Abstract
BACKGROUND:
Prognostic factors are not well exploited in childhood T-cell acute lymphoblastic leukemia (T-ALL).
OBJECTIVE:
The aim of this study was to analyze the prognostic role of CD38 as well as minimal residual disease (MRD) and other biological factors in T-ALL.
METHODS:
Immunophenotyping of bone marrow (BM) at diagnosis and MRD levels were determined using a standard panel of antibodies by 4-colour flow cytometry. A total of 96 children with T-ALL were enrolled.
RESULTS:
The results showed that 97.9% of T-ALL patients were positive for CD38 with a median level of 85.3%. CD38-high group had a worse early treatment response than the CD38-low group. However, CD38 levels were not associated with prognosis, albeit CD38-high group had a worse 5-year event free survival rate (55.1% vs. 66.6%,
CONCLUSIONS:
We conclude that T-ALL has a high level of CD38 expression which is not associated with prognosis. Very high MRD level (
Introduction
Outcomes of childhood T-cell acute lymphoblastic leukemia (T-ALL) have been steadily improved with current risk-adapted therapy, as T-ALL patients are normally assigned to the intermediate-risk (IR) or high-risk (HR) arms of contemporary clinical trials. However, relapse is still the most common cause of treatment failure and patients with refractory and relapsed T-ALL have a particularly dismal outcome compared to the B-cell ALL (B-ALL) patients [1, 2, 3, 4]. Few effective therapies exist for refractory and relapsed T-ALL patients yet. So far, the most independent determinant in risk classification and prognostic prediction is minimal residual disease (MRD). Nevertheless, a number of patients with favourable MRD still experience relapse. Therefore, it is necessary to further improve the identification of patients with high risk of relapse.
CD38 is a 45 kDa type II transmembrane protein expressed on thymocytes, activated T cells and terminally differentiated B cells, etc. [5]. Some hematologic malignancies also express different levels of CD38 [6, 7, 8], such as multiple myeloma (MM), chronic lymphocytic leukemia (CLL), non-Hodgkin lymphoma (NHL), acute lymphoblastic leukemia (ALL) and acute myeloid leukemia (AML). In hairy cell leukemia, CD38 is a marker of poor prognosis [9], and CD38
However, the reports regarding CD38 expression and the association with progression in pediatric ALL are scarce, and even less in T-ALL. In 1993, St. Jude Children’s Research Hospital reviewed 325 ALL patients, found T-cell ALL cases were more likely than B-lineage ALL cases to express CD38 and the expression of CD38 was not related to any clinical or biologic feature [17]. Bras et al. [6] showed CD38 was significantly higher in the pediatric B-ALL than that of AML patient and lower than that of NHL patients. They evaluated 28 pediatric T-ALL patients and indicated that CD38 expression levels were comparable to T-cell lymphoblastic lymphoma. However, there are also some different opinions. Karawajew et al. [18] reported that CD38 was generally under-expressed in childhood ALL.
In this study, the flow cytometric data of 96 cases of childhood T-ALL was analyzed and the prognostic role of CD38 expression was assessed. The results showed that 97.9% of T-ALL patients were positive (
Methods
Patients
Between January 2009 and December 2017, 96 evaluable children with newly diagnosed T-ALL treated at our hospital were enrolled. The diagnosis of T-ALL was based on morphology and immunophenotyping. The genetic abnormalities were examined by PCR and the conventional karyotypes were determined by standard methods at diagnosis. Patients who had white blood cell (WBC) count more than 100
Immunophenotyping and minimal residual disease detection
Flow cytometric immunophenotyping of bone marrow (BM) at diagnosis was performed by 4-colour flow cytometry (FACSCalibur with CellQuest software, Becton Dickinson, San Jose, CA, USA) using a standard panel of antibodies. The panel of monoclonal antibodies (BD Biosciences) included the following: CD10, CD19, CD20, cytoplasmic (c) and surface (s) CD22, sIgM for B-lineage, CD1a, CD2, cCD3, sCD3, CD4, CD5, CD7, and CD8 for T-lineage, CD11b, CD13, CD14, CD15, CD33, CD117, and MPO for myeloid lineage, CD34, CD38, CD45, HLA-DR, CD56, and nTdT as non-lineage markers. Isotype antibodies were used as negative controls. CD45 gating strategy was used to identify the blast population. The threshold of
The characteristics of the T-ALL, B-ALL and AML patients
The characteristics of the T-ALL, B-ALL and AML patients
After the bone marrow was labelled with antibodies, a lysing reagent (BD lyse, Becton Dickinson, USA) was added to the bone marrow and then washed twice by PBS, followed by adding 0.5% paraformaldehyde to fix the cells. Then the BM minimal residual disease (MRD) in mononuclear cells was determined by 4-colour flow cytometry [19] at the following time points (TP): TP1, on day 15 of remission induction; TP2, on day 33 at the end of remission induction and before consolidation therapy; TP3, on week 10 before the early intensification; TP4, on month 6 before the first late intensification; TP5, on month 12 before the second late intensification; TP6, on month 24 during the maintenance therapy; TP7, on month 36 of maintenance therapy for those remaining in clinical remission.
All the patients were treated in our hospital with National Protocol of Childhood Leukemia in China (NPCLC)-ALL2008 protocol [20], which was modified from protocol NPCAC97. After 7 days of prednisone pre-test, all patients received a 4-week remission induction regimen with vincristine, daunorubicin, L-asparaginase and dexamethason (VDLD). Additional doses of L-asparaginase were given to HR patients. A 2-week consolidation therapy consisting of cyclophosphamide, cytarabine, 6-mercaptopurine (6-MP) and a 4-week extramedullary leukemia prophylaxis including 3 courses of high-dose methotrexate (HD-MTX, 5 g/m
Statistical analysis
The survival rates of event-free survival (EFS) and overall survival (OS) were estimated with Kaplan-Meier analysis and log-rank test. The duration of EFS was defined as the time from the date of diagnosis until the first event (including relapse, development of a second malignancy or death) or until the date of last contact. The duration of EFS of patients who did not reach CR was considered as 0. Cumulative incidence of relapse (CIR) was compared with Gray’s test. Comparison of continuous and categorical variables was performed by Mann-Whitney U test and Chi-square test. Multivariate analysis was conducted by using Cox proportional hazards regression. A
Results
For the cohort of 96 T-ALL patients, 72 male and 24 female, the median age was 9.5 years, range from 1.0 year to 16.0 years. The majority of the characteristics were different compared to the B-ALL patients and AML patients (Table 1). T-ALL had more male patients (
The survival of T-ALL patients. A, The 5-year EFS and OS rates of the T-ALL patients; B, The 5-year OS rate of the T-ALL patients compared between CD38-high and CD38-low groups; C, The 5-year EFS rate of the T-ALL patients compared between CD38-high and CD38-low groups; D, The 5-year CIR rate of the T-ALL patients compared between CD38-high and CD38-low groups.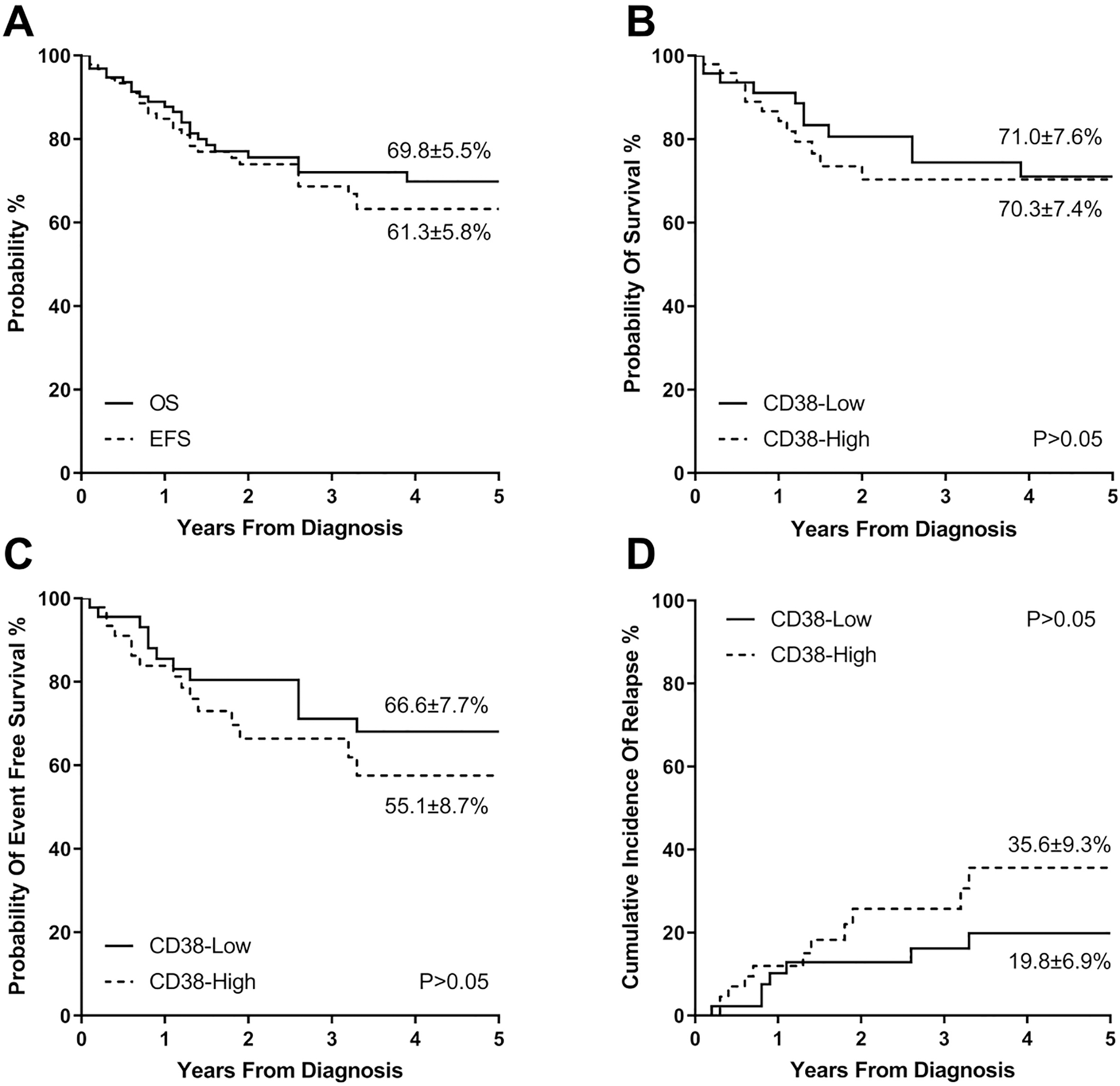
Of the 96 patients, 92 (95.8%) patients achieved CR, of whom, 74 (77.1%) achieved CR on the Day 15 of remission induction and 88 (91.7%) on the Day 33 after remission induction and before consolidation therapy. The 5-year EFS and OS rates were 61.3
Comparison of CD38 expression in the T-ALL, B-ALL and AML patients, 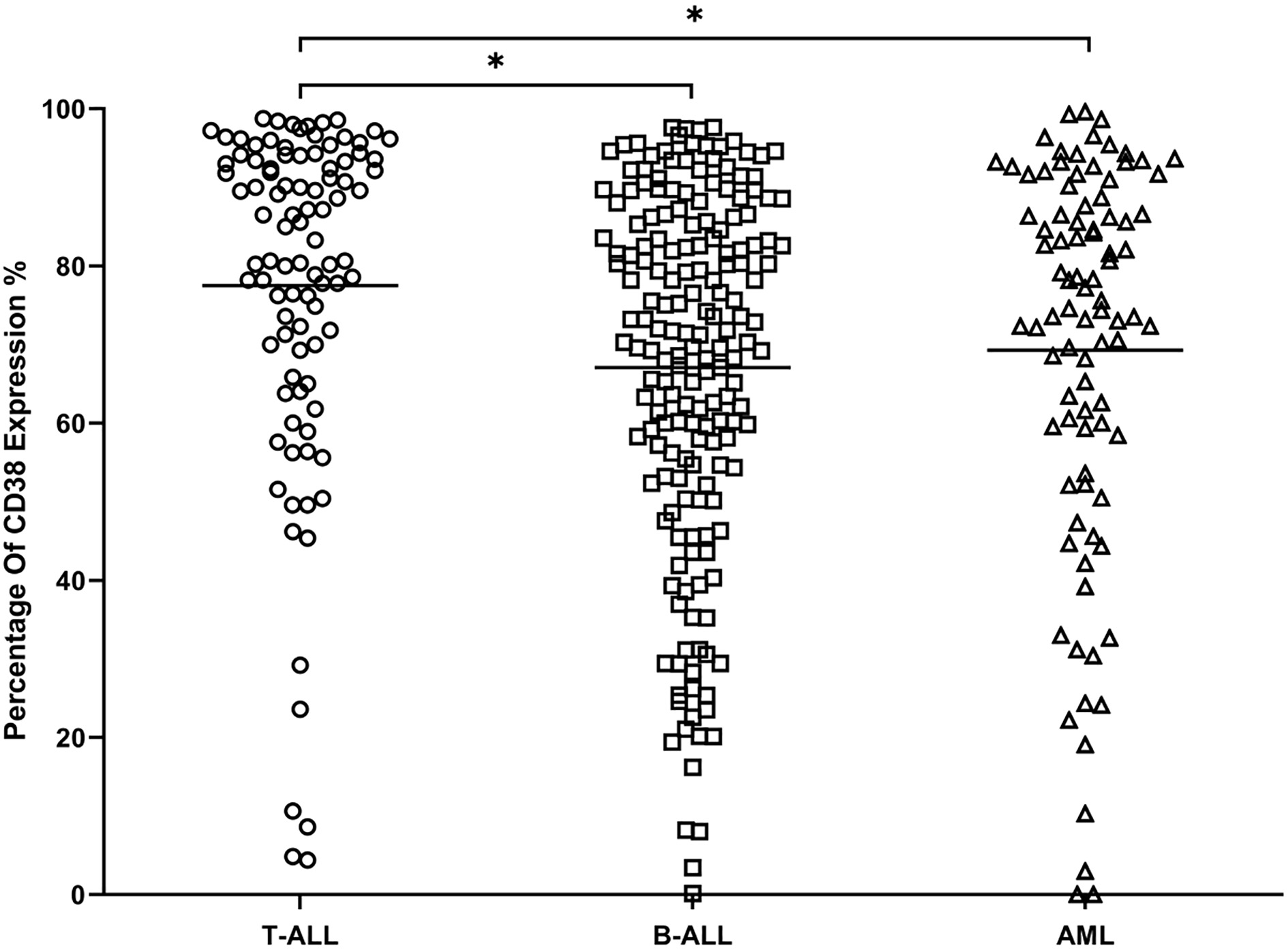
The comparison of CD38 expression levels between the relapsed (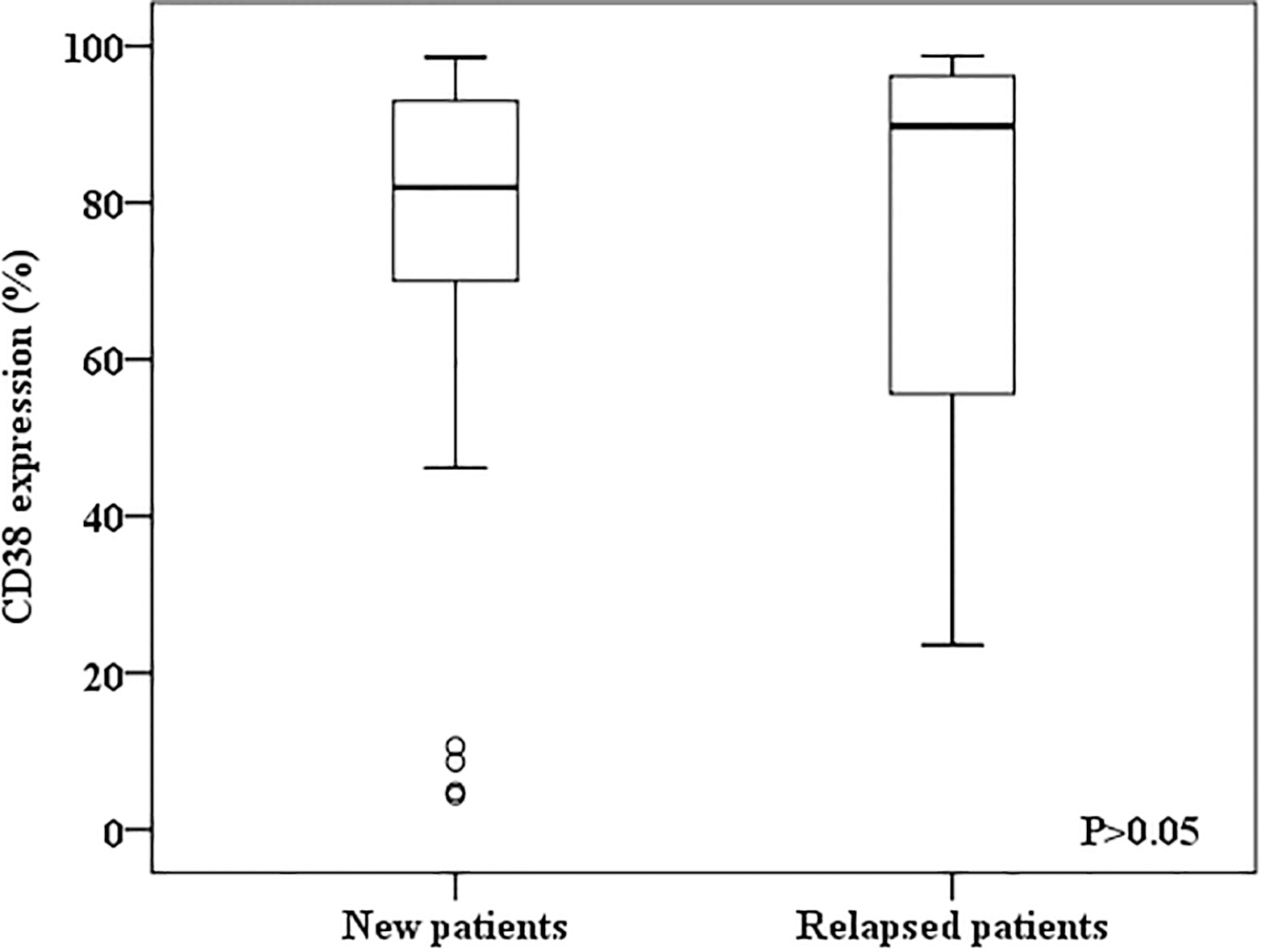
CD38 expression was not associated with gender, age, WBC at diagnosis, relapsed or not. In relapsed cases, the CD38 expression levels were comparable to the newly diagnosed patients (Median levels were 82.0% and 89.8%, respectively,
There was also no statistically significant difference in the expression of the markers CD1a, CD2, CD3, CD4, CD5, CD7, CD8, CD13, CD33, CD34, CD56 and CD117 with regard to inferior survival (
CD38 expression on T-ALL detected by flow cytometry
The cut-off point for CD38 expression as high or low was 85%.
The association analysis of high MRD levels and the survival. A, The 5-year EFS compared between the patients with MRD 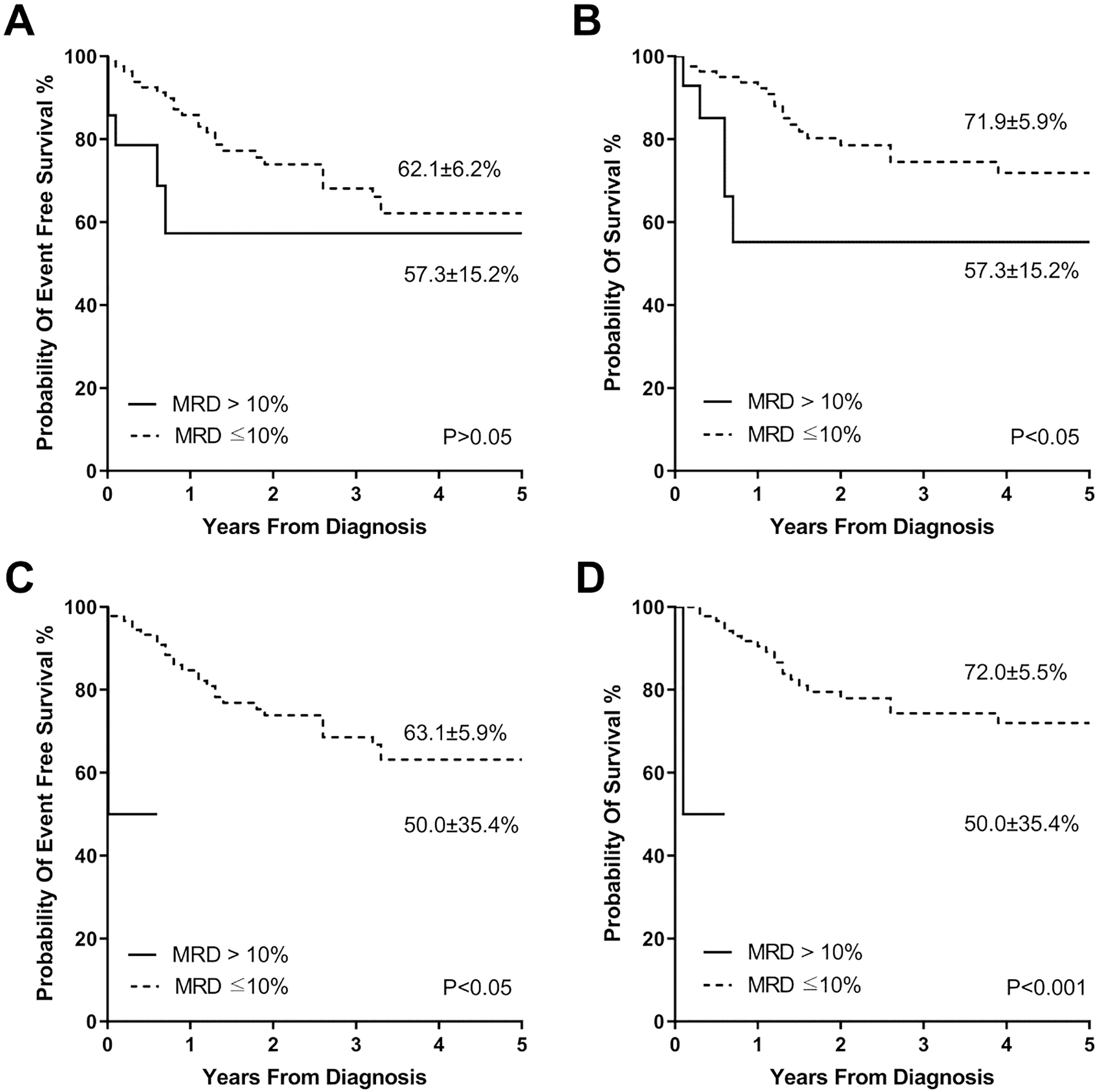
We further analysed the relationship between the very high MRD levels (
Multivariate cox regression analysis was performed for the risk of relapse in T-ALL patients. However, none of the clinical and biological features including gender, age, WBC at diagnosis, risk group, response to prednisone, MRD levels, or immunophenotype, including CD38 expression level showed significant relation to the hazard of relapse (
Patients with T-ALL especially refractory and relapsed T-ALL have particularly dismal outcomes compared to the B-ALL. Considering the high incidence of relapse and poor survival after relapse, it is important to identify patients early who are at risk of relapse. MRD monitoring has been proven valuable to predict the prognosis in ALL by many studies, especially in B-ALL [21, 22, 23]. The importance of MRD analyses is exemplified by the AIEOP-BFM-ALL 2000 study that showed that MRD
Recently, CD38 targeting monoclonal antibodies, such as Daratumumab and Isatuximab, have been successfully applied in the therapy of multiple myeloma patients. CD38 has a rather unique pattern of expression in lymphocytes. Naïve lymphocytes and peripheral B and T lymphocytes have weak to negative expression of CD38 while this molecule is strongly expressed on lymphocyte precursors and activated lymphocytes. The high level expression of CD38 on T-ALL cell surface makes the molecule an attractive target for antibody-based treatment strategy in childhood T-ALL.
Our study have showed that 97.9% of the T-ALL patients are positive (
In conclusion, CD38 levels are not associated with the survival in pediatric T-ALL and the high expression of CD38 on this malignancy indicates that CD38 might be a novel molecular target for the antibody-based treatment of this disease.
Footnotes
Acknowledgments
This study was supported in part by grants from National Natural Science Foundation of China (Nos. 81770202 and 81470304), the Pediatric Leukemia Diagnostic and Therapeutic Technology Research Center of Zhejiang Province, the Doctoral Program of Higher Education (No: 20130101120057), and the General Program of Health department of Zhejiang Province (2014KYB145). We would also like to thank Mr. Hong-Qiang Shen, Mrs. Bai-Qin Qian and Dr. Si-Si Li for their excellent technical support on flow cytometry.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-190946.
