Abstract
Background:
Although renal cell carcinoma remains one of the most malignant cancers, our understanding of progression and recurrence of this disease is limited. The present study explored the precise role of miR-155-5p in renal cancer metastasis.
Methods:
The expression of miR-155-5p in renal carcinoma clinical tissues and cells was determined using quantitative real time-polymerase chain reaction. The role of miR-155-5p on tumor cell growth were examined using CCK-8 and colony formation assays. Transwell assay was utilized to identify the role of miR-155-5p on the invasion and migration of renal cancer cells. Markers of epithelial-mesenchymal transition were determined using western blot. The in vivo effects of miR-155-5p on renal cancer cell growth, apoptosis, and metastasis were explored using xenograft mice. Luciferase reporter assay was performed to identify the potential target of miR-155-5p.
Results:
Levels of miR-155-5p were significantly elevated in renal cancer tissues and cell lines. Suppression of miR-155-5p decreased the growth, colony formation, migration, and invasiveness of renal cancer cells. In contrast, overexpression of miR-155-5p led to opposite effects on renal cancer cells. Mechanically, the apoptosis-inducing factor was identified as the target of miR-155-5p. Interference of miR-155-5p significantly increased mRNA and protein expression of the apoptosis-inducing factor, whereas overexpression of miR-155-5p remarkably suppressed the apoptosis-inducing factor levels in renal cancer cells. The xenograft model identified that suppression of miR-155-5p restrained tumor growth and promoted apoptosis, whereas overexpression of miR-155-5p decreased apoptosis and accelerated tumor growth. Moreover, the number of lung metastasis nodules were decreased following injection with anti-miR-155-5p transfected cells, whereas the nodules were remarkably increased after overexpression of miR-155-5p. In addition, in vitro and in vivo assays both confirmed that suppression of miR-155-5p increased the expression of E-cadherin and decreased levels of N-cadherin and Snail, whereas overexpression of miR-155-5p accelerated epithelial–mesenchymal transition progression in renal cancer cells.
Conclusion:
These findings demonstrate that miR-155-5p enhances metastasis and epithelial–mesenchymal transition by targeting the apoptosis-inducing factor, suggesting that miR-155-5p represents a novel therapeutic target for renal cancer.
Introduction
Renal cell carcinoma (RCC), a high-risk kidney cancer with a high-mortality rate, originates from the lining of the proximal convoluted tubules and accounts for approximately 2%–3% of human cancers worldwide. 1 In China, RCC is the second most frequent genitourinary malignant cancer, demonstrating a steady increase in frequency in recent years. 2 Although progress has been made in cancer-diagnostic technology, the majority of patients are diagnosed in the advanced metastatic stage resulting in dramatic decrease of 5-year survival. It is critical to unravel the pathogenesis of the advanced aggressive stage of this disease and to further develop novel therapeutic options for RCC.
Epithelial-to-mesenchymal transition (EMT) is closely associated with the aggressiveness of cancer during which EMT promotes cell migration and invasion, and suppresses apoptosis and senescence.3,4 EMT is characterized by the loss of cell adhesion of epithelial cells and the gain of mesenchymal phenotype with potentiated migratory and invasive capability. Increasing evidence confirms that the acquisition of EMT is mechanistically associated with metastasis, drug resistance, and tumor recurrence.5,6 Elucidating the mechanism that controls RCC metastasis is crucial to develop novel therapeutic options for the disease.
miRNAs are 19- to 25-nucleotide, non-coding RNAs that are indispensable upstream regulators of gene expression at the posttranscriptional level. Diverse biological functions have been confirmed to be influenced by miRNAs, including proliferation, apoptosis, metastasis, and invasion.7,8 Indeed, aberrant expression of miRNAs is detected in multiple cancers, including RCC. Some of these miRNAs function as oncogenic or tumor suppressor molecules involved in oncogenesis, progression, and prognosis. 9
miR-155-5p plays a critical role in immune responses and oncogenesis. 10 Interestingly, miR-155-5p is found to be elevated in various types of tumors, including RCC. 11 In addition, high expression of miR-155-5p positively correlates with recurrence and a shorter median survival time in RCC patients, representing an oncogenic miRNA in this disease. 12 However, the precise role of miR-155-5p and the associated mechanism remains unknown in RCC. Therefore, our present study identified the expression profile and biological role of miR-155-5p in RCC. In addition, the regulatory mechanism of miR-155-5p on EMT during RCC oncogenesis was explored, aiming to identify novel miRNAs that may act as novel therapeutic targets for RCC.
Materials and methods
Tissue specimens
In total, 16 pairs of RCC and adjacent normal tissue specimens were collected from the Haikou Municipal Hospital. All participants had no chemotherapy or any other medications before surgery. This study was in accordance with the Helsinki declaration and approved by the ethics committee of Haikou Municipal Hospital. Informed consent was obtained from the participants.
Cell culture
The normal human renal cell line (HK-2), and four RCC cell lines (786-O, 769-P, Caki-1, and ACHN) were purchased from the Chinese Academy of Sciences Cell Bank of Type Culture Collection (Shanghai, China). Cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM) (Gibco, Langley, OK, USA) containing 10% fetal bovine serum (FBS) (Gibco, Langley, OK, USA). Cells were cultured in a humidified incubator with 5% CO2 at 37°C.
miRNA transfection
Cells at the density of 2×105 cells were seeded into each well of a 6-well plate. When the cells were 70%–80% confluent, they were transfected with anti-miR-control, anti-miR-155-5p, pCDH, and pCDH-miR-155-5p using Lipofectamine 2000 reagent (Invitrogen, USA) according to the manufacturer’s protocol.
Cell viability assay
CCK-8 assay was performed for determining cell viability, using the CCK-8 assay kit according to the manufacturer’s protocol (CCK-8; Dojindo Laboratories, Japan). In brief, cells were seeded into 96-well plates. Then, 10 µL of CCK-8 solution were added into each well and incubated at 37°C for 2 h. The optical absorbance was measured using an ELISA reader at a wavelength of 450 nm.
Migration and invasion assays
To measure cell migration and invasion, 5×104 786-O and Caki-1 cells transfected with either anti-miR-155-5p or pCDH-miR-155-5p were seeded into Transwell chambers (Corning, NY, USA) uncoated or coated with Matrigel (BD Biosciences, Bedford, MA, USA) and incubated for 24h at 37°C in a humidified incubator with 5% CO2. Medium containing 10% FBS in the lower chamber served as the chemoattractant. Non-migrated cells were mechanically removed, and the migrated cells were fixed in methanol and stained with 0.5% crystal violet. Finally, all migrated or invaded cells were counted microscopically at a magnification of ×400.
Quantitative real-time polymerase chain reaction
Total RNA of clinical tissues and cells were prepared using TRIzol (Invitrogen, USA) according to the manufacturer’s instructions. Briefly, 1 μg RNA was used to synthesize cDNA using M-MLV Reverse Transcriptase (Promega, Fitchburg, WI). Polymerase chain reaction (PCR) was performed using the SYBR Green PCR Master Mix (Applied Biosystems, Foster City, CA). The relative expression levels of each gene were determined using the comparative cycle threshold (Ct) method through calculating the 2(-ΔΔCt). U6 and GAPAH were used as the endogenous controls.
Western blot
Whole cells were lysed in 1× SDS sample buffer and the supernatants were transferred into fresh tube. Then, protein samples were separated by SDS-PAGE and transferred to nitrocellulose membranes (Millipore, Billerica, MA, USA). The membranes were probed with primary antibodies against apoptosis-inducing factor (AIF), E-cadherin, N-cadherin, and Snail (1:1000, Abcam, CA, USA) overnight, and then incubated with appropriate horseradish peroxides–conjugated secondary antibodies for 1h at room temperature. Finally, protein signals were detected using a Super Signal Enhanced Chemiluminescence kit (Pierce, Rockford, IL, USA).
Xenograft model
All nude mice were housed in SPF-grade facilities with a controlled humidity and room temperature. They were fed in the isolation cages with access to food and water ad libitum. Anti-miR-155-5p or pCDH-miR-155-5p transfected cells were subcutaneously inoculated into nude mice. The tumor volume was calculated using the following formula: length × width2/2. For lung metastasis assay, cells were injected into mice via tail vein, and the number of metastatic nodules into the lungs were counted. Animal experiments were approved by the Institutional Animal Care and Use Committee of Haikou Municipal Hospital, which is affiliated to the Xiangya School of Medicine of the Central South University.
Luciferase reporter assay
RCC cells at 1 × 105 was cultured into 6-well plate for 24h. The luciferase reporter vector for the wild type (WT) or mutant type (MUT) 3ʹ-UTR of AIF was inserted into the pMIR-reporter luciferase system (Thermo Fisher Scientific, USA). Anti-miR-155-5p or pCDH- miR-155-5p together with WT or MUT 3ʹ-UTR of AIF was co-transfected into 786-O and Caki-1 cells. After 24h, the luciferase activity was detected using the dual-luciferase reporter assay (Promega, Madison, WI, USA).
TUNEL staining
For apoptosis assay in vivo, TUNEL staining was performed using an in-situ cell death detection kit (Roche, Mannheim, Germany) according to the manufacturer’s instructions. Briefly, sections were fixed in ethano/lacetic acid (2:1), incubated in 3% H2O2, permeabilized with 0.5% Triton X-100, and then incubated in the TUNEL reaction mixture. Sections were rinsed, stained with DAPI, and observed under fluorescence microscopy.
Statistical analysis
Data were presented as mean ± SD for the three experiments in each group. Differences between groups were analyzed using one-way ANOVA with post hoc (Tukey’s) test using GraphPad Prism 6 (GraphPad Software). A P value of <0.05 was considered statistically significant.
Results
MiR-155-5p regulates RCC cell growth and colony formation
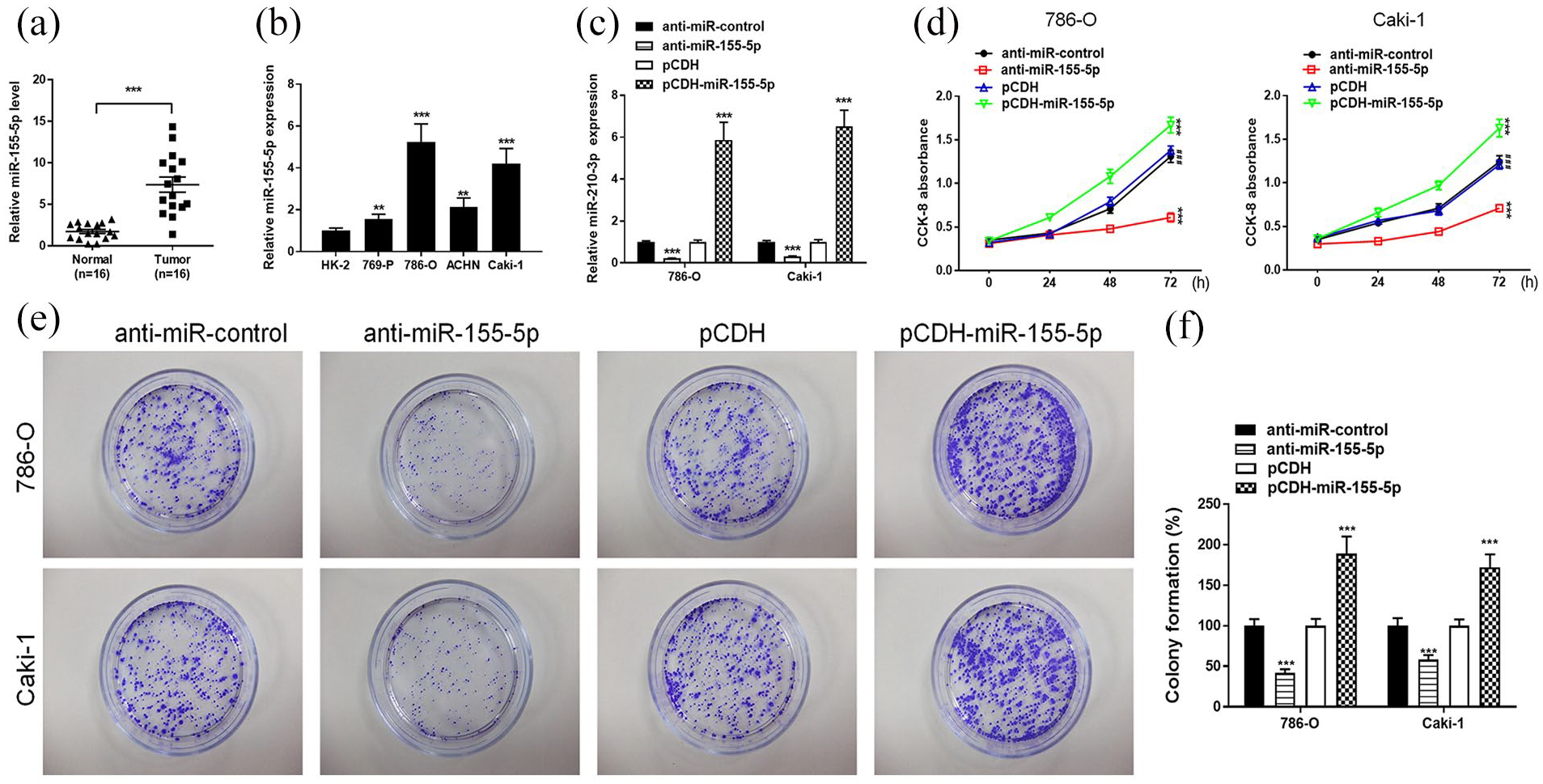
First, we explored the expression profile of miR-155-5p in RCC tissues and the adjacent normal tissues. A total of 16 cases of RCC and the adjacent normal tissues were collected for determination of miR-155-5p expression. Data from quantitative real time-polymerase chain reaction (qRT-PCR) showed that levels of miR-155-5p were significantly increased in RCC tissues compared to the adjacent normal tissues (Figure 1(a)). The level of miR-155-5p in RCC cell lines and the normal human renal cell line (HK-2) was also detected by qRT-PCR. Consistently, the expression of miR-155-5p was also elevated in four RCC cell lines, including 769-P, 786-O, ACHN, and Caki-1, compared to that in the control HK-2 cell line (Figure 1(b)). RCC cell lines 786-O and Caki-1 with highest levels of miR-155-5p were chosen for further investigation. To investigate whether miR-155-5p is involved in RCC cell proliferation and colony formation, we constructed the miR-155-5p downexpression and upregulation system in 786-O and Caki-1 cells using miR-155-5p inhibitor and miR-155-5p mimics, respectively. We found that miR-155-5p was successfully down-regulated or up-regulated in 786-O and Caki-1 cells (Figure 1(c)). Strikingly, transfection with anti-miR-155-5p decreased cell proliferation while overexpression of miR-155-5p significantly suppressed the proliferation in 786-O and Caki-1 cells (Figure 1(d)). We next examined the colony formation by transfecting 786-O and Caki-1 cells with anti-miR-155-5p and pCDH-miR-155-5p. Notably, transfection by anti-miR-155-5p dramatically decreased colony number, whereas upregulation of miR-155-5p enhanced the colony formation ability in 786-O and Caki-1 cells (Figure 1(e) and (f)). These data suggest that miR-155-5p may serve as a cancer promoter in RCC in vitro.
MiR-155-5p regulates RCC migration and invasion via EMT
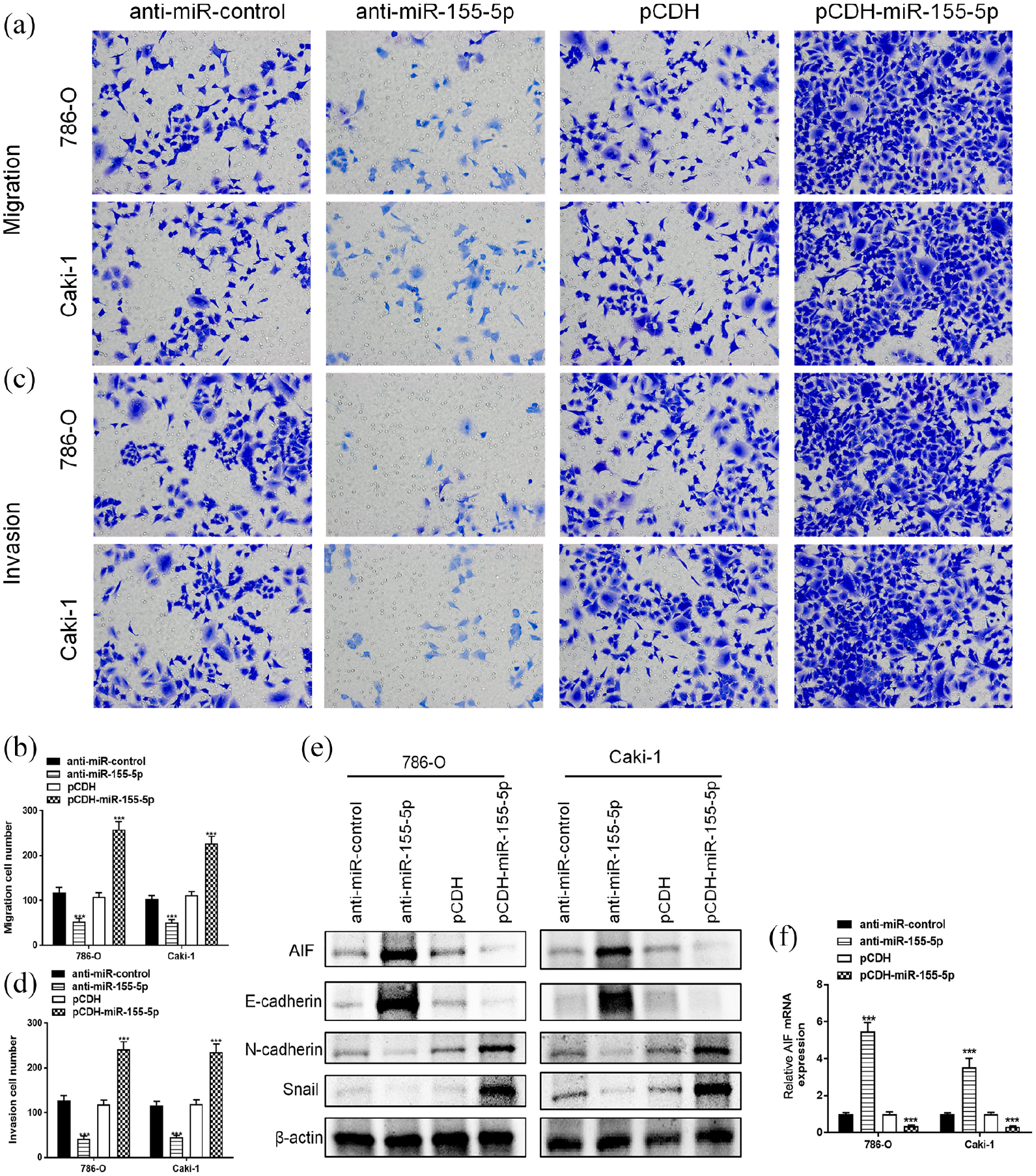
Given that metastatic potential is critical for RCC progression, we asked whether miR-155-5p could regulate the migration and invasion in 786-O and Caki-1 cells. The cell migration test showed that the migration cell number of 786-O and Caki-1 cells in the anti-miR-155-5p group was obviously lower than that anti-miR-control group. Meanwhile, overexpression of miR-155-5p significantly promoted the migratory ability in RCC cells (Figure 2(a) and (b)). In addition, we found that suppression of miR-155-5p also hampered the invasiveness, whereas upregulation of miR-155-5p dramatically enhanced the invasive potential in 786-O and Caki-1 cells (Figure 2(c) and (d)). Furthermore, we asked whether AIF is involved in this process; the levels of AIF were determined using qRT-PCR and western blot. Strikingly, interference of miR-155-5p significantly increased mRNA and protein expression of AIF, whereas overexpression of miR-155-5p remarkably suppressed AIF levels both at mRNA and protein levels in 786-O and Caki-1 cells (Figure 2(e) and (f)). Also, we evaluated the critical role of EMT in miR-155-5p-mediated metastasis and invasion in RCC. Western blot results showed that suppression of miR-155-5p increased the expression of E-cadherin, a marker of epithelial phenotype, and decreased mesenchymal markers N-cadherin and Snail. By contrast, upregulation of miR-155-5p dramatically suppressed epithelial E-cadherin and enhanced the protein levels of N-cadherin and Snail in 786-O and Caki-1 cells (Figure 2(e)). Collectively, these findings reveal that miR-155-5p mediates migration and invasion via inducing EMT progression in RCC.
MiR-155-5p binds to the 3ʹ-UTR of AIF
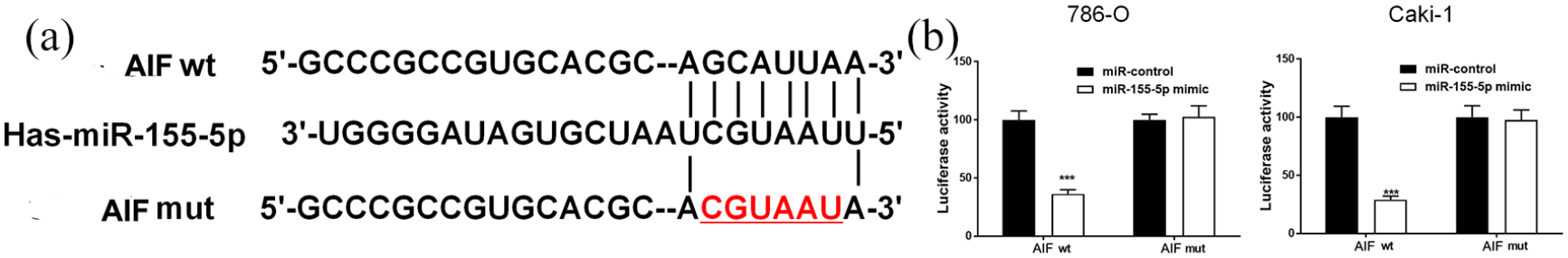
Commonly, miRNAs exert the biological role by regulating their target genes and we utilized bioinformatics analysis tools to predict the targets of miR-155-5p. Consequently, the 3′-UTR of AIF gene contained binding sites partially complementary to miR-155-5p (Figure 3(a)). As previously mentioned, interference of miR-155-5p increased the mRNA and protein expression of AIF while the overexpression of miR-155-5p inhibited AIF in RCC cells. To verify that the AIF gene was the potential target of miR-155-5p, the WT 3′-UTR of AIF or MUT 3′-UTR of AIF that contained the miR-155-5p binding sites was inserted into the pMIR-REPORT luciferase system (Figure 3(a)). Then, we co-transfected 786-O and Caki-1 cells with miR-155-5p mimic and wild or mutant AIF. The luciferase activity in 786-O and Caki-1 cells that were transfected with WT 3′-UTR of AIF was inhibited by miR-155-5p; however, miR-155-5p had no inhibitive effects on luciferase activity in 786-O and Caki-1 cells transfected with MUT 3′-UTR of AIF (Figure 3(b)). Taken together, these results indicated that AIF was the target of miR-155-5p.
MiR-155-5p regulates RCC growth and metastasis in vivo
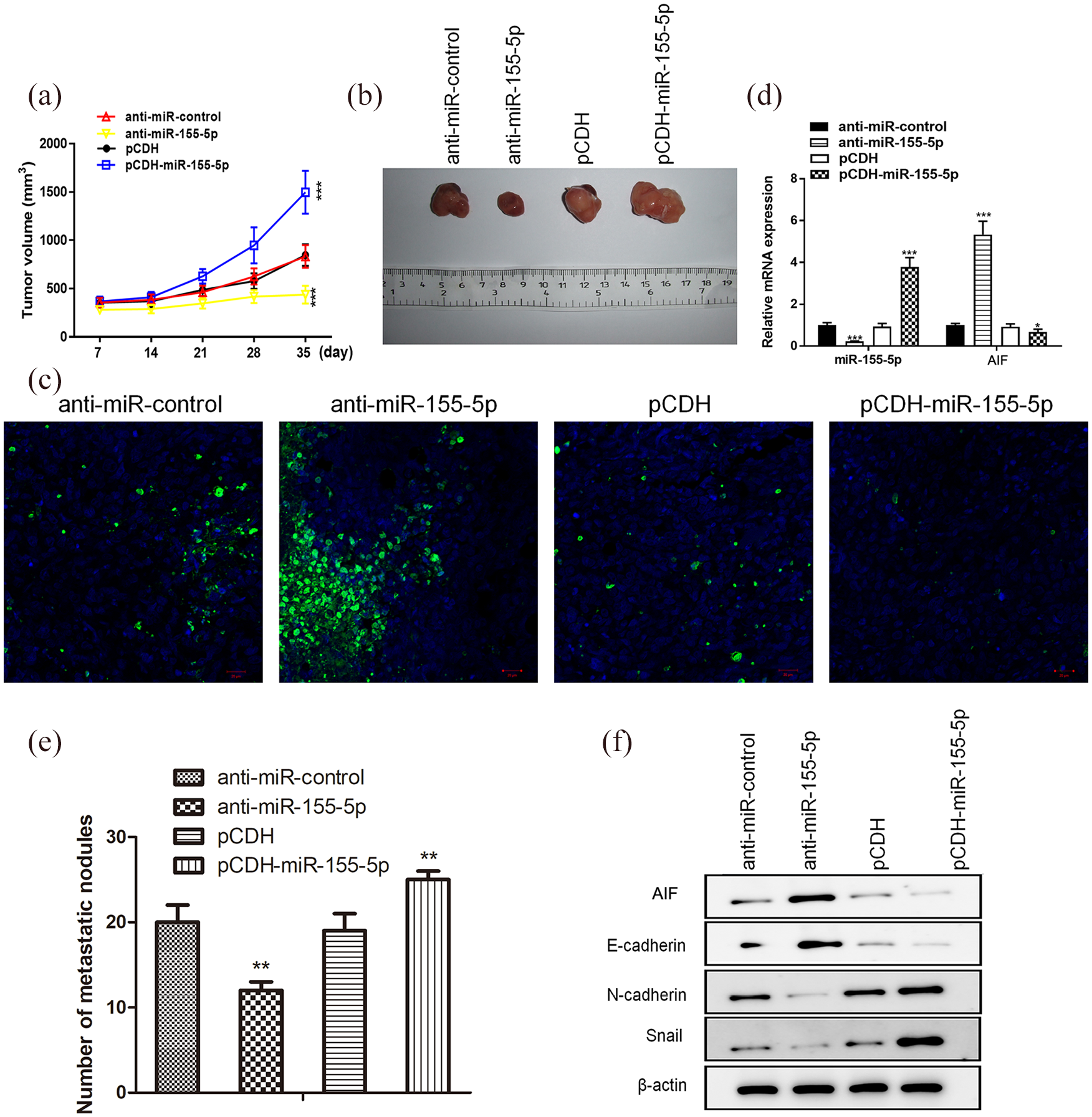
In vitro, miR-155-5p promoted RCC cell proliferation and aggressiveness. We further verified whether miR-155-5p could regulate the progression of RCC in vivo. RCC 786-O cells transfected with anti-miR-155-5p or pCDH-miR-155-5p were subcutaneously inoculated into nude mice. In vivo, the suppression of miR-155-5p significantly restrained tumor cell growth, whereas overexpression of miR-155-5p dramatically accelerated the growth of tumor cells (Figure 4(a) and (b)). Then, mice were sacrificed and tumor tissues were subjected to TUNEL staining assay. The level of TUNEL+ cells was remarkably increased in tumor tissues formed by anti-miR-155-5p transfected cells. Conversely, TUNEL+ cells were decreased in tumor tissues that originated from pCDH-miR-155-5p transfected cells (Figure 4(c)). In order to evaluate the metastatic potential of RCC, 786-O cells transfected with anti-miR-155-5p or pCDH-miR-155-5p were injected into nude mice via the tail vein. As expected, the number of lung metastasis nodules in mice injected with anti-miR-155-5p transfected cells were obviously decreased, whereas lung metastatic nodules were remarkably increased in mice injected with pCDH-miR-155-5p transfected cells (Figure 4(e)). Furthermore, qRT-PCR showed that interference of miR-155-5p significantly increased AIF levels, whereas overexpression of miR-155-5p remarkably suppressed AIF in vivo (Figure 4(d)). Furthermore, we evaluated the role of EMT in miR-155-5p-mediated metastasis in vivo. Western blot results showed that suppression of miR-155-5p increased E-cadherin, and decreased N-cadherin and Snail, whereas upregulation of miR-155-5p suppressed epithelial E-cadherin and enhanced levels of N-cadherin and Snail (Figure 4(f)). Taken together, these data validate that miR-155-5p could induce EMT progression and promote metastasis in vivo.
Discussion
RCC is one of the most aggressive malignancies, and its metastasis results in an unsatisfactory overall survival of patients. miRNAs play crucial roles in tumor cell invasion and metastasis, thus representing a potential therapeutic target for human cancers.13,14 In the present study, we identified that miR-155-5p functions as an oncogenic miRNA and accelerates tumor cell growth, migration, and invasiveness in RCC.
Previous studies have revealed that miR-155-5p was markedly overexpressed in various human malignancies, such as cervical cancer, melanoma and colon cancer.15-17 In contrast, decreased levels of miR-155-5p in gastric cancer have also been reported. 18 In the present study, we identified that miR-155-5p was significantly elevated in RCC tissues compared to the adjacent normal tissues. In addition, the expression of miR-155-5p was also increased in RCC cell lines compared to the normal human renal cell line HK-2. Metastasis is a complicated event orchestrated by loss of adhesion, detachment, migration, colonization, and growth. 19 Deregulated miRNAs play vital roles in the metastatic process of cancer cells. In colorectal and cervical carcinoma, overexpression of miR-155-5p has been confirmed to accelerate tumor cell growth and metastasis.20,21 Additionally, miRNA-155-5p facilitates the growth and invasion of oral squamous cell carcinoma by targeting the chromatin remodeling gene AT-rich interactive domain 2 (ARID2). 22 However, the effect of miRNA-155-5p on metastasis in renal cancer has not been well studied. Herein, we proved that miR-155-5p was upregulated in renal cancer tissues and cell lines. Strikingly, miR-155-5p promoted tumor cell growth, colony formation, and aggressiveness in vitro, whereas the suppression of miR-155-5p restrained such malignant behaviors. These findings identify that miR-155-5p may serve as an oncogenic miRNA in the progression and metastasis of renal cancer. Furthermore, the mice xenograft model revealed that downregulation of miR-155-5p restrained tumor growth and induced apoptosis, whereas overexpression of miR-155-5p promoted tumor growth and decreased apoptosis in renal cancer. In addition, the lung metastasis model revealed that miR-155-5p could promote tumor metastasis in vivo. In summary, these findings verified the crucial oncogenic functions of miR-155-5p in the progression of renal cancer.
One of the key molecular events in distant metastasis includes EMT, which confers great aggressiveness and is associated with poor prognosis in human cancers. 5 During EMT, the loss of epithelial cell–cell adhesion is mediated by decreased adhesion molecules, such as E-cadherin, and the gain of the highly motile mesenchymal phenotype is associated with N-cadherin and Snail. 6 Interestingly, our data showed that knockdown of miR-155-5p promoted E-cadherin, and suppressed levels of N-cadherin and Snail in renal cancer cells. By contrast, upregulation of miR-155-5p could promote EMT progression both in vitro and in vivo, suggesting that miR-155-5p promotes migration and invasion via inducing EMT progression in RCC.
It is well known that miRNAs regulate gene expression by binding to the 3ʹ-UTR of the target genes. 8 Currently, many targets of miR-155-5p have been identified in human cancers. For instance, miR-155-5p controls colon cancer cell migration via post-transcriptional regulation of human antigen R (HuR). 20 Additionally, miR-155-5p accelerates the metastasis of cervical cancer cell via targeting the tumor protein p53-inducible nuclear protein 1 (TP53INP1). 21 In this study, the target gene of miR-155-5p was identified using the bioinformatics analysis tools, and AIF was screened as the candidate target. AIF plays a critical role in caspase-independent cell apoptosis by triggering chromatin condensation and DNA fragmentation. 23 Moreover, accumulating evidence reveals that AIF is also involved in tumor initiation, metastasis, and recurrence. Critically, downregulation of AIF has been shown to mediate hypoxia-induced EMT progression in colon cancer. Our previous study has demonstrated that AIF was decreased in RCC tissues and was negatively associated with tumor node metastasis staging, serving as a poor factor in patients with this disease. In the current study, interference of miR-155-5p promoted AIF, and overexpression of miR-155-5p remarkably suppressed AIF levels. In addition, the luciferase reporter assay confirmed that miR-155-5p directly binds to 3′-UTR of AIF, verifying that AIF was the target of miR-155-5p. Consistently, in vivo assay revealed that the interference of miR-155-5p could promote the expression of AIF, whereas overexpression of miR-155-5p suppressed AIF levels in tumor tissues. Taken together, these data indicated that miR-155-5p enhances renal cancer progression through targeting AIF and inducing EMT.
In summary, our study demonstrates that miR-155-5p promotes the growth, invasion, and metastasis of renal cancer cells both in vitro and in vivo via targeting AIF. These findings might provide a novel therapeutic target combating the metastasis of renal cancer.
Footnotes
Acknowledgements
We thank all the patients who agreed to participate in this study.
Author contributions
Qing-Qing Lei and Cai Lv participated in the design of this study. Qing-Qing Lei, Yuan Huang, Bin Li, and Lu Han carried out the study and collected important background information. Qing-Qing Lei drafted the manuscript. All authors read and approved the final manuscript. Qing-Qing Lei and Cai Lv performed the statistical analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was supported by the Natural Science Foundation of Hainan Province (Grant No. 819MS136).
