Abstract
BACKGROUND:
Breast cancer has become a major threat to women’s physical and mental health. Many long noncoding RNAs (lncRNAs) have been reported to exert effect in the progression of breast cancer. However, the regulatory mechanism of HAND2-AS1 remains unclear. Therefore, this study aimed to elucidate the role of HAND2-AS1 in breast cancer.
METHODS:
The expression of HAND2-AS1, miR-1275 and SOX7 was assessed using RT-qPCR and Western blot analysis. CCK-8, Transwell and luciferase reporter assays were used to investigate their regulatory mechanisms.
RESULTS:
Downregulation of HAND2-AS1 was detected in breast cancer and was associated with adverse clinical features and prognosis. Furthermore, overexpression of HAND2-AS1 restrained cell viability, migration and invasion in breast cancer. In addition, reciprocal suppression between HAND2-AS1 and miR-1275 was identified in breast cancer cells. Further, SOX7, a target of miR-1275, strengthened the effect of HAND2-AS1 in breast cancer.
CONCLUSION:
LncRNA HAND2-AS1 sponging miR-1275 restrains proliferation and metastasis of breast cancer cells by regulating SOX7 expression.
Introduction
Breast cancer is a common malignant tumor that seriously affects women’s physical and mental health and even life-threatening. Moreover, its incidence is usually related to heredity [1], Surgery and radiotherapy alone can cure most breast cancer patients before tumor metastasis. Once metastasis occurs, active treatment can only cure a small number of patients [2]. In addition, the prognosis of breast cancer patients is mainly related to the extent of tumor invasion and pathological type. Patients with triple-negative breast cancer have the worst prognosis [3]. The 5-year survival rate of patients at stage I is 88%. However, the survival rate of patients at stage IV is only 15% [4]. Besides that, postoperative recurrence is another major threat to the health of breast cancer patients [5]. However, due to clinical and molecular heterogeneity, the molecular mechanism of breast cancer recurrence is unclear [6]. Now, a more accurate prediction of clinical outcomes and recurrence is a huge challenge for breast cancer treatment [7]. Therefore, it is still urgent to explore and develop effective breast cancer treatments.
Long non-coding RNAs (lncRNAs) are involved in the regulation of various biological processes in human diseases and cancers [8]. It has become the hottest research area for messenger RNA (mRNA) and microRNA post functional genomics. Moreover, many lncRNAs have been found to play important roles in the pathogenesis of breast cancer. Li et al. has found the carcinogenic potential and diagnostic significance of lncRNA LINC00310 in breast cancer [9]. LncRNA p10247 has been demonstrated to be high expressed in breast cancer and was correlated with metastasis [10]. Inversely, lncRNA CASC2 was found to be downregulated in breast cancer and restrained cell growth and metastasis by modulating the miR-96-5p/SYVN1 pathway [11]. Recently, the dysregulation of lncRNA HAND2-AS1 has been investigated in some malignant tumors other than breast cancer. You et al. found that HAND2-AS1 was downregulated in papillary thyroid cancer and may play a role in its progression [12]. Moreover, lncRNA HAND2-AS1 was found to repress HIF1
Precious studies have shown that miR-1275 was upregulated in head and neck squamous cell carcinoma and glioma, promoting cell migration, invasion and proliferation [15, 16]. In addition, lncRNA miR143HG was proposed to suppress the development of bladder cancer by modulating miR-1275/AXIN2 axis [17]. These studies indicate that the interaction between lncRNAs and miR-1275 is involved in tumorigenesis. Besides that, miR-1275 is involved in human cancers via regulating target genes, such as IGF2 [18]. In this study, SOX7 was selected as a target for miR-1275, which has not been confirmed in previous studies. It is well known that SOX7 can be used as an inhibitor of malignant tumors, such as hepatocellular carcinoma and lung adenocarcinoma [19, 20]. In particularly, SOX7 was downregulated and exerted tumor suppressive effect in breast cancer [21]. But the functional mechanism of lncRNA HAND2-AS1/miR-1275/SOX7 is still unknown in breast cancer. Therefore, this study aims to confirm their relationship and explain its regulatory mechanisms in breast cancer.
Materials and methods
Clinical tissues
Experimental tissues were obtained from 88 patients with breast cancer in Linyi Cancer Hospital. The clinicopathological features of these patients, including age, tumor size, lymph node metastasis, ER status, PR status and Her-2 status, are shown in Table 1. Informed consents were obtained from all breast cancer patients. Furthermore, breast cancer patients did not receive any treatment prior to surgery. This study was approved by the Institutional Ethics Committee of Linyi Cancer Hospital.
Relationship between HAND2-AS1 expression and clinic-patholo- gical characteristics in breast cancer patients
Relationship between HAND2-AS1 expression and clinic-patholo- gical characteristics in breast cancer patients
Statistical analyses were performed by the
Human breast epithelial cell line MCF10A and breast cancer cells MDA-MB-231, MDA-MB-468, MCF-7 and UACC812 were purchased from the American Type Culture Collection (ATCC, Manassas, VA, USA). The growth conditions for these cells were 5% CO
RNA isolation, reverse transcription and RT-qPCR
Total RNA isolation was performed using TRIZOL reagent (Invitrogen, USA). The cDNA solution was synthesized using PrimeScript RT reagent kit (Takara, Dalian, China). RT-qPCR assay was performing on ABI 7500 Sequence Detection system by Real-time PCR Mixture assays (Takara). The internal reference for HAND2-AS1, SOX7 or miR-1275 is GAPDH or U6. The expression level was quantified by the 2
CCK-8 assay
First, the transfected cells were incubated in 96-well plates for 24 hours (at 37
Colony formation
The transfected cells were digested by trypsin, and then collected and seeded into 6-well plates at a density of 2
Wound healing assays
A Wound-healing assay was performed to examine the migratory ability of the transfected cells. The cells were washed with serum free media and treated with mitomycinc (10
Transwell assay
First, cell invasion was detected in the upper chamber with Matrigel. After 30 min, transfected cells (3
Western blot analysis
Protein sample extraction was performed by RIPA lysis buffer (Beyotime). Next, 25
Dual luciferase reporter assay
Reporter plasmids of HAND2-AS1 (wt-HAND2-AS1 and mut-HAND2-AS1) and SOX7 (wt-SOX7 and mut-SOX7) were obtained from RiboBio (Guangzhou, China). Next, the reporter plasmids were transfected into MDA-MB-231 cells with miR-1275 mimics (RiboBio, Guangzhou, China). After 48 h, luciferase activity was assessed by the dual-luciferase reporter assay system (Promega, USA).
HAND2-AS1 was involved in pathogenesis of breast cancer. (A) The mRNA HAND2-AS1 expression in breast cancer tissues. (B) The expression of HAND2-AS1 at different breast cancer subtypes (luminal A, luminal B, normal-like and basal-like). (C) The mRNA HAND2-AS1 expression in breast cancer cell lines. (D, E) High HAND2-AS1 expression patients showed longer overall survival (OS) and (recurrence-free survival) RFS. *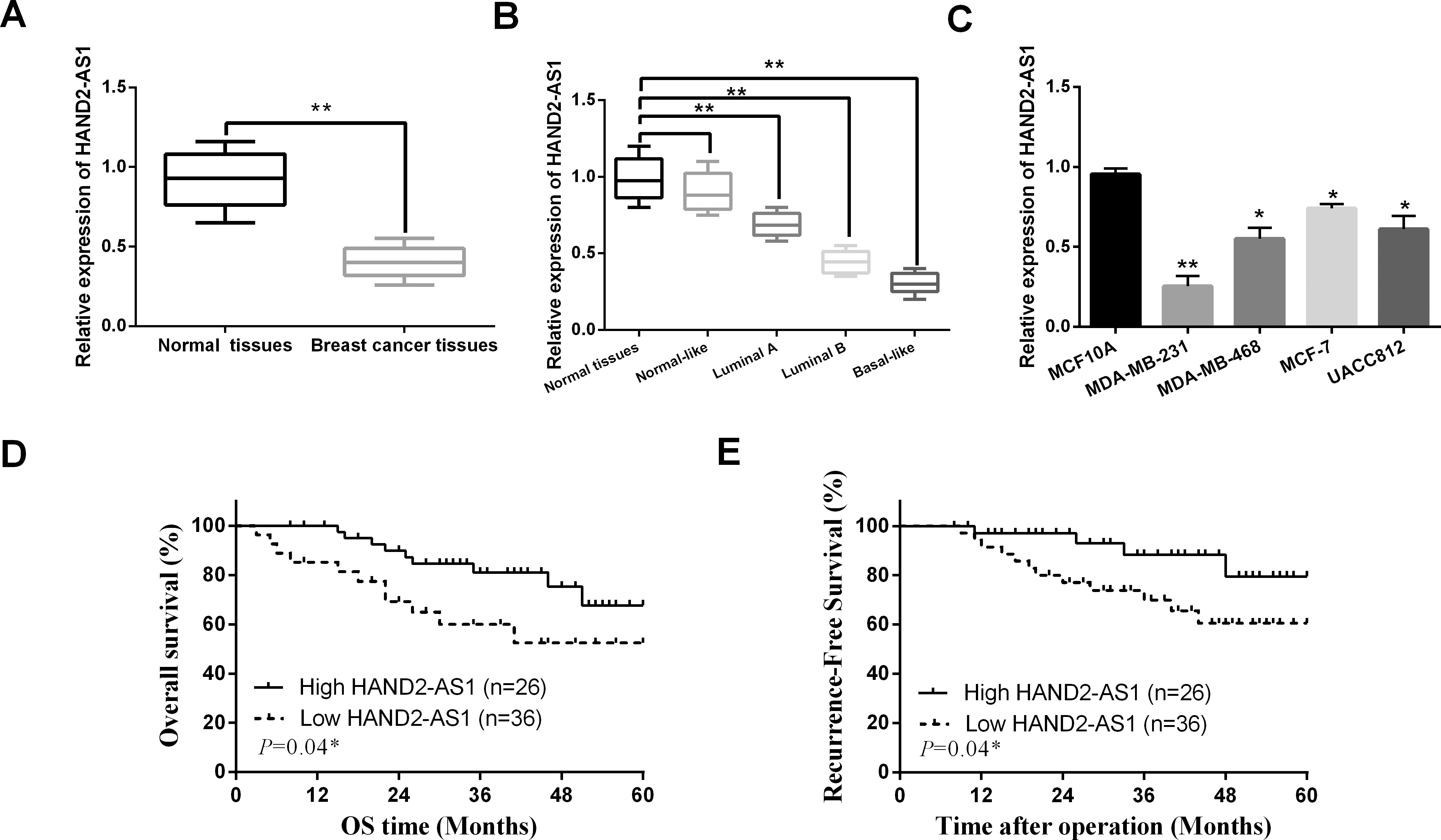
HAND2-AS1 restrained cell proliferation and metastasis in breast cancer. (A) HAND2-AS1 expression in MDA-MB-231 and MCF10A cells with its overexpression vector or siRNA. (B, C, D) Cell proliferation and colony formation in MDA-MB-231 and MCF10A cells with HAND2-AS1 overexpression vector or siRNA. (E, F) Cell migration in MDA-MB-231 and MCF10A cells with HAND2-AS1 overexpression vector or siRNA detected by Transwell and Wound healing assays. (G) Cell invasion in MDA-MB-231 and MCF10A cells with HAND2-AS1 overexpression vector or siRNA detected by Transwell assay. *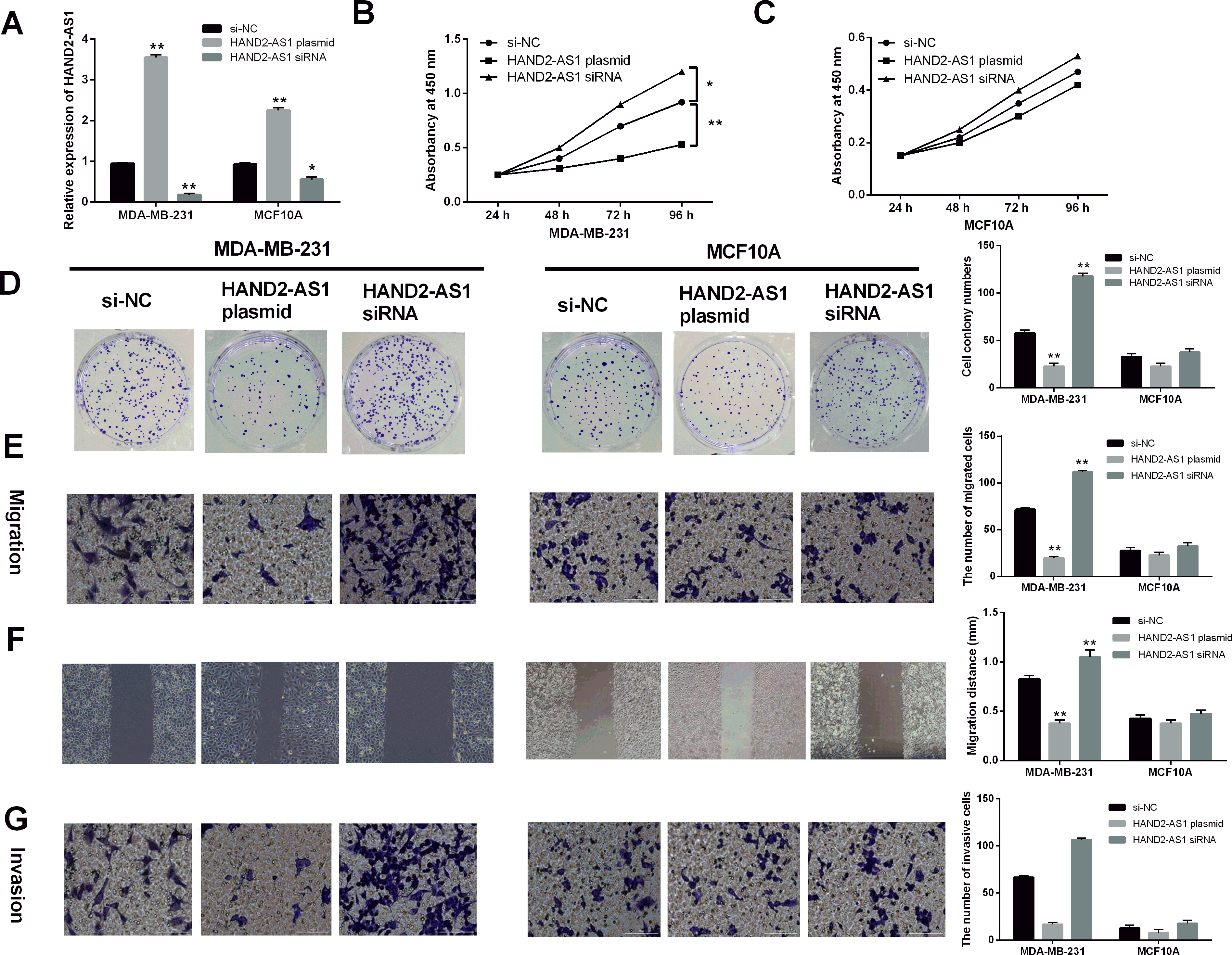
Reciprocal suppression between HAND2-AS1 and miR-1275 was identified in breast cancer. (A) The binding sites between HAND2-AS1 with miR-1275. (B) Luciferase reporter assay. (C) RNA pull-down experiment was performed to determine the binding between HAND2-AS1 and miR-1275. (D) MiR-1275 expression regulated by HAND2-AS1 overexpression vector or siRNA in MDA-MB-231 cells. (E) HAND2-AS1 expression regulated by miR-1275 mimic or inhibitor (30 nM). (F) A negative correlation between HAND2-AS1 and miR-1275 was identified in breast cancer tissues. (G) MiR-1275 expression in MDA-MB-231 cells with its mimics or inhibitor (30 nM). (H, I, J) Cell proliferation, migration and invasion in MDA-MB-231 cells with miR-1275 mimics or inhibitor. *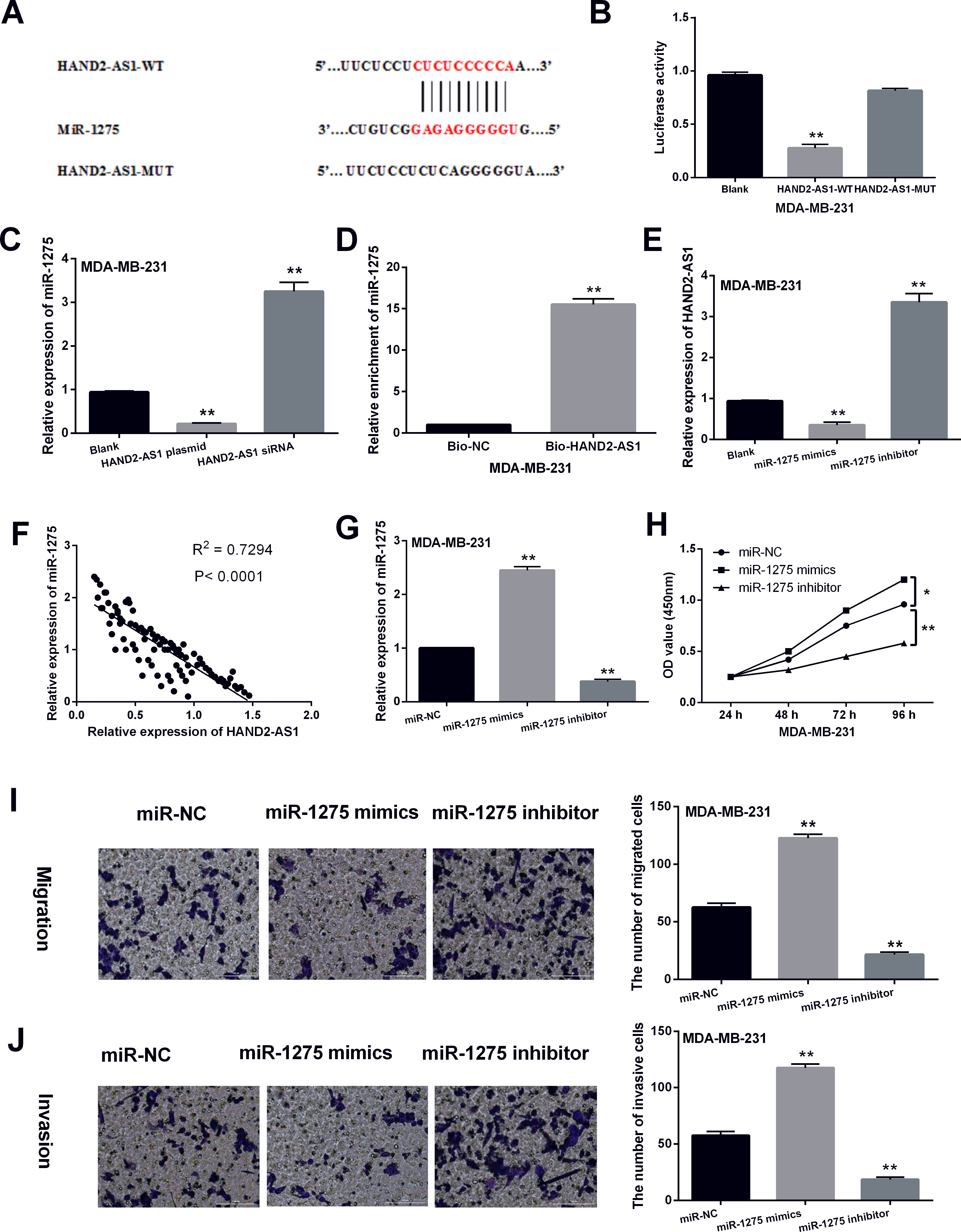
SOX7 targeted by miR-1275 regulated the effect of HAND2-AS1 in breast cancer. (A) The binding sites between SOX7 with miR-1275. (B) The mRNA SOX7 expression in breast cancer tissues. (C) SOX7 expression regulated by miR-1275 mimic or inhibitor. (D) Luciferase activity assay. (E, F) SOX7 expression regulated by different transfections in MDA-MB-231 and MCF10A cells. (G) SOX7 expression regulated by HAND2-AS1. (H, I) Cell proliferation, migration and invasion in MDA-MB-231 cells with different vectors. *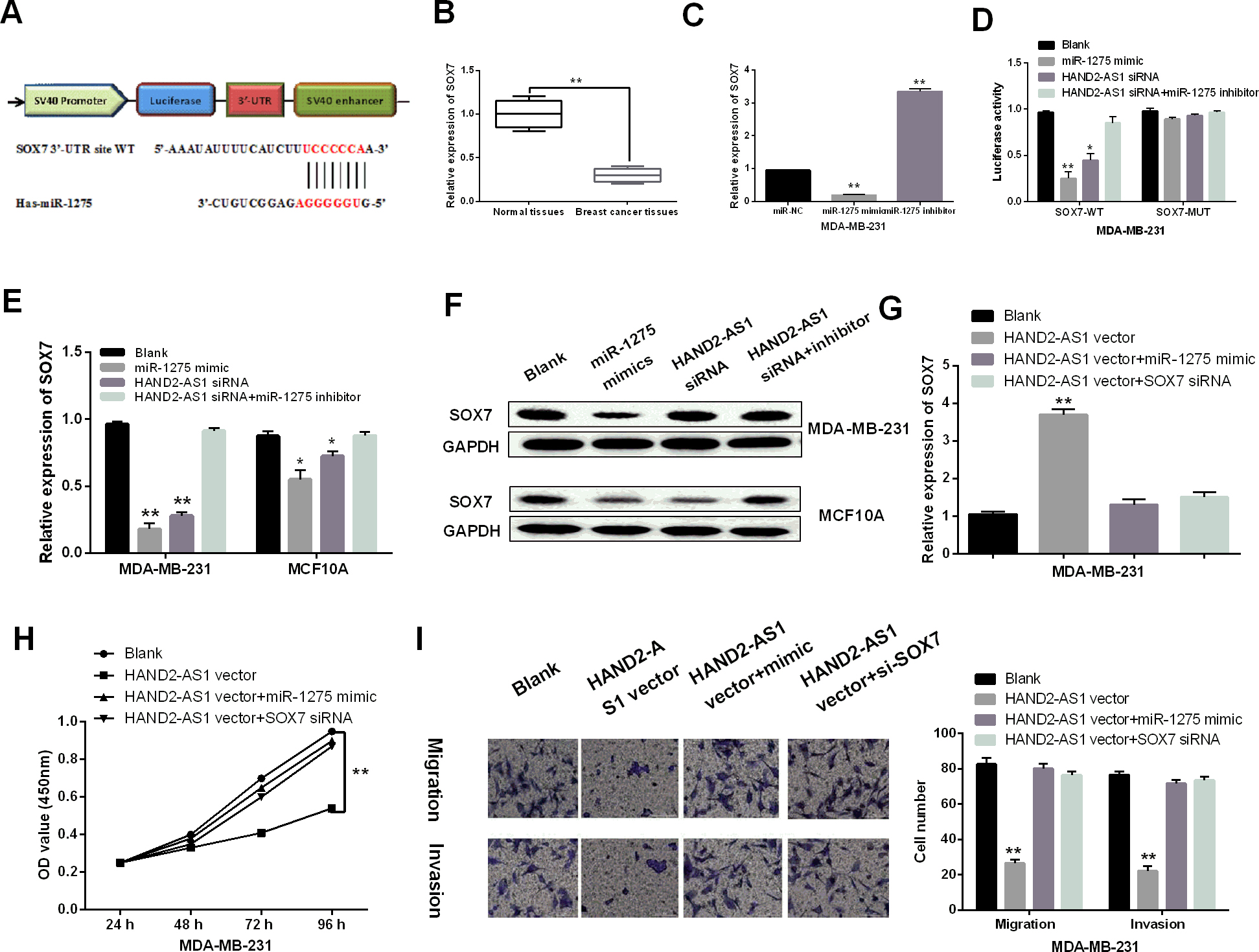
Biotinylated HAND2-AS1 was transfected into MDA-MB-231 cells. A biotinylated lncRNA not complementary to miR-1275 was employed as a negative control. After 48 hours, the cell lysate was incubated with M-280 streptavidin magnetic beads (Invitrogen), followed by RT-qPCR assay to measure miR-1275 level.
Statistical analysis
Data were analyzed using Student’s t-test, one-way ANOVA, Univariate Kaplan-Meier method with log-rank test and Chi-squared test in SPSS 17.0 or Graphpad Prism 6 software. Data are shown as mean
Results
HAND2-AS1 was involved in pathogenesis of breast cancer
First, we detected abnormal expression of HAND2-AS1 in breast cancer. We found that HAND2-AS1 was downregulated in breast cancer tissues compared to normal tissues (
HAND2-AS1 restrained cell proliferation and metastasis in breast cancer
To explore the functional role of HAND2-AS1 in breast cancer, gain-loss functional experiments were performed in MDA-MB-231 and MCF10A cells with HAND2-AS1 plasmid or siRNA. RT-qPCR was used to assess transfection efficiency. It showed that HAND2-AS1 plasmid significantly increased its expression, while HAND2-AS1 siRNA reduced its expression in MDA-MB-231 and MCF10A cells (
Reciprocal suppression between HAND2-AS1 and miR-1275 was identified in breast cancer
Next, the downstream target of HAND2-AS1 was searched in the starBase database (
SOX7 targeted by miR-1275 regulated the effect of HAND2-AS1 in breast cancer
Finally, the target gene of miR-1275 was searched in the TargetScan database (
Discussion
Recently, many lncRNAs have been found to serve as tumor promoters or inhibitors in the progression of breast cancer. For example, lncRNA CASC2 was downregulated in breast cancer, and upregulation of CASC2 restrained cell proliferation and metasta- sis [22]. Here, downregulation of HAND2-AS1 was also found in breast cancer, which has not been demonstrated in previous studies. In addition, this downregulation was associated with poor clinical features and prognosis in breast cancer patients. Functionally, HAND2-AS1 overexpression restrained cell viability, migration and invasion in breast cancer. Further, HAND2-AS1 sponging miR-1275 was found to impede breast cancer progression by regulating SOX7 expression. All of these findings imply that HAND2-AS1 is involved in the development of breast cancer by acting as an inhibitor.
Consistent with our results, HAND2-AS1 has been identified to be downregulated in hepatocellular carcinoma and HPV-negative head and neck squamous cell carcinoma [23, 24]. Huang et al. proposed that downregulation of HAND2-AS1 predicted a poor prognosis in patients with colon adenocarcinoma [25]. In our study, poor prognosis in breast cancer patients was also identified to be associated with low HAND2-AS1 expression. Functionally, lncRNA HAND2-AS1 has been demonstrate to repress invasion and migration of non-small cell lung cancer cells [26]. The same effect of HAND2-AS1 was also found in breast cancer. Additionally, Yan et al. showed that HAND2-AS1 restrained cell proliferation in esophagus squamous cell carcinoma by regulating microRNA-21 [27]. Here, it was observed that HAND2-AS1 restrained breast cancer cell proliferation by sponging miR-1275. Similarly, Zhou et al. revealed that HAND2-AS1 sponging miR-1275 hindered the progression of colorectal cancer [28]. Besides that, reciprocal suppression between HAND2-AS1 and miR-1275 was identified in breast cancer cells.
More and more studies have shown that miR-1275 is involved in human cancers. For instance, miR-1275 was downregulated and repressed the growth of nasopharyngeal carcinoma [29]. However, upregulation of miR-1275 was detected in glioma, which promoted the progression of glioma [16]. These findings suggest that the dysregulation of miR-1275 has tissue specificity in human cancers. Moreover, the function of miR-1275 is still dim in breast cancer. Here, we found that overexpression of miR-1275 inhibited the progression of breast cancer. Furthermore, our rescue experiments indicated that upregulation of miR-1275 weakened the inhibitory effect of HAND2-AS1 in breast cancer. All of these results reflect that miR-1275 serves as a carcinogenic miRNA in breast cancer progression.
Further, downstream target of miR-1275 was explored in this study. SOX7 was identified as a direct target of miR-1275, which has not been investigated in other studies. We found that SOX7 expression was downregulated by miR-1275 and upregulated by HAND2-AS1 in breast cancer cells. Downregulation of SOX7 and its inhibitory effect have been extensively investigated in human cancers, such as lung cancer and colorectal cancer [30, 31]. Here, SOX7 was found to be downregulated and exert tumor suppressive effects in breast cancer [21]. In addition, knockdown of SOX7 was found to restore the inhibitory effect of HAND2-AS1 in breast cancer. It also suggests that SOX7 serves as a suppressive regulator in breast cancer, consistent with previous studies. Moreover, this is the first report that SOX7 involves in breast cancer progression through interacting with the HAND2-AS1/miR-1275 axis. However, we have only initially elucidated the regulatory mechanism of the HAND2-AS1/miR-1275/SOX7 axis in the pathogenesis of breast cancer. The specific functional mechanisms of HAND2-AS1/miR-1275/SOX7 axis and their roles in vivo have not been investigated in this study. Another drawback is that we currently have a small number of clinical samples. In the future, we will increase the number of samples and further explore the function of HAND2-AS1/miR-1275/SOX7 axis in vivo.
Conclusion
In conclusion, this study first proposed the downregulation of HAND2-AS1 in breast cancer. Furthermore, downregulation of HAND2-AS1 was associated with adverse clinical features and prognosis in breast cancer patients. Importantly, HAND2-AS1 restrained breast cancer progression and acted as a competing endogenous RNA to regulate SOX7 expression by sponging miR-1275.
Footnotes
Conflict of interest
The authors declare that they have no competing interests.
