Abstract
BACKGROUND:
This study aimed to determine the prevalence and clinical impact of neurogenic locus notch homolog protein 1 (NOTCH1) mutations among patients with T cell acute lymphoblastic leukemia (T-ALL).
PATIENT AND METHODS:
A cohort of 60 T-ALL cases was included in this study. Sanger sequencing were done for NOTCH1 exon 26, 27, and distal part of exon 34 expanding the sequences encoding transcription activation domain (TAD) and a peptide sequence rich in proline, glutamic acid, serine, threonine (PEST) domains in all studied T ALL patients at diagnosis.
RESULTS:
NOTCH1 mutations was detected in 40 out of 60 T-ALL patients (66%). Mutations in T-ALL patients are deletions (22 mutations) and point mutation (10 mutations). NOTCH1 mutations was found to have no significant impact on clinical outcome and prognosis in T-ALL including overall survival, progression free survival, relapse and mortality (
CONCLUSION:
NOTCH1 mutations were frequently detected in T All patients; however, these mutations did not affect the T ALL patient’s outcome. The high prevalence of NOTCH1 mutations at diagnosis could be used for detection of minimal residual disease in T ALL.
Introduction
T cell acute lymphoblastic leukemia (T-ALL) is an aggressive hematologic tumor associated with poor outcome and represent 25% and 15% of adult and childhood ALL respectively. It results from multistep transformation during which accumulation of genetic abrasion corrupt tumor suppressor genes and developmental pathways implicated in perfect control of cell growth, proliferation, differentiation and survival [1]. In contrast to B-ALL, patients with T ALL always presented by heavy tumor burden and usual have poor outcome. The factors determining T ALL aggressiveness and poor response to therapy is not understood and remains to be explained [1].
NOTCH1 is a class I transmembrane (TM) protein which directly transduce extracellular signal into gene expression changes which function as ligand activated transcription factors. Interaction of the receptor with D like and Jagged ligands expressed on the surface of neighboring cells initiate NOTCH1 signal. This ligand receptor interaction cleaves extracellular domains of the NOTCH1 receptor by the metalloprotease (ADAM10), that facilitates subsequent proteolytic cleavage by g secretase complex in the transmembrane region of the receptor [2]. NOTCH1 pathway is an evolutionarily conserved signaling pathway that regulates cell fate specification differentiation, and patterning. Mutations of NOTCH1 ligands and receptors are implicated in numerous congenital and acquired human diseases [3]. Multiple studies revealed that NOTCH1 plays a prominent role in T-ALL, choriocarcinoma and breast cancer [4, 5], and chronic lymphocytic leukemia [6]. Favorable outcome was observed in adult T-ALL patient samples with NOTCH1 and/or FBXW7 mutations [7].
The data regarding the prevalence and impact and types of NOTCH1 mutations in T-ALL patients among Egyptian is not previously identified. The aim of the current study was to detect the prevalence of NOTCH1 mutations among Egyptian patients with T-ALL, as well as, it’s relation to disease behavior, and patient outcome.
Patients and method
This study was carried out on 60 newly diagnosed patients with T-ALL attending Mansoura University Oncology Center (MUOC) before start of therapy; after giving the informed consent from all participating patients. Local ethical committee approved this study. The Age of T-ALL patients was ranging from 15–66 years. T-ALL patients was diagnosed by complete blood counts including blood smear; cytological examination of bone marrow smear, and flowcytometry for characteristics immunophenotyping. Median follow up of T-ALL cases was 15 months ranging from 10 to 32 months. The exclusion criteria include age less than 15 years; the diagnosis of T-lymphoblastic lymphoma according to WHO classification; and patients presenting with lymphomatous signs including isolated mediastinal mass. During follow up we recorded induction of remission response, overall survival (OS), disease free survival (DFS).
Induction in adult T-ALL cases, was by either augmented Berlin-Frankfurt-Münster (aBFM) protocol [8] or modified hyper CVAD (cyclophosphamide, vincristine, dexamethasone, doxorubicin) (anthracycline intensification) [9] in relapsed or high risk ALL cases and gemcitabine was used in pediatric T-ALL cases [10].
Follow up and prognostic criteria
Hematological remission was defined as restoration of normal hematopoiesis with a blast cells
Methods
Three ml of peripheral blood was taken on EDTA tube for complete blood counts (CBC), flowcytometry. Peripheral blood films were prepared for morphological examination. 2 ml of bone marrow was aspirated on EDTA; 1 ml for immunophenotyping and 1 ml for DNA extraction. DNA amplification was performed by conventional PCR with NOTCH1 mutation analysis by direct Sanger sequencing of PCR products encompassing NOTCH1 exon 26, 27, and the distal part of exon 34 including the sequences encoding TAD and PEST domain. Sequencing Primers were as follows: Exon 26 Forward (FW): 5-GGAAGGCGGCCTGAGCGTGTC-3; Exon 26 Revers (RV): 5-ATTGACCGTGGGCGCCGGGTC-3; with annealing temperature of 67.5, C Exon 27 FW: 5-GCCTCAGTGTCCTGCGGC-3; Exon 27 RV: 5-GCACAAACAGCCAGCGTGTC-3; with annealing temperature of 60 C, Exon 34 FW1 (TAD): 5-GCTGG CCTTTGAGACTGGC-3; Exon 34 RV1 (TAD): 5-GC TGAGCTCACGCCAAGGT-3; with annealing temperature of 63 C, Exon 34 FW2 (Pest): 5-CAGATGCA GCAGCAGAACCTG-3; and exon 34 RV2 (Pest): 5-AAAGGAAGCCGGGGTCTCGT-3 with annealing temperature of 64 C.
Statistical analysis
The statistical analysis of data was done using excel program (Microsoft Office 2013) and SPSS (statistical package for social science) program (SPSS, Inc, Chicago, IL, USA) version 20. Qualitative data were presented as frequency and percentage. Chi square and Fisher’s exact tests were used to compare groups. Quantitative data were presented by mean, SD or median and range. Comparisons between two groups were done using t-test or Man Whitney (for non-parametric). Kaplan – Meier test was used for survival analysis and the statistical significance of differences among curves was determined by Log-Rank test. Cox regression analysis was used for prediction of OS.
Age and gender in NOTCH1 mutated T ALL cases versus those that have wild type
Age and gender in NOTCH1 mutated T ALL cases versus those that have wild type
Hematological data in NOTCH1 mutated T ALL cases versus those that have wild type
A: The Point mutation A5023T in HD domain (exon 27) of NOTCH1 gene. B: Wild NOTCH1 sequence in the same position.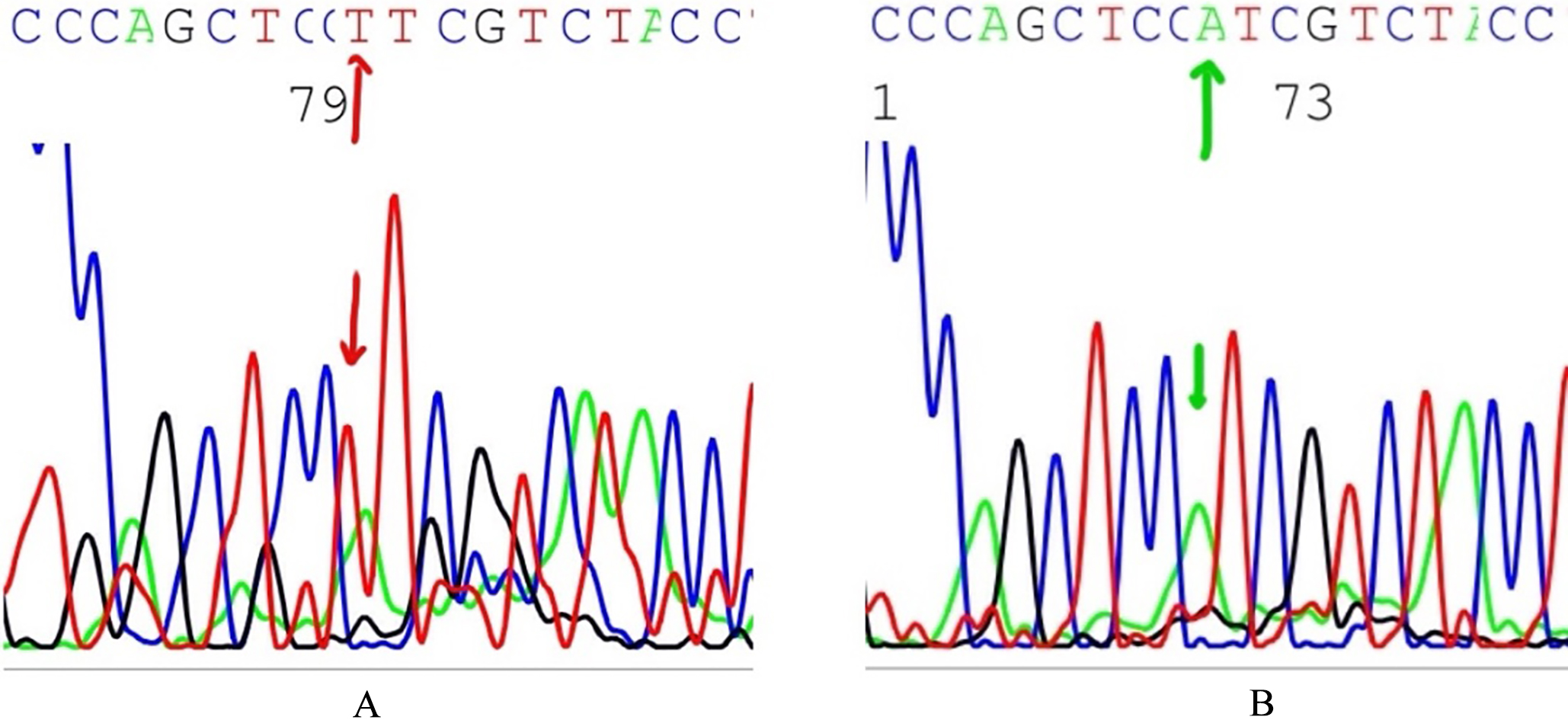
NOTCH1 mutations was detected in 40 out of 60 of T-ALL patients (66%). The highest frequency of mutations was detected in exon 26 (40%) (24 out of 60 cases) and exon 27 (40%) (24 out of 60 cases), followed by exon 34 (Pest domain) 6.6% (4 out of 60 cases). Four have mutations in exon 26 only, and 12 cases have mutations in exon 27 only, and 8 cases have mutations in both exon 26 and exon 27, and only 4 cases were mutated in both exon 27 and exon 34 (best domain). Twelve cases had single mutation and 16 cases had 2 mutations and only 4 cases had 3 mutations. Most of these mutations are deletions (22 mutations) (4609 Del A) and point mutations (10 mutations) (A5023T), see Fig. 1.
The frequency of NOTCH1 mutations in T-ALL did not differ according to sex or age (
In T-ALL cases, no significant differences in clinical outcome in relation to NOTCH1 mutation. Cases who achieved complete remission in wild type cases were 60% but in mutated cases were 100% (
Clinical course in NOTCH1 mutated T ALL cases versus those that have wild type
Clinical course in NOTCH1 mutated T ALL cases versus those that have wild type
The presence of NOTCH1 mutations in T ALL patients did not have impact on either overall survival or disease-free survival (
Impact of NOTCH1 mutations on T ALL patient’s OS. No statistically significant differences in OS in relation to NOTCH1 mutation within T-ALL cases (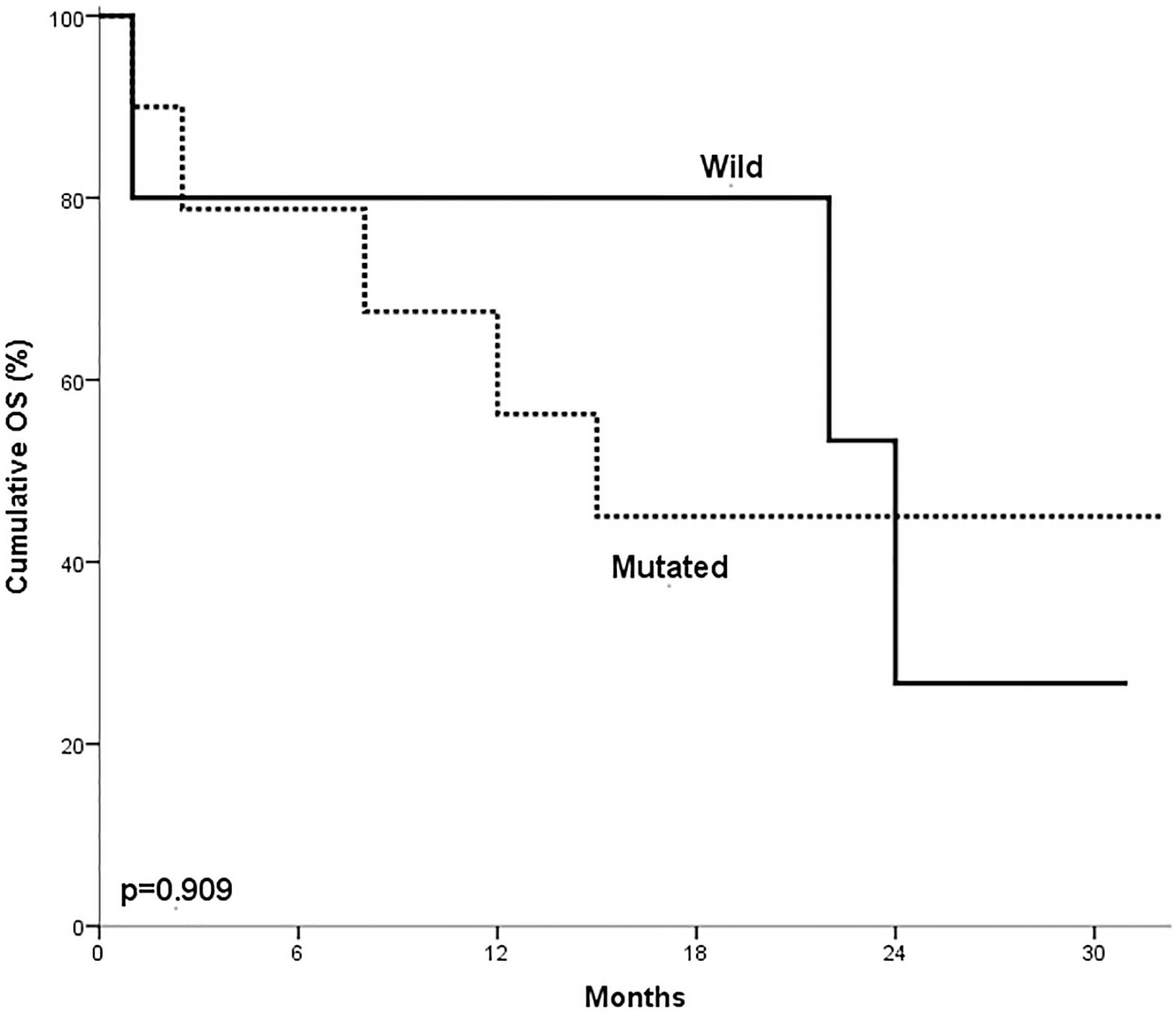
Impact of NOTCH1 mutations on T ALL patients DFS. No statistically significant differences in DFS in relation to NOTCH1 mutation within T-ALL cases (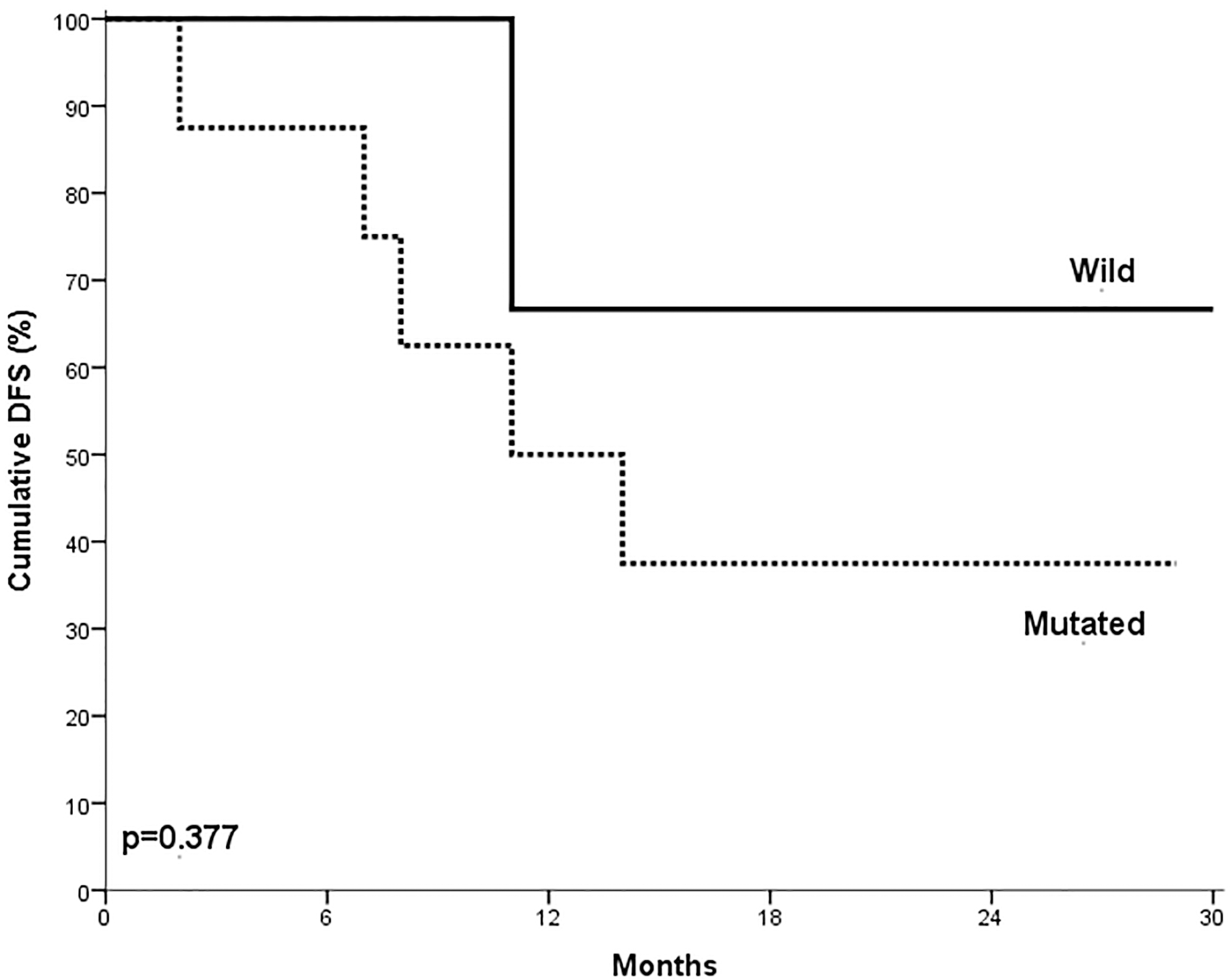
Notch signaling pathway is critical for the T-cell ontogeny and also in T-ALL. The Notch pathway regulates many important cell processes that control cellular transformation, cell-cycle progression and apoptosis. Expression of NOTCH1 gene has been implicated in promotion of T cell lineage choice and commonly it is found mutated in T-ALL [15]. In our study, NOTCH1 mutations were found in 66% of T-ALL cases that was somewhat lower than that was previously reported 71% [16] and 76% [17]. This could be attributed to the difference in race and in the pathogenesis of T-ALL in our cohort of patients [7].
No statistically significant differences were detected in the age and gender in relation to NOTCH1 mutations in T-ALL patients. These findings were consistent with the findings reported by Zhu et al. [15]. However, Fogelstrand et al. [18] reported that the frequency of NOTCH1 mutations increased with age.
NOTCH1 mutations were significantly associated with lower BM blasts count in T-ALL cases with no significant variations in the other hematologic data. This data consistent with that reported by Fogelstrand et al. [18]. However, Zhu et al. [15] reported significant association of NOTCH1 mutations with higher WBCs count. The lower blast cell counts in the bone marrow in T-ALL patients with mutated NOTCH1 could be attributed to increased egress of blast cells from the bone marrow to peripheral tissues.
NOTCH1 mutations did not have significant impact on the overall survival (OS) in our studied T-ALL cases. This result was in agreement with the finding reported by Fogelstrand et al. [18] and Baldus et al. [12]. On the other hand, Zhu et al. [15] reported that NOTCH1 mutations were associated with shorter OS in T-ALL cases. However, Breit et al. [19] reported that NOTCH1 mutations in T-ALL patients had better OS when compared with wild type cases.
DFS was not significantly different in NOTCH1 mutated and unmutated T ALL patients. This was consistent with the data reported by Fogelstrand et al. [18] and Baldus et al. [12]. On the other hand, Breit et al. [19] demonstrated that patients with NOTCH1 mutations had better DFS when compared with wild type cases. However, Zhu et al. [15] reported that NOTCH1 mutations were associated with shorter DFS in T-ALL patients.
No statistically significant differences were found in remission induction rates in relation to NOTCH1 mutation in studied T-ALL patients. This observation was in agreement with Fogelstrand et al. [18] and Baldus et al. [12]. There were no statistically significant variations in relapse rates according to NOTCH1 mutation in T-ALL patients. This was in agreement with the results of Fogelstrand et al. [18] and Baldus et al. [12]. On the other hand, relapse rate was significantly increased in wild type T-ALL cases than NOTCH1 mutated cases in Breit et al. [19]. No statistically significant differences in remission induction in relation to NOTCH1 mutation in T-ALL cases. This was consistent with that found by Fogelstrand et al. [18].
No statistically significant differences in prognosis in relation to NOTCH1 mutation in T-ALL cases. This was consistent with the data reported by Zuurbier et al. [20] and Baldus et al. [12]. However, Breit et al. [19] and Van Vlierberghe et al. [7] showed good prognosis for NOTCH1 mutated T-ALL patients. On the other hand, Zhu et al. [15] reported that NOTCH1 mutated T-ALL case showed poor prognosis due to the NOTCH1 mutations which disturb interaction of HD domain subunits, with constitutive NOTCH1 activation resulting in cell transformation or by truncating NOTCH1 mutations in PEST domain which are suggested to increase half-life of the intracellular portion of NOTCH1 receptor (ICN1).
Conclusion
NOTCH1 mutations had no significant impact on patient outcome in T-ALL patients. The high prevalence of NOTCH1 mutations at diagnosis could be used for detection of minimal residual disease in T ALL.
Footnotes
Conflict of interest
The authors declare that there is no conflict of interest.
