Abstract
BACKGROUND:
Recently, hepatocellular carcinoma (HCC) has been ranked as the second leading cause of cancer-associated death. However, the underlying molecular mechanisms of HCC progression remain unclear. Vesicular overexpressed in cancer pro-survival protein 1 (VOPP1) could be upregulated in a quantity of human cancers, including squamous cell carcinoma (SCC), gastric cancer, and glioblastoma. However, the precise functional mechanism of VOPP1 in HCC remains poorly understood. The present study aimed to investigate the role of VOPP1 in HCC proliferation.
METHODS:
Immunohistochemistry (IHC), Western blot and Reverse-transcription polymerase chain reaction (RT-PCR) were used to analyze the protein and mRNA expressions of VOPP1, mitogen-activated protein kinase (MAPK) 14, ribosomal protein S6 kinase
RESULTS:
The expression of VOPP1 in HCC samples was higher than that in adjacent noncancerous tissues by immunohistochemistry. In addition, the down-regulation of VOPP1 using shRNA inhibited cell proliferation and tumour growth, and induced cell apoptosis in vitro and in vivo. Furthermore, VOPP1 silencing increased the expression of MAPK14 and RPS6KB1, indicating that the MAPK and mTOR signalling pathways might be involved in VOPP1-mediated cancer cell proliferation.
CONCLUSION:
The present data indicate that VOPP1 may play an important role in the progression of HCC by targeting the MAPK and mTOR signalling pathways, and that VOPP1 may potentially be a candidate as a novel molecular target for HCC therapy.
Background
Hepatocellular carcinoma (HCC) is the most commonly diagnosed hepatic cancer worldwide, and has been ranked as the second leading cause of cancer-associated death [1, 2, 3]. Liver cancer is often diagnosed in the late stages, bringing about a poor survival rate in HCC patients [4, 5, 6]. At present, the main treatments for HCC include chemotherapy, hepatectomy and liver transplantation [7]. Hepatectomy and liver transplantation can lead to high recurrence rates, and recurrence is correlated to frequent intrahepatic metastasis. Chemotherapy can induce severe side effects. Thus, the treatment for HCC remains unsatisfactory [8]. Based on research and clinical data, different signalling pathways are involved in the initiation and regulation of HCC, which provide a strong rationale to develop anti-cancer drugs for key components. However, the underlying molecular mechanisms of HCC progression remain unclear. Therefore, investigations involved in the molecular mechanism of HCC are necessary to develop novel avenues for targeted therapies.
In order to explore the mechanism of Rpb3- dependent tumour growth, HCC-specific molecules mediated by Rpb3 were searched in previous trial via screening genes differentially expressed in cells transfected with Rpb3 shRNA. It was found that 571 transcripts were upregulated and 768 transcripts were downregulated in shRNA-mediated Rpb3 silencing cancer cells (fold change
VOPP1, which is also perceived as EGFR- coamplified and overexpressed protein (ECOP) and glioblastoma-amplified secreted protein (GASP) [10, 11, 12], is upregulated in a quantity of human cancers, including squamous cell carcinoma (SCC), gastric cancer, and glioblastoma [10, 13, 14, 15]. Evidence suggests that ECOP is a key regulator of nuclear factor kappa B signalling, and that ECOP expression could be involved in apoptosis resistance [11]. However, the precise functional mechanism of VOPP1 in HCC remains poorly understood.
Recently, the underlying molecular mechanisms of HCC progression remain unclear. Although VOPP1 could be upregulated in a quantity of human cancers, including SCC, gastric cancer, and glioblastoma, the precise functional mechanism of VOPP1 in HCC remains poorly understood. Therefore, we conducted this study to investigate the role of VOPP1 in HCC.
Materials and methods
Subjects
Surgical specimens of HCCs and adjacent hepatic tissues were obtained from 39 patients with HCC who underwent surgical resection from September 20, 2010 to January. 22, 2017. These 39 patients with HCC included 34 men and 5 women with a median age of 57.8 years old (range, 43–79 years old). All 39 tumour specimens were histologically classified as HCC. The PrimeView Human Gene Expression Array was obtained from Affymetrix Thermo Scientific (Shanghai, China). None of these patients had received radiotherapy or chemotherapy. All patients were histopathologically confirmed, and the samples were obtained with an informed consent from the patients. This study was approved by the Medical Ethics Committee at the Taizhou Hospital of Zhejiang Province. The ethical number was GSZE0125682.
Animal experiments
Four-week-old female BALB/c nude mice were purchased from the Shanghai Laboratory Animal Center (Shanghai, China). BEL7404 (5
Materials
The specific information about the main equipments and materials were listed as follow. The PrimeView Human Gene Expression Array was obtained from Affymetrix Thermo Scientific (Shanghai, China). Four-week-old female BALB/c nude mice were purchased from the Shanghai Laboratory Animal Center (Shanghai, China). IVIS Lumina LT: PerkinElmer, Waltham, MA, USA. VOPP1 antibody: Rabbit, Polyclonal, diluted at 1:600; Bioss, Beijing, China. TRIzol reagent: Pu Fei Biotechnology Co., Ltd., Shanghai, China. Light Cycler 480 Real Time System with the SYBR green reagent (TaKaRa, Beijing China), High sensitivity: detect single copy gene. Wide range of kinetics: 1–1010 copies of DNA can be detected simultaneously. High repeatability: CV%
Immunohistochemistry
The living imaging apparatus is equipped with a gas anesthesia system for isoflurane gas anesthesia. Tissue slides (5-
Cell lines, culture, and RT-PCR
The human HCC-derived cell lines (SMMC-7721, BEL-7402 and BEL-7404) and hepatoblastoma cell line (HepG2) were grown in PRMI1640 with 10% fetal bovine serum (Gibco, MA, USA) at 37
Gene silencing transfection
Lentiviral vectors encoding VOPP1 shRNA (shVOPP1) were designed. The synthesized DNA oligonucleotides, which contained these sequences, were annealed and inserted into the GV115-GFP vector (Genechem, Shanghai, China). Then, cells were seeded in 6-well plates at a concentration of 2
Western blot analysis
Cells including the human HCC-derived cell lines (SMMC-7721, BEL-7402 and BEL-7404) and hepatoblastoma cell line (HepG2), were lysed with precooled and sonicated lysis buffer (50 mmol/L_Tris-Cl_PH8.0, 150 mmol/L NaCl, 0.1% sodium dodecyl sulfate [SDS], 1% NP-40, 0.5% DOC, 0.02% NaN3, 100 ug/ml_PMSF, and 1 ug/ml_Aprotinin). The protein concentration was determined using the bicinchoninic acid (BCA) method (Beyotime, Shanghai, China). Cell lysates (20
Annexin V-APC apoptosis assay
The cell apoptosis assay was performed using an annexin V-APC apoptosis detection kit (eBioscience, MA, USA), according to manufacturer’s instructions. The cell suspensions were incubated with 10
Colony-forming cell assay
Cells infected with a lentivirus expressing shVOPP1 or scrambled shRNA were seeded in 6-well plates (1,000 cells per well) and cultured at 37
Statistical analysis
The results were presented as mean
Results
Correlation of VOPP1 expression in HCC lesions to clinicopathological parameters
Compared with adjacent tissues, the expression of VOPP1 in HCC lesions was increased, and the difference was statistically significant (
The relative expression level of VOPP1 expression
The relative expression level of VOPP1 expression
Data are presented as numbers and percentages (in parentheses) based on the total number of patients.
Association of VOPP1 expression with clinicopathological parameters
Data are presented as numbers and percentages (in parentheses) based on the total number of patients with tumours expressing high and low VOPP1 levels.
The expression of VOPP1 is upregulated in human HCC surgical specimens: (a) A boxplot showing the relative expression of VOPP1 in normal and HCC specimens. (b) The immunohistochemical staining of VOPP1 in HCC tissue samples (T) and matched adjacent normal tissues (N). Two representative clinical cases of immunohistochemical staining are shown (upper panel with 200 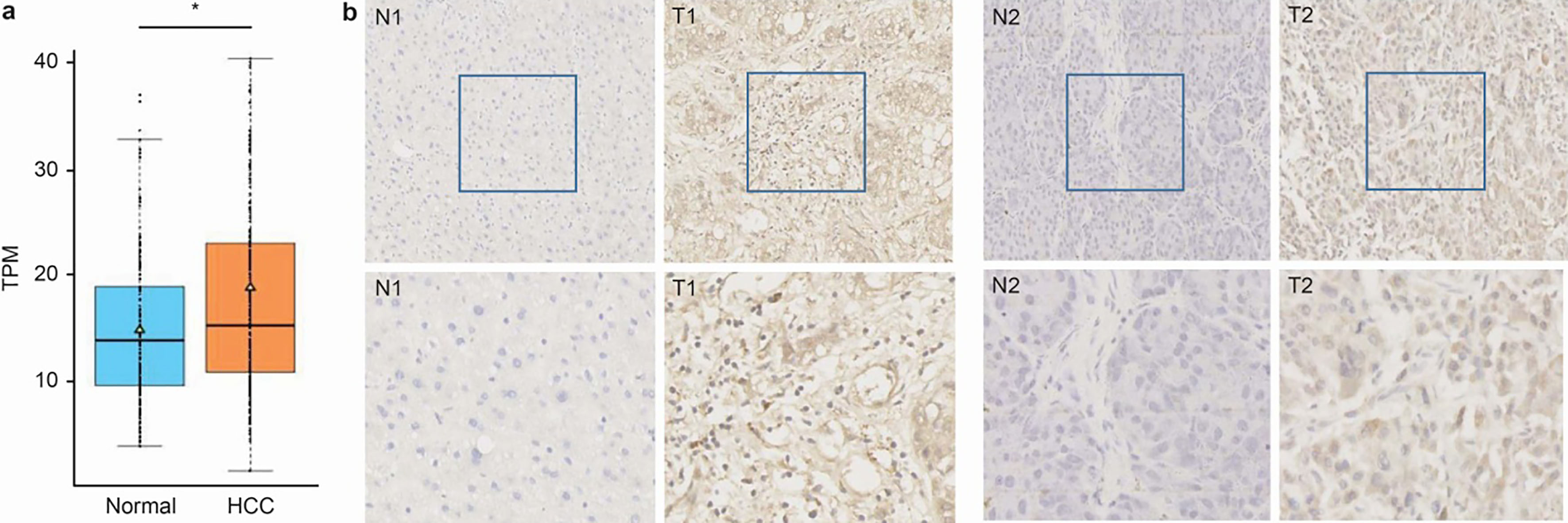
The gene expression analysis from the Cancer Genome Atlas (TCGA) database revealed that VOPP1 expression was tightly correlated with HCC (Fig. 1a). Therefore, specimens were collected and the expression of VOPP1 in HCC tissue samples and adjacent normal tissue samples were examined by immunohistochemistry (IHC) staining. The IHC results revealed the strong staining of VOPP1 in cancer tissues, when compared with adjacent liver tissues (Fig. 1b) (Table 1). These results suggest that VOPP1 is overexpressed in patients with HCC.
QRT-PCR was performed to detect the VOPP1 expression in liver cancer cell lines BEL-7402, BEL-7404, SMMC-7721 and HepG2. The relative expression levels of VOPP1 in the liver cancer cell lines were 9.79
The silencing of VOPP1 inhibited HCC cancer cell proliferation and tumorigenesis in vitro
The analysis of VOPP1 expression and its lentivirus-mediated shRNA knockdown: (a) The real-time PCR analysis of VOPP1 expression in HCC cell lines. (b)–(d) The transfection and knockdown efficiency of lentivirus shRNA using fluorescence microscopy (b), real-time PCR (c), and western blotting (d) in SMMC-7721 cells. (e) and (f) The effect of VOPP1 knockdown on cell growth potential in SMMC-7721 cells. Cell growth was measured using a Celigo cell imaging analyzer for five days. The data was presented as mean 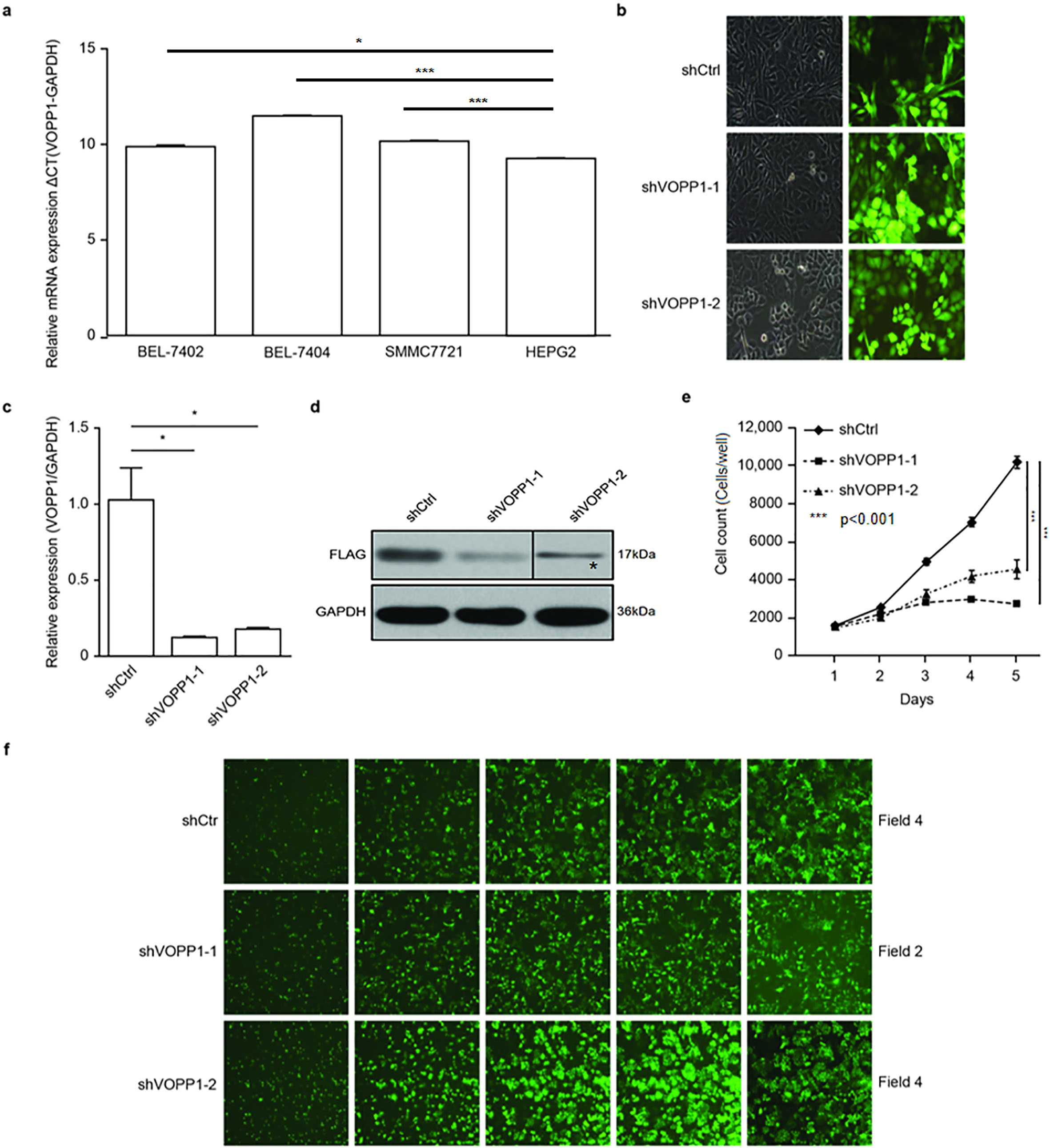
The positive correlation between VOPP1 expression and HCC suggests that VOPP1 may be involved in HCC progression. In order to further study the biological function of VOPP1 in HCC, two shRNAs that were cloned into a GFP-expressing lentivirus vector in SMMC-7721 cells were screened. On account of the GFP expression, the lentivirus infection efficiency was above 88% (Fig. 2b). The qRT-PCR analysis revealed that cells infected with the shVOPP1 lentivirus exhibited a significantly reduced VOPP1 expression, when compared with that in cells infected with the NC lentivirus (
VOPP1 knockdown inhibited the cell proliferation in HCC cell lines: VOPP1 knockdown attenuated the cell growth potential in SMMC-7721 (a) and BEL-7404 (b) cells. Cell growth was measured using a Celigo cell imaging analyzer for five days. The data was presented as mean 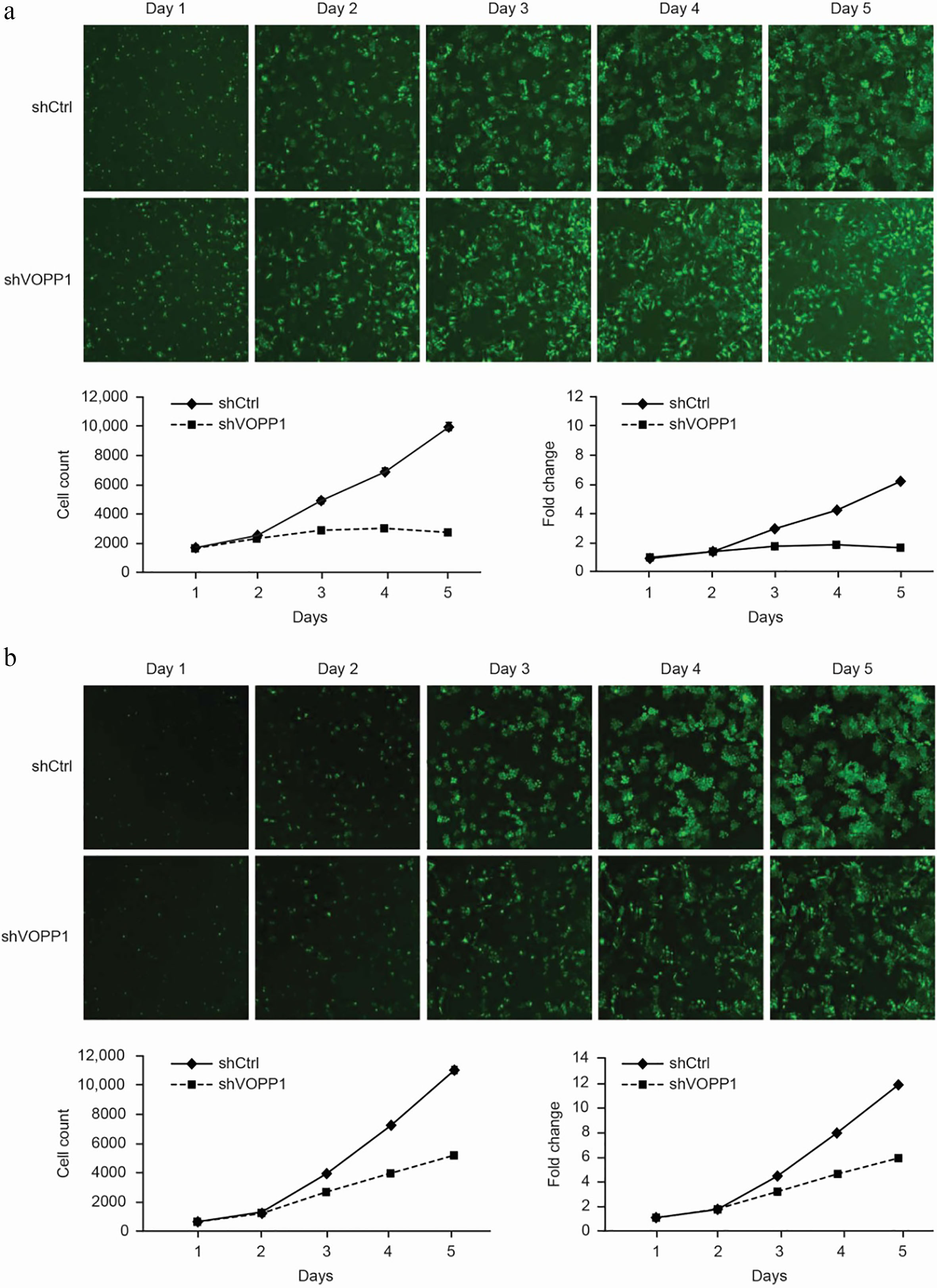
VOPP1 is involved in cell proliferation. The role of VOPP1 on cell tumorigenesis was analysed by colony formation assay. The results revealed that the colony formation ability drastically declined following VOPP1 knockdown in both SMMC-7721 and BEL-7404 cells. The number of colonies was 111
VOPP1 knockdown reduced HCC cell colony formation: VOPP1 knockdown reduced the quantity of SMMC-7721 (a) and BEL-7404 (b) colonies. The left panel presents the crystal violet dye-stained colonies in well plates; 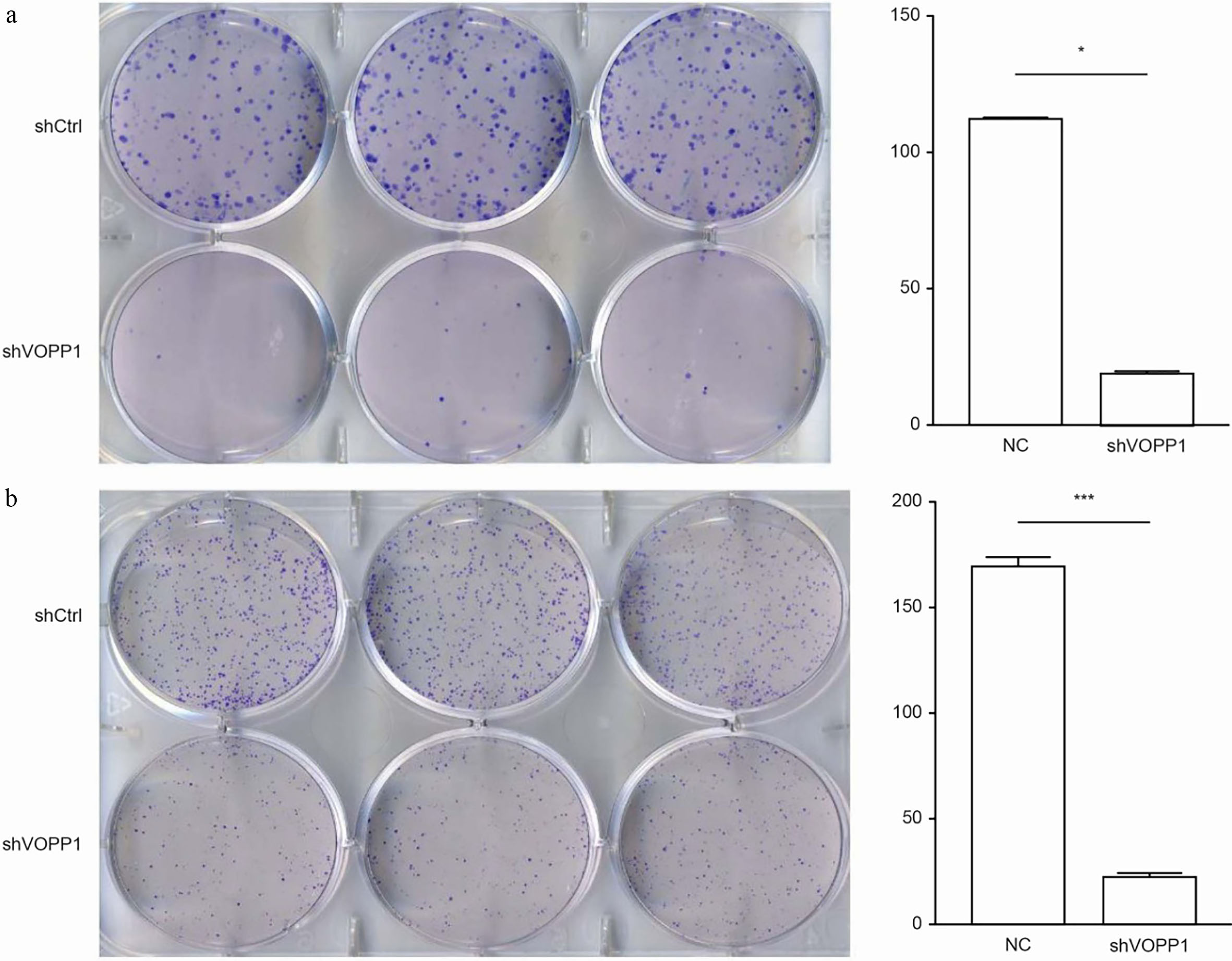
In order to examine the role of VOPP1 in primary tumour growth in vivo, an heterotopic xenograft model was used. The BEL-7404 xenograft tumor efficiency rate in nude mice was 100%. Hence, these nude mice were randomly selected and subcutaneously injected with BEL-7404 cells transfected with shVOPP1 or NC shRNA for 23 days. In accordance with the results in vitro, the tumors harvested from mice that received shVOPP1 cells were smaller than those harvested from mice that received NC shRNA cells. Both the volumes and weights of tumors from the shVOPP1 group were significantly smaller, when compared to those from the control group (
Silencing of VOPP1-induced apoptosis in HCC
Cell proliferation is usually affected by cell cycle change or cell apoptosis. Therefore, annexin V-APC staining was further performed to determine whether the apoptosis was triggered by VOPP1 downregulation. As presented in Fig. 6, the apoptosis rate of cells treated with shVOPP1 significantly increased, when compared to that of the scrambled shRNA. The percentage of apoptotic cells detected in the SMMC-7721 cell line was 4.48
Silencing of VOPP1-induced apoptosis in HCC
Silencing of VOPP1-induced apoptosis in HCC
Note: The percentage of apoptotic cells is presented mean
The knockdown of VOPP1 inhibited HCC cell growth in the heterotopic xenograft model: (a) BEL-7404 cells infected with lentiviral vectors encoding a control shRNA (NC shRNA) and shRNAs targeting VOPP1-1 (shVOPP1) were subcutaneously injected into nude mice. The representative bioluminescent images captured from subcutaneous tumors are presented. (b) The representative images of tumors are shown. (c) The growth curves of tumours in nude mice injected with cells are presented; 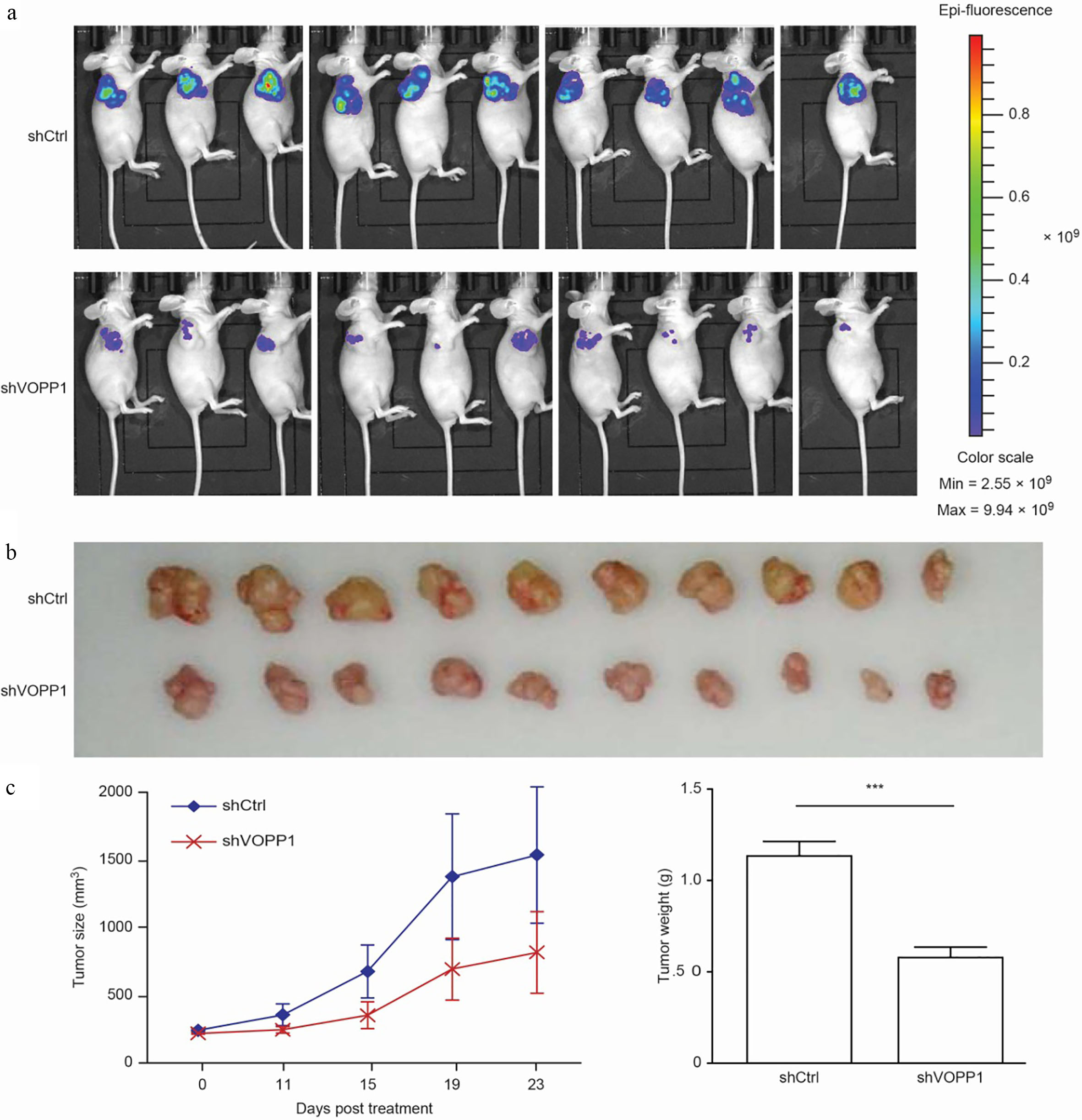
VOPP1 knockdown induced apoptosis in HCC cells: VOPP1 knockdown increased the incidence of apoptosis in SMMC-7721 (a) and BEL-7404 (b) cells. The percentage of apoptosis was calculated based on the triplicate experiments. The results were expressed as mean 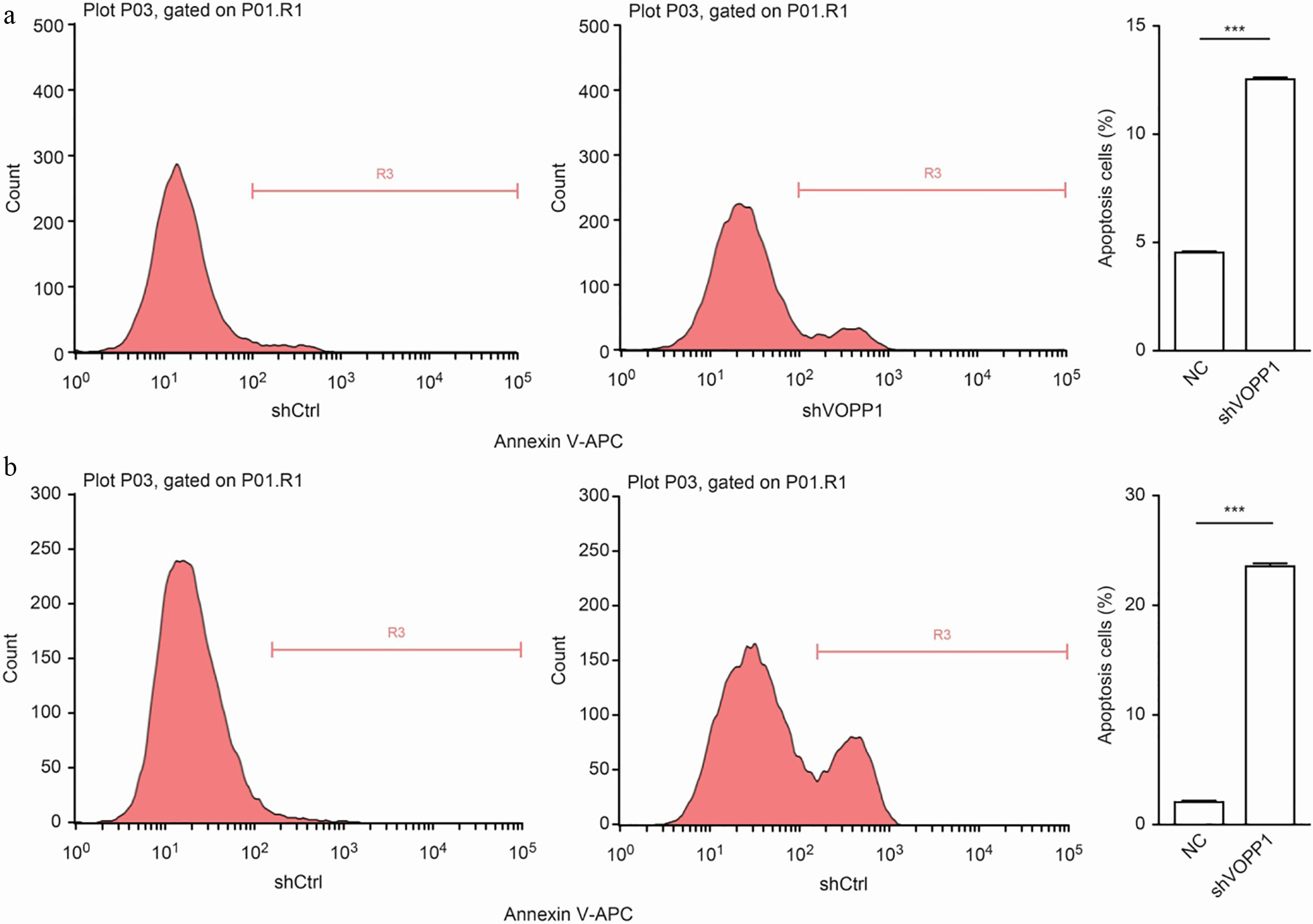
VOPP1 regulates the expression of MAPK14 in HCC cells: (a) The quantitative reverse transcription PCR analysis of MAPK14, RPS6KB1, CYLD and TWIST1 expression following VOPP1 knockdown in HCC cells. (b) The Western blot analysis of MAPK14, RPS6KB1, CYLD and TWIST1 expression following VOPP1 knockdown in HCC cells. GAPDH served as the loading control (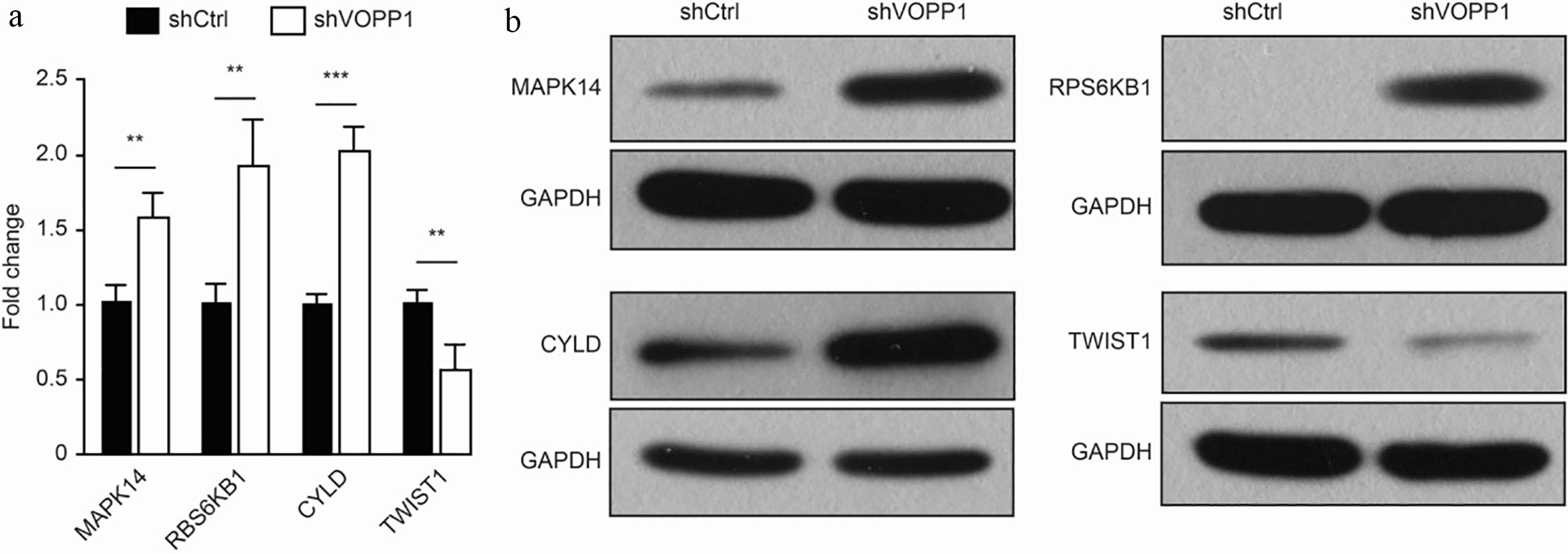
In order to investigate the potential molecular mechanism by which VOPP1 promotes HCC cell proliferation, microarray analysis was performed using cells that were either transfected with shVOPP1 or scrambled shRNA. A total of 330 upregulated genes and 679 downregulated genes were authenticated along with the VOPP1 knockdown. Based on the bioinformatics analysis, tumor necrosis factor-mediated signalling pathways were significantly activated. Therefore, the present study focused on genes that might be correlated to the tumor necrosis factor. First, the expression of four genes (MAPK14 [16] RPS6KB1 [17], CYLD [18] and TWIST1 [19]) was analysed using qRT-PCR. The qRT-PCR data revealed that the mRNA levels of MAPK14, RPS6KB1 and CYLD were upregulated in VOPP1-silenced cells (Fig. 7a). The Western blot results also revealed that the levels of MAPK14, RPS6KB1 and CYLD increased, the levels of TWIST1 decreased (Fig. 7b). These results indicate that the activation of MAPK and mTOR may be responsible for the VOPP1-regulated HCC proliferation and apoptosis.
Discussion
The molecular mechanisms of HCC have gradually become clearer in recent years, and aberrant activation of a number of cellular signaling pathways were shown to contribute to liver cancer, such as wnt/
Recent studies have implicated VOPP1 in the regulation of tumorigenesis [25]. The role of VOPP1 has been found in various cancers, including squamous cell carcinoma (SCC), gastric cancer and glioblastoma [10, 13, 14, 15]. However, there is little knowledge about its expression and role in HCC. In the present study, the expression of VOPP1 in HCC tissues and adjacent liver tissues was investigated, and the association between VOPP1 expression and clinical manifestations was analysed. In addition, the role of VOPP1 on HCC cell proliferation, colony formation tumorigenesis, and apoptosis in vitro and tumorigenesis in vivo was further evaluated.
These present results indicate that VOPP1 was upregulated in HCC samples by IHC. VOPP1 knockdown dramatically inhibited HCC cell proliferation and tumorigenesis in vitro. These results highlight that VOPP1 may play a vital role in the promotion of HCC progression. Furthermore, evidence has shown that VOPP1 enhanced HCC cell tumorigenesis in a nude mouse model.
The MAPK and mTOR signalling pathway plays important roles in tumor progression [16, 26, 27]. In the present study, MAPK14 and RPS6KB1 were found to be upregulated following VOPP1 knockdown, suggesting that VOPP1 knockdown mediates cell proliferation, apoptosis and migration via the MAPK and mTOR signalling pathways, and the multi-targeting regulation of VOPP1 [28, 29, 30, 31].
Limitations
Firstly, the effect of the inhibition of VOPP1 expression on HCC with HCC cells stably transfected with MAPK and/or mTOR expression vector(s) remains unknown, which should be further researched. Secondly, the expression level of MAPK14, RBS6KB1, CYLD, and TWIST1 in HCC compared to non-cancer cells remains unknown. Thirdly, the present trial just evaluated one element for each pathway, more elements for each pathway still need investigation. Fourthly, there were no enough data about the cell cycle, which should be further researched. Fifthly, no comparison between liver cancer cells and other cancer cells was conducted in this study.
Conclusions
The present data indicate that VOPP1 may play an important role in the progression of HCC by targeting the MAPK and mTOR signalling pathways, and that VOPP1 may potentially be a candidate as a novel molecular target for HCC therapy.
Footnotes
Acknowledgments
This study was supported by grants from the National Natural Science Foundation of China (Grant No. 81872237), the Science Technology Program of Zhejiang Province on the Scientific Research Project (Grant No. LGF19H160018, LQ18H160028 and LY17 H160069), Zhejiang Provincial Health Department Project (Grant No. 2017KY161 and 2015KYB434).
