Abstract
Recent studies have shown that long noncoding RNAs (lncRNAs) have profound impacts on cancer development. In our previous study, we have confirmed that lncRNA small nucleolar RNA host gene 16 (SNHG16) is associated with poor prognosis and malignant phenotype of gastric cancer (GC). However, the biological function of lncRNA SNHG16 is still unclear. Here, we aimed to investigate the mechanisms underlying the roles of SNHG16 in GC. In this work, SNHG16 knockdown could inhibit epithelial-mesenchymal transition (EMT) and invasion of GC cells. Moreover, our results revealed that SNHG16 could promote EMT via down-regulation of Dickkopf WNT signaling pathway inhibitor 3 (DKK3) in GC cells. In addition, SNHG16 was found to be upregulated whereas DKK3 was downregulated in tumor tissues compared with adjacent normal tissues. It showed that the expressions of SNHG16 and DKK3 were negatively correlated in clinical GC tissues.
All these results suggested that SNHG16 promotes GC progression via regulation of DKK3 directly or indirectly. SNHG16 might be used as a putative biomarker for metastatic prediction in GC patients.
Introduction
Gastric cancer (GC) is one of the common malignant tumors in the world. According to the report, the new cases of GC have increased to 410,000 in 2014 in China, accounting for 40% of all the GC cases around the world [1]. Curative treatment of GC generally depends on surgical resection. Unfortunately, the overall effect is poor due to recurrence and metastasis. Therefore, it is urgent to explore new clinical biomarkers for targeted treatment for GC patients. Currently, epithelial mesenchymal transition (EMT) is a hot topic associated with the metastasis of carcinomas. Recent reports have revealed that activation of EMT leads to invasion and metastasis in multiple cancers [2, 3]. A considerable amount of research showed that lncRNAs with the length of more than 200 nucleotides play a key role in the development of tumors including involvement in EMT [4, 5]. LncRNA SNHG16, the nuclear small RNA host gene 16, which is located on human chromosome 17q25.1, was firstly found in neuroblastoma [6]. Our previous work has clarified that lncRNA SNHG16 was highly expressed in GC cell lines HGC-27 and AGS, and participated in the invasion and metastasis of GC [7]. However, the function of SNHG16 in the GC metastasis remains uncharacterized and even less is known about the potential mechanisms responsible for SNHG16-mediated EMT.
In addition, we surprisingly found through GeneChip analysis that SNHG16 knockdown could upregulate the level of Dickkopf WNT signaling pathway inhibitor 3 (DKK3) in HGC-27 cells. DKK3, an antagonist of Wnt ligand activity, is a member of the human DKK family encoding secreted proteins, which comprises four members DKK1-4 and a unique DKK3-related protein termed Soggy [8]. Recently, more evidence has emerged that DKK3 is low expressed in many cancers and the overexpression of DKK3 in tumor cells could partially reverse EMT [9, 10, 11]. DKK3 might be considered as a potential target in cancer therapy [12].
In this study, we discovered that SNHG16 could promote EMT, and part of the mechanism was through down-regulation of DKK3 in GC cells. In addition, we also found that SNHG16 was negatively correlated with DKK3 in clinical GC tissues. In summary, our results suggested that SNHG16 may be involved in GC progression and might be used as a putative biomarker for metastatic prediction in GC patients.
Materials and methods
Tissue collection and storage condition
A total of 20 GC tissues and their paired adjacent tissues (located
Cell lines and culture
Human GC cell lines HGC-27 and AGS were purchased from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China). All cells were cultured in a 1640 medium (Gibco, USA) supplemented with 10% fetal bovine serum (Lonsa Science SRL, Lonsera) and 1% penicillin/streptomycin (Invitrogen, Shanghai, China). The cells were incubated at 37
Total RNA extraction and qRT-PCR assay
Total RNA was extracted from tissues or cells using Trizol reagent (Sangon, Shanghai, China). The RNA samples were reverse transcribed to cDNA using a Reverse Transcription Kit (Takara, Dalian, China). The cDNA was measured by SYBR Premix Ex Taq
Small interfering RNA (siRNA) transfection and lentivirus vector construction
The siRNAs specifically targeting SNHG16 and DKK3 were synthesized and purchased from GenePharma (Shanghai, China). The siRNA sequence of SNHG16 and DKK3 were listed in Supplementary Table 2. The GC cell lines HGC-27 and AGS were transfected with the si-SNHG16 or scrambled control si-NC using Lipofectamine
SNHG16 knockdown attenuated EMT in GC HGC-27 and AGS cells. A: The SNHG16 expressions in HGC-27 and AGS cells were detected by qRT-PCR assay after si-SNHG16 or si-NC transfection. B–D: The EMT-associated proteins in HGC-27 and AGS cells were detected by western blot assay after si-SNHG16 or si-NC transfection. 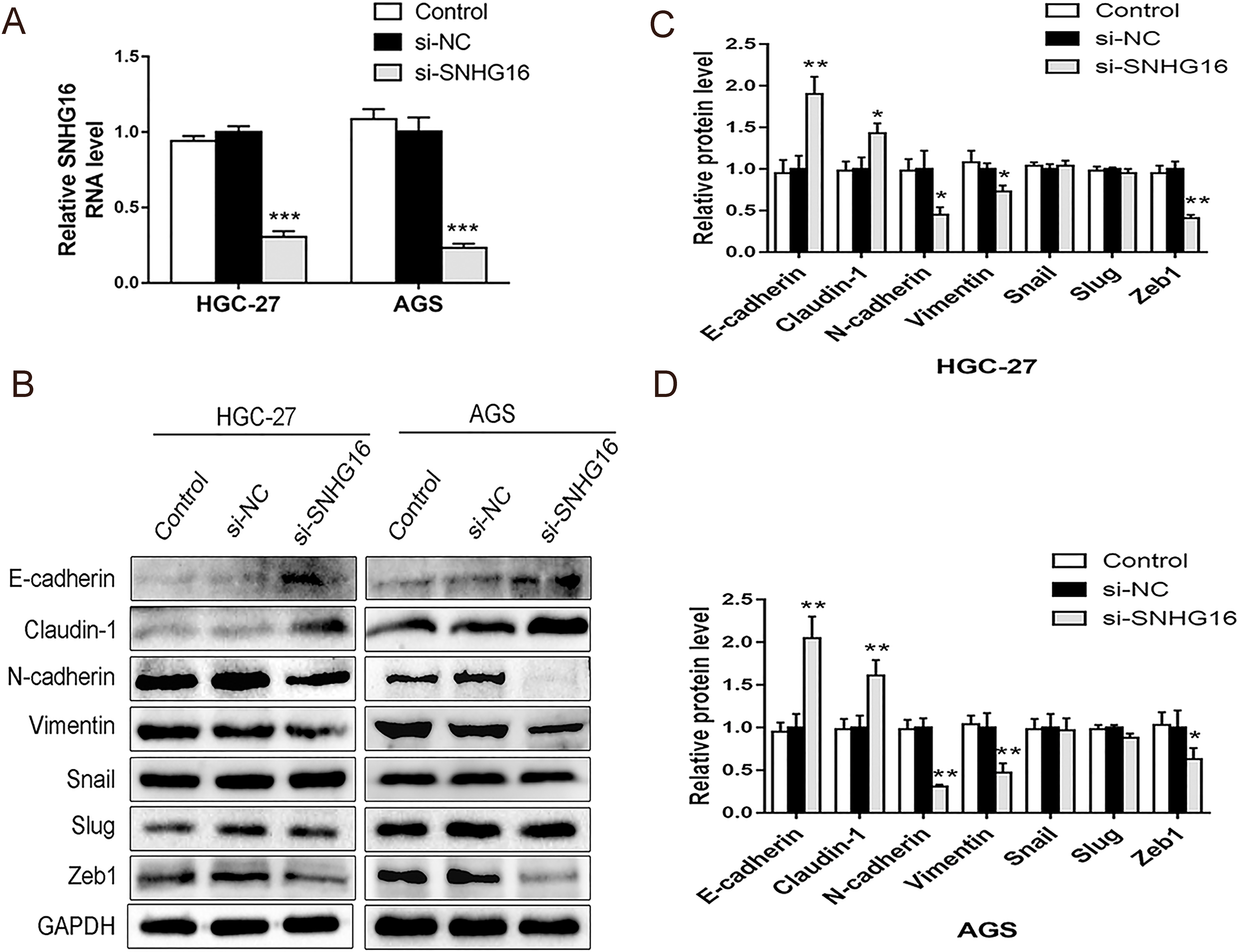
SNHG16 knockdown inhibited migration and invasion of HGC-27 and AGS cells. A–D: The role of SNHG16 on migration and invasion was evaluated by transwell assays. 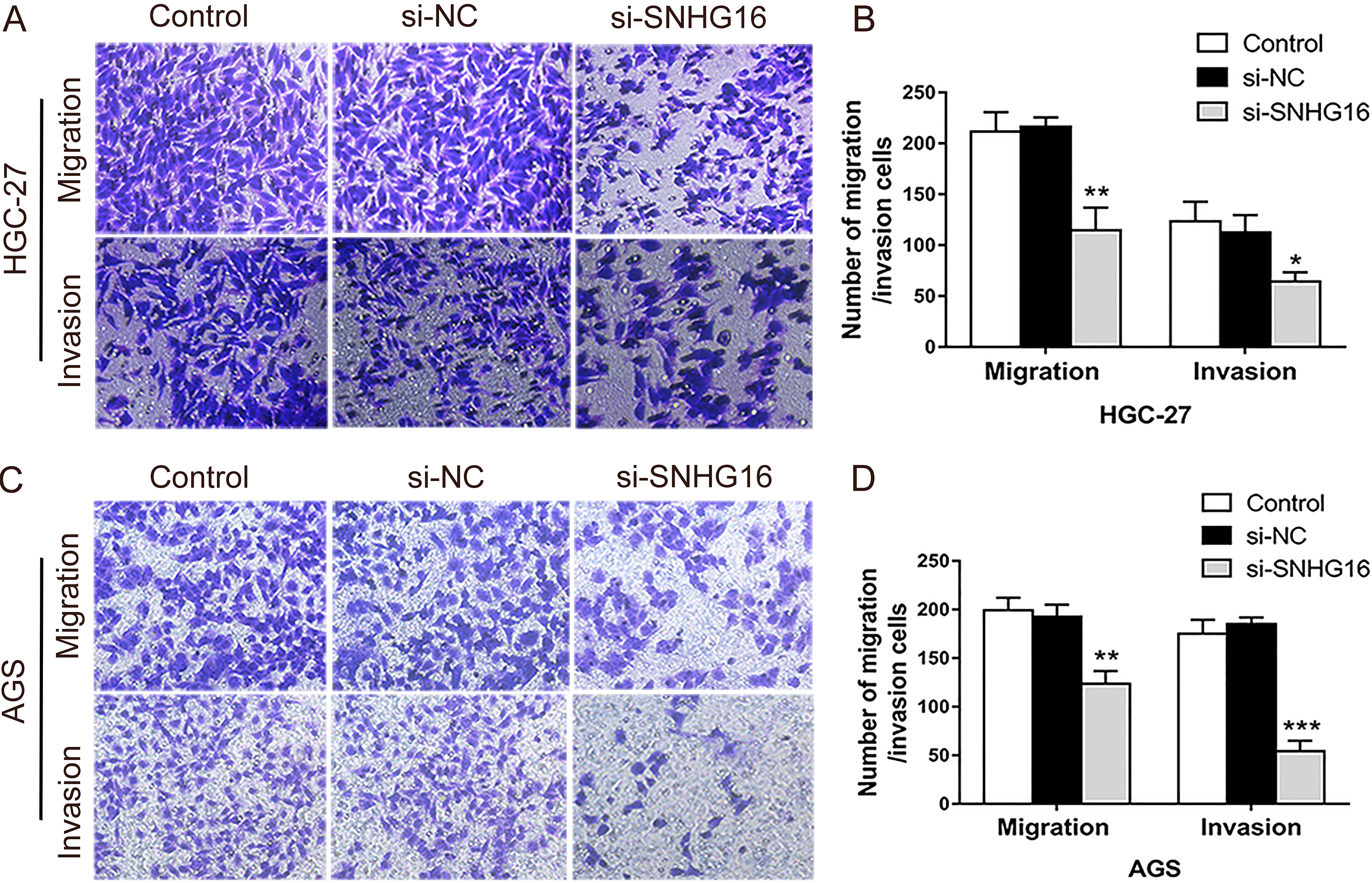
SNHG16 knockdown increased the level of DKK3 protein. A: Differential gene expressions were displayed as a heatmap through Genechip analysis after SNHG16 knockdown in HGC-27 cells. Downregulated genes are shown in green and upregulated genes in red. B: The expression of DKK3 mRNA was evaluated by qRT-PCR after transfected with si-SNHG16 or si-NC into HGC-27 and AGS cells. C and D: The level of DKK3 protein was detected by western blot assay after transfected with si-SNHG16 or si-NC into HGC-27 and AGS cells. E and F: The expression of SNHG16 and DKK3 at RNA level were detected by qRT-PCR assay in GC tissues and the adjacent tissues (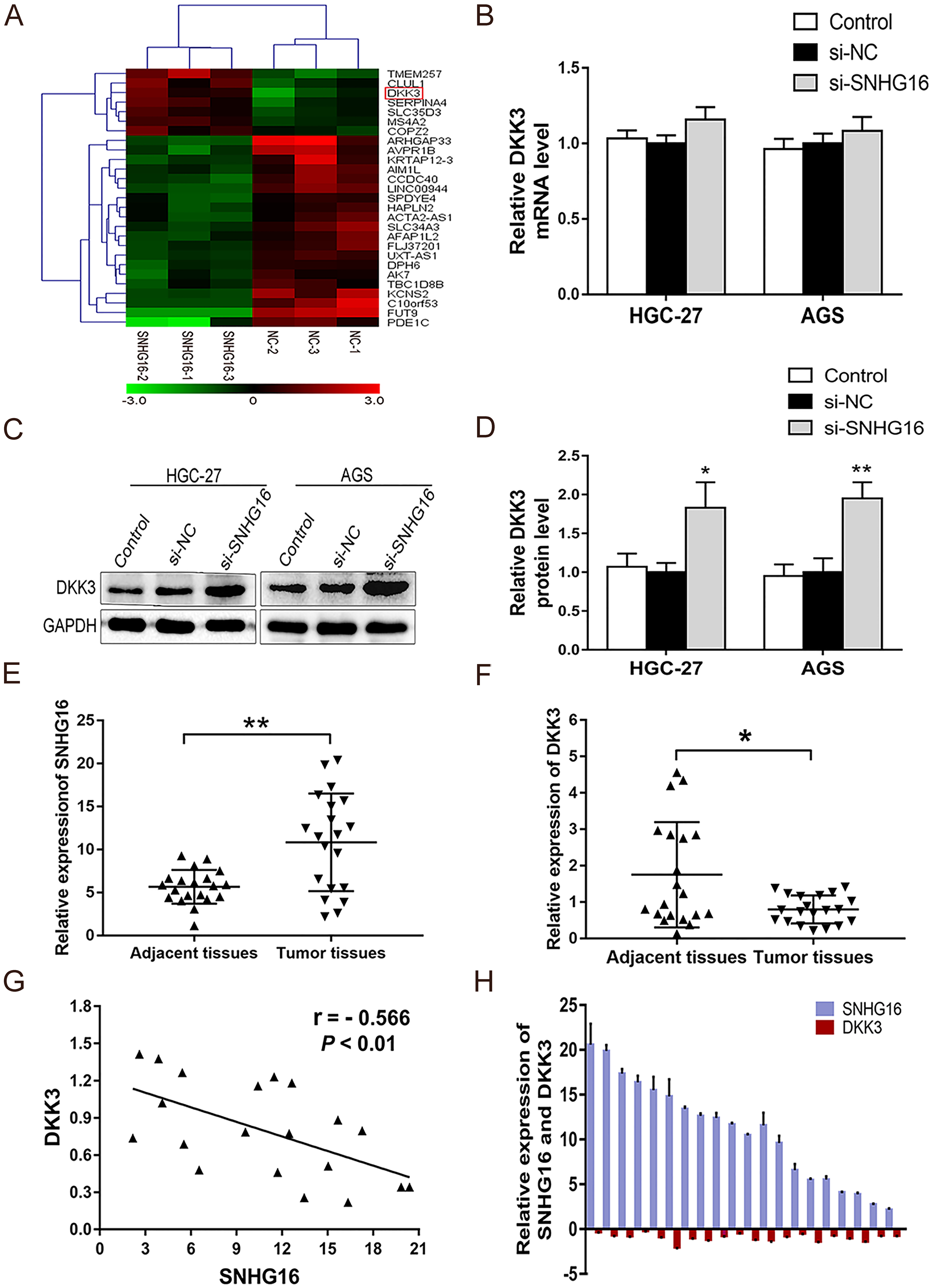
For migration and invasion assay, transwell experiment was performed with chambers with 8
Western blot assay
Cell lysates were mixed with 1
Statistical analysis
The cell data was presented as 2
Results
SNHG16 knockdown attenuated EMT in HGC-27 and AGS cells
It is well known that EMT is involved in metastatic events in tumors. To evaluate the role of SNHG16 on EMT in GC, si-SNHG16 or si-NC were transfected into HGC-27 and AGS cells. The interference efficiency of SNHG16 was over 70%, as detected by qRT-PCR assay (Fig. 1A). Next, we detected EMT-associated proteins using Western blot assay. At protein (Fig. 1B–D) levels, SNHG16 knockdown upregulated the expressions of epithelial markers E-cadherin and Claudin-1, reduced the expressions of mesenchymal markers, such as N-cadherin, Vimentin. In addition, it could reduce the level of transcription factors Zeb1 protein, but there were no obvious differences in Slug and Snail proteins.
SNHG16 knockdown inhibited migration and invasion of HGC-27 and AGS cells
As known, the metastasis is the key factor leading to death in many tumors, and EMT is the initial stage of tumor metastasis. Consistent with the alterations in the EMT-associated markers, the results of transwell assays indicated that lack of SNHG16 inhibited the migration and invasion of HGC-27 (Fig. 2A and B) and AGS cells (Fig. 2C and D).
Knockdown of SNHG16 upregulated the DKK3 protein expression
To further explore the target of SNHG16 in GC, Genechip assay was performed following SNHG16 knockdown (Fig. 3A), which displayed 7 upregulated genes and 21 downregulated genes in HGC-27 cells. Among them, a remarkable 3.26-fold increase of DKK3 gene expression was detected. qRT-PCR and western blot assays were performed to verify the authenticity of Genechip results. Unexpectedly, the transcription level of DKK3 was only slightly increased by SNHG16 knockdown in both HGC-27 and AGS cells, as detected by qRT-PCR assay (Fig. 3B), but the DKK3 protein abundance was significantly increased (Fig. 3C and D). The discrepancy may be caused by the methodological differences between Genechip and qRT-PCR or the involvement of other regulatory mechanisms. To explore the correlation between SNHG16 and DKK3 clinically, the RNA expressions of SNHG16 and DKK3 in 20 pairs of GC tissues and their adjacent normal tissues were examined by qRT-PCR assay. It indicated an inverse correlation between the enhanced SNHG16 (Fig. 3E) and the declined DKK3 expressions (Fig. 3F), with
SNHG16 induced cell EMT through DKK3-dependent mechanism, and SNHG16 regulated
-catenin protein expression
To verify whether SNHG16 was engaged in EMT via the regulation of DKK3, a rescue experiment was performed to explore the correlation between SNHG16 and DKK3 in EMT. HGC-27 and AGS cells were transfected with sh-SNHG16 to gain stable cell lines by lentivirus-mediated SNHG16 knockdown. An interference efficiency above 65% was achieved in HGC-27 and AGS stable cells (Fig. 4A). The DKK3-transcript levels, as detected by qRT-PCR, were not obvious changed by SNHG16 knockdown in HGC-27 and AGS stable cells (Fig. 4B). Meanwhile, DKK3 gene was silenced by RNAi technology. Shown by qRT-PCR and western blot results, the si3-DKK3 was the most optimal among the three siRNAs and was chosen for the next experiment (Fig. 4C and D). The SNHG16 knockdown-induced inhibition of EMT was reversed by co-transfection of sh-SNHG16 and si3-DKK3 into HGC-27 and AGS cells (Fig. 4E), implying that SNHG16 promoted EMT via down-regulation of DKK3 protein in GC cells.
SNHG16 induced cell EMT through a DKK3-dependent mechanism. A: qRT-PCR assay was used to verify the effect of lentivirus-mediated SNHG16 knockdown in HGC-27 or AGS stable cells. B: The change of DKK3 expression was detected by qRT-PCR assay after lentivirus-mediated SNHG16 knockdown in HGC-27 and AGS cells. C and D: The effects of different si-DKK3 were detected in HGC-27 or AGS cells by qRT-PCR assay or western blot assay. E: Downregulation of DKK3 reversed the inhibition of EMT by SNHG16 knockdown in GC cells, as assessed by western blot assay. F: The levels of 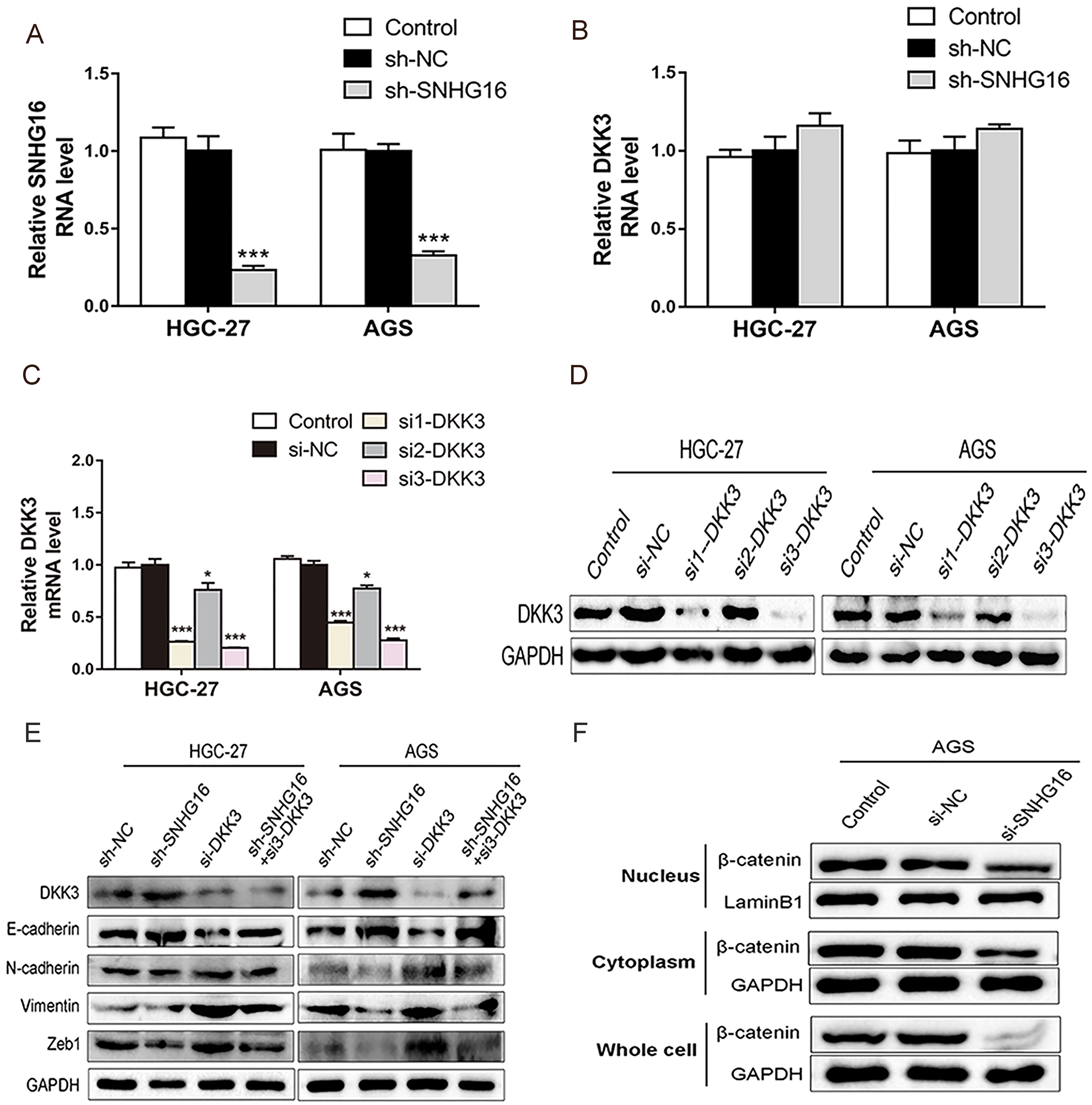
Next, whether SNHG16 was associated with translocation of
Increasing evidence indicated that lncRNA was involved in the different aspects of GC progression, including proliferation, metastasis and apoptosis of tumor cells [13, 14]. In our previous study, we have shown that the elevation of lncRNA SNHG16 expression was associated with GC invasion and metastasis [7]. It has been reported that SNHG16 participated in the process of invasion and metastasis in colorectal cancer, hepatocellular carcinoma (HCC), and breast cancer [15, 16, 17].
Recently, it was reported that EMT plays an important role in the initial stage of cancer metastasis which refers to the evolution of epithelial cells transdifferentiated into motile mesenchymal cells [18, 19]. EMT is accompanied by the downregulation of epithelial cell junction proteins, such as E-cadherin, Claudin-1 and the upregulation of mesenchymal markers such as N-cadherin, Vimentin. Meanwhile, EMT-inducing transcription factors, including Twist, Snail, Slug and Zeb1 proteins, also contribute to EMT [20]. Our results in the present work implied that SNHG16 knockdown could attenuate EMT by inhibition of Zeb1 protein which was the key transcription factor driving EMT. Besides, SNHG16 knockdown reduced cell migration and invasion in GC cells. Previous studies also confirmed that SNHG16 could induce EMT and invasion via Zeb1 protein in several cancers [21, 22, 23]. These results provide evidence that SNHG16 is involved in the EMT and invasion, suggesting SNHG16 may act as an oncogene in tumorigenesis.
DKK3, a secreted Wnt antagonist, belongs to DKK family but its function is different from other members [9, 10]. The promoter methylation of DKK3 is a common event leading to repressed DKK3 expression in GC [24, 25]. In the current work, we found an inversed relationship between the expressions of DKK3 and SNHG16 in GC tissues. These results indicated that SNHG16 may play an important role in the evolution by inhibition of DKK3 level in GC. Furthermore, we discovered that SNHG16 could promote EMT, and part of the mechanism was through down-regulation of DKK3 in GC cells. Several studies have suggested that DKK3 overexpression could inhibit EMT and invasion of cancer, including GC [9, 26, 27]. Therefore, we speculated that SNHG16 induces EMT by downregulation of DKK3 in GC cells.
DKK1, DKK2 and DKK4 could interact with LRP6/Kremen protein to destroy the Wnt complex, leading to blocked Wnt signaling and inhibition of Wnt pathway activation [28]. However, the function of DKK3 was unique in its family as DKK3 does not interact with LRP6/Kremen proteins. Its exact mechanism is still unclear. Yet, considerable studies have reported that DKK3 may act as tumor suppressor against Wnt signaling pathway [29, 30].
In conclusion, our findings revealed that there was a negative correlation between SNHG16 and DKK3 expressions in some extent in GC tissues. And SNHG16 could promote EMT via regulation of DKK3 in GC cells. Despite the limitations in our experiments, this study provides evidence for the role of SNHG16 in GC metastasis.
Footnotes
Acknowledgments
This study was supported by research grants from the National Natural Science Foundation of China to H. Huang (Grant No. 81760429) and Guizhou Provincial Innovative Talents Team to H. Huang (Grant No. 2019-5610). And we really appreciate the assistance of Jin Weilin professor in Shanghai Jiao Tong University.
Conflict of interest
No potential conflicts of interest were disclosed.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-190497.
