Abstract
BACKGROUND:
Contactin1 (CNTN1), a member of the immunoglobulin superfamily, is known to correlate with tumor development and progression. Although recent studies have found that elevated CNTN1 has been demonstrated in some types of cancers, the expression and prognosis of CNTN1 in colorectal cancer (CRC) are unclear. Here, we aimed to determine the clinicopathological characteristics and prognostic role of CNTN1 in CRC patients.
METHODS:
The protein expression of CNTN1 in tumor tissues was evaluated by immunohistochemistry. In addition, the mRNA and protein expressions of CNTN1 were examined by qRT-PCR and Western blotting analysis in 40 matched adjacent normal mucosa samples. The relationships of CNTN1 with clinicopathological data and prognosis significance were analyzed.
RESULTS:
Immunohistochemical consequence suggested that the protein level of CNTN1 was obviously raised in CRC compared with adjacent normal mucosa tissues (56.9% vs 10.3%,
CONCLUSION:
Taken together, the findingsindicate that CNTN1 plays a significant role and serve as a potential biomarker for the prediction of adverse prognosis in CRC. Intriguingly, high express of CNTN1
Introduction
Colorectal cancer (CRC) is one of the most frequent malignancies with incidence ranked second in women and third in men worldwide [1]. In China, both the incidence and mortality of CRC have been raising [2]. The 5-year survival rate of CRC patients ranges between 50 and 60% [3]. Although, the mortality rate has declined in recent decades due to the advancements in diagnostic and therapeutic methods, some individuals with CRC always occur recurrence and the clinical results of traditional chemotherapies are still dissatisfied [4]. Therefore, recognition of newfangled prognostic biomarkers and therapeutic targets is of great importance to enhance the survival of CRC patients.
Contactin-1 (CNTN-1), a member of the immunoglobulin family, is a glycosyl phosphatidylinositol (GPI) anchored neuronal membrane protein that functions as the neuronal cell adhesion molecule. Loss of CNTN-1 damages communication or adhesion between muscle and nerve and was therefore considered to be a cause of severe myopathies [5]. Recently, several studies have revealed that CNTN-1 was expressed in diseases outside of the nervous system, particularly cancer. Furthermore, the mounting evidence suggested that CNTN1 was found to mediate tumor invasion and metastasis.
Reports have demonstrated CNTN1 to be up-regulated in many types of cancer such as lung cancer, oesophageal squamous cell carcinomas, gastric cancer, thyroid cancer, prostate cancer and hepatocellular carcinoma, suggesting its contribution to carcinoma progression, invasion, and metastasis. Although evidences have revealed that CNTN1 was acted as a mediator in invasion and metastasis of several tumors, the role of CNTN1 in CRC remains unclear.
In this study, we investigated both the protein and mRNA expressions of CNTN1 in CRC tumor tissue and adjacent non-tumor tissue by quantitative real-time polymerase chain reaction (qRT-PCR) and Western blotting. Furthermore, immunohistochemistry was used to assess the protein expression of CNTN1 in CRC tissues and to evaluate the correlation between the protein expression level of CNTN1 and clinicopathological characteristic as well as patient prognosis. Our data suggested that CNTN1 expression in CRC tissue samples may be used as a potential prognostic factor for CRC patients and increased protein level of CNTN1 expression was correlated with poor prognosis in CRC patients.
Materials and methods
Patients and specimens
During January 2009 and December 2011, 116 cases of CRC that underwent surgical resection at The Second People’s Hospital of Wuhu, along with the corresponding adjacent tissues were converged. The age of the patients ranged from 28 to 85 years, with an average of 61.2
Immunohistochemistry analysis
The expression levels of CNTN1 were detected by immunohistochemistry as described according to the manufacturer’s protocol. Serial tissue sections (4-
The protein expression of CNTN1 was evaluated using a semiquantitative method [7], and the percentage of staining-cell scores (0, no staining; 1,
Quantitative real-time PCR
Total RNA was extracted from frozen tissues using Trizol reagent according to the manufacturer’s instructions. 2
Western blot analysis
Protein extraction was performed using radio-immunoprecipitation assay (RIPA) lysis buffer (Beyotime Institute of Biotechnology, Shanghai, PRC). Protein concentration was determined using BCA assay kit. Equal amounts of proteins were loaded on 10% SDS-PAGE gels. Proteins were then transferred to polyvinylidene fluoride membranes. After being blocked with 5% nonfat milk, membranes were incubated with rabbit primary antibodies against human CNTN1 (Abcam PLC, Cambridge, UK) and GAPDH proteins (Zhongshan Golden Bridge Biotechnology Co Ltd, Beijing, PRC) overnight at 4
Statistical analysis
SPSS 19.0 (SPSS, Inc., Chicago, IL, USA) was used for statistical analyses. Continuous variables were reported as mean and standard deviation (SD). Differences in CNTN1 mRNA and protein expression levels between CRC tissue and non cancerous tissue were examined by the independent Student
Results
Elevated expression of CNTN1 in CRC tissues at mRNA levels
mRNA expression levels of CNTN1 were analyzed for 40 paired tumor and adjacent non-tumor tissues by qRT-PCR. As given in Fig. 1, the median mRNA expression level of CNTN1 was overexpressed in CRC tissues compared with that in the paired adjacent non-cancerous tissues (3.46
Quantitative real-time PCR analysis of CNTN1 expression in 40 pairs of CRC and matched adjacent normal mucosa tissues. *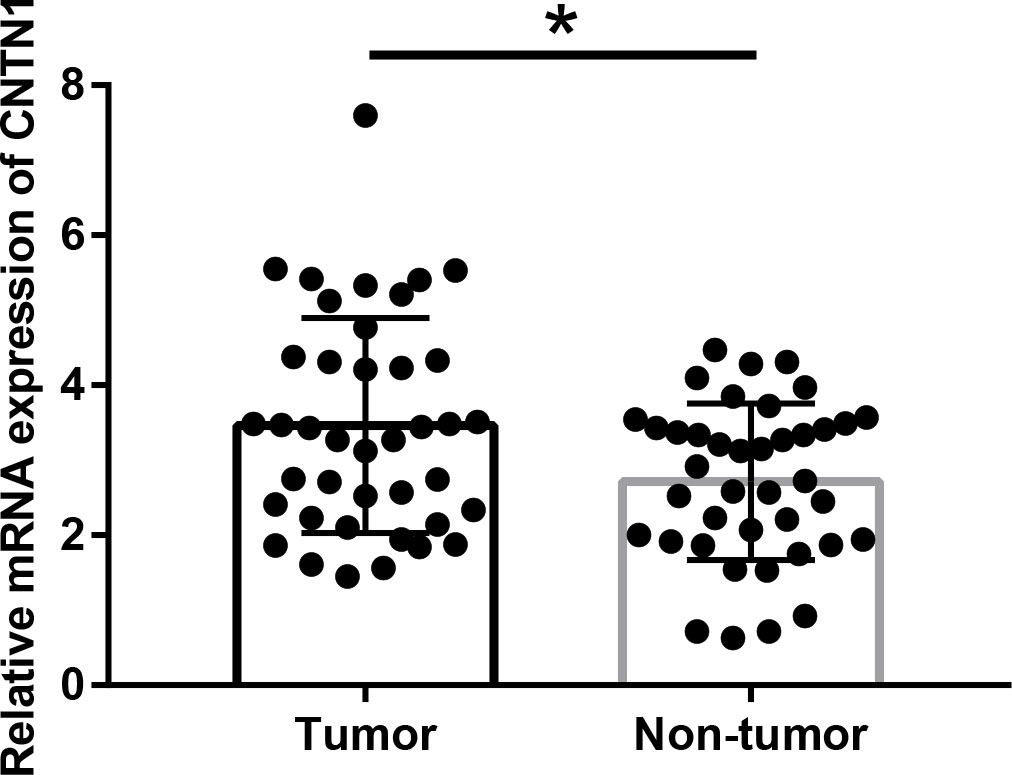
Analyses of CNTN1 protein expression in specimens of tumor tissue and the corresponding non-tumor tissue were performed using samples from 40 patients whose mRNA expression levels were measured for western blotting. Studies revealed that CNTN1 protein levels in CRC and adjacent non-cancerous tissues were 1.39
(a) Western Blotting analysis of CNTN1 expression in 40 pairs of CRC and matched adjacent normal mucosa tissues. **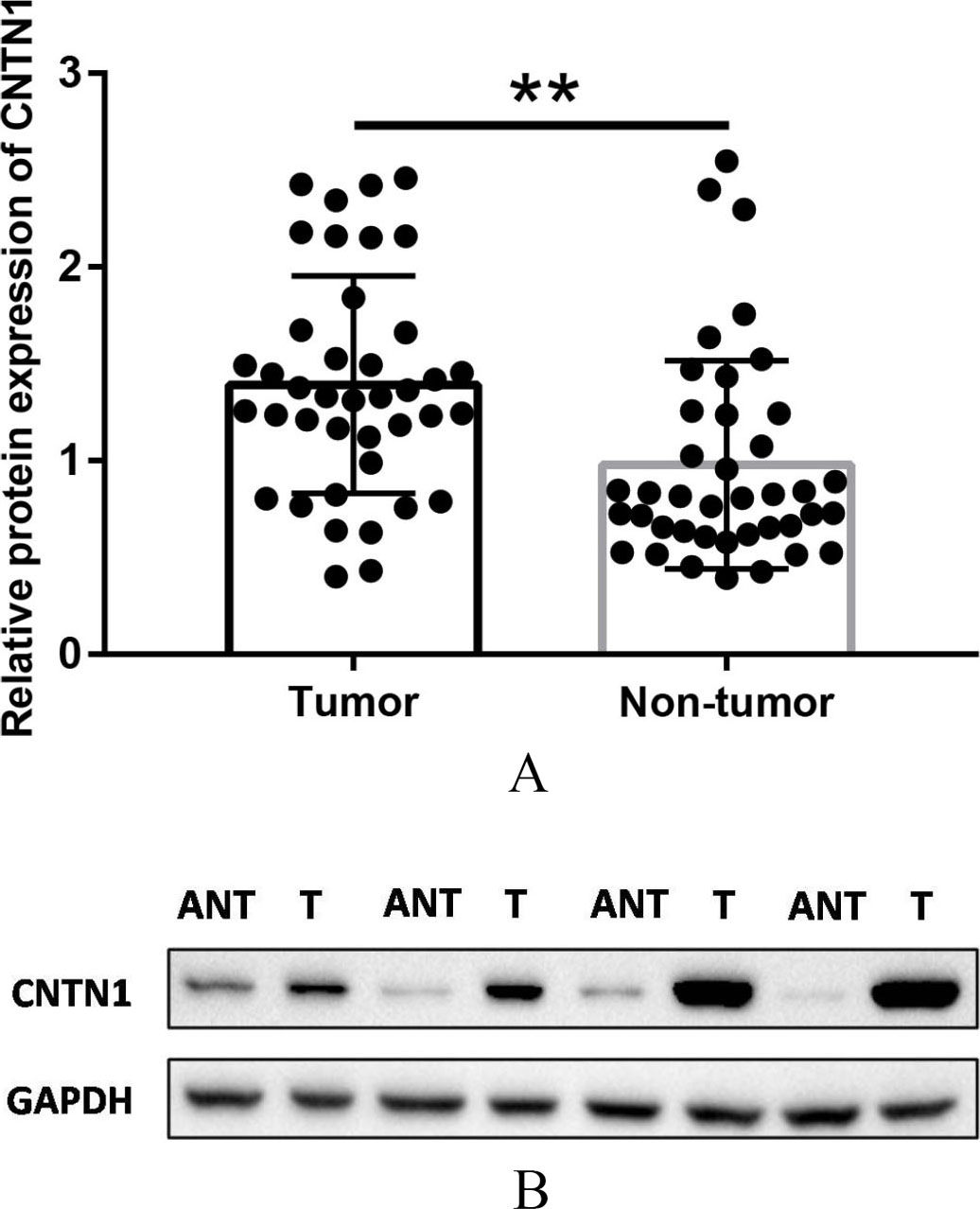
CNTN-1 protein expression in relation to clinicopathologic features
Abbreviations: CNTN-1, contactin-1; LNM, Lymph node metastasis; TNM, tumor node-metastasis; CEA, carcino-embryonic antigen.
Representative immunohistochemical staining of CNTN1 in CRC and matched adjacent normal mucosa tissues. A. Positive expression of CNTN1 in CRC tissues. B. Negative expression of CNTN1 in CRC tissues. C. Positive expression of CNTN1 in matched adjacent normal mucosa tissues. D. Negative expression of CNTN1 in matched adjacent normal mucosa tissues. A-D with 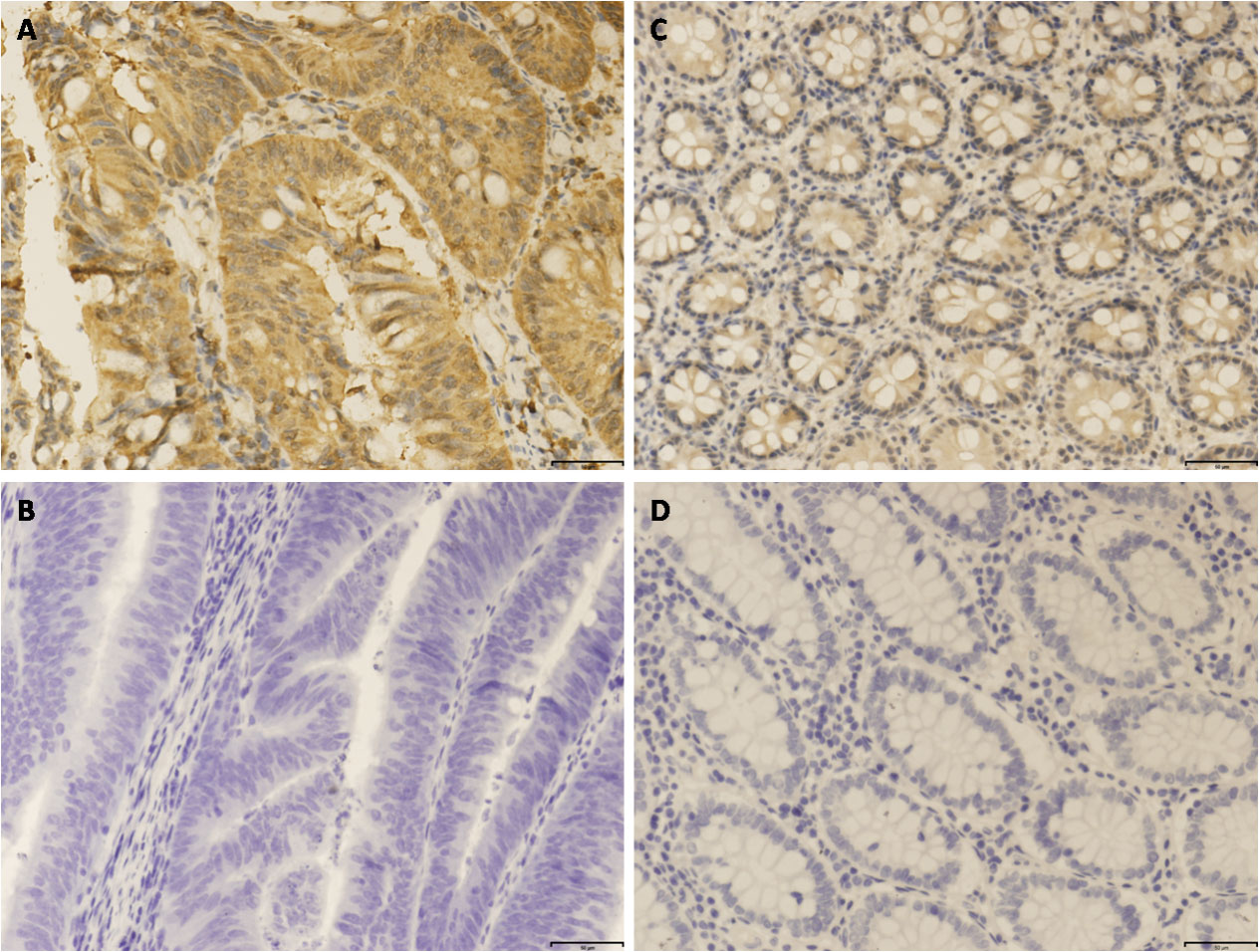
In the present study, the expression of CNTN1 protein was analyzed in 116 CRC samples and corresponding paracarcinomatous samples by immunohistochemistry. Results showed that CNTN1 protein staining is primarily located at cytoplasm with various staining intensity (Fig. 3). The positive rate of CNTN1 protein expression was 56.9 % (66/116) in cancerous samples and 10.3 % (12/116) in adjacent non-cancerous tissues. CNTN1 protein expression was significantly elevated in CRC tissues than that in adjacent non-cancerous CRC tissues (
CNTN1 protein expression correlates with CRC patients’ prognosis
Kaplan-Meier survival analysis was used to assess the relationship between CNTN1 protein expression and patients’ survival. The log-rank test revealed that the overall survival rates of CNTN1-positive group (49.92
Kaplan-Meier analysis of OS in patients with CRC based on CNTN1 positive or negative expression. The CRC patients with CNTN1 positive expression showed notably worse OS rates than those with CNTN1 negative expression. Abbreviations: CNTN1, contactin 1; CRC, colorectal cancer; OS, overall survival.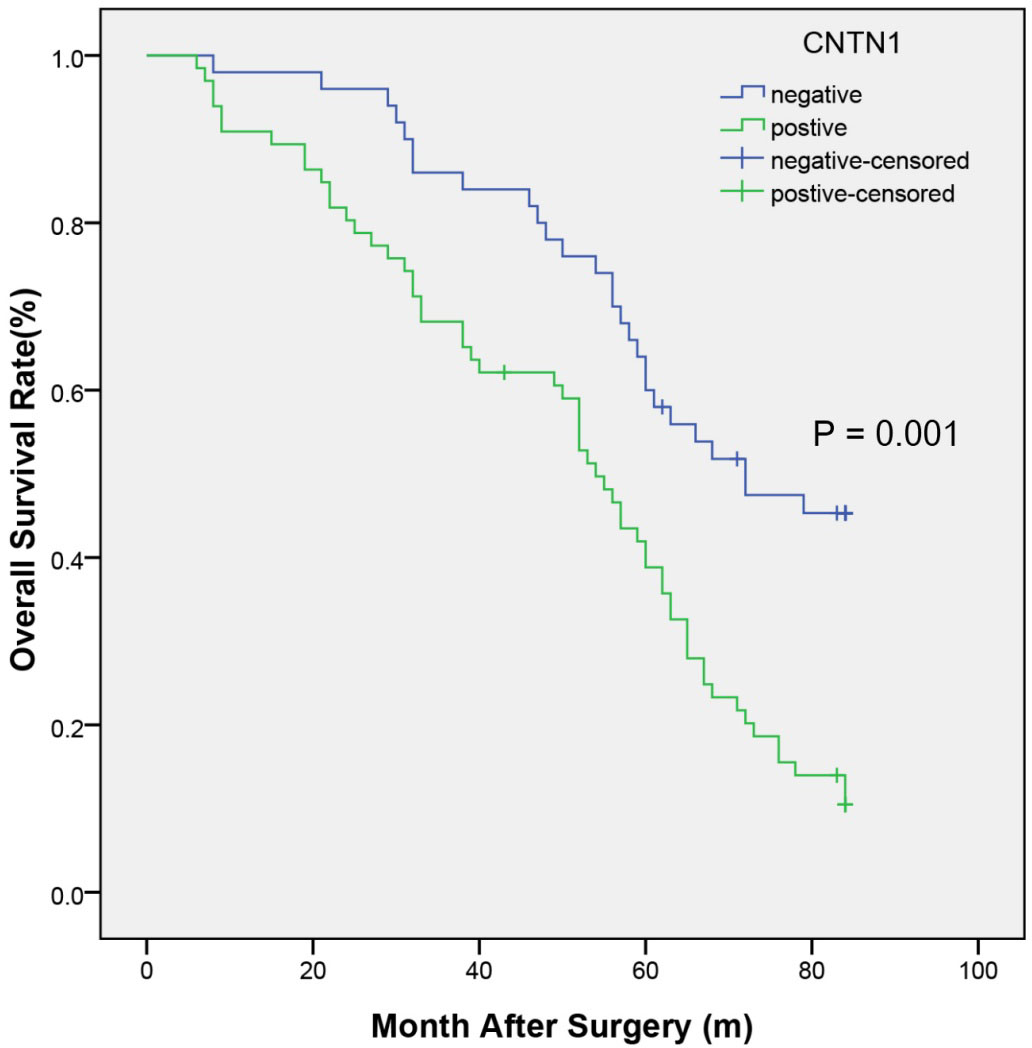
Univariate and multivariate analysis of factors associated with OS
Abbreviations: LNM, Lymph node metastasis; TNM, tumor node-metastasis; CEA, carcino-embryonic antigen; CNTN-1, contactin-1.
Multivariate Cox regression analysis of the risk factors for OS. Distant metastasis is not shown in the figure as its HR is too high. Abbreviations: OS, overall survival; HR, Hazard Ratio.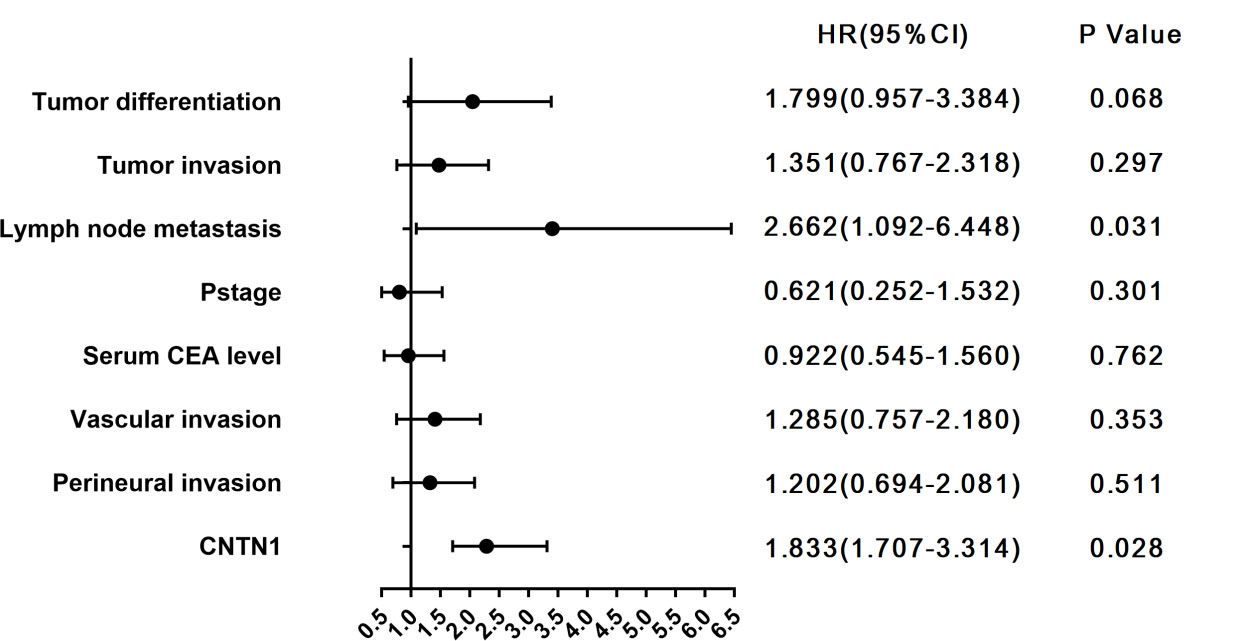
clinocopathological characteristics of patients in each group
Abbreviations: LNM, Lymph node metastasis; TNM, tumor node-metastasis; CEA, carcino-embryonic antigen.
Kaplan-Meier analysis of OS of patients with CRC based on the combination of CNTN1 expression and LNM. The CRC patients with CNTN1 positive and LNM present showed notably worse OS rates than other groups. Abbreviations: CNTN1, contactin 1; CRC, colorectal cancer; OS, overall survival.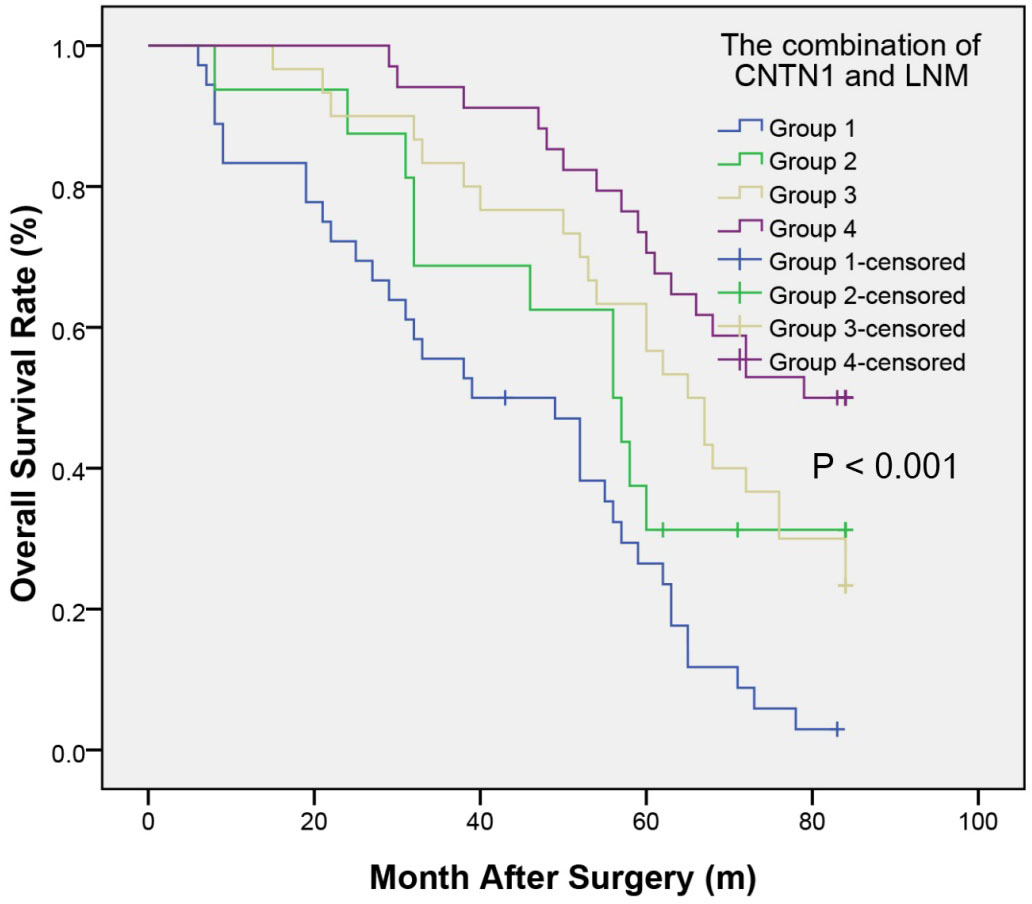
To illuminate the prognostic value of CNTN1 associated with LNM in CRC, patients were divided into four groups: Group 1, the CNTN1-positive and LNM-present group, included 36 patients; Group 2, the CNTN1-positive and LNM-absent, included 16 patients; Group 3, the CNTN1-negative and LNM-present group, included 30 patients; Group 4, the CNTN1-negative and LNM-absent, incuded 34 patients(Table 3). Group 4 had the best OS rates, and Group 1 had the worst OS rates. Group 4 (compared with Group 1) had significantly higher 1-, 3-, and 5-year OS rates (100%, 94.1%, and 75% vs 83.3%, 55.6%, and 25%,
Discussion
Despite upgrades in diagnostic techniques, efficient therapy and evidence based staging systems, the prognosis of CRC remains poor largely based on the depth of tumor invasion and the presence of lymph node metastasis. Currently, molecular mechanisms underlying CRC pathogenesis remain poorly understood. Therefore, early detection and a deeper molecular pathogenesis understanding of CRC are needed to improve diagnosis and treatment and the discovery of the current prognostic molecular predictors is of primary importance.
CNTN-1 is a member of the contact in subgroup of the immunoglobulin superfamily and located in the 12q11–q12 chromosomal region [8]. Previous studies have showed that CNTN-1 is a glycosyl phosphatidylinositol anchor neural cell adhesion molecule (NCAM) exceptionally expressed in many neuronal tissues and is involved in the management of synapse formation, neurite cultivation, fasciculation, and myelin organization [9, 10, 11]. Although increasing research has been focused on the regulatory function of CNTN1 in the nervous system, some of the most recent findings suggest that CNTN1 plays a crucial role in diseases not related to the nervous system, its most remarkable function is its involvement in malignant tumor progression in various cancers such as lung adenocarcinoma [12, 13, 14], esophageal squamous cell carcinoma (ESCC) [15], gastric cancer (GC) [16, 17, 18], oral squamous cell carcinoma (OSCC) [19], thyroid cancer [20], hepatocellular carcinoma [21] and prostate cancer [22]. Su et al. first explained that CNTN-1 may play an important role through RhoA-mediated mechanisms in invasion and metastasis of lung adenocarcinoma cells [12]. Moreover, researches provide evidence that up-regulation of CNTN-1 was partially mediated by VEGF-C/Flt-4-induced enhancement of cancer cell invasiveness and metastasis [13]. Additionally, CNTN1 ablation prominently suppressed the invasion potential of OSCC cell lines [19]. The expression level of CNTN-1 is upregulated in the OSCC tissue and correlated with tumor stage, lymphatic metastasis and invasion [15]. Similarly, CNTN-1 is connected with the expression of VEGFR-3 and VEGF-C in gastric cancer and related to lymphatic node metastasis, which may plays a substantial role on the lymphatic vessel invasion by means of lymphangiogenesis pathway [16]. To illuminate the underlying mechanism by which CNTN-1 accelerates tumor metastasis, Yan et al. observed that CNTN-1 improves AKT activation by preventing PHLPP2-mediated AKT dephosphrorylation in a way [14]. In prostate cancer, CNTN1 knockdown reduced prostate cancer stem-like cells (PCSC)-mediated cancer initiation, whereas CNTN1 overexpression accelerated prostate cancer cell invasion in vitro and facilitated xenograft tumorigenesis and pulmonary metastasis in vivo, which demonstrated that CNTN1 promotes prostate cancer progression and metastasis [22]. Recent studies have proposed that CNTN-1 accelerates cisplatin-resistance in human cisplatin-resistant lung adenocarcinoma through induction of EMT process by activating the PI3K/Akt signaling pathway. These findings gave us clue that CNTN-1 may be a potential therapeutic target to reverse chemo- resistance in cisplatin-resistant lung adenocarcinoma [23]. Kok-Sin et al. successfully identified a group of genes including CNTN1, which show methylation and gene expression changes in colorectal cancer tissues with high purity via integrative epigenomics and genomics data [24]. The integrated analysis gives additional insight regarding the regulation of colorectal cancer-associated genes and their underlying mechanisms that contribute to colorectal carcinogenesis.
In the present study, we are the first to demonstrate CNTN1 protein expression and the prognostic relevance of CNTN1 in CRC tissues, as well as the correlation between CNTN1 expression and clinicopathological features. The immunohistochemical analysis suggested that expression levels of CNTN1 protein were evidently higher in CRC tissues compared to adjacent non-cancerous tissues. Besides, we found that CRC tumors with CNTN1
Besides, previous studies have confirmed the prognostic value of CNTN1 expression in different malignant tumor. Wu et al. found that CNTN1 expression of patients with oral squamous cell carcinoma was dramatically connected with OS [18]. Equally, Yu et al. reported that CNTN-1 expression was significantly associated with poorer prognosis in gastric cancer [15]. Our previous research suggested that CNTN1 may play crucial role in hepatocellular carcinoma and acts as an independent unfavorable prognostic factor for OS and DFS [20]. In the present study, results obtained from the Kaplan-Meier survival analysis suggested that the CRC patients with high CNTN1 expression tended to have poor OS. Multivariate survival analysis based on the Cox regression analysis manifested that CNTN1 was an independent predictor of poor prognosis for both unfavorable OS of CRC patients following curative resection, which revealed that overexpression of CNTN1 had an adverse outcome in CRC, and detection of CNTN1 may be used as a predictive marker of CRC patient survival. Nevertheless, the underlying mechanisms that mediate the function of CNTN1 in CRC progression is unknown and need further research.
In the present study, combining CNTN1 with LNM solved the problem of the low accuracy of CNTN1 alone as a predictor of prognosis. In the first place, it was defined that CNTN1 was positively correlated with LNM. Next patients of CRC were divided into four groups according to positive and negative CNTN1 and absent and present LNM. Research suggested that patients with negative CNTN1 and absent LNM had the best outcome, while those in the positive CNTN1 and present LNM, negative CNTN1 and present LNM, and positive CNTN1 and present LNM had progressively worse outcome. Research results support our hypothesis that combining CNTN1 and LNM would enhance the prognostic accuracy of each one individually in CRC.
Some limitations should be noted. First, the sample size of the single-institute study was relatively small. Besides, there may be a potential selection bias of the cohort due to the sampling method of specimens. Thirdly, we did not clarify the mechanisms how CNTN1 promote tumor angiogenesis of CRC. Consequently, further studies with a larger, multicenter, and detailed mechanism should be necessary to corroborate the present findings and to provide evidence for the potential role of CNTN1 in angiogenesis and CRC progression.
In conclusion, the current findings showed that the upregulation of CNTN1 was significantly associated with tumor size, LNM and TNM stage of CRC.
Overexpression of CNTN1 was an independent risk factor for poor prognosis in CRC patients. Moreover, CNTN1 combined with LNM improved the accuracy of prognosis prediction, suggesting that this combination may be used as a prognostic indicator for CRC patients. Further researches are needed to clarify the function and mechanism of CNTN1 in the progress of CRC.
Footnotes
Acknowledgments
This research is supported by the Clinical Medicine Special Project of Nantong University (No. 2019JQ015) and the Science and Technology Bureau Project of Wenzhou (No. Y20180847).
Conflict of interest
The authors declare that they have no conflict of interest.
