Abstract
BACKGROUND:
Histone deacetylases (HDACs) are considered as an essential regulator of cellular proliferation, differentiation, and apoptosis. The HDAC2 enzyme of Class I HDACs plays an important role in tumor progression of human malignancies.
OBJECTIVE:
The aim of the present study was to analyze the HDAC2 gene expression in pre-oral cancer and oral squamous cell carcinoma (OSCC), and its association with clinico-pathological features.
METHODS:
The HDAC2 protein expression was analyzed through the immunohistochemistry and western blot techniques in 82 oral pre-malignant, 90 OSCC, and 16 normal control tissues. qRT-PCR was used to quantify the mRNA fold change in all groups.
RESULTS:
The HDAC2 protein and mRNA levels were significantly higher in OSCC and pre-oral cancer groups compared to the controls. Immunostaining of HDAC2 protein was enhanced in 84.4% of OSCC and 67.1% of pre-cancerous tissue sections (
CONCLUSIONS:
The upregulated HDAC2 gene in pre-cancer and OSCC tissues indicates its crucial role in the transformation of pre-malignant to malignant carcinoma. It could be a potential cancer biomarker of prognosis and targeted therapy in OSCC.
Introduction
OSCC is the most prevalent type of cancer worldwide with an estimated (including lip cancer) 354,864 new cases and 177,384 deaths in 2018 [1]. The GLOBOCAN 2018 data presented oral cancer as the second most common cancer in India with incidence 119,000 and 72,500 deaths [2]. Mostly, oral carcinoma develops from pre-malignant lesions such as leukoplakia, erythroplakia, and lichen planus. Mixed lesion or erythroleukoplakia is also diagnosed in most of the pre-oral cancer patients [3]. Alcohol and tobacco use are often predisposing risk factors of oral cancer in which tobacco and betel quid chewing habits are more significant in Asian subcontinents. Recently, human papillomavirus was also identified as a causative agent of oropharyngeal and oral cancers [4, 5, 6].
There are multiple genetic aberrations that must occur in normal tissues to proliferate to dysplasia and carcinoma. Indeed, overexpression of Aldehyde dehydrogenase 1 and CD133 proteins showed a significant relationship with the progression of oral leukoplakia to squamous cell carcinoma [7]. Some other predictive markers such as mutated EGFR, VEGF, cyclin D, p53, and specific DNA methylation participate in oral carcinogenesis [8, 9]. Altered levels of H-Ras GTPase and c-Fos protein contribute to the development of oral carcinoma, reported in our previous findings [10, 11]. Moreover, it has been postulated that the deregulated function of HDACs leads to proliferation and progression in different kinds of malignancies [12].
The acylation of DNA-bound histone protein is typically related to gene transcription, regulated by two antithetical enzyme classes: histone acetyltransferases (HATs) and HDACs [13]. HATs decrease the affinity of histone complexes to DNA by adding acetyl groups to specific lysine residues of protein and help in the DNA accessibility for transcription of genes. However, HDACs tend to catalyze the acetyl group of histone by which chromatin becomes more condensed and generally repress the transcription [14]. Considerably, the earlier reports showed that HDACs may also alter the transcriptional activity by regulating dynamic acetylation of DNA sequence-specific transcriptional factors (p53, STAT3, E2F, pRB, NF-
To date, there are 18 different HDACs enzymes are known in humans which are classified into four classes; class I (HDAC1-3, 8), class II (HDAC4-7, 9, 10), class III (SIRT1-7) and class IV (HDAC 11). HDAC1-10 are primarily located in the nucleus of cells and associated with transcriptional repressors and co-factors. All the HDAC isoforms of class I, II and IV have a common enzymatic mechanism, Zn dependent hydrolysis of acetyl-lysine amide bond [18, 19]. HDAC1 and HDAC2 are the most distinguished HDACs and ubiquitously expressed in various transcriptional repression complexes which to be important in the regulation of cell proliferation [20].
The earlier studies depicted about the association of higher level of HDAC2 protein with gastric, colorectal, prostate, cervical, bladder, ovarian and breast cancers [21, 22, 23, 24, 25, 26, 27]. However, broad data of HDAC2 protein expression in oral carcinoma has not been presented so far. Previously, Chang and colleagues examined the immunohistochemical expression of HDAC2 protein in tissue sections from oral epithelial dysplasia and SCC in a Taiwan cohort [28]. Therefore, in this context, we aimed to provide an overview of HDAC2 protein and mRNA expression patterns in well-characterised tissue of oral pre-malignant and malignant patients using immunohistochemistry (IHC), Western blot (WB), and quantitative Real-Time PCR (qRT-PCR) techniques. Association of protein level was further observed in respect of clinico-pathological characteristics of patients to clarify its future therapeutic values in OSCC.
Materials and methodology
Tissue samples and histopathology
A total of 172 pre-cancerous, cancerous and 16 normal tissue samples were obtained from patients with oral non-malignant, pre-malignant and malignant lesions who underwent biopsy/surgery in Surgical Oncology, Oral and Maxillofacial Surgery departments of the King George’s Medical University (K.G.M.U), Lucknow, U.P, India. All patients or their kin signed the informed consent prior to the collection of samples. This study has been approved by the Institutional Ethics Committee, K.G.M.U, Lucknow, U.P, India (no. 9382/Ethics/R.Cell-16; Feb 04. 2016). Tissue samples were aliquoted in three parts; first part was fixed in 4% buffered formaldehyde solution for histopathological examination and IHC, second in RIPA buffer (G-Biosciences, USA) for western blotting and third in RNA later solution (Invitrogen, USA) for qRT-PCR. Typing, grading, and staging of formaldehyde-fixed tissue sections were done using Hematoxylin and Eosin staining in accordance with WHO guidelines and AJCC manual for the histological classification of oral lesions [29, 30]. Demographic and risk habit details were recorded on a pre-designed standard questionnaire. Moreover, clinical and pathological profiles were obtained from the case history-sheet. Patients with secondary malignancy or completed/undergoing therapy were excluded from the study.
Immunohistochemistry
Paraffin-embedded tissue sections (4
Western blot assay
Proteins were extracted from tissue samples by homogenization in cell lysis buffer. The homogenate was centrifuged at 12,000 rpm for 15 minutes. Protein concentration was determined in the supernatant by BCA protein assay kit (Merck, USA). Thirty micro gram of total protein was separated on 12% SDS-polyacrylamide gel and transferred on PVDF membrane by following standard protocol. Further, PVDF membrane was blocked in TBS Tween-20 (TBST) including 5% skim milk for 1.5 hrs following mix the primary antibodies (HDAC2; dilution 1:1000,
RNA isolation and qRT-PCR
RNA isolation from all tissue samples was carried out by total RNA isolation kit (#74104, Qiagen, Germany) and cDNA was synthesized by reverse transcriptase PCR using cDNA synthesis kit (#4368814, Applied Biosystems, USA) through following manufacturer protocol. Relative mRNA expression of HDAC2 genes (NM001527.3) was done by qRT-PCR (Light cycler 96 System, Roche, USA) using specific primer; sense 5’-GGGAATACTTTCCTGGCACA-3’ antisense 5’-ACGGATTGTGTAGCCACCTC-3’ (product size 314 bp) [26] and SYBR Green PCR master mix (#4367659, Applied Biosystem, USA) in triplicates. The thermal profile for HDAC2 gene was 94
Statistical analysis
The categorical data is summarized in number and percentage whereas continuous data in mean
Results
Out of 172 oral tissue samples, 82 samples of pre-cancerous lesions were diagnosed with mild to severe epithelial dysplasia and 90 samples presented squamous cell carcinoma during histopathological diagnosis. Moreover, 16 tissue samples of non-malignant oral lesions with normal epidermal cell morphology in the histological investigation were considered as controls.
Demographic characteristics and risk habits of patients
Most of the patients with oral pre-malignant and OSCC groups were observed in the age group of 41–60 years. The frequency of male patients was higher both pre-oral cancer and OSCC groups. Tobacco chewing habit was observed in 81.1% of OSCC and 67.1% of pre-malignant patients (
Association of demographic characteristics and risk habits with pre-cancer, OSCC and control groups
Association of demographic characteristics and risk habits with pre-cancer, OSCC and control groups
HDAC2 protein expression in pre-cancer, OSCC and control groups
Pre-malignant and malignant lesions were mostly identified in the buccal mucosa, lower and upper alveolus, tongue, lip, and palate regions of the oral cavity. The clinical evaluations of pre-cancer patients presented that out of 82 cases, 43 cases (52.4%) were identified with leukoplakia, 27 cases (32.9%) with erythroplakia whereas 12 cases (14.7%) with mixed type lesions. Of total pre-cancerous biopsies, 52 biopsies (63.5%) showed mild dysplasia (Mild Dys.), 18 biopsies (21.9%) moderate dysplasia (Mod.Dys.) while 12 biopsies (14.6%) confirmed for severe dysplasia (Sev.Dys.) in the histopathological diagnosis. However, out of the total number of OSCC cases, 47 patients were reported in stage IV, 30 cases in stage III, and 13 cases in early stages (Stage I
Microphotograph showing immunohistochemistry image of HDAC2 protein: A. Negative immunoexpression in normal tissue of oral cavity, B. Brown color stained cells showed weak expression in dysplastic epithelia of oral pre-malignant tissue, C. Strong immunoexpression of several nuclei in carcinoma tissue. (DAB, digital magnification, X100).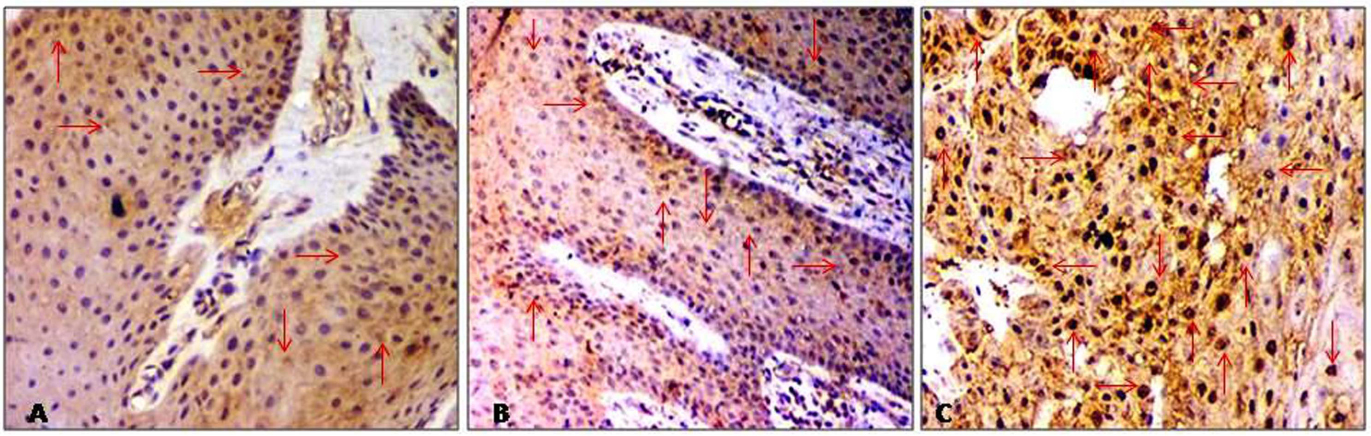
Immunohistochemical expression pattern of HDAC2 protein in all tissue sections of pre-cancer, OSCC and control groups are presented in Table 2 and Fig. 1. Of total 47.6% pre-cancerous tissues were found with moderate expression (score
HDAC2 gene was frequently upregulated in OSCC and pre-malignant cancer
Frequency of HDAC2 protein expression in different tissue categories of the oral cavity (Neg.Exp; Negative expression, Weak Exp; Weak expression; Mod.Exp; Moderate expression; Str.Exp; Strong expression).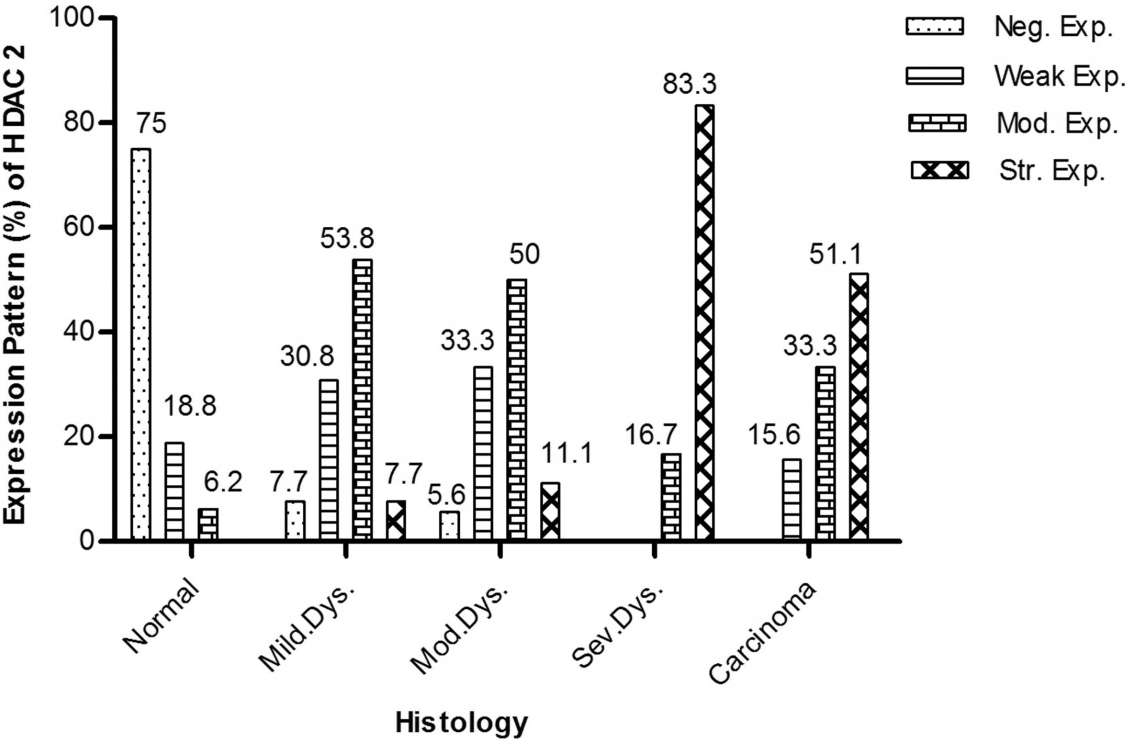
Significant higher expression of HDAC2 protein and mRNA in oral pre-cancer and OSCC groups compared to the normal control group: A. Western blot of HDAC2 protein in control, dysplastic epithelium, and differentiated carcinoma tissues. 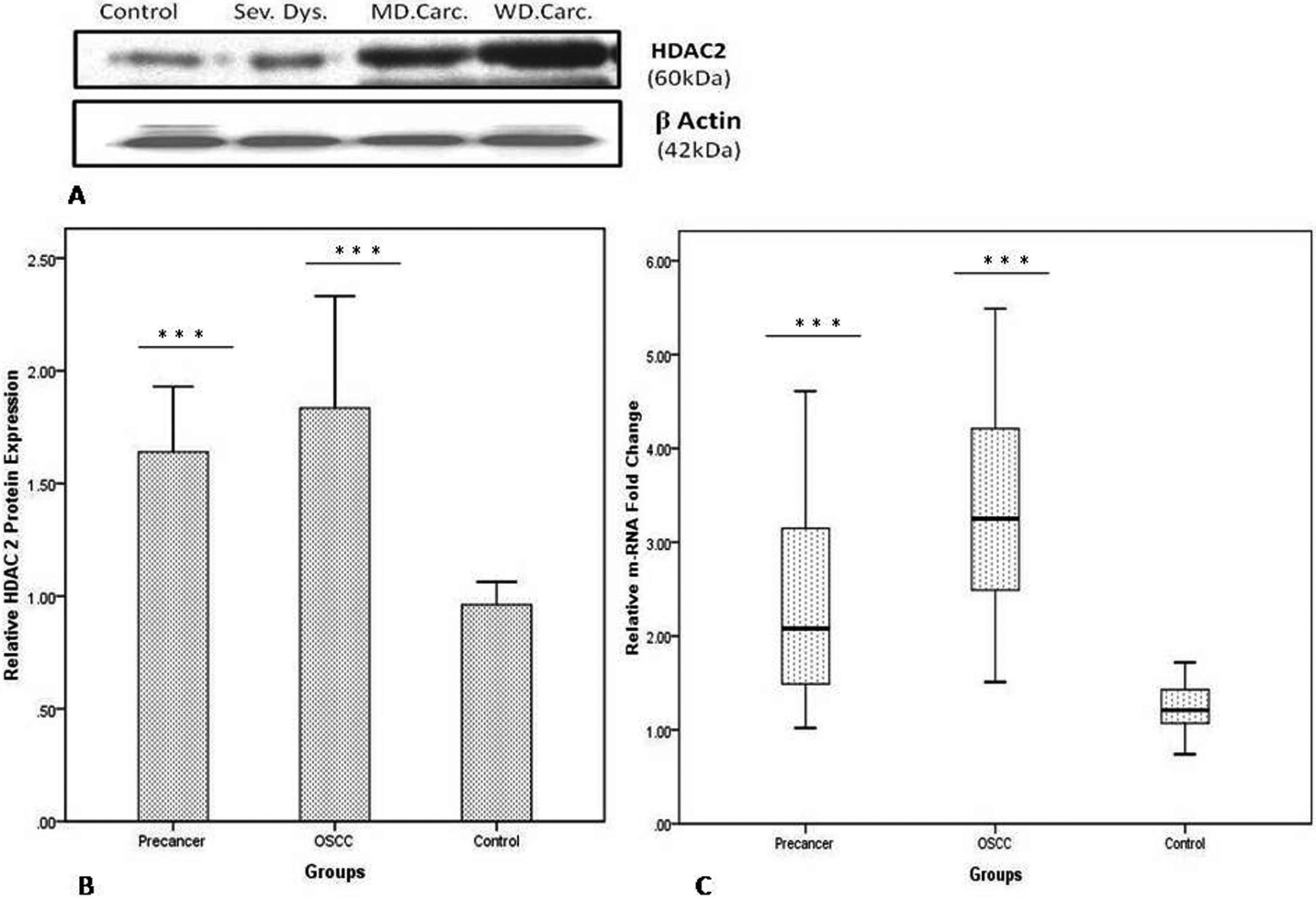
Further, western blot analysis was also performed to validate the expression levels of HDAC2 protein in all tissue samples. Most of pre-cancerous (64 of 82, 78%) and cancerous (84 of 90, 93.3%) tissues showed significant overexpression of HDAC2 in comparison to normal mucosal specimens (
Correlation between protein and mRNA expression levels of HDAC2
Association of HDAC2 protein expression with clinico-pathological features of patients with oral pre-cancer and SCC
The association of HDAC2 protein expression with clinicopathological parameters is summarized in Table 4 In the control group, mean RD value of HDCA2 was 0.96 (
Expression of HDAC2 was significantly higher in OSCC than pre-cancerous tissues of the oral cavity in estimation by both IHC (OR 2.6,
Discussion
The present study illustrates the expression of HDAC2 gene using WB, IHC, and qRT-PCR in different histological grades of the oral cavity tissues. With our best knowledge, this is the first report on the Indian population which analyzed the expression status of HDAC2 and its role in the progression of oral pre-cancerous and cancerous lesions. The study groups were neither significantly associated with the demographical characteristics nor pattern of risk habits except tobacco chewing. In agreement with the previous findings, our study also confirmed that chewing tobacco either alone or in combination with other risk habits often develop pre-malignant and malignant lesions of the oral cavity [31, 32]. In the clinical investigations, buccal mucosa was the most frequently affected site for leukoplakia and erythroplakia lesions and leads to the well-developed tumor as the previous observations by Singhania et al. [33].
This study shows that HDAC2 protein expression was significantly higher in both OSCC and pre-cancer groups compared to the normal group, however, the expression pattern differed in histological grades of epithelial dysplasia and carcinoma tissues of the oral cavity. We observed the increased expression of HDAC2 protein in 51 of 58 of carcinoma and 12/12 of severe dysplasia tissues of the oral mucosa. The positive immunostaining for HDAC2 protein increased significantly from normal oral epithelium to dysplastic and squamous cell carcinoma as well. Additionally, these outcomes were further validated and finding reveals the high expression of HDAC2 in pre-cancer and cancerous tissues at both transcriptional and translational levels. Further, overexpression of HDAC2 in moderate to severe dysplasia and higher grade carcinoma signify that changes in this protein level might be involved in oral carcinogenesis and abnormal cellular proliferation. These findings are in concordance with Chang et al. who found HDAC2 protein increased significantly from normal oral mucosa through dysplasia to squamous cell carcinoma. They also analyzed that patients with high HDAC2 expression tumors had significantly shorter overall survival than patients with low expression of protein [28]. Similarly, our results also support some earlier studies in other tumors. A study on expression arrays of HDAC2 in urothelial carcinoma showed the upregulation of HDAC2 mRNA and protein in tumors compared to normal urothelium [34]. Jin et al. detected increased relative mRNA fold change and protein in immunoblot and IHC staining of HDAC2 in ovarian carcinoma than normal tissues [26]. These investigations are in line with our study which presents 1.5 higher mRNA fold change difference in the pre-cancer group (
The immunoblotting estimation in our study showed a significant association between higher HDAC2 protein level and histological grades (
Interestingly, we observed the moderate level of HDAC2 nuclear staining in the majority of pre-malignant cancer tissue samples (47.6%) vs. OSCC tissue samples (33.3%) whereas strong immunoreactivity analyzed mostly in carcinoma group (51.1%). Despite this, a significant positive correlation between HDAC2 mRNA and protein level in the pre-cancer group indicates the transformation of epithelial dysplasia to carcinoma may be promoted by upregulated HDAC2 gene and participates in cellular proliferation.
Various studies have presented a basic understanding of the mechanisms by which HDACs modulate the expression of important genes of transcriptional regulation in endometrial, esophageal and colorectal carcinomas [22, 38, 39, 40]. The p21, an inhibitor cyclin-dependent kinase, is one of the most studied targets of HDAC inhibitor-mediated de-repression. Overexpression of HDACs is examined in many cancer types, with corresponding decreases in p21 expression. For instance, Huang et al. showed that increased level of HDAC2 correlates with reduced p21 expression and correspondingly, HDAC2 knockdown increases p21 expression in colorectal cancer at the polyp stage. It is well established that p53 is a regulator of p21 protein expression. Therefore they also determined the p53 gene expression after HDAC2 knockdown and found the changes in p21 mRNA and protein levels without a change in p53 protein levels. This analysis suggesting that HDAC2 knockdown increases p21 gene expression and apoptosis, independent of p53. Eventually, these outcomes signifying that HDAC2 is an important enzyme involved in the early events of carcinogenesis by alteration of p21 expression, as a decreased level of p21 may cause cell proliferation and its higher expression may direct the cell to an apoptotic pathway [22]. Similarly, Zhu et al. also ascertained the elevated expression of HDAC2 participates in the HDAC-dependent transcriptional repression machinery in colonic cancerous cells. In cases with human colon cancer, intestinal mucosa and polyps of adenomatosis polyposis coli (APC) deficient mice, it was found that the increased HDAC2 expression is frequently associated with aberrant signaling through the Wnt pathway, due to the loss of both functional copies of the tumor suppressor APC and/or mutations in the
Additionally, the results of the present study (Table 4) suggest that immunobloting is requisite for relevant HDACs protein estimation which can also reduce the chances of the inherent limitation of false positivity interpretation in IHC staining. Ultimately, findings of the present report confirmed that HDAC2 protein level could be a potential biomarker of solid tumors in human.
Recently, HDACs have been demonstrated as therapeutic targets of HDAC inhibitors (HDACi) for the treatment of cancer. The HDACi have pleiotropic effects and induce cellular differentiation, apoptosis and/or cell cycle arrest [42]. The novel HDACi; panobinostat and combination of vorinostat and carboplatin are being applied in phase I and II clinical studies against advanced stages of solid and hematological malignancies [43]. Eventually, the present study might open a new vista for targeted HDACi therapy in the response of HDAC2 for oral carcinoma.
The present study has some limitations, including less number of normal oral mucosa tissues in comparison with pre-malignant and OSCC groups. We did not analyze the survival of the patients. So, the prognostic value of HDAC2 protein in oral carcinoma remains to evaluate in future study.
Conclusions
Overexpression of HDAC2 protein and mRNA showed a significant association in pre-malignant and malignant lesions of the oral mucosa. The protein quantifications indicate that HDAC2 aggressively expressed in tumor transformation and in advance clinical stages. In the present study, the higher level of this protein in pre-malignant oral lesions compared with normal controls could be a sign of a suitable biomarker for the prognosis and targeted therapy of solid tumors. So, further studies of HDAC2 inhibitors on a large cohort are warranted for systemic treatment of OSCC.
Footnotes
Acknowledgments
We are grateful to Dr. Pratima Verma, Dr. Pravin Kumar Gangwar and Dr. Sujit Mohanty for their support and evaluation of the article. We would like to thank all the patients and volunteers who participated in this study. We apologize to those investigators whose findings are omitted in this article due to the knowledge or space limitation. Our research has been supported by the grant of the Indian Council of Medical Research (ICMR), New Delhi, India (Letter no. 3/2/2/312/2015-NCD-III Dated 28/03/2016).
Conflict of interest
There is no conflict of interest.
