Abstract
BACKGROUND:
Female carriers of BRCA1 or BRCA2 germline mutations are at a substantially increased risk for developing breast and ovarian cancer. The lack of effective early detection schemes for ovarian cancer, mandate surgical removal of adnexa at age 35–40 years in these high-risk women. The role of circulating cell-free DNA (cfDNA) levels as a marker for early detection in high-risk women has rarely been reported.
OBJECTIVE:
To quantify cfDNA levels in BRCA1 BRCA2 carriers.
METHODS:
Serum cfDNA levels, measured by direct fluorometric assay in cancer-free female BRCA1 BRCA2 mutation carriers were compared with cancer-free controls recruited from among women undergoing breast biopsy or routine colonoscopy.
RESULTS:
Overall, 10 BRCA1 (185delAG) and 10 BRCA2 (6174delT) mutation carriers, 20 breast biopsy controls, and 20 colonoscopy controls participated. cfDNA levels [Median (95% CI)], were 472 (317–589) ng/ml and 525 (339–621) ng/ml in breast biopsy and colonoscopy controls, respectively. Median levels of cfDNA in BRCA1 and BRCA2 mutation carriers combined were 921 (835–1087) ng/ml, significantly higher than in both controls (
CONCLUSIONS:
cfDNA levels are significantly higher in BRCA1 and BRCA2 mutation carriers compared with non-carriers. This finding, if validated, may facilitate development of early detection breast/ovarian cancer biomarker in high-risk women.
Introduction
Circulating cell-free DNA (cfDNA) levels originating from healthy or tumorous tissues [1, 2] are reportedly increased in several clinical scenarios: cancer, inflammation, autoimmune disease smoking, pregnancy and even intense exercise [3, 4]. Increased amounts of cfDNA collectively referred to as “liquid biopsy” are appealing as a possible additional diagnostic tool in cancer [5, 6, 7]. Indeed, several studies have examined the potential of liquid biopsy for early detection of breast cancer [8, 9], and in assessing recurrence risk [10]. Madhavan et al., reported the ability to apply a specific assay of cfDNA level determination for early detection of breast cancer in a cohort of 383 clinically distinct breast cancer cases [11]. Several other studies [12, 13, 14] and a meta-analysis [15] focused on the potential use of cfDNA as a biomarker in ovarian cancer diagnosis. Kamat et al. [13, 14] reported that cfDNA levels in advanced ovarian cancer samples were elevated compared with controls. Yet, No and coworkers [12] detected no significant difference between cfDNA levels of ovarian cancer patients and patients with benign disease. While most studies focused on the utility of liquid biopsy and cfDNA quantification in cancer affected individuals (reviewed by Bronkhorst et al. [16]), there is a paucity of data on cfDNA analysis in high risk BRCA germline mutation carriers. Dey and colleagues [17] reported that using a specifically designed assay to quantify telomere derived cfDNA, levels in unaffected BRCA1
BRCA2 mutation carriers (
Materials and methods
Participant identification and recruitment
Healthy BRCA1 BRCA2 mutation carriers
Healthy BRCA1 BRCA2 mutation carriers were identified and recruited from amongst women attending the Meirav high-risk clinic, the Sheba Medical Center. All women carry a BRCA1 or a BRCA2 germline mutation that was clinically tested in the context of a formal oncogenetic counseling. All were offered participation in this study in the course of a routine visit to a high-risk clinic that serves as a one-stop shop surveillance clinic that includes semiannual clinical breast exam by a surgeon, breast imaging (MRI alternating with mammograms) and a gynecological screen. The Sheba Ethics committee approved the study for analysis of serum markers in these high-risk women, and all participants gave a written informed consent.
Cancer-free controls
Breast biopsy group
This part of the study (
Colonoscopy controls
Subjects who underwent routine population-based screening colonoscopy were offered participation and volunteers signed a written informed consent. Of this group of participants, 20 women with no pathological findings on colonoscopy were randomly selected to serve as controls.
Serum extraction
Serum was obtained from a whole blood sample (3 ml) that was withdrawn using venipuncture and was centrifuged at 4000 Xg for 30 minutes. Serum was immediately removed from the tube and 200 microliters were aliquoted into freeze resistant tubes and stored at
cfDNA assay
cfDNA levels were measured on coded samples, according to the previously described method [18]. Briefly, 20
Statistical analyses
Statistical analysis was performed using GraphPad Prism (Version 6.01, La Jolla, CA, USA). ANOVA, Kruskal-Wallis test followed by Dunn’s multiple comparisons test was used to compare between BRCA carriers and controls. Results are presented as median and 95% confidence interval (CI).
Results
Cases and controls
Overall, 10 BRCA1 (185delAG) and 10 BRCA2 (6174delT) cancer-free mutation carriers (mean age 46.8
cfDNA levels analysis
No differences were noted between cfDNA levels by specific BRCA gene mutation: cfDNA levels in BRCA1 carriers were 923 (681–1223) ng/ml compared with BRCA2 with 921 (787–1162) ng/ml (
Circulating cell free DNA (cfDNA) concentration in BRCA mutation carriers. Levels of cfDNA were measured in healthy BRCA carriers (10 BRCA1 and 10 BRCA2) and compared to the levels of age-matched Negative Breast Biopsy group (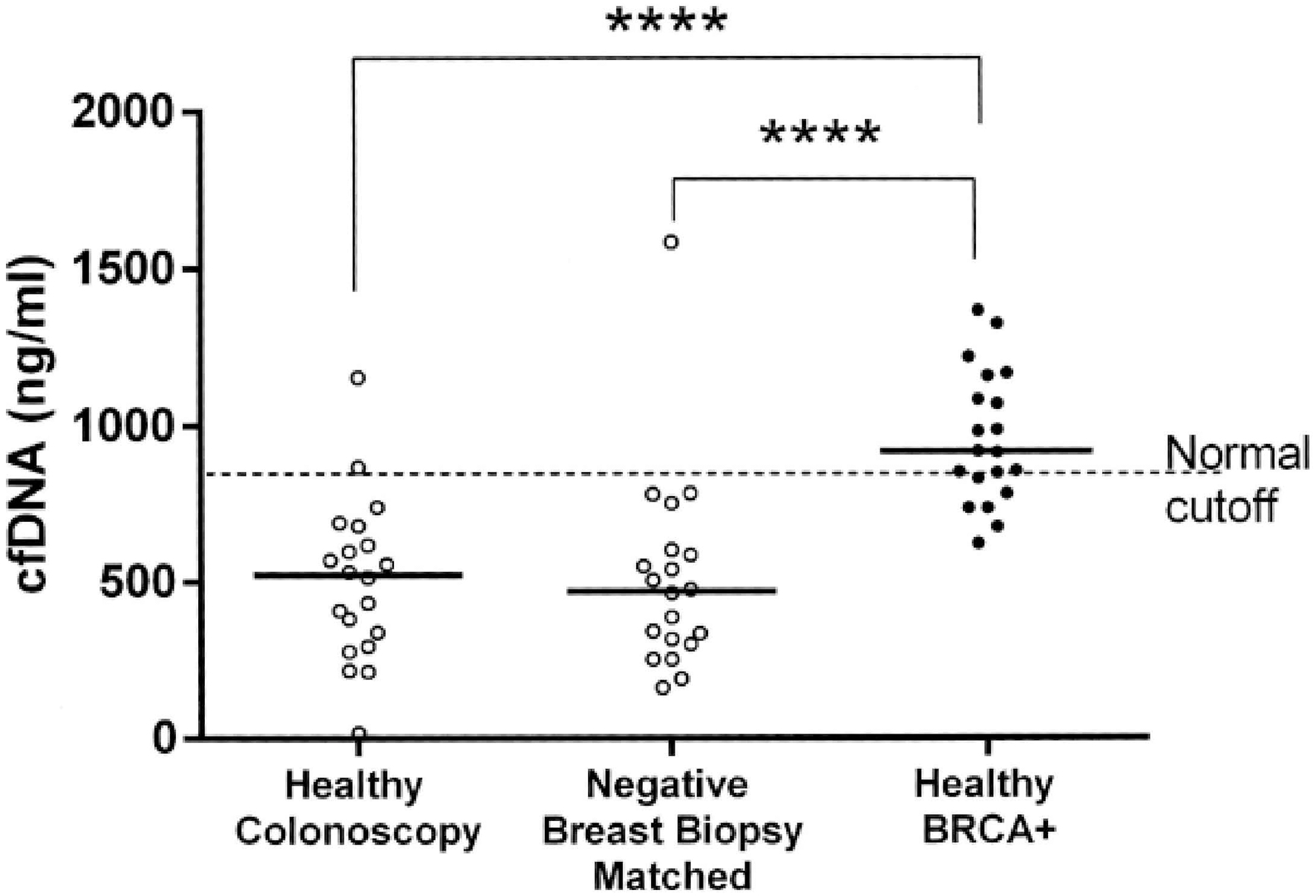
Receiver operator curve (ROC) analysis of Healthy BRCA vs age-matched Negative Breast Biopsy controls.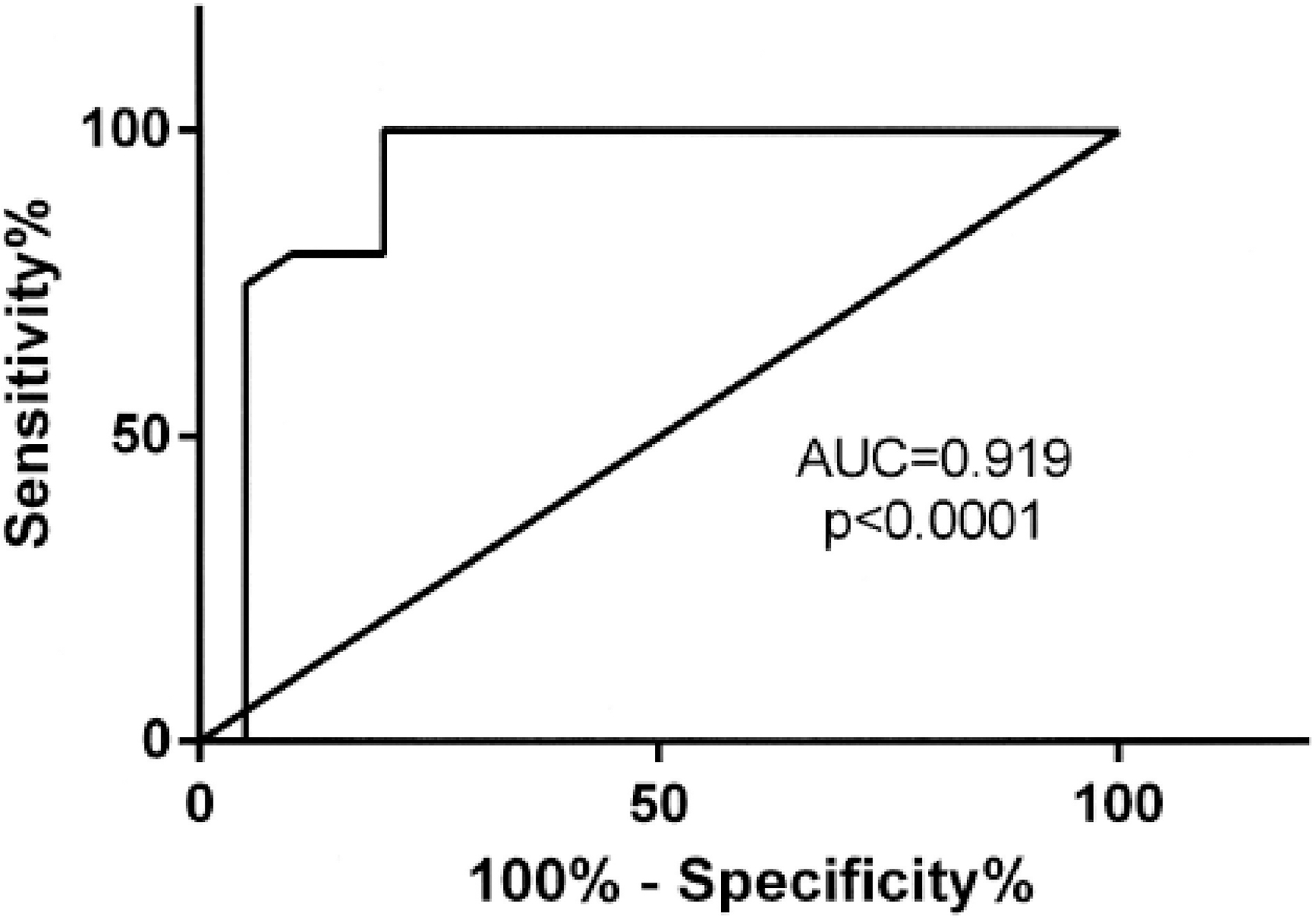
In this preliminary study, cfDNA levels measured using a robust assay, were significantly higher in cancer-free BRCA1/BRCA2 mutation carriers compared with cancer-free controls. The biological basis for this phenomenon may be associated with defective DNA repair mechanisms. Both BRCA genes encode large proteins that play an essential role in DNA damage response (DDR) and homologous repair (HR) of double stranded DNA damage [19]. In BRCA-associated cancers there are no active BRCA proteins somatically, a fact that may lead to accelerated apoptosis that could result in elevated levels of cfDNA. Indeed, Park et al. [20] reported that baseline cfDNA, measured by the same assay applied in the current study, is an effective predictor of relapse in triple negative breast cancer (TNBC) patients, a subtype of breast cancer much more prevalent amongst BRCA1 mutation carriers [21]. Dey and coworkers reported the presence of shorter telomere in 28 BRCA mutation carriers compared with controls [17]. In agreement with these studies, in non-BRCA carrier women with infertility problem we previously found an inverse relationship between cfDNA levels and telomere lengths [22]. Elevated cfDNA and high rate telomeres shortening are probably both caused by accelerated cell turnover.
Thus, it is plausible to assume that BRCA haploinsufficiency, a feature shared in all cells of a BRCA mutation carrier, may result in an increased apoptotic rate of non-cancer cells and cause cfDNA elevation. Noteworthy, this is presently a speculative explanation to account for the results of this study that should be viewed cautiously.
The assay employed herein is simple, robust, easy, and cheap. It has also been shown to be applicable in several clinical scenarios: cfDNA level had better discriminatory ability than the gold standard APACHE II score and procalcitonin (PCT) for 28-day mortality of patients with severe sepsis [23]. Post-dialysis cfDNA concentration was an independent predictor of all-cause mortality in patients undergoing hemodialysis [24]. Baseline pre-surgery cfDNA levels measured in patients with colorectal cancer were good predictors of disease-free survival at 5 years: all patients who died within 5 years had elevated cfDNA levels, while normal cfDNA levels (
There are inherent limitations to this study that should be enumerated. The numbers of cases and controls is small, the spectrum of BRCA1 and BRCA2 mutations is limited, there is no long-term follow up of mutation carriers, the mutational status of the controls was not determined. Given the rates of germline BRCA1 BRCA2 mutations in the Jewish Ashkenazi population (2.5%) [26], it seems that at the most we may have missed one possible mutation carrier in the controls. Lastly, we do not have all possible factors that may have affected the final cfDNA levels-smoking habits menstrual cycle, pregnancy status, to mention a few.
In conclusion, cfDNA levels are seemingly elevated in cancer-free BRCA mutation carriers. The results presented in this preliminary and admittedly limited scope study should be validated in a larger set of BRCA mutation carriers’ cases of diverse ethnicities. These studies should evaluate the sensitivity and specificity of sequential cfDNA measurements as a surrogate marker for early detection of breast or ovarian cancer in healthy BRCA1/2 carriers.
Footnotes
Acknowledgments
We thank Valeria Frishman for excellent technical assistance. This work was supported by the Dr. Montague Robin Fleisher Kidney Transplant Unit Fund and by The Goldman Family Foundation Grant, Research Excellence Initiative, the Goldman Medical School at the Faculty of Health Sciences, Ben-Gurion University of the Negev.
