p < 0.05). In multivariate analysis, EBV DNA positivity (hazard ratio (HR): 4.715; 95% CI: 1.662–13.377; p = 0.004) was confirmed as an independent risk factor for shorter 6-month OS. Additionally, age > 50 years (HR: 1.881; 95% CI: 1.006–3.520; p = 0.048), APTT > 40 s (HR: 2.096; 95% CI: 1.137–3.861; p = 0.018), and ferritin > 15,000 ng/mL (HR: 2.684; 95% CI: 1.381–5.215; p = 0.004) were also identified as independent predictors of shorter 6-month OS.
Univariable and multivariable analysis of the effect of clinical features on 6-month OS.
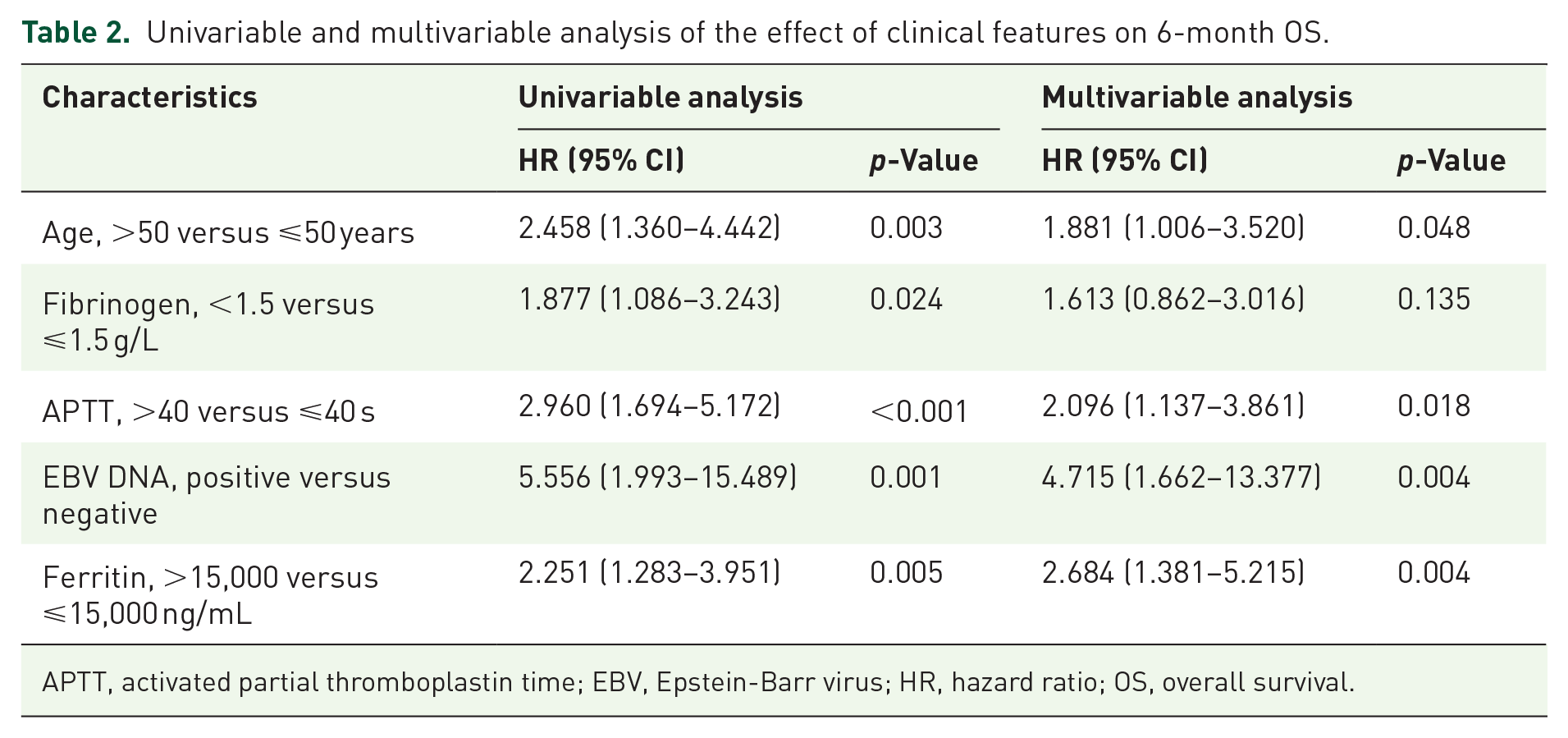
APTT, activated partial thromboplastin time; EBV, Epstein-Barr virus; HR, hazard ratio; OS, overall survival.
Sensitivity analysis results
After excluding cases with undefined pathological subtypes (n = 25), the sensitivity analysis demonstrated that the primary findings remained consistent. EBV DNA status was significantly associated with the prognosis of T/NK-LAHLH (Figure S1) and remained an independent risk factor for shorter 6-month OS, even after excluding these cases (HR: 5.980; 95% CI: 1.403–25.494; p = 0.016). In addition, age was no longer identified as an independent risk factor for 6-month OS in the adjusted Cox regression model (HR: 1.430; 95% CI: 0.703–2.908; p = 0.323; Table S1). These findings indicate that the exclusion of undefined cases did not alter the robustness of the primary results but may have affected the significance of secondary factors such as age.
Relationship between EBV DNA load, diagnosis, and prognosis
To further investigate the relationship between EBV viral load, diagnosis, and prognosis, we performed subgroup analyses within the two major subtypes: NKTL-associated HLH and PTCL-associated HLH. In both subgroups, EBV viral load was not significantly associated with 6-month OS (Table S2).
In addition, a direct comparison of prognosis between NKTL-associated HLH and PTCL-associated HLH revealed no significant differences in survival outcomes (p > 0.05; Figure S2). These findings suggest that neither EBV viral load nor diagnosis independently predicts prognosis in this cohort.
Discussion
This study investigated the clinical features and prognostic significance of whole-blood EBV DNA status in patients with T/NK-LAHLH. Our findings highlight that EBV DNA positivity is strongly associated with poorer survival outcomes and correlates significantly with adverse laboratory parameters. Multivariate analysis confirmed that whole-blood EBV DNA positivity is an independent prognostic factor for T/NK-LAHLH.
EBV plays a central role in the pathogenesis of both lymphoma and HLH. In EBV-associated lymphomas, such as NKTL and EBV-positive diffuse large B-cell lymphoma, EBV directly infects lymphoma cells, driving disease development. In PTCL, EBV infection causes immunodeficiency by impairing the T-cell antigen pool, creating a chronic inflammatory environment that fosters free radical production and promotes lymphomagenesis. 8 For EBV-positive lymphomas, higher EBV loads can exacerbate or even trigger HLH by activating T lymphocytes and macrophages, resulting in the secretion of proinflammatory cytokines such as IFN-γ and TNF-α. These cytokines contribute to a cascade of inflammatory responses, increasing disease severity.9–11 Consistent with these mechanisms, our findings demonstrated that EBV DNA-positive T/NK-LAHLH patients exhibited significantly lower platelet counts, reduced globulin levels, prolonged APTT, and elevated IL-10 levels compared to EBV DNA-negative patients. This further underscores the close relationship between EBV DNA load and disease activity, tumor burden, and treatment resistance in T/NK-LAHLH.
Several studies have previously established the prognostic impact of EBV infection in LAHLH. Song et al. identified EBV infection as an independent risk factor in 66 cases of LAHLH, including 34 T/NK-LAHLH patients. 12 Qiu et al. analyzed a cohort of 26,527 lymphoma patients and found EBV DNA universally present in HLH cases, underscoring the role of active EBV infection in increasing HLH risk (p < 0.001). 3 Zhang et al. reported that in 162 T/NK-LAHLH cases, EBV DNA loads > 16,100 copies/mL were significantly associated with reduced 60-day OS. 10 Similarly, in our cohort, EBV-positive patients had a median OS of just 52 days, significantly shorter than that of EBV-negative patients (p < 0.001). These findings align with prior evidence, reinforcing the prognostic importance of EBV DNA status while providing new insights into its clinical significance in T/NK-LAHLH. However, further analyses in our cohort revealed complexities in the prognostic role of EBV viral load. Subgroup analyses within NKTL-associated HLH and PTCL-associated HLH showed that EBV viral load was not significantly associated with prognosis in either group. Moreover, direct comparisons of prognosis between NKTL-associated HLH and PTCL-associated HLH revealed no significant differences, suggesting that diagnosis itself does not independently influence prognosis. These findings imply that while EBV viral load is closely associated with clinical outcomes, its prognostic value may diminish when controlling for the lymphoma subtype. This may be explained by the overlapping clinical and biological features shared by different subtypes, as well as the uniformly aggressive disease course in HLH. Collectively, these observations underscore the complexity of the prognostic landscape in T/NK-LAHLH and highlight the need for larger, multicenter studies to evaluate the synergistic or independent roles of EBV viral load and specific lymphoma subtypes. Future research should also incorporate molecular and immunophenotypic data to better delineate the prognostic implications of EBV viral load across diverse subtypes and improve risk stratification in this challenging population.
The high prevalence of EBV DNA positivity in our study (78.8%) is consistent with prior reports of approximately 75%.4,5 This is likely attributable to the predominance of NKTL, as nearly all Asian NKTL cases are associated with EBV infection. 13 These results emphasize the need for comprehensive screening for underlying lymphomas in adult HLH cases, even when EBV infection is already confirmed. 9 Given the significant impact of EBV DNA status on prognosis, its incorporation into risk assessment and treatment planning is essential.
Patients with T/NK-LAHLH and EBV DNA positive face a dismal prognosis, highlighting the need for more effective therapeutic approaches. Hematopoietic stem cell transplantation (HSCT) is widely regarded as the only curative option following induction therapy in LAHLH patients. While data on HSCT efficacy in EBV-positive T/NK-LAHLH remain limited, our findings provide promising evidence. All four patients who underwent allo-HSCT in our cohort, including three EBV-positive cases, achieved CR and long-term survival. These results suggest that allo-HSCT has the potential to cure T/NK-LAHLH and should be strongly considered for eligible patients.
Patients with T/NK-LAHLH and high EBV DNA loads face a dismal prognosis, highlighting the need for more effective therapeutic approaches. HSCT is widely regarded as the only curative option following induction therapy in LAHLH patients.14,15 While data on HSCT efficacy in EBV-positive T/NK-LAHLH remain limited, our findings provide promising evidence. All four patients who underwent allo-HSCT in our cohort, including three EBV-positive cases, achieved CR and long-term survival. These results suggest that allo-HSCT has the potential to cure T/NK-LAHLH and should be strongly considered for eligible patients.
Emerging therapies, such as anti-PD-1 antibodies combined with chemotherapy, have also shown favorable outcomes in certain cases of NKTL-associated HLH. These therapies have demonstrated significant reductions in EBV DNA levels and improved survival, warranting further investigation as a potential therapeutic option in EBV-positive T/NK-LAHLH. 16
This study has several limitations. First, as a single-center retrospective study, it is subject to inherent biases. The relatively small sample size (85 patients) limits the generalizability of the findings and may affect statistical power. Second, variability in treatment strategies, even when managed by a single medical team with consistent diagnostic criteria, could have influenced outcomes. Finally, our reliance on clinical manifestations and laboratory parameters for prognostic assessments underscores the need for external validation in larger, multicenter cohorts.
Despite these limitations, our study represents one of the largest cohorts of T/NK-LAHLH patients to date, providing valuable insights into the prognostic role of whole-blood EBV DNA status. To address the potential heterogeneity introduced by cases with undefined pathological subtypes, we conducted a sensitivity analysis, which further supports the robustness of our findings. Excluding these cases did not alter the primary conclusion that whole-blood EBV DNA positivity is an independent prognostic factor for shorter OS in T/NK-LAHLH patients. This underscores the reliability of our results and highlights the critical role of EBV DNA in risk stratification and therapeutic decision-making for this aggressive disease. Interestingly, the sensitivity analysis also revealed that age was no longer an independent risk factor after excluding these cases. This finding suggests that the reduced sample size and the decreased heterogeneity of the cohort may have influenced the significance of age as a prognostic factor. Alternatively, interactions between age and other key prognostic factors, such as EBV DNA status or ferritin levels, may have contributed to this result. These observations highlight the complexity of prognostic factors in T/NK-LAHLH and the importance of considering the interplay of multiple variables when assessing patient outcomes.
Moreover, our findings emphasize the importance of EBV DNA as a key factor in risk stratification and treatment planning for this challenging condition. In addition, our results support allo-HSCT as a potentially curative option, while underscoring the need for further research to optimize therapeutic strategies and explore emerging treatments, such as anti-PD-1 antibody regimens. These findings contribute to advancing the understanding and management of T/NK-LAHLH and highlight the urgency of developing more effective treatments for this aggressive disorder.
Supplemental Material
sj-docx-1-tah-10.1177_20406207251319604 – Supplemental material for Significance of whole-blood EBV DNA status in T/NK-cell lymphoma-associated hemophagocytic lymphohistiocytosis: a single-center retrospective analysis
Supplemental material, sj-docx-1-tah-10.1177_20406207251319604 for Significance of whole-blood EBV DNA status in T/NK-cell lymphoma-associated hemophagocytic lymphohistiocytosis: a single-center retrospective analysis by Mengqi Xiong, Li Li, Lulu Wang, Lixia Zhu, Rongrong Chen, Jingsong He and Xiujin Ye in Therapeutic Advances in Hematology
Footnotes
Acknowledgements
We sincerely thank all the patients and their families for their participation and cooperation in this study. Their contributions were invaluable to the success of this research.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
