Abstract
This article has been retracted, and the online PDF replaced with this retraction notice.
Introduction
Oral cancer (OC) is the most common malignant tumor in oral and maxillofacial region. Its incidence accounts for about 5% of systemic malignant tumors and 20% of head and neck malignant tumors [1]. Among the head and neck malignant tumors, the incidence of oral cancer ranks second only after nasopharyngeal carcinoma [2]. Although surgical resection, radiotherapy, chemotherapy, and other comprehensive treatments have been widely used in the treatment of oral cancer, the treatment effect is still unsatisfactory. The patient’s prognosis is poor and the survival rate is low.
FoxO3a is an important member of the forkhead transcription factor O subfamily (FoxO) and is a tumor suppressor gene [3, 4, 5]. As a transcription factor, FoxO3a can regulate the transcription and expression of various genes, thus affecting various biological processes, such as cell proliferation, cycle, apoptosis, migration, and invasion. Abnormal expression and dysfunction of Foxo3a is associated with tumor occurrence, progression, metastasis, and drug resistance [6, 7, 8].
MicroRNA is an endogenous non-coding small RNA molecule in eukaryotes that can be complementarily band to the 3’-untranslated region (3’-UTR) of the target gene mRNA, thus degrading mRNA or inhibiting mRNA translation to regulate the expression of target genes, thereby participating in the regulation of biological processes such as cell survival, proliferation, apoptosis, and migration. MicroRNA expression and dysfunction receive more and more attention in colon cancer [9], esophageal cancer [10], and breast cancer [11]. A number of studies showed that elevated expression of miR-155 is associated with the development, progression, and prognosis of oral cancer, suggesting that miR-155 acts as a pro-oncogene in oral cancer [12, 13, 14]. Bioinformatics analysis revealed a targeted complementary binding site between miR-155 and the 3’-UTR of FoxO3a mRNA. This study investigated whether miR-155 plays a role in regulating FoxO3a expression and affecting proliferation, apoptosis, and cisplatin (DDP) resistance in oral cancer cells.
Materials and methods
Main reagents and materials
Human normal oral epithelial cell line and special medium were purchased from Wuhan procell. Human oral cancer cell line KB was purchased from Shanghai Huiying Biotechnology. HEK293 T cells were purchased from Beijing Beina Biotechnology. DMEM medium was purchased from Hyclone. Opti-MEM and fetal Bovine serum (FBS) were purchased from Gibco. Trizol and Lipo 2000 transfection reagents were purchased from Thermo. ReverTra Ace qPCR RT Kit and SYBR dye were purchased from Toyobo. MiR-155 mimic, inhibitor, and miR-NC were purchased from RiboBio. Rrabbit anti-human polyclonal antibody Foxo3a antibody was purchased from Abcam. Rabbit anti-human polyclonal antibody
Cell culture
Human normal oral epithelial cell line was cultured in special medium (including M199, FBS, epithelial cell growth additive, Hydrocortisone, Insulin, Transferrin, Epinephrine, and penicillin-streptomycin). KB cells were cultured in DMEM medium containing 10% FBS and 1% penicillin-streptomycin. Both cells were cultured in a chamber containing 5% CO
DDP drug resistant cell line establishment
KB cells in logarithmic phase were treated by DDP from 0.1
KB and KB/DDP cells were treated by different concentrations (0, 0.1, 1, 10, 100, and 1000
Dual luciferase reporter gene assay
The PCR product of the FoxO3a 3’-UTR full-length fragment or mutant fragment was double-digested and then ligated into the pMIR vector. After sequencing, the plasmid was designated as pMIR-FoxO3a-WT and pMIR-FoxO3a-MUT. The HEK293 T cells were transfected with pMIR-FoxO3a-WT (or pMIR-FoxO3a-MUT) together with miR-155 mimic (or miR-NC) by Lipofectamine 2000. After incubated for 48 h, luciferase activity was detected by Dual-Glo Luciferase Assay System kit according to the manual.
Cell transfection and grouping
KB/DDP cells were divided into miR-NC group and miR-155 inhibitor group. The cells were seeded in 6-well plate and treated by 1.6
qRT-PCR
Trizol reagent was used to extract RNA and reverse transcribe RNA to cDNA. The reverse transcription system contained 1
Western blot
Total protein was extracted from the cells by RIPA. After quantified by BCA method, a total of 40
Cell apoptosis detection
The cells were digested by trypsin and collected. After resuspension in 100
Cell proliferation detection
The cells were added with EdU solution at 10
Statistical analysis
SPSS 18.0 software was applied for data analysis. Measurement data was presented as mean
Results
The targeted regulatory relationship between miR-155 and Foxo3a mRNA
Bioinformatics analysis showed the complementary binding site between miR-155 and the 3’-UTR of FoxO3a mRNA (Fig. 1A). Dual luciferase reporter gene assay exhibited that miR-155 mimic transfection significantly reduced the relative luciferase activity in HEK293 T cells transfected by pMIR-Foxo3a-WT but not by pMIR-FoxO3a-MUT, confirming that FoxO3a was the target gene of miR-155a (Fig. 1B).
The targeted regulatory relationship between miR-155 and FoxO3a. (A) The complementary binding site between miR-155 and the 3’-UTR of FoxO3a mRNA. (B) Dual luciferase reporter assay. *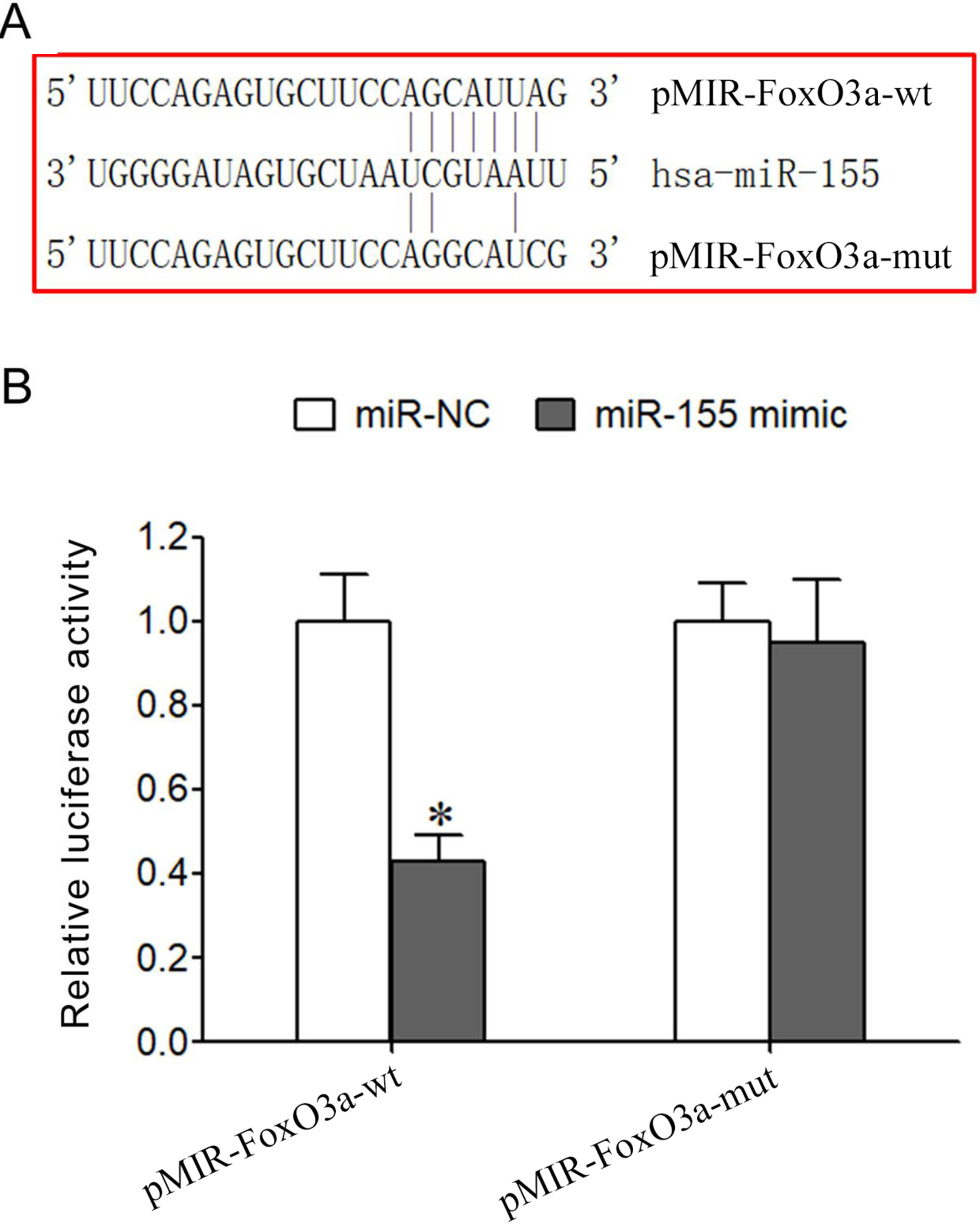
CCK-8 assay showed that the proliferative activities of parental KB cells were significantly lower than those of KB/DDP cells under the same dose treatment of DDP (Fig. 2). The IC
RI comparison
RI comparison
CCK-8 detection of KB and KB/DDP cell viabilities. *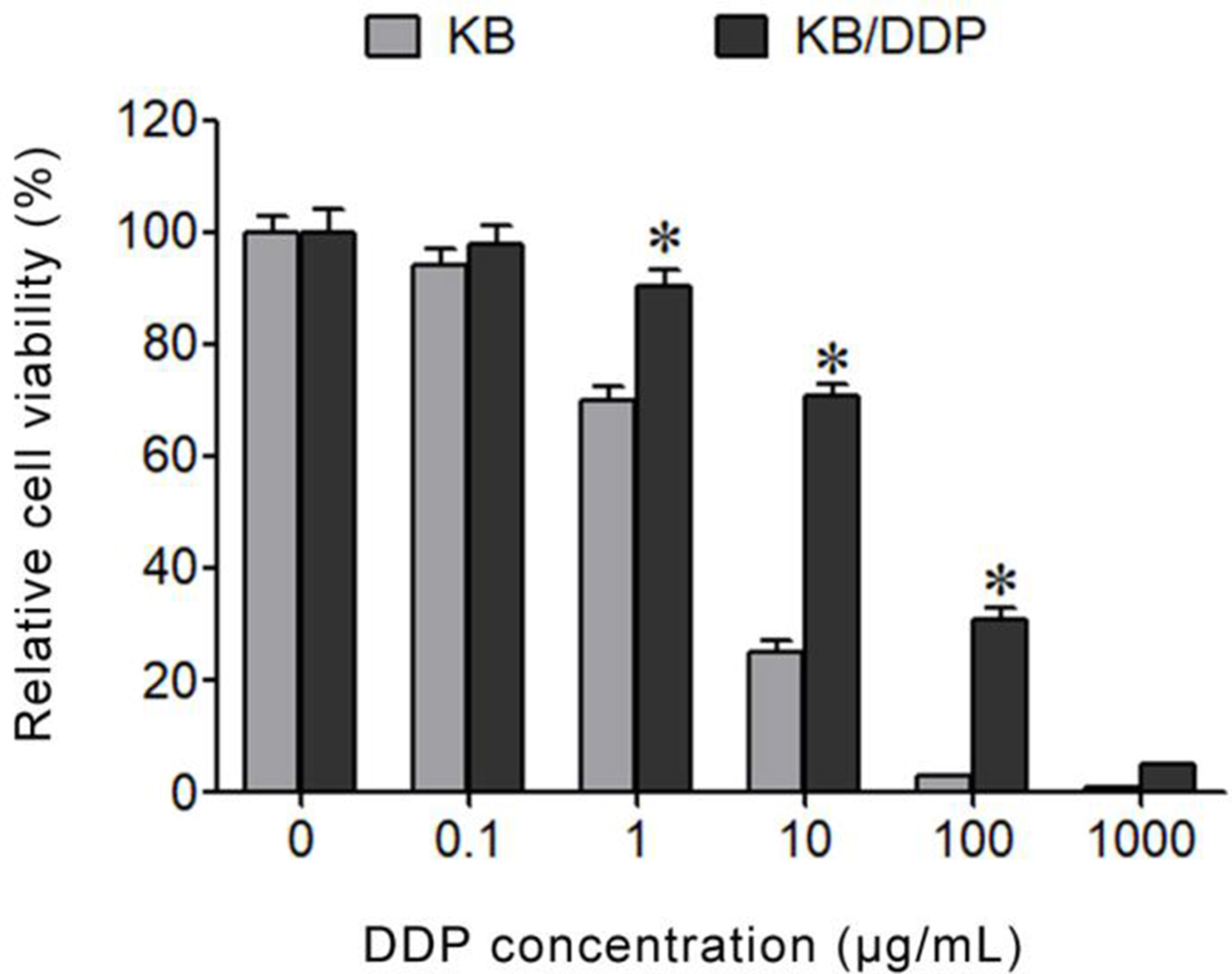
qRT-PCR demonstrated that compared with human normal oral epithelial cells, miR-155 expression was significantly elevated in KB cells, and its level was obviously higher in KB/DDP cells than that of parental KB cells (Fig. 3A). qRT-PCR revealed that the FoxO3a mRNA expression in KB cells was markedly lower than that of normal oral epithelial cells, and it was apparently lower in KB/DDP cells compared with the parental KB cells (Fig. 3A). Western blot exhibited that compared with normal oral epithelial cells, FoxO3a protein expression in KB cells was significantly downregulated, and its level in KB/DDP cells obviously attenuated (Fig. 3B).
MiR-155 elevated, while FoxO3a downregulated in drug resistant cells. (A) qRT-PCR detection of miR-155 and FoxO3a expressions; (B) Western blot detection of Foxo3a protein expression; * 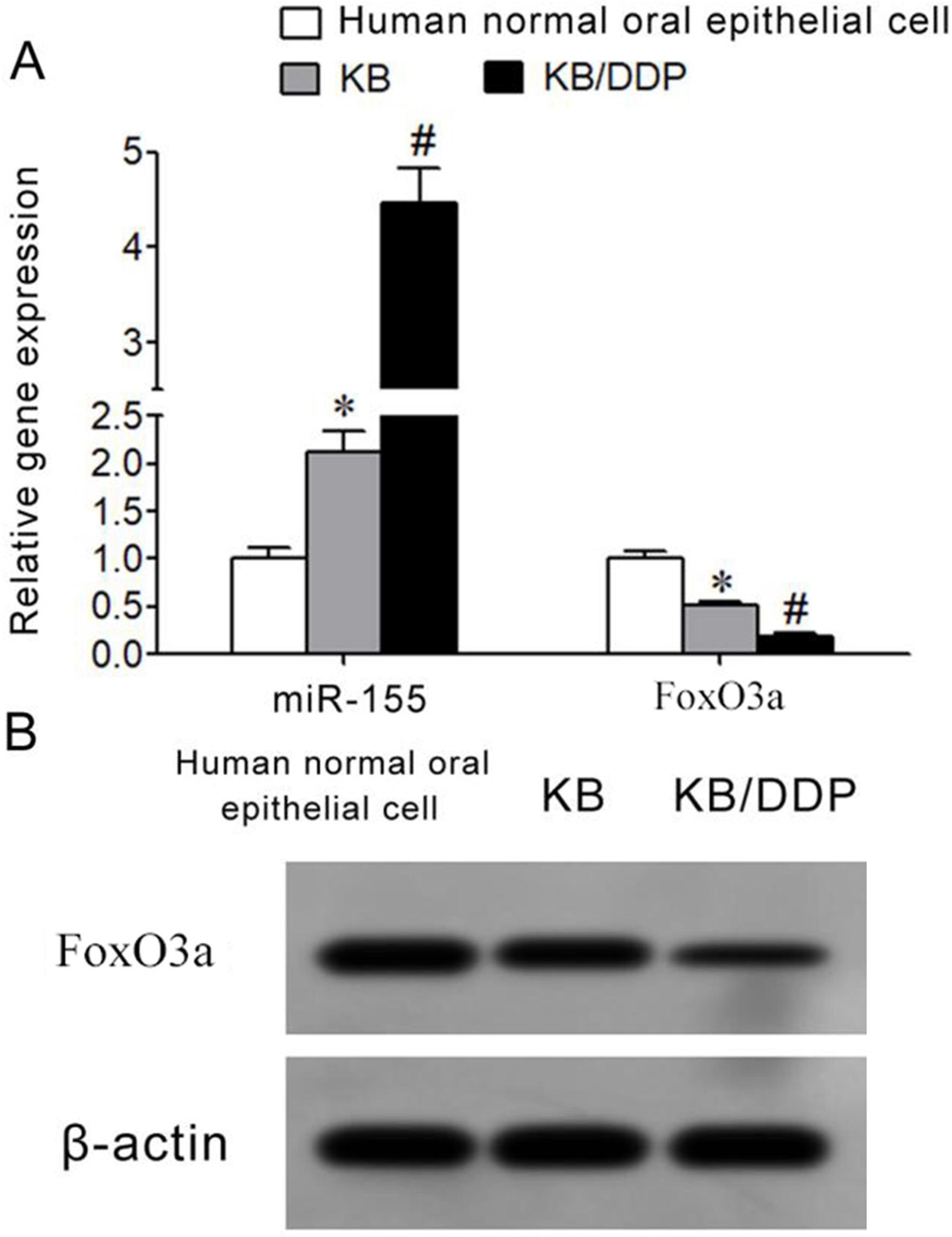
qRT-PCR showed that miR-155 inhibitor transfection significantly downregulated miR-155 expression and elevated Foxo3a mRNA level in KB/DDP cells compared with miR-NC group (Fig. 4A). Western blot demonstrated that miR-155 inhibitor transfection obviously elevated FoxO3a protein level in KB/DDP cells (Fig. 4B). Flow cytometry revealed that transfection of miR-155 inhibitor markedly enhanced cell apoptosis, while inhibiting cell proliferation in KB/DDP cells (Fig. 4C and D).
MiR-155 inhibitor transfection promoted KB/DDP cell apoptosis and reduced DDP resistance. (A) qRT-PCR detection of miR-155 expression; (B) Western blot detection of FoxO3a protein expression; (C) Flow cytometry detection of cell apoptosis; (D) Flow cytometry detection of cell proliferation; *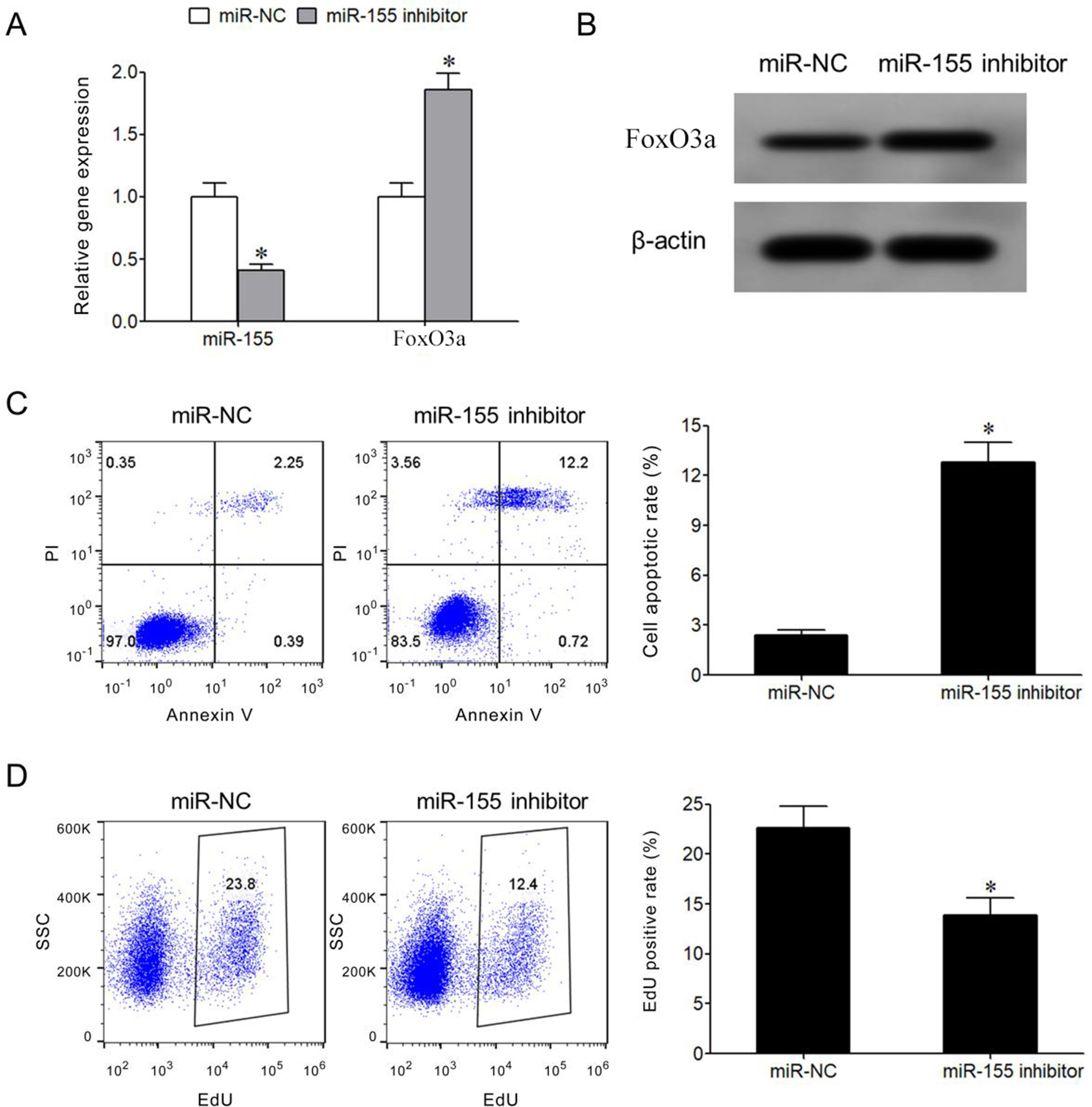
In recent years, the incidence of oral cancer keeps increasing. About 300,000 oral cancer patients were diagnosed each year worldwide [15]. The incidence of oral cancer has ranked 9th, and the mortality rate ranks 8th in global malignant tumors. The pathogenesis of oral cancer is not fully understood, and is related to smoking, drinking, viral infection, eating habits, and other factors. Smoking and drinking are the biggest risk factors for oral cancer [16]. Although surgical resection, radiotherapy, chemotherapy and other comprehensive treatments have been widely used in the treatment of oral cancer, the treatment effect is still unsatisfactory. The patient’s prognosis is still poor, and the survival rate is low [17, 18, 19].
FoxO is a class of evolutionarily highly conserved transcription factors that are widely involved in the regulation of various biological processes, such as embryonic development, cell proliferation, cell cycle, and apoptosis. The FoxO transcription factor family contains four members, including FoxO1, FoxO3a, FoxO4, and FoxO6, of which FoxO3a is mostly investigated. FoxO3a is a well-defined tumor suppressor gene that regulates a variety of genes involved in cell proliferation, cell cycle, and apoptosis, such as BIM (Bcl-3 mediator of cell death, BIM), PUMA (p53 up-regulated modulator of apoptosis), CKI (cyclin-dependent kinase inhibitor), p27Kip1, and cyclin D1 (cyclin-dependent kinase D1). It participates in the regulation of multiple signaling pathways in tumor cells and various biological processes, such as proliferation, cycle, apoptosis, migration, and invasion [5, 20, 21, 22, 23, 24]. The abnormal expression and function of FoxO3a are related to the occurrence, progression, metastasis, and drug resistance of ovarian cancer [6], breast cancer [7], and lung cancer [8]. However, whether the abnormality of FoxO3a is related to the pathogenesis of oral cancer is unclear.
MiR-155 is a well-researched microRNA that is closely related to the occurrence and progression of various tumors, such as breast cancer [25], intestinal cancer [26], lung cancer [27]. It was shown that miR-155 overexpression is related to the occurrence, progression, and prognosis of oral cancer, suggesting that miR-155 plays an oncogene role in oral cancer [12, 13, 14]. Bioinformatics analysis revealed a targeted complementary binding site between miR-155 and FoxO3a. In this study, we explored whether miR-155 regulates the expression of FoxO3a and affects the proliferation and apoptosis of oral cancer cells.
In this study, the dual luciferase gene reporter assay demonstrated that transfection of miR-155 mimic significantly reduced the relative luciferase activity in pMIR-FoxO3a-wt transfected HEK293 T cells, but not in pMIR-FoxO3a-mut transfected HEK293 T cells, indicating that miR-155 can target the 3’-UTR region of FoxO3a mRNA and inhibit its expression. The CCK-8 assay showed that DDP treatment can significantly inhibit the proliferation of KB cells, but exhibited less effect on KB/DDP cells, indicating the successful establishment of DDP-resistant oral cancer cells. Compared with normal oral epithelial cells, miR-155 expression in oral cancer KB cells was significantly increased. Its level in drug-resistant oral cancer KB/DDP cells was obviously higher than that of parental KB cells. Compared with normal oral epithelial cells, FoxO3a expression in oral cancer KB cells was markedly reduced. Its level in drug-resistant oral cancer KB/DDP cells was apparently lower than that in parental oral cancer KB cells. This indicated that miR-155 plays a role in targeted inhibition of FoxO3a, and that increased expression of miR-155 and decreased expression of FoxO3a are related to the pathogenesis and DDP resistance of oral cancer cells.
This study further explored whether miR-155 plays a role in regulating the proliferation, apoptosis, and DDP resistance of oral cancer cells through FoxO3a. It was observed that the transfection of miR-155 inhibitor increased the expression of FoxO3a in KB/DDP cells, enhanced sensitivity to DDP, aggravated cell apoptosis under DDP treatment, and attenuated cell proliferative capacity. Bano et al. [28] found that the expression of miR-155 was significantly increased in oral cancer stem cells, and was related to the malignant biological characteristics of cancer stem cells. Baba et al. [13] showed that the expression of miR-155 was obviously upregulated in tumor tissues of patients with oral cancer, especially in metastatic tumor tissues. In oral cancer HSC-3 cells, transfection of miR-155 inhibitor markedly enhanced the expression of E-cadherin, inhibited EMT process, and attenuated the migration and metastasis of oral cancer cells. Kim et al. [12] revealed that the expression of miR-155 was apparently elevated in tumor tissues of patients with oral cancer, and was associated with tumor TNM stage and poor prognosis. Rather et al. [14] reported that the expression of miR-155 was significantly increased in oral cancer KB cell line and oral cancer patients. Overexpression of miR-155 in oral cancer KB cells obviously reduced CDC73 expression, promoted cell proliferation, and reduced cell apoptosis; inhibiting the expression of miR-155 markedly upregulated the expression of CDC73 in oral cancer cells, promoted cell apoptosis, inhibited cell proliferation, and reduced the tumorigenicity of KB cells in animals. This study combined the targeted regulation between miR-155 and FoxO3a, revealing that elevated expression of miR-155 plays a role in inhibiting FoxO3a expression, promoting oral cancer cell proliferation, reducing apoptosis, and enhanced DDP resistance. However, whether miR-155 regulates FoxO3a in relation to drug resistance in patients with oral cancer is still unclear.
Conclusion
Increased expression of miR-155 and decreased expression of FoxO3a were associated with DDP resistance in oral cancer cells. Inhibition of miR-155 expression upregulated FoxO3a expression, restrained oral cancer cell proliferation, promoted cell apoptosis, and enhanced DDP sensitivity in oral cancer cells.
