Abstract
OBJECTIVE:
To investigate the expression pattern, prognostic value and biological functional of LINC00174 in glioma.
METHODS:
In total, 140 glioma samples were collected as discovery cohort. TCGA RNA sequence dataset was obtained as validation set. Kaplan–Meier survival and multivariate Cox analysis were performed to evaluate survival difference. Furthermore, the biological function of LINC00174 was analyzed by clonogenic and intracranial tumor model assays.
RESULTS:
Overexpressed LINC00174 was significantly correlated with tumor grade as well as the higher mortality in survival analysis both in the discovery and the validation GBM cohorts. Besides, LINC00174 served as an independent prognostic indicator in glioblastoma patients. Additionally, knock down of LINC00174 expression significantly suppressed GBM cells’ proliferation both in vitro and vivo.
CONCLUSION:
LINC00174 acts as an oncogene in glioma and may be a new potential therapeutic target.
Introduction
Glioblastoma multiforme (GBM), the most frequent (
LncRNAs are class of RNAs with a length over 200 nucleotides, modulating downstream gene expression at transcriptional, post-transcriptional and epigenetic level without coding any protein [4]. Dysregulated lncRNAs has been found in many solid cancers, acting as oncogenes or anti-oncogenes in the process of oncogenesis and malignant progression of various cancers, including glioma [5, 6, 7]. Previous study reported that long intergenic non-protein coding RNA 174 (LINC00174) acted as an oncogene in colorectal carcinoma (CRC), which was significantly up-regulated and conferred poor survival in CRC patients [8]. However, the expression pattern, prognostic value and functional role of LINC00174 in glioma remains to be elucidated.
In the present study, we demonstrated that LINC00174 expression was positively correlated with glioma grade and could serve as an independent prognostic marker. Antagonism of LINC00174 expression suppressed aggressive proliferation of GBM cells. All these results indicated that LINC00174 may be a novel therapeutic target in GBM.
Methods
Glioma samples and patients
Glioma samples were collected and confirmed by pathologists according to the 2016 WHO classification scheme. All tumor tissues were immediately frozen in liquid nitrogen after resection until RNA extraction. Overall survival (OS) was calculated from the date of histological diagnosis to death or the last follow-up.
LINC00174 expression analysis in TCGA dataset
The Cancer Genome Atlas (TCGA) glioma RNA sequence database (
Gene ontology (GO) analysis of LINC00174 associated genes
Pearson correlation analysis of LINC00174 and other genes in whole genome gene expression profile was performed in TCGA dataset. LINC00174 positively correlated genes (
Cell culture and transfection
Cell culture and transfection were done as previously discribed [9]. In brief, Human glioma cell line U87, U251 were purchased from the Chinese Academy of Sciences Cell Bank. All cell lines were maintained in a 37
RNA extraction and quantitative real-time reverse transcription PCR (qRT-PCR)
Total RNA was extracted by using the TRIzol reagent (Invitrogen, Carlsbad, USA). The amount of RNA was quantified by absorbance reading at A260/A280
Clonogenic assays
The clonogenic assays were performed as previous discribed [10]. Briefly, cells in the log phase of growth were digested with 0.25% pancreatin. Cells were then dissociated and a single cell suspension was obtained. Fifty cells were seeded onto a culture plate, which after adherence, were transfected with LINC00174 siRNA or NC for 48 h. Six hours later medium was replaced. Plates were incubated in a 37
Intracranial tumor assay
Intracranial tumor assay was done as previously discribed [11]. Briefly, Six-week-old female nude mice (animal center of the Cancer Institute of the Chinese Academy of Medical Science) were used in this assay. U87 cells which were transfected with siRNA or NC, and then 1.0
(A) LINC00174 expression was positively correlated with tumor grade. This result is validated by TCGA RNA sequence database. A single spot is the LINC00174 expression value of an individual patient. Lines in the middle indicate the mean expression value. Error bars represent standard deviation (SD). (B) Higher LINC00174 expression conferred worse survival in 60 GBM patients. TCGA database showed a similar trend. High group, patients with higher LINC00174 level than the median value or equal to the median one. Low group, patients with lower LINC00174 value than the median one.
Statistical analysis was performed using SPSS Graduate Pack 16.0, R 3.2.1 and GraphPad Prism 5.0 statistical software. Descriptive statistics were shown as mean
Results
The expression of LINC00174 was correlated with glioma grade
To explore LINC00174 expression pattern in glioma, 140 tumor samples (40 grade II, 40 grade III, and 60 grade IV) were collected and qRT-PCR was performed, as shown in Fig. 1A, LINC00174 expression was positively associated with the grade of glioma (
High LINC00174 predicted poor survival in GBM patients
To investigate the relationship between LINC00174 expression and GBM patients’ OS, Kaplan-Meier survival curve analysis with a log-rank test of 60 patients in the above discovery dataset was applied. As shown in Fig. 1C, patients with higher LINC00174 expression values (median survival is 315 days) had a worse survival compared to those with lower LINC00174 expression values (median survival is 481 days) (
LINC00174 acted as an independent prognostic biomarker in GBM patients
The prognostic value of LINC00174 in GBM patients was further evaluated through a univariate Cox regression analysis. As shown in Table 1, high LINC00174 expression level was determined as a risk factor for GBM patients’ survival. Additionally, non-methylation of MGMT, non-radiotherapy, and non-TMZ treatment were all risk factors for GBM patients’ survival. Furthermore, we performed multivariate Cox proportional hazards analysis incorporating the above factors. The results indicated that LINC00174 expression level was an independent prognostic biomarker for the overall survival time of GBM patients.
Clinicopathological factors affecting GBM patients’ overall survival
Clinicopathological factors affecting GBM patients’ overall survival
TMZ, temozolomide. HR, hazard ratio.
To assess the potential biological processes of LINC00174 in glioma, Pearson correlation analysis between LINC00174 expression and other genes in whole genome gene profiling were performed in 699 TCGA glioma samples. We found 874 positively correlated (
(A) GO analysis of the biological processes of LINC00174 positively correlated genes in TCGA dataset. (B) GO analysis of the KEGG pathway of LINC00174 positively associated genes. The upper X-axis indicates gene counts, and the lower X-axis indicates adjusted (-log10) 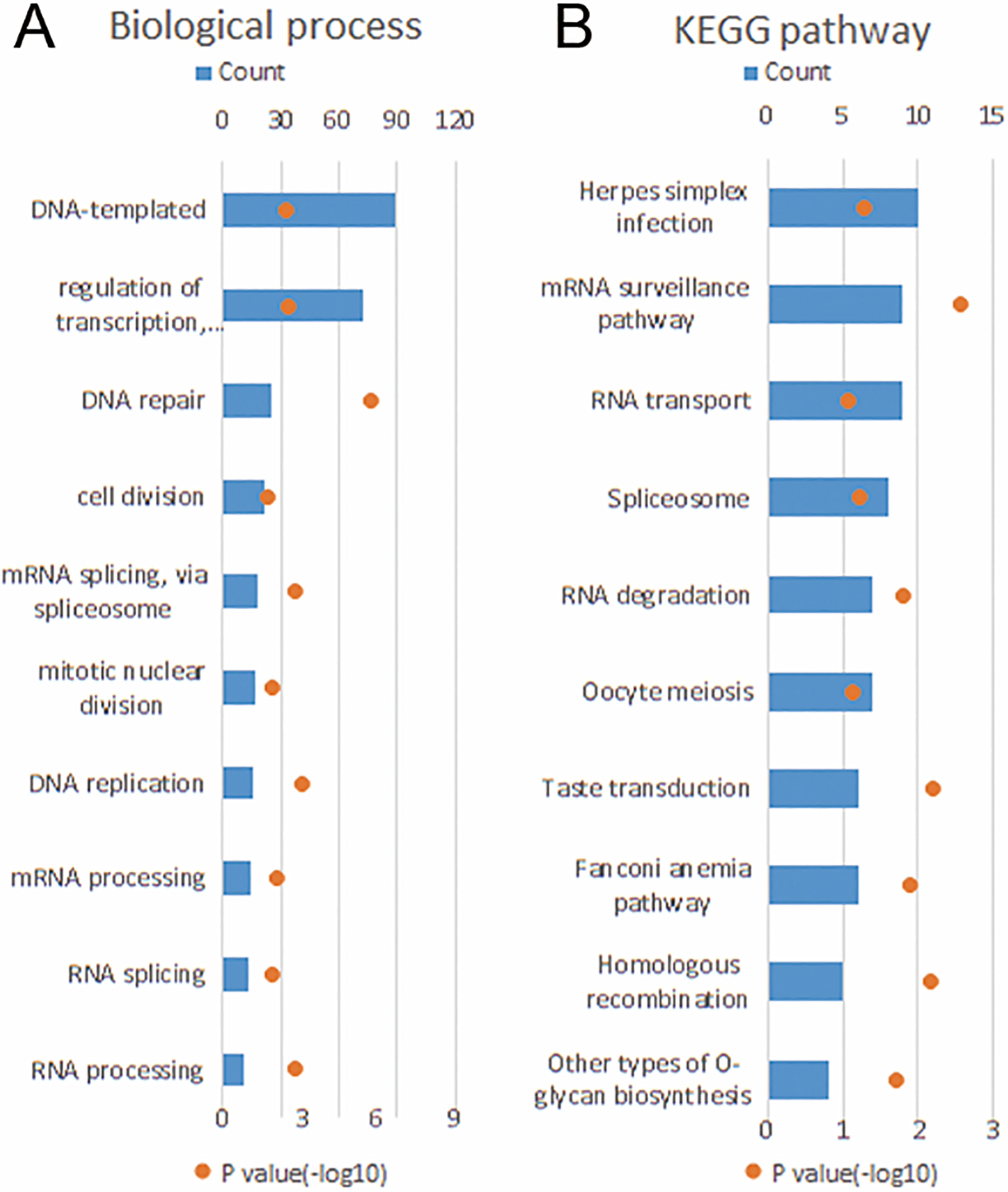
To further identify the functional role of LINC00174 in GBM, loss-of-function assay was employed by transfecting LINC00174 small interference RNA (siRNA) into U87 and U251 GBM cells to silence LINC00174 expression (Fig. 3A), and we found that antagonism of LINC00174 expression suppressed the formation of cell clones (Fig. 3B). Next, mouse intracranial tumor model was used to assess the effect of LINC00174 siRNA on tumor growth in vivo. The siRNA group showed significantly decrease of tumor mass, compared to the control group as shown by bioluminescence imaging (Fig. 3C). Additionally, reduction of LINC00174 expression prolongs intracranial glioma mice’s overall survival days in compared with the control group.
LINC00174 acted as an oncogene in glioma. (A&B) LINC00174 siRNA decreased LINC00174 expression in both U87 and U251 glioma cells. (C&D) Clonogenic assay indicated that reduction of LINC00174 expression suppressed cell proliferation in vitro. (E) Intracranial tumor model assay showed that LINC00174 siRNA significantly inhibited tumor growth in vivo. (F) Reduction of LINC00174 expression prolonged intracranial glioma mice’s overall survival days. NC means negative control treatment group, siRNA means LINC00174 small interference RNA treatment group.
LncRNA is a class of non-coding RNA, with a length range from 200 to 10000 nucleotides. These RNAs have been found to be dysregulated and play essential roles in the origin and progress of many human tumors [12, 13, 14, 15, 16]. LncRNA functions as tumor oncogene or suppressor by regulating the target gene expression at the transcriptional level, post-transcriptional level and epigenetic level. The underlying functional mechanisms of LncRNA are various, including: (1) acts as miRNA sponges by binding the target miRNA competitively [17], (2) modulates the DNA methylation state through binding the target gene promoter region [18], (3) the anti-sense LncRNA protects mRNA from RNase or miRNA caused degradation through binding to the target mRNA [19], (4) interact with and destabilize the pri-miRNA transcript directly [20], (5) direct enhance pri-miRNA processing [21].
In glioma, abnormal expression of LncRNAs also have been reported, and the dysregulated LncRNAs can affect glioma cells’ abilities of proliferation, invasion, apoptosis, drug resistance, and so on [6, 22]. Zhang et al. reported that HOTAIR serves as a prognostic marker and a critical regulator of cell cycle progression in glioma [23], as well as enhancing angiogenesis by induction of VEGFA expression and transmission to endothelial cells via glioma cell derived-extracellular vesicles. [24]. MALAT1was revealed to be associated with poor prognosis in glioma [25], promoting cell proliferation and suppressing apoptosis as well as decrease the sensitivity of resistant GBM cells to temozolomide [26, 27]. Cui et al. found that knockdown of CCAT1 notably suppressed proliferation, migration and the epithelial-mesenchymal transition (EMT) process, and promoted the apoptosis of glioma cells [28]. Moreover, LncRNA H19 was showed to be overexpressed and negatively associated with glioma patient survival. [29], and inhibited LncRNA-H19 expression could decrease chemoresistance of human glioma cells to temozolomide [30].
LINC00174 also known as NCRNA00174, located on chromosome 7q11.21 at 5 exon count, with a transcript length of 4426 nucleotides. Shen et al. showed that LINC00174 was overexpressed both in CRC tissues and cells. CRC patients with high LINC00174 expression had short survival time, and silence of LINC00174 inhibited CRC cell growth both in vitro and in vivo. Finally, they found that LINC00174 played as a competitive endogenous RNA (ceRNA) for miR-1910-3p, which further suppressed the TAZ expression and promoted CRC cell proliferation [8]. However, little was known about the expression pattern of LINC00174 in glioma, as well as the clinical significance, prognostic value and functional role of LINC00174 in glioma. A better understanding of the expression pattern and functional role of LINC00174 in glioma progressive process may provide better diagnose, prognosis of patients, as well as advances the development of personalized therapeutics to improve the clinical outcome. So, further analyzing on large glioma samples are urgent and valuable.
We first evaluated LINC00174 expression in 140 glioma samples, the results showed that LINC00174 expression was positively correlated with tumor grade. Next, we explored the prognostic value of LINC00174 in 60 GBM patients, Kaplan-Meier analysis showed that GBM patients with higher expression of LINC00174 had a worse survival than those with lower LINC00174 expression. The above results suggested that LINC00174 was related to glioma malignancy, in accordance with the expression pattern and prognosis value of LINC00174 in CRC. To further estimate whether LINC00174 is an independent prognostic biomarker for GBM patients, a stepwise multivariate Cox regression analysis was employed in the above GBM patients. This analysis revealed that LINC00174 expression level was an independent prognostic factor for the overall survival of GBM patients. Taken together, these results indicate that LINC00174 is a potential biomarker for predicting the malignancy of GBM and prognosis of patients.
In order to investigate the biological processes of LINC00174 in glioma, LINC00174 correlated genes were analyzed by Pearson correlation in TCGA dataset. We observed a positive correlation of LINC00174 expression with a series of tumor oncogenes, driving DNA replication, and cell division, including MAPK, SOX and BMP family numbers. Additionally, positively associated genes of LINC00174 were analyzed by GO analysis, the results denoted that the main biological function of LINC00174 associated gene sets were to modulate cell proliferation. Finally, to verify the GO analysis result, clonogenic and intracranial tumor model assays were applied to decipher the functional role of LINC00174 in GBM, and the assays showed that reduction of LINC00174 expression significantly suppressed U87 and U251 cells proliferation both in vivo and vitro. Additionally, antagonism of LINC00174 expreion can prolong the overall survival of intracranial glioma mouse models. All these results are in concordance with the previous reports that LINC00174 is an oncogene in tumor original and development processes.
Conclusion
In conclusion, this study showed that the expression value of LINC00174 increased with the ascending grade of glioma. Moreover, it conferred a worse overall survival and served as an independent prognostic biomarker in GBM patients. Finally, we demonstrated that LINC00174 acted as an oncogene in GBM cells. All these results suggest that LINC00174 is a novel biomarker and a potential target for GBM treatment.
Footnotes
Acknowledgments
This work was supported by Beijing Talents Foundation from Organization department of Municipal committee of the CPC (No. 2017000021223ZK32) and National Natural Science Foundation of China (No. 81902528).
Conflict of interest
The authors declare that no competing interests exist.
