Abstract
OBJECTIVE:
In this study, we determined the prognostic values of magnetic resonance imaging (MRI)-based primary tumor regression and serum squamous cell carcinoma antigen (SSCC-Ag) levels 4 weeks after definitive radiotherapy (RT) in cervical squamous cell carcinoma (CSCC) patients.
METHODS:
This was a retrospective study involving 218 patients with histologically confirmed CSCC (stages IB-IVA). All the patients received definitive RT. Pre- and post-RT pelvic MRI and SSCC-Ag levels were measured. Locoregional control (LRC), distant metastasis-free survival (DMFS), disease-free survival (DFS), and overall survival (OS) were evaluated, and possible OS prognostic factors were analyzed.
RESULTS:
The median follow-up time was 25.57 (1.73–58.93) months. Thirty-six and 68 patients died and experienced recurrence, respectively, and the primary tumors of 130 (59.6%) and 88 (40.4%) patients exhibited complete response (CR) and non-CR, respectively. The 3-year OS, DFS, LRC, and DMFS rates were significantly higher in the CR than in the non-CR patients (85.2% vs. 67.9%, 78.9% vs. 39.0%, 93.4% vs. 63.8%, and 83.4% vs. 54.5%, respectively;
CONCLUSION:
Post-RT SSCC-Ag levels are OS independent prognostic factors in CSCC patients receiving RT. Timely and optimized treatment plans for CSCC patients after 4 weeks of RT are necessary when patients with persistent tumor and/or positive SSCC-Ag.
Introduction
Cervical cancer (CC) is a common malignancy that adversely affects women’s health in China [1, 2]. Definitive concurrent chemoradiotherapy (CCRT) is an effective treatment for locally advanced CC [3]. Advances in diagnostic techniques and treatments have improved the survival rate of CC patients. However, no breakthrough has been made in the treatment of advanced CC, and the 5-year survival rates of stage II and stage IV patients are 50–65% [4] and 20–25% [5], respectively.
Locoregional recurrence and distant metastasis are the main causes of treatment failure in patients with CC, and recurrence mainly occurs within 2 years of the first treatment [6, 7]. Therefore, it is necessary to evaluate the tumor response after radiotherapy (RT) for CC. In addition, early detection of tumor recurrence is also needed as it enables the delivery of effective salvage therapies in order to improve long-term survival. However, how to evaluate the extent of residual tumor burden remains controversial. Patients are reluctant to undergo biopsy after radiation therapy because the procedure is distressing and painful despite the administration of local anesthesia [8]. Positron emission tomography with 18F-fluorodexyglucose [9, 10] can be used to detect residual or recurrent tumors in CC patients after completion of the treatment, but its high cost has precluded its widespread use in resource-limited countries. Magnetic resonance imaging (MRI) is the preferred modality for determining the extent of soft tissue or perineural infiltration of CC [11]. After RT, most CC patients in China are regularly monitored using MRI. However, there is still no consensus about the optimal time point to evaluate treatment response using MRI after RT. Lee et al. [12] reported that following up patients with MRI for a median of 35 days after the beginning of CCRT can effectively predict distant tumor metastasis and disease-specific survival. Another study showed that detection of residual tumor volume by MRI 4 weeks after RT was an important prognostic factor for local control of CC [13]. In some hospitals, response evaluation by MRI is performed at 8–12 weeks after RT [14, 15, 16]. In our hospital, MRI is used to detect residual diseases 4 weeks after the end of the treatment, mainly due to our desire to diagnose residual tumor as soon as possible, so that patients could receive timely salvage therapy, which might improve their survival.
Certain studies have assessed the relationship between tumor regression and survival outcome in patients with CC [17, 18]. Another study [19] reported that a tumor volume reduction rate of 90% or less after RT was a significantly adverse factor for progression-free survival in CC patients. However, that study had a few shortcomings, such as the sample size was small, only the mid-RT response of the patients was measured, and conventional external beam RT was used instead of intensity-modulated radiation therapy (IMRT).
Besides gross tumor regression, the serum squamous cell carcinoma antigen (SSCC-Ag) level was reported to be a reliable diagnostic marker for cervical squamous cell carcinoma (CSCC) and monitoring CSCC treatment in clinical practice [20]. Numerous studies have confirmed that the SSCC-Ag level before treatment is a key prognostic factor [21, 22, 23] and an indicator of possible tumor relapse during the follow-up of CC patients [24, 25]. However, the predictive value of SSCC-Ag levels after RT in the survival of CC patients remains unknown.
In the present retrospective study, we analyzed primary cervical tumor regression and SSCC-Ag levels 4 weeks after RT and evaluated the prognostic value of SSCC-Ag levels in CSCC patients.
Materials and methods
Patients
A total of 218 patients who received new CSCC diagnoses and definitive IMRT at our cancer center from Oct 2013 to February 2017 were analyzed. Patients who had histologically confirmed CSCC, received IMRT and intracavitary brachytherapy, did not show evidence of distant organ metastasis, and had acquired data from a pelvic MRI examination and SSCC-Ag levels both before RT and 4 weeks after RT at our institution were included. Patients without clinical records, with distant metastases other than at the paraaortic lymph nodes at diagnosis, with histology other than squamous cell carcinoma, and those who had received surgery treatment were excluded. We collected the following patient data: age, histopathological findings, stage, tumor size as determined through imaging, SSCC-Ag levels before and after the completion of treatment, and tumor response.
Treatment methods
All the patients were treated with a combination of IMRT and high-dose-rate intracavitary brachytherapy (HDR-ICBT). All the patients received a total dose of 45–50 Gy applied in daily fractions of 1.8–2.0 Gy for IMRT. The doses administered to treat grossly enlarged lymph nodes were boosted up to 60–70 Gy. After IMRT, three-dimensional computed tomography-based ICBT was performed using an
Study procedures
MRI scans and SSCC-Ag levels were obtained at the beginning of RT (pretreatment) and 4 weeks after the completion of RT (post treatment). All the patients underwent routine pelvic MRI, SSCC-Ag level testing, and gynecologic examination after RT. Two radiation oncologists at our center who were blinded to the clinical findings defined the tumor areas in each slice. The tumor size on MRI scans was defined as the maximum width on axial T2-weighted sequences using a measurement tool on the Picture Archiving and Communications System. Positive lymph node involvement was indicated by lymph nodes with a short axial diameter of
Response evaluation
Post treatment pelvic MRI scans were routinely performed 4 weeks after the completion of ICBT, and the maximum width of the tumors on the MRI scans was measured. Tumor response was evaluated according to revised Response Evaluation Criteria in Solid Tumors [26] version 1.1, which are based on the final tumor size on MRI scans after the completion of RT. Tumor responses were classified as complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD). Any lymph node with a short-axis diameter of
Follow-up
Patients were followed up for 4 weeks after treatment, every 3 months during the first 3 years and then every 6 months until death. Locoregional recurrence was defined as identification of recurrence within the pelvis three months after definitive treatment, whereas metastasis to distant sites was defined as recurrence outside the pelvis. All events from the date of the end of definitive RT to the date of first recurrence or death were recorded.
Statistical analysis
The Kaplan-Meier method was used to analyze over-all survival (OS), locoregional control (LRC), disease-free survival (DFS), and distant metastasis-free survival (DMFS). Univariate analysis was performed using the log-rank test. Multivariate analysis was performed using the Cox proportional hazards model to identify independent predictors. Receiver operating characteristic (ROC) curves were used to identify the optimal cutoff points of tumor size and SSCC-Ag levels before and after treatment. The Youden index of each ROC curve and the area under the curve (AUC) were calculated. The SPSS software (version 23, SPSS Inc., Chicago, IL, USA) was used for the statistical analysis and
Characteristics of patients with cervical squamous carcinoma cancer
Characteristics of patients with cervical squamous carcinoma cancer
Abbreviations: FIGO
Prognostic comparison of 218 patients with complete response (CR) and non-complete response (Non-CR) of primary tumor (PT). (A) Overall survival. (B) Disease-free survival. (C) Loco-regional control. (D) Distant-metastasis free survival.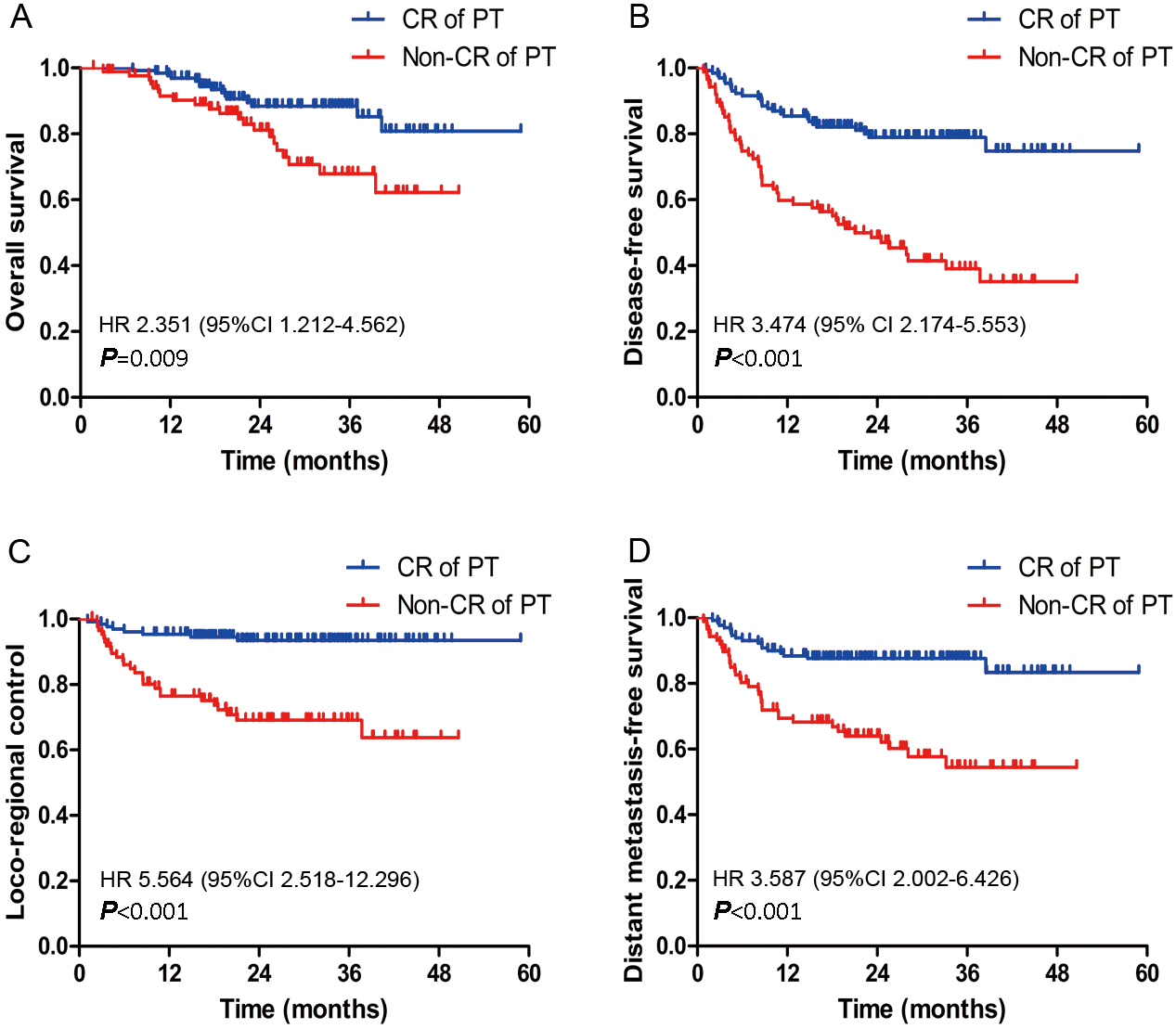
Patient characteristics and treatment outcomes
During the study period, 218 patients with CSCC were included in the study. The clinicopathological characteristics of the patients are presented in Table 1. The median follow-up duration for the entire cohort was 25.57 (range, 1.73–58.93) months. During the follow-up period, 36 patients died and 68 experienced recurrences. Locoregional failure, distant failure, and both pelvic and distant failure was observed in 34, 51, and 17 patients. In 62 patients, recurrence was detected within 2 years of treatment. The 3-year OS, LRC, DMFS, and DFS rates for the entire cohort were 80.5%, 81.4%, 74.4%, and 62.4%, respectively (Supplementary Fig. 1).
As indicated in Supplementary Table S1, in the univariate analysis, the factors of Age (
Post-RT gross tumor regression and SSCC-Ag level
At 4 weeks after completion of treatment, the primary tumors of 130 (59.6%) patients exhibited CR, 62 (28.4%) patients exhibited PR, 25 (11.5%) patients exhibited SD, and one patient exhibited PD. Residual PLN were observed in 19 (8.7%) patients. A significant positive correlation was observed between the pre-RT primary tumor size and pre-RT SSCC-Ag levels (
Among the 218 patients, before treatment, a high SSCC-Ag level was noted in 181 (83.0%) patients and the levels were normal in 37 patients (17.0%). After completion of RT, high SSCC-Ag levels were noted in 20 (9.2%) patients. Of the 181 patients with high pretreatment SSCC-Ag levels, 162 achieved a normal SSCC-Ag status after 4 weeks of treatment. The number of patients with high SSCC-Ag levels and non-CR of primary tumors was significantly higher than that of patients with high SSCC-Ag levels and CR at the end of RT (15.9% vs. 4.6%;
Prognostic comparison of 218 patients with post-radiation (Post-RT) negative SCC and positive SCC. (A) Overall survival. (B) Disease-free survival. (C) Loco-regional control. (D) Distant metastasis-free survival.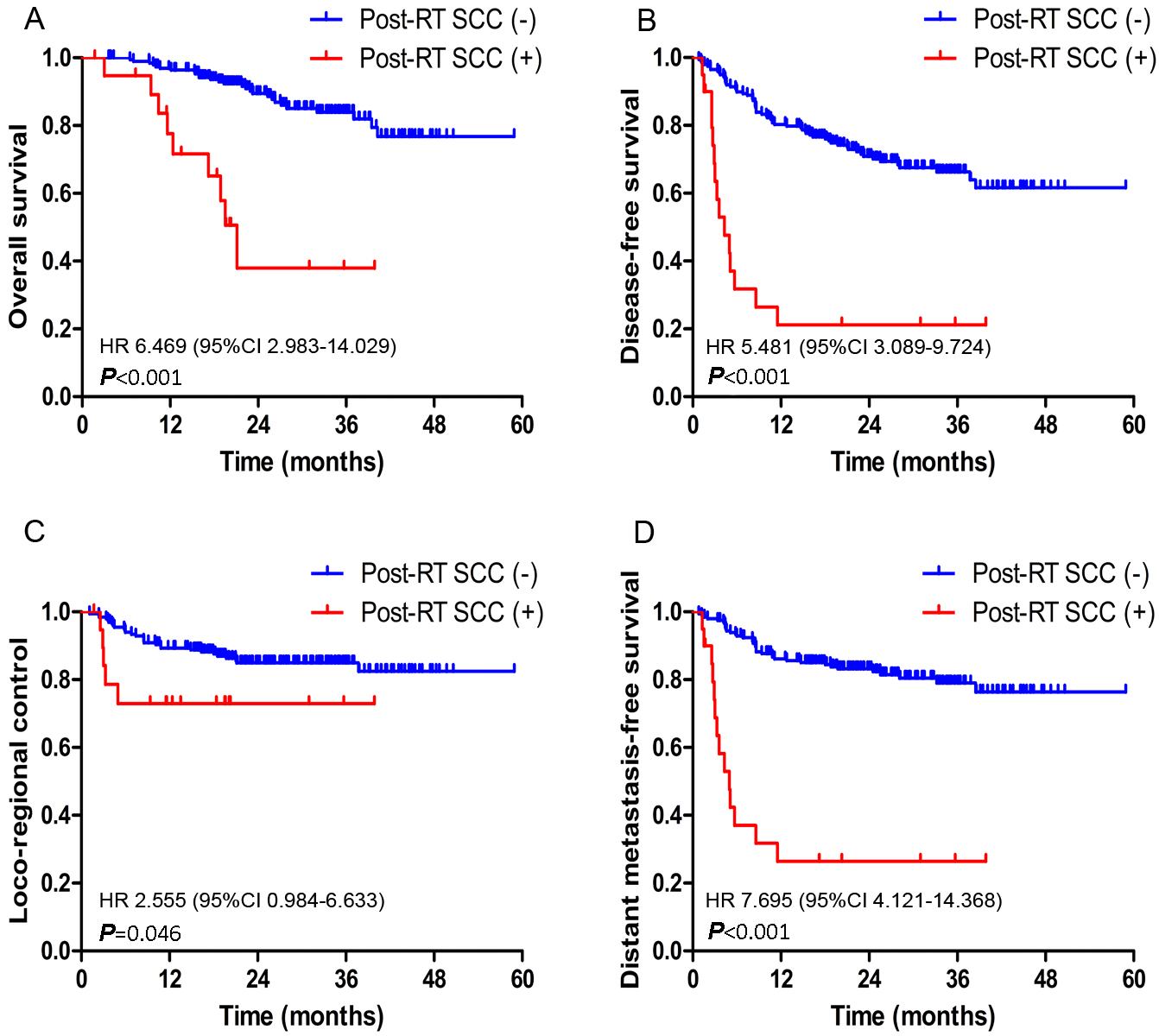
In the assessments of primary tumors 1 month after RT, 130 and 88 patients exhibited CR and non-CR, respectively. The 3-year OS rates differed significantly between the patients with CR and non-CR (85.2% vs. 67.9%; HR, 2.351; 95% CI, 1.212–4.562;
Prognostic value of post-RT SSCC-Ag level
After 4 weeks of RT, 218 patients were classified into two categories, namely 198 patients with low SSCC-Ag levels (
Receiver operating characteristics (ROC) curves of (A) post-RT SSCC levels and (B) post-RT MRI based tumor diameter and (C) combination of both factors.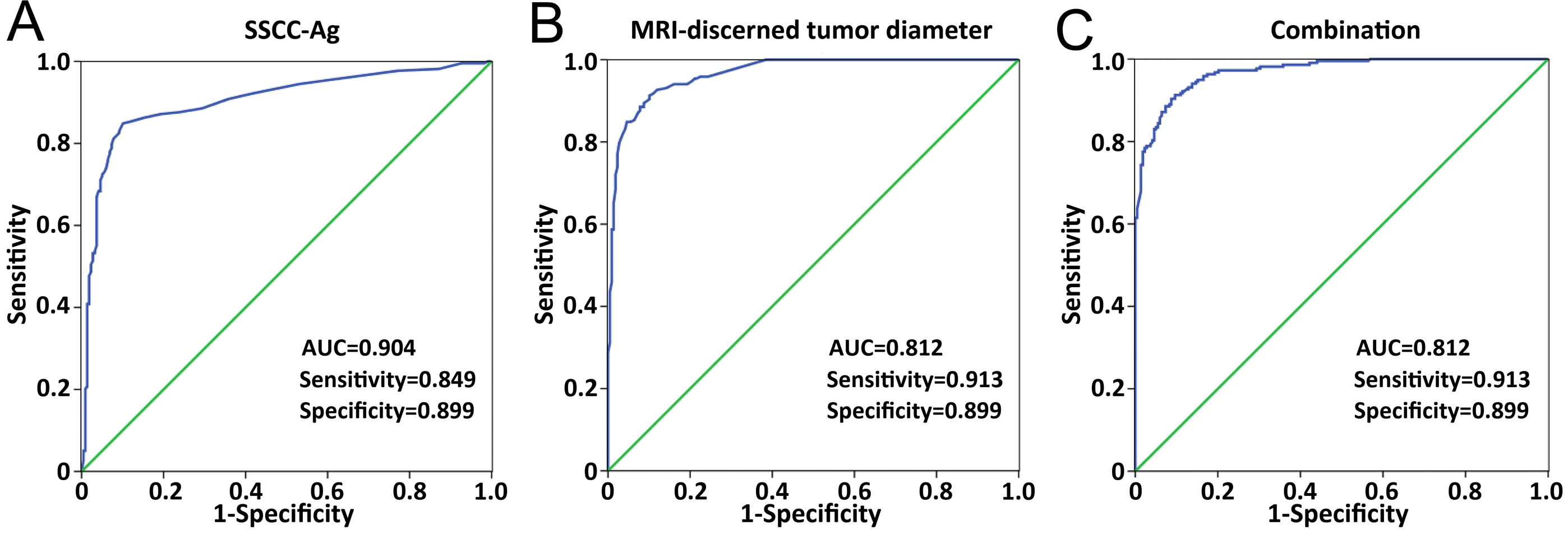
Among the 218 patients, 199 (91.3%) and 19 (8.7%) patients exhibited no pelvic lymph node (PLN) involvement (PLN
Evaluation of diagnostic potential of post-RT SSCC-Ag levels and tumor size
The ROCs were used to evaluate the performance of MRI-discerned tumor diameter and SSCC-Ag levels in distinguishing between the pretreatment CSCC patients and post-RT patients. As depicted in Fig. 3 and Supplementary Table S2, the SSCC-Ag levels achieved the highest accuracy in discriminating pretreatment CSCC patients from post-RT patients. The observed AUC of the SSCC-Ag level panel was 0.904 with a sensitivity and specificity of 84.9% and 89.9%, respectively.
The sensitivity and specificity of MRI-discerned tumor diameter at 4 weeks after RT in discriminating between pre- and post-RT patients were 91.35% and 89.9%, respectively. The ROC analysis of MRI-discerned tumor diameter (AUC
Upon combining these two factors in the ROC analysis, the AUC increased to 0.97 (95% CI, 0.957–0.983;
Discussion
In this study, we measured both the size of the primary tumor and the tumor marker SSCC-Ag at the pre-RT and post-RT time points and evaluated the prognostic value of these two factors. The results revealed that patients with non-CR and high SSCC-Ag levels after RT were significantly associated with relatively low OS, DFS, LRC, and DMFS rates. Furthermore, post-RT residual PLN was related to poor OS and DFS rates in CSCC patients.
Although CC is radiosensitive, residual disease is detected after RT in approximately 35–60% of patients [27, 28], which leads to a poor prognosis. Accurate prediction of early recurrence is imperative to be able to determine whether additional treatment strategies are required and to improve the survival rate among these patients. Some studies have reported that tumor volume [29], nodal volume [30] and FIGO stage [31] are independent prognostic factors in the survival outcome of CC patients treated with RT. High SSCC-Ag levels also predict early tumor recurrence in CC patients [32]. However, few studies have focused on the potential for using tumor size and SSCC-Ag levels after RT to predict the survival rate in CC patients.
The SSCC-Ag is currently recognized as a useful tumor marker for CSCC detection, although high levels of SSCC-Ag have also been observed in other SCCs, such as lung, head and neck, esophagus, and anal canal [33]. In addition, SSCC-Ag levels have been used to monitor the recurrence in patients with CC. For instance, Salvatici et al. [25] reported that elevated SSCC-Ag levels reflected early disease recurrence of early-stage CC. In another study, Ohara et al. [17] found that nonnormalization of SSCC-Ag 3 months after RT was associated with a high recurrence rate of CC. However, limited data are available on the negative and positive predictive value of SSCC-Ag analysis for the OS of CC patients. Ohno et al. also determined that the SSCC-Ag level can predict the response at the pre-RT, mid-RT, and end of RT in CC patients and that it was a risk factor for local recurrence and distant metastasis [34]. They also reported that normalization of the SSCC-Ag level after treatment was significantly correlated with CR. In this study, high pre-RT SSCC-Ag level was observed in 181 patients and was restored to a normal SSCC-Ag level in 162 (89%) patients after treatment, which was a higher rate than that (60.6%) observed in a previous study [35]. High SSCC-Ag levels after RT were observed in 9.2% patients 1 month after treatment and high levels of SSCC-Ag after RT were significantly associated with poor OS, DFS, LRC, and DMFS. In addition, the SSCC-Ag levels in the patients with non-CR of PT were significantly higher than those in the other patients (
The clinical pelvic examinations for clinical staging of CC often produce inaccurate results due to the performance of a limited radiographic evaluation. MRI is an essential tool for diagnosis, allowing a clinician to evaluate local tumor extension and determine lymph node involvement [36]. According to previous studies, post-treatment MRI was performed around 2–3 months after RT. In the current study, we used MRI 4 weeks after treatment to evaluate treatment response, in order to diagnose residual diseases as soon as possible and allow the sooner consideration of alternative treatment options. Although edema and acute inflammation may sometimes occur after RT, diffusion-weighted imaging may contribute to identify the local residual tumor [15]. Besides, delaying salvage treatment may have potential risks, as radiation-resistant cancer stem cells may proliferate if there is actual residual disease. At 4 weeks after RT, CR of primary tumors was noted in 130/218 (59.6%) patients, which was significantly higher than the 40/231 (17.3%) rate reported in a previous study [35]. This was probably the result of treating all the patients in our study with IMRT. A retrospective analysis of 81 patients with CC by Nam et al. [13], using pelvic MRI to evaluate the tumor volume regression rate, revealed that the CR rates in the RT- and CCRT-treated patients were 11.6% and 7.9%, respectively. They also found that 25.6% patients had a residual primary tumor after 3 months and the mid-RT tumor volume regression rate was significantly associated with the LRC rate. MRI was also reported to be useful for evaluating the response to RT, and the study suggested that MRI might be a crucial predictor of LRC and long-term survival. A study by Wang et al. [37] determined that combined pretherapy volumes and mid-RT volume regression ratios were highly accurate in predicting LRC failure and OS. However, the sample size in most of the aforementioned studies was relatively small and in most studies the volume regression rate was calculated to predict the prognostic value of CC [38]. In our study, the CSCC patients with CR after RT exhibited significantly higher OS, DFS, LRC, and DMFS rates than did the patients with non-CR. Additionally, the MRI-discerned tumor diameter was found to have highly significant predictive value for identifying treatment response after RT with high sensitivity (91.3%), specificity (89.9%), and an area under the curve (AUC) of 0.967 with a cutoff value of 23.5 mm. For the SSCC-Ag level, the predictive ability for the treatment response remained high, with 84.9% sensitivity and 89.9% specificity, and a cut-off value of 1.45 ng/mL. When the MRI data were combined with the SSCC-Ag level, the predictive ability increased slightly (AUC
Regarding the effects of lymph nodes, a study by Park et al. [39] reported that the presence of residual lymph node 3 months after CRT was associated with poor survival in patients with node-positive CC. Our results also indicated that the patients with residual lymph node after RT had worse survival outcome than did those without residual lymph node, suggesting that the measurement of the lymph node response has a high prognostic value for CC.
Patients with CC may develop tumor recurrence and/or distant metastasis after RT, and their prognosis is poor, with a median OS of less than 1 year [40]. Accordingly, long-term follow-up is still required after treatment. Early detection of recurrent CC can enable patients get appropriate treatment on time, and prolong the survival rate. The management for recurrent CC after RT represents a challenge for clinicians. RT is an effective salvage modality for CC patients with in-field recurrent lesions, but the previously administered RT dose, duration and recurrent site should be carefully considered to avoid severe toxicity. Salvage surgery is another curative treatment for patients with pelvic recurrence after RT. Palliative platinum-based chemotherapy combined with molecular targeting treatment also plays an important role for most recurrent/distant metastasis CC patients. There is currently no standard second-line treatment for recurrent CC patients, although a combination of multiple treatment modes to prolong the survival time and improve the life quality is also the direction of future efforts by researchers.
Conclusion
In summary, in definitive RT for patients with CSCC, determination of post-RT SSCC-Ag levels and MRI-discerned tumor regression was useful for predicting survival outcome. The SSCC-Ag level was an independent prognostic factor. Some effective measures should be taken, and appropriate treatments administered if patients have persistently high SSCC-Ag levels and non-CR of tumors.
Footnotes
Conflict of interest
The authors declare no potential conflicts of interest concerning this manuscript.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-190934.
