Abstract
Recently, Arsenic trioxide (ATO) has been reported as an efficient drug for suppression of cancer cell growth. Existing studies revealed the extensive involvement of microRNAs (miRNAs) in initiation and development of hepatocellular carcinoma (HCC). However, the potential correlation between ATO and miRNAs in HCC progression remains to be explored. To conduct our research, we applied a qRT-PCR analysis to find miRNAs that were upregulated in HCC cells treated with ATO. In our present study, miR-1294 was found to be significantly upregulated in ATO-treated HCC cells. To confirm the function of ATO and miR-1294 in HCC progression, gain-of function assays were designed and conducted. As expected, proliferative ability of ATO-treated HCC cells was markedly weakened compared to DMSO-treated HCC cells. More importantly, proliferation was further suppressed in ATO-induced HCC cells after overexpression of miR-1294. Through bioinformatics analysis, some potential targets of miR-1294 were predicted. Further investigation revealed that Pim-1 proto-oncogene (PIM1) and TEA domain transcription factor 1 (TEAD1) were two downstream targets of miR-1294 and could be negatively regulated by ATO. Functionally, we determined that cell proliferation and apoptosis resistance suppressed by miR-1294 and ATO were recovered by introduction of TEAD1 and PIM1. Collectively, this study revealed that a novel ATO-miR-1294-TEAD1/PIM1 axis regulated HCC cell growth, offering a potential insight into the HCC therapy.
Introduction
Hepatocellular carcinoma (HCC) is acknowledged as the 3rd primary cause of cancer-induced death, ranking the 5th among the most prevalent malignancies worldwide [1, 2, 3]. According to some statistics, HCC caused over six hundred thousand deaths annually [4, 5]. In spite of the development of personalized cancer treatment protocol, the overall prognosis of HCC patients has not been improved. For this reason, more valuable diagnostic and therapeutic biomarkers for HCC remain to be explored.
Arsenic trioxide (ATO) is known as the most toxic compound in Chinese medicine [6], which has been demonstrated to be effective in the suppression of cancer cellular processes [7, 8, 9, 10]. Recent studies have elucidated that ATO could induce the differential expression of miRNAs in tumor progression [11, 12]. However, it is unclear whether there is close combination between ATO and miRNAs. This notion prompted us to further elucidate the association between ATO and miRNA expression, and function of miRNAs in HCC cellular processes.
As a type of small endogenous non-coding RNA, microRNAs (miRNAs) could bind to 3’-untranslated regions (UTRs) of target genes, thereby post-transcrip- tionally modulating mRNAs expression [13]. Mounting evidences uncovered that extensive miRNAs played indispensable regulatory roles in numerous cancerous biological processes, such as cell proliferation, apoptosis and migration [14, 15, 16, 17]. Additionally, the diagnostic and prognostic values of miRNAs were previously studied in human tumors [18, 19]. Therefore, investigation of functional miRNAs in HCC progression may be meaningful.
In the present study, we focused on investigating ATO-regulated miRNAs in HCC. At first, miR-1294 was found to be significantly upregulated in HCC cells after treatment of ATO. Recently, tumor-suppressive role of miR-1294 has been reported in ovarian cancer [20, 21], esophageal squamous cell carcinoma [22] and osteosarcoma [23]. However, the mechanism and function of miR-1294 in HCC were rarely reported. Functionally, we detected the effect of ATO or combination of ATO and miR-1294 on HCC cell growth. Mechanistically, two targets of miR-1294 were searched out: Pim-1 proto-oncogene (PIM1) and TEA domain transcription factor 1 (TEAD1). At length, the function of ATO-miR-1294-TEAD1/PIM1 axis in HCC was verified by rescue assays. To sum up, this study aimed to explore the functional role and underlying mechanism of miR-1294 in ATO-induced HCC cells.
Materials and methods
Cell culture, reagents and antibodies
Human HCC cell lines, SMMC7721, HepG2, Hep3B, Huh7 and Huh6, were commercially acquired from American Type Culture Collection (ATCC, Rockville, MD, USA). Cell lines were incubated in Dulbecco’s Modified Eagle’s Medium (DMEM, Gibco Diagnostics, Madison, WI, USA) with 10% fetal bovine serum (FBS, Gibco) and 1% penicillin- streptomycin (Hyclone, Logan, UT, USA). Cells were allowed to grow in a humidified incubator with 5% CO
ATO (Sigma Aldrich, Saint-Louis, MO, USA) was treated with 1.0 N NaOH at room temperature. Stock solution (0.25 mM) was obtained and diluted in DMEM. Primary antibodies (Abcam, Cambridge, MA, USA) and the secondary antibodies labeled with horse-radish peroxidase (HRP) used in western blotting analysis were purchased from Abacm (Cambridge, MA, USA). The methyl thiazolyl tetrazolium (MTT) was acquired from Sigma Aldrich.
Quantitative real-time polymerase chain reaction (qRT-PCR)
As described previously [24], Trizol
Cell transfection
SMMC-7721 and HepG2 cells were planted into 6-well plates containing DMEM and 10% FBS, followed by incubation at 37
Heatmap plotting
Twenty-four reported miRNAs with repressive function in cancers were examined in HepG2 cells with or without ATO treatment through qRT-PCR analysis. The results were analyzed and plotted as the heatmap (
MTT assay
HCC cells in 96-well culture plates (2
Colony formation assay
After treating with 8
Detection of caspase-3 activity
Sigma colorimetric caspase kit was applied to determine caspase-3 activity. HCC cells were placed in a 96-well plate for 1 d of culture. After centrifugation (1200 rpm) for just 5 min, cells were lysed in cell lysis buffer (15
In vivo animal study
Four-week-old female BALB/C athymic nude mice (
Luciferase reporter assay
The amplified 3’UTR of TEAD1 or PIM1 was cloned into firefly luciferase reporter gene downstream in pSi-check2 vector (Promega Corporation, Madison, WI, USA). TEAD1 or PIM1 wild type (termed TEAD1-WT or PIM1-WT, Genepharma, Shanghai, China) was generated. The mutant type of TEAD1 or PIM1 (termed TEAD1-MUT or PIM1-MUT, Genepharma) was simultaneously constructed using GeneTailor
Western blotting
Total protein was isolated from cell lines through RIPA buffer reagent (Thermo Fisher Scientific, Rochester, New York, USA). Lysates concentration was measured using BCA Protein Assay Kit (Beyotime, Guangzhou, China). Afterwards, 10% SDS-PAGE (Thermo Fisher Scientific) was utilized to separate protein samples. After transferring onto polyviny- lidene fluoride (PVDF) membranes, specimens were cultured on ice all night. Membranes were sealed in 5% bovine serum albumin solution (BSA, Beyotime) for two hours. After incubation with specific primary antibodies (1:1000) at 4
Statistical analysis
Data were all presented as the mean
ATO induced the upregulation of miR-1294 in human HCC cell lines. A. Fold changes of 24 miRNAs in HCC cells treated with or without ATO. B. Cell viability was measured in SMMC-7721 and HepG2 cells treated with increased dose of ATO by MTT assay. C. MiR-1294 expression in HCC cells treated with 8 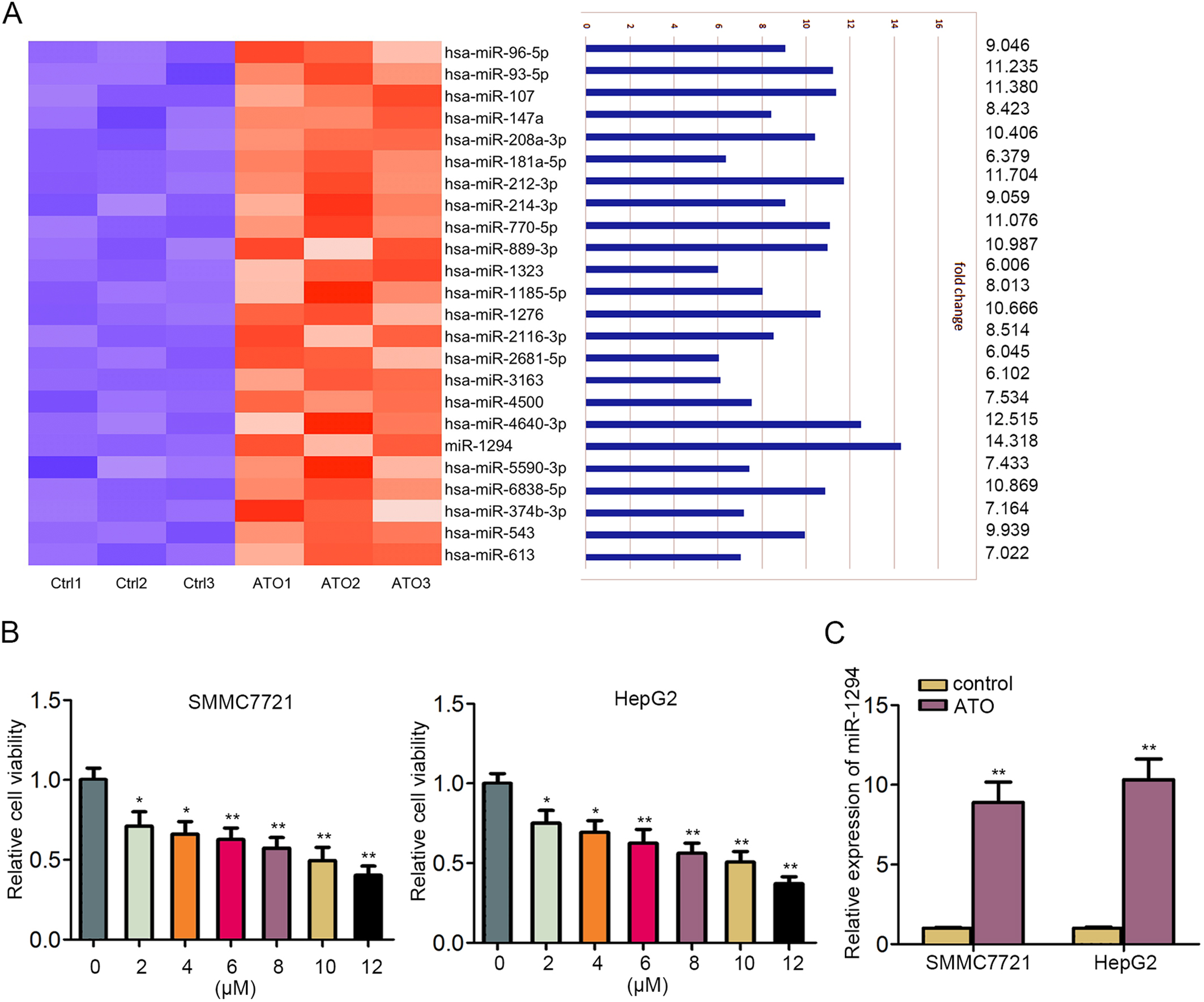
A. MTT assay estimated cell viability of Hep3B, Huh7 and Huh6 cells treated with increased dose of ATO. B–C. Western blot results and semi-quantitative analyses in SMMC7721 and HepG2 cells of four groups. D–F. Tumor size, volume and weight derived from Hep3B cells-injected nude mice were shown and calculated. 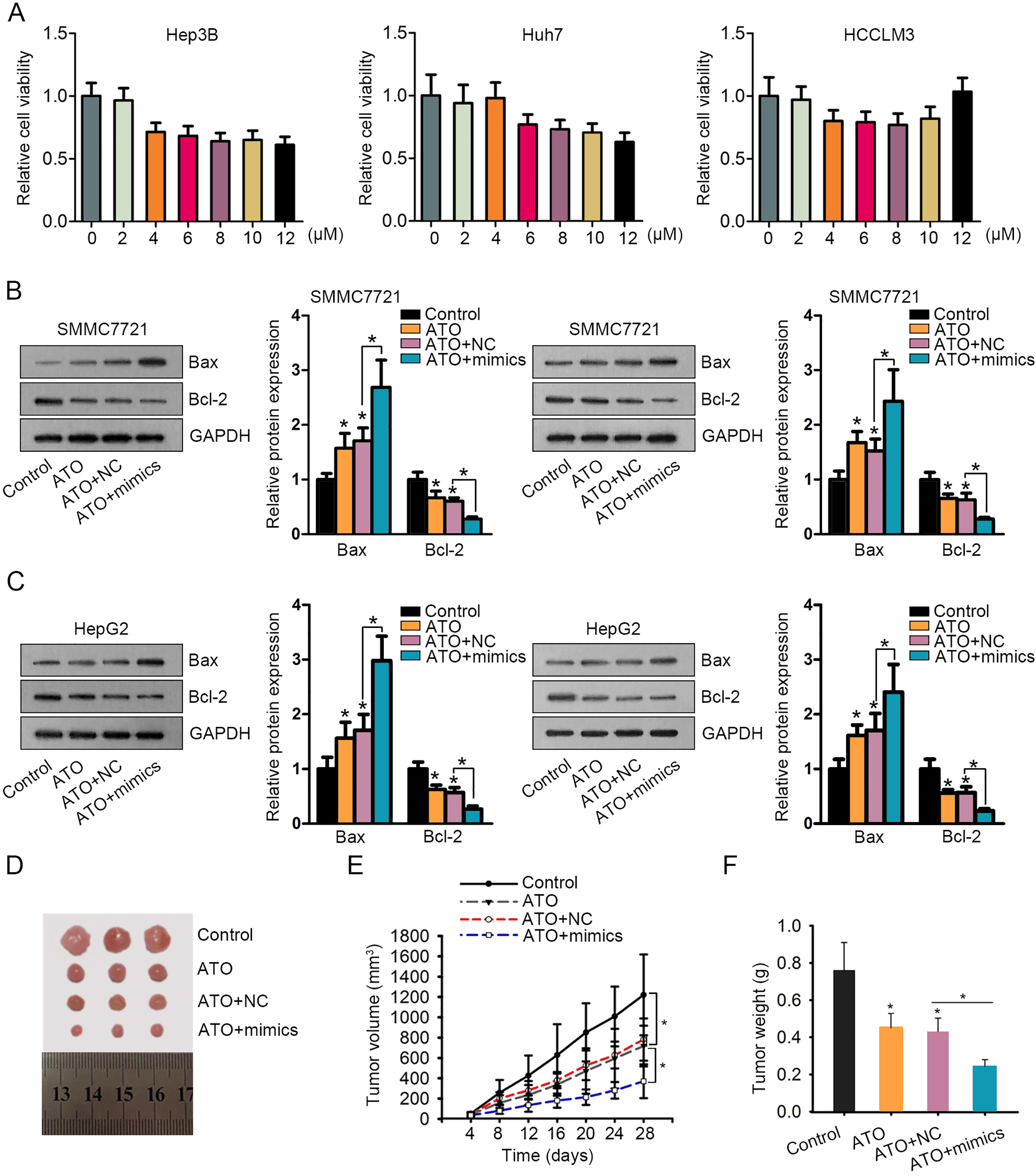
ATO and miR-1294 synergistically regulated HCC cell proliferation and apoptosis. A. Overexpression efficiency of miR-1294 in SMMC-7721 and HepG2 cells was determined by qRT-PCR. B–C. MTT and colony formation assays were used to measure the cell proliferation in SMMC-7721 and HepG2 cells treated with Control, ATO, ATO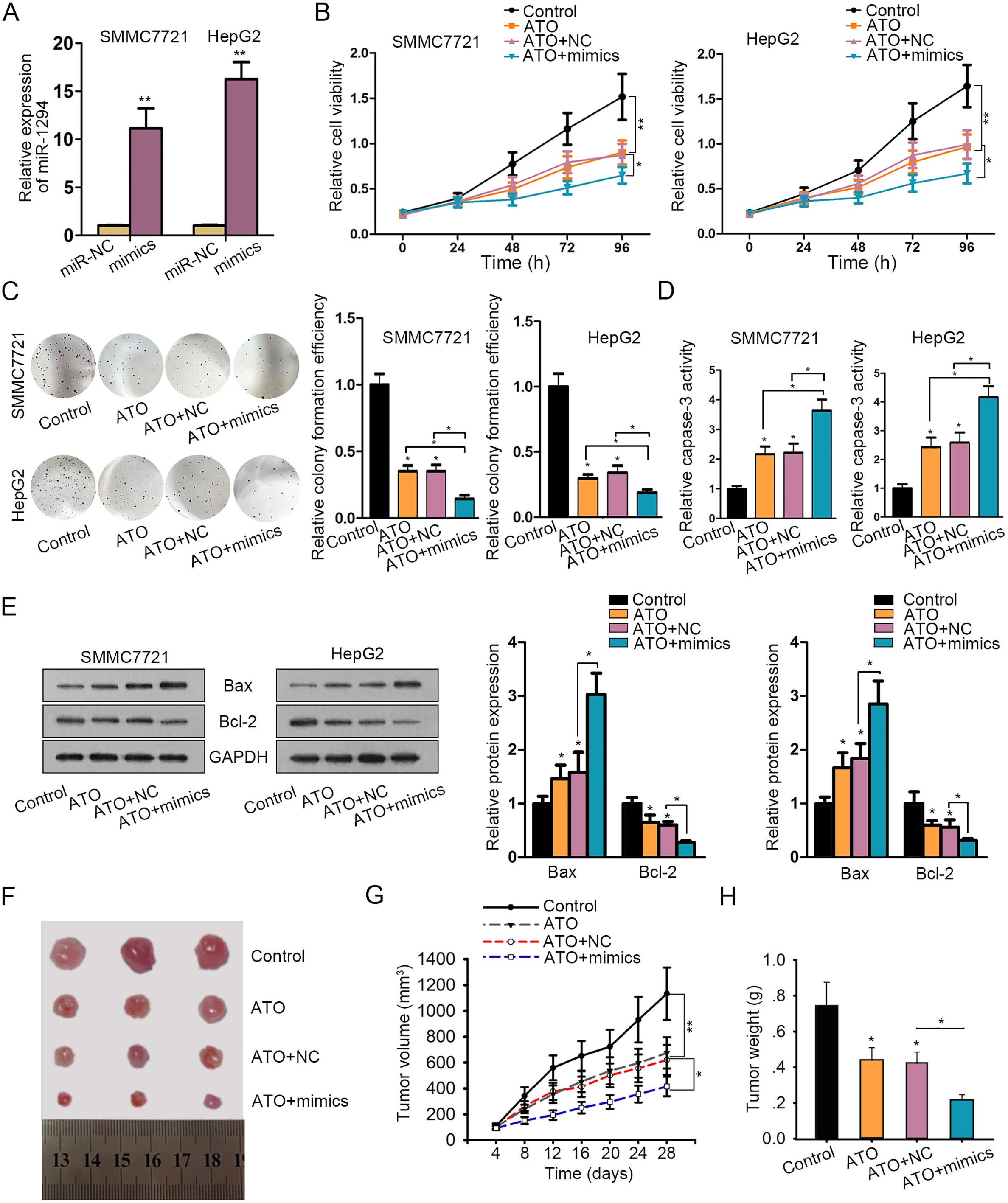
TEAD1 and PIM1 are two downstream targets of miR-1294. A. Sixty-eight target mRNAs of miR-1294 were predicted from four bioinformatics websites. B. Venn diagram revealed that only TEAD1 and PIM1 were down-regulated in response to the treatment of ATO ang miR-1294 mimics. C. The binding sites of miR-1294 in 3’UTR of TEAD1 or PIM1. D–E. Luciferase reporter assay was utilized to validate the interaction between miR-1294 and TEAD1 or PIM1. F–G. Both mRNA and protein levels of TEAD1 or PIM1 were analyzed in miR-1294-upregulated HCC cells, as measured through qRT-PCR and western blot. 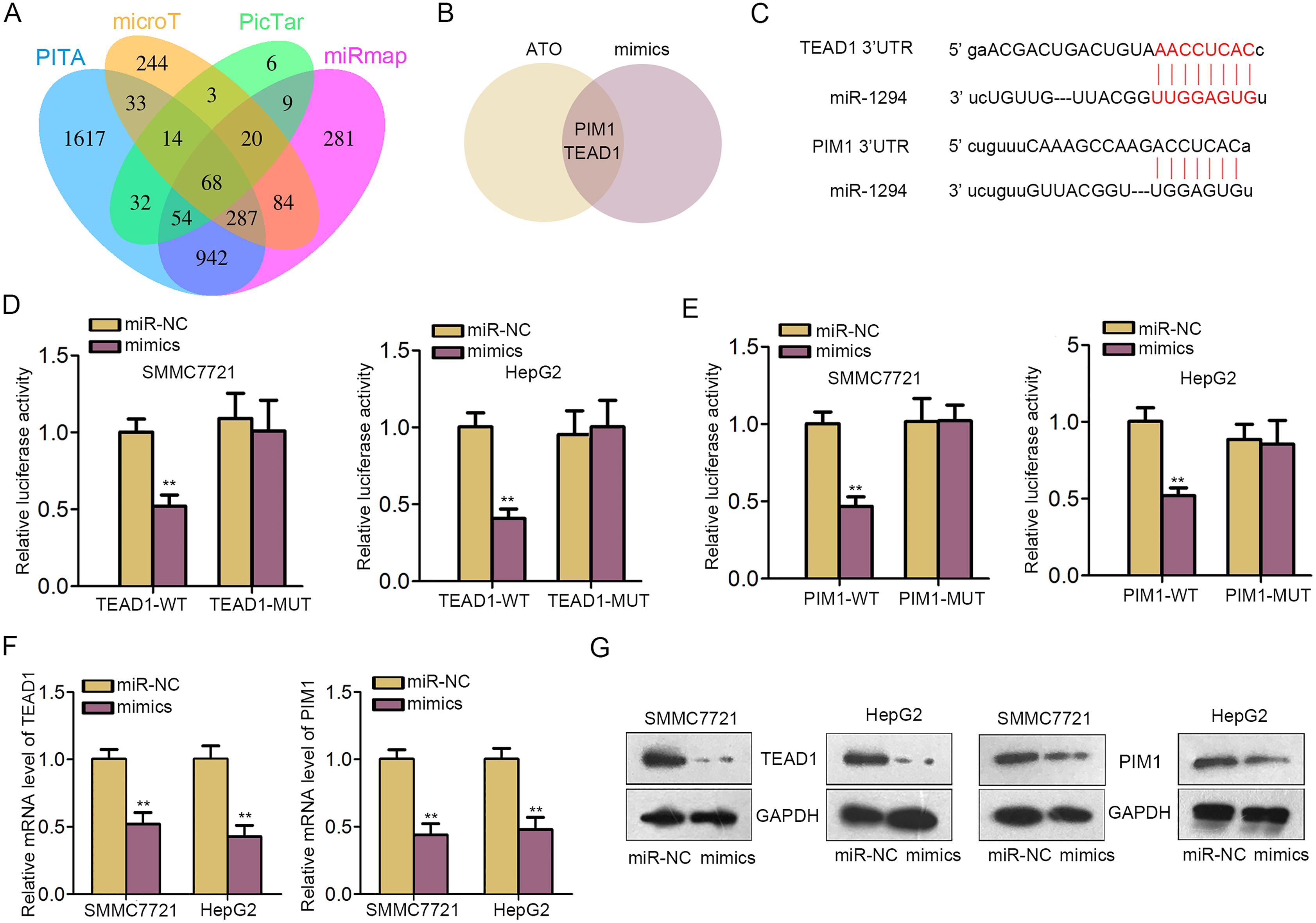
TEAD1 and PIM1 reversed miR-1294-mediated cell growth in ATO-induced HCC cells. A–C. Cell proliferation was detected in ATO-treated HCC cells after transfection with NC, miR-1294 mimics, or miR-1294 mimics and TEAD1 or PIM1 expression vector. D–E. Caspase-3 activity by specific kit and apoptosis-related proteins by western blot were measured in indicated cells under four treatments. 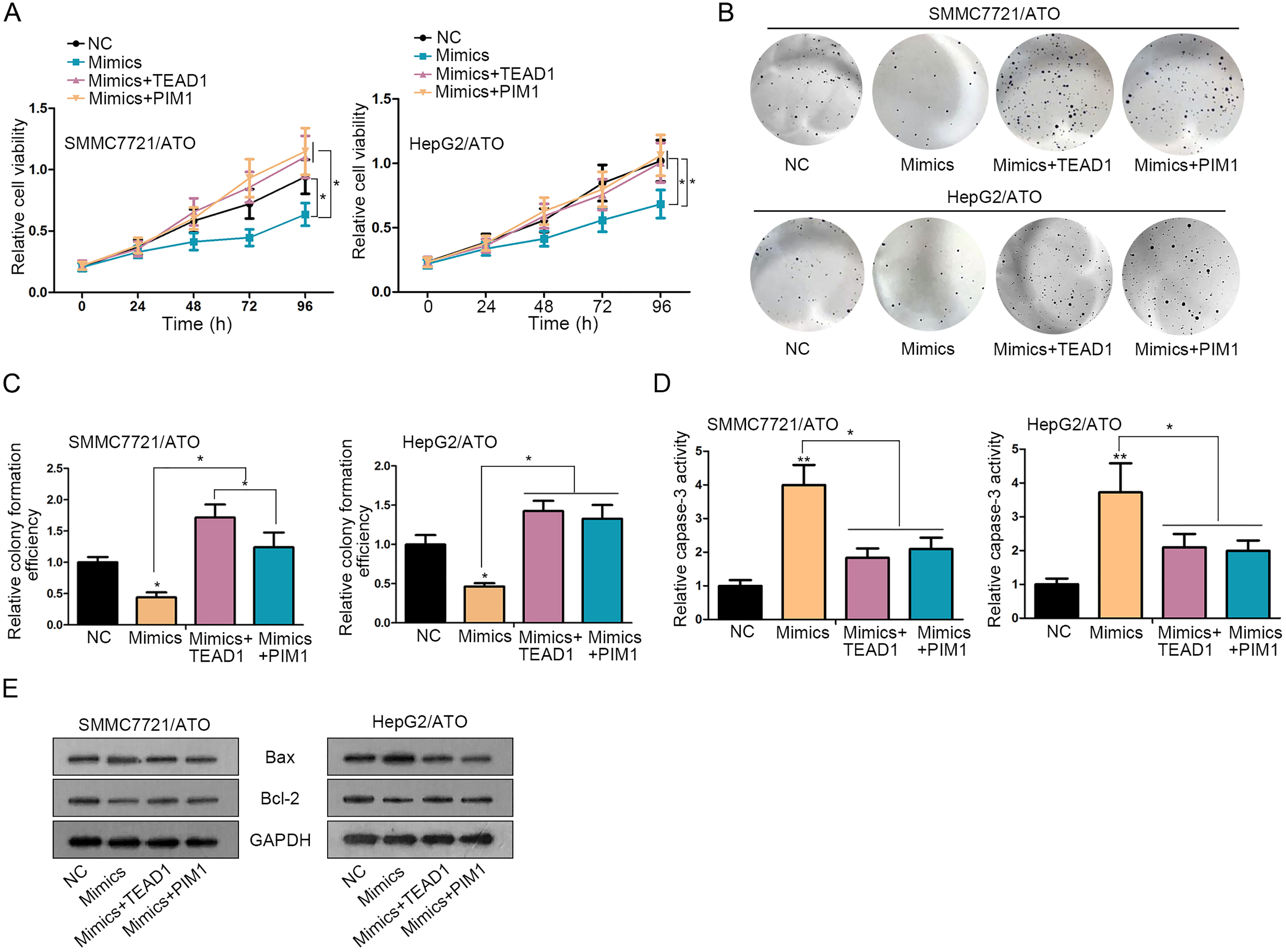
ATO induced the upregulation of miR-1294 in human HCC cell lines
To determine a certain miRNA that may function in HCC cell growth by combination with ATO, we screened out miRNAs that were upregulated in ATO-induced HCC cells by using qRT-PCR analysis. As illustrated in Fig. 1A, among 24 miRNAs that were upregulated by ATO treatment, miR-1294 presented the highest up-regulation. To observe whether ATO regulated HCC cell growth in a dose-dependent manner, five HCC cell lines were treated with increasing dose of ATO. MTT assay indicated that cell growth of SMMC7721 and HepG2 cells was more inhibited than that of Hep3B, Huh7 and Huh6 cells (Fig. 1B, Supplementary Fig. 1A). Hence, SMMC7721 and HepG2 cells were selected for later experiments. Remarkably, 8
ATO and miR-1294 synergistically regulated HCC cell proliferation and apoptosis
In order to determine the role of ATO-induced miR-1294 in HCC cell growth, miR-1294 was overexpressed in SMMC-7721 and HepG2 cells by transfecting miR-1294 mimics or miR-NC (Fig. 2A). According to the experimental results of MTT and colony formation assays, proliferative ability of HCC cell lines treated with ATO was suppressed compared with control cells. Noticeably, proliferation was suppressed more efficiently after combination of ATO and miR-1294 mimics (Fig. 2B and C). Afterwards, cell apoptosis was measured by detecting caspase-3 activity or apoptosis-related proteins. The caspase-3 activity and protein levels of Bax were enhanced, and the protein levels of Bcl-2 were decreased in ATO group. Such effects were further strengthened in ATO
TEAD1 and PIM1 are two downstream targets of miR-1294
As mentioned in previous reports, miRNAs could play functional roles in various human tumors via modulating their downstream target genes [26, 27]. In our current study, we predicted 68 target mRNAs of miR-1294 by using four bioinformatics analysis tools (microT, miRmap, PITA and PicTar), as displayed in Fig. 3A. Among these putative targets, only PIM1 and TEAD1 were significantly downregulated after treatment with ATO and miR-1294 mimics (Fig. 3B). Thus, we chose these two mRNAs for further study. The binding sequences of miR-1294 in 3’UTR of TEAD1 or PIM1 were obtained and shown (Fig. 3C). Subsequently, pSi-check2 luciferase reporter assay was conducted to verify the interaction between miR-1294 and TEAD1 or PIM1. As presented in Fig. 3D and E, luciferase activity of wild type TEAD1 or PIM1 (TEAD1-WT or PIM1-WT) was markedly decreased by miR-1294 mimics, while no significant change was observed in the luciferase activity of mutant type TEAD1 or PIM1 (TEAD1-MUT or PIM1-MUT). Finally, qRT-PCR and western blot assays indicated that miR-1294 overexpression efficiently impaired the mRNA and protein levels of TEAD1 and PIM1, separately (Fig. 3F and G). All these data indicated that TEAD1 and PIM1 were two targets of miR-1294 in HCC cells.
TEAD1 and PIM1 reversed miR-1294-mediated cell growth in ATO-induced HCC cells
To verify the involvement of TEAD1 or PIM1 in miR-1294-mdeiated HCC cell growth, rescue assays were carried out in ATO-treated HCC cells. MTT and colony formation assays revealed that cell proliferation inhibited by miR-1294 mimics were restored by the overexpression of TEAD1 or PIM1 (Fig. 4A–C). Besides, cell apoptosis increased by miR-1294 mimics was lessened after co-transfection with TEAD1 or PIM1 expression vector, as detected through caspase-3 activity and apoptosis-associated proteins (Fig. 4D and E). Collectively, TEAD1 and PIM1 involved in miR-1294-mediated cell growth in ATO-induced HCC cells.
Discussion
Therapeutic potential of ATO has been reported in chronic myeloid leukemia [28] and HCC [29, 30]. As a kind of chemotherapeutic drug used in oncological practice, ATO has been proved to suppress tumor cell proliferation and migration [31]. According to the existing evidence, ATO could modulate miRNAs expression to affect tumor cell activities [32, 33]. MiRNAs were acknowledged as indispensable factors in diagnosis and prognosis for HCC patients [34]. Previously, miR-1294 has been proved to be a prognostic factor in gastric cancer [35] and a tumor suppressor in ovarian cancer [36]. To our knowledge, it is unclear whether miR-1294 exerts function in hepatocellular carcinoma. Since ATO induced upregulation of miR-1294 in HCC cells, we designed experiments to investigate the role of miR-1294 in ATO-induced HCC cells. Functionally, we uncovered that ATO treatment obviously suppressed HCC cell growth. More importantly, growth inhibition was found to be more efficient after overexpression of miR-1294 in ATO-induced cells. In vivo experiments further verified the synergistic suppression of ATO and miR-1294 on HCC tumor growth. It was for the first time that miR-1294 and its function role were investigated in ATO-induced HCC cells.
In mechanism, miRNAs exert function though interacting with the 3’UTR of mRNAs, thus regulating gene expression [37]. Through bioinformatics analysis, we predicted some downstream targets of miR-1294. After qRT-PCR examination, PIM1 and TEAD1 were screened out due to the downregulation of them in response to ATO and miR-1294 mimics. Thus, we chose TEAD1 and PIM1 for subsequent experiments. TEAD1 has previously proved to be a tumor facilitator in human laryngeal squamous cell carcinoma and colorectal cancer [38]. Moreover, its oncogenic properties have been revealed in HCC [39]. In our current study, we also assessed the oncogenic role of TEAD1 and its upstream molecular mechanism in HCC cells. In addition, PIM1 was also known as a positive regulator in malignant progression of human cancers through a variety of ways. For instance, PIM1 facilitated tumor formation and metastasis in lung adenocarcinoma via c-MET signaling pathway [40]. Silencing of PIM1 modu- lated glycolysis and inhibited tumor progress in gallbladder cancer [41]. Through interacting with MYC, PIM1 could regulate ovarian cancer progress [42]. Mechanism investigation further validated the interaction between miR-1294 and PIM1 or TEAD1. Through luciferase activity analysis, we verified the affinity of miR-1294 to the 3’UTR of TEAD1 and PIM1. Further, the expression of both TEAD1 and PIM1 was negatively regulated by miR-1294. Herein, we confirmed that TEAD1 and PIM1 were two downstream targets of miR-1294 in HCC cells. At length, the functional role of miR-1294-TEAD1/PIM1 axis was determined in ATO-induced HCC cells by rescue assays. Intriguingly, overexpression of TEAD1 and PIM1 rescued cell growth which was suppressed by miR-1294 mimics in ATO-induced cells.
Based on all these experimental data, we concluded that ATO-induced miR-1294 exerted tumor-suppressive role in HCC via targeting TEAD1 and PIM1. Taken all together, our study reported a novel molecular pathway in ATO-inhibited HCC cell growth. Our research findings may contribute to the exploration of novel therapeutic targets and enhancement of the therapeutic effect of ATO for HCC.
Footnotes
Acknowledgments
Authors sincerely appreciate all supports to this study. Our study was supported by Wenzhou Science and Technology Bureau 356 (Y20150153).
Conflict of interest
No conflicts of interest to disclose.
