Abstract
BACKGROUND AND OBJECTIVE:
Fucosyltranferase 8 (FUT8), which catalyzes core fucosylation of glycopeptides, plays important roles in cancer development. In this study, we aimed to explore the influence of FUT8 expression on migration ability of human breast cancer cells and its potential mechanisms.
METHODS:
The core fucosylation levels in normal and FUT8 deficient MCF-7 cells were analyzed by lectin LCA blots. Then, the cell adhesion assay, transwell and wound healing experiments were conducted. The phosphorylation of FAK and core fucosylation of E-cadherin and its downstream integrins in the FAK/integrin pathway were measured. Moreover, the expression levels of nuclear
RESULTS:
The core fucosylation levels were significantly reduced by inhibited FUT8. FUT8 deficiency suppressed the adhesion, migration and invasion of MCF-7 cells; the potential mechanisms might involve three aspects. FUT8 deficiency inhibited FAK/integrin pathway by suppressing core fucosylation of E-cadherin. In addition, FUT8 deficiency reduced nuclear
CONCLUSIONS:
FUT8 deficiency suppressed migration of MCF-7 cells by impacting core fucosylation of E-cadherin and the downstream FAK/integrin pathway. Therefore, FUT8 is a potential biomarker for breast cancer detection and treatment.
Introduction
Breast cancer has become a vital cause of cancer-associated morbidity and mortality among woman worldwide [1] and the incidence of breast cancer has been increasing at a fast pace for the past few decades [2]. Although some improvements have been achieved in oncological immunotherapies, gene therapies and target-oriented therapies, large amounts of patients still suffer from tumor proliferation, metastasis and malignancy [3]. Therefore, it is necessary to identify effective biomarkers that can be used for prevention, early detection and treatment in breast cancer.
Core fucosylation is a post-translational modification process that plays an important role in the pathological progression of breast, hepatocellular carcinoma and colon carcinoma [4, 5]. Fucosyltranferase 8 (FUT8) is the only pivotal enzyme that can transfer the fucose residue to the innermost N-acetylglucosamine (GlcNAc) residue of the N-sugar chain in glycoproteins [6] to form
In the present study, to explore the effect of FUT8 expression on breast cancer cell migration, FUT8 deficient MCF-7 cells were constructed according to our previous work [12]. The core fucosylation levels, migration and invasion capabilities of breast cancer cells with normal or deficient FUT8 expression were investigated. To probe the potential mechanism, the phosphorylation of FAK and core fucosylation of E-cadherin and its downstream integrins in FAK/integrin pathway were compared in MCF-7-NC, shFUT8-1 and shFUT8-2 cells. In addition, the expression of MMP-2 and MMP-9 was also monitored.
Materials and methods
Materials
Antibodies against FUT8, EGFR, integrin-
Cell culture
Human breast cancer MDA-MB-231, MCF-7, ZR-75-1 and Bcap-37 cell lines were acquired from the American Type Culture Collection (Manassas, VA, USA). MDA-MB-231 and Bcap-37 cells were cultured in DMEM medium. ZR-75-1 cells were maintained in RPMI 1640 medium and MCF-7 cells were propagated in MEM medium. All cell culture mediums were supplemented with 10% (V/V) fetal bovine serum (FBS), 0.1 mg/mL streptomycin and 100 U/mL penicillin. The cells were cultured in a 37
Transfection FUT8 gene lentiviral RNA interference vectors
The FUT8 gene lentiviral RNA interference vectors were transfected into MCF-7cells to construct FUT8 deficient MCF-7 cells (shFUT8-1 and shFUT8-2 with different short hairpin RNA sequence (5’-3’) as TG GAGTGATCCTGGATATA and CTATAATGACGGA TCTATA) according to our previous work [12]. The linear PGC-LV-GFP carriers were also transfected into MCF-7 cells (MCF-7-NC) for negative control.
Real-time PCR
Total RNA in cells were extracted using TRIzol
Western blot analysis and immunoprecipitation analysis
Total proteins in cells were extracted by ice cold RIPA buffer with PMSF. Cells were lysed for 30 min and then centrifuged at 15000 rpm at 4
LCA lectin blot analysis
Protein extraction steps were the same as 2.5. The protein samples were fractioned by SDS-PAGE and then transferred to PVDF membranes. After blocking with 0.1% gelatin and 0.05% tween 20 in PBS (pH 7.5) for 1 h at room temperature, the PVDF membranes were incubated overnight in 4
Cell adhesion assay
Cells were plated in Matrigel coated 24-well plates (Corning Inc., USA) at the density of 1
Transwell assay
For invasion assay, cells were incubated on the top chambers of 24-well transwell plates (8.0
Wound healing assay
Cells were plated in 6-well plates with a density of 2
Statistical analysis
Data were expressed as mean
Results
MCF-7 cells highly expressed FUT8 among four breast cancer cell lines
The FUT8 relative expressions in MDA-MB-231, MCF-7, ZR-75-1 and Bcap-37 cell lines were examined. The results of RT-PCR and Western blot revealed that FUT8 mRNA and protein expression was highest in MCF-7 cells, followed by MDA-MB-231, ZR-75-1, and Bcap-37 as the lowest (Fig. 1A and B). MCF-7 cells were chosen to construct FUT8 deficient cells due to the high levels of FUT8 expression relative to the other breast cancer cell lines.
MCF-7 cells express higher FUT8 than other human breast cancer cells. (A) RT-PCR and Western blot. (B) analysis of gene and protein expressions of FUT8 in MDA-MB-231, MCF-7, ZR-75-1 and Bcap-37 cell lines. (C) Lectin blot analysis of 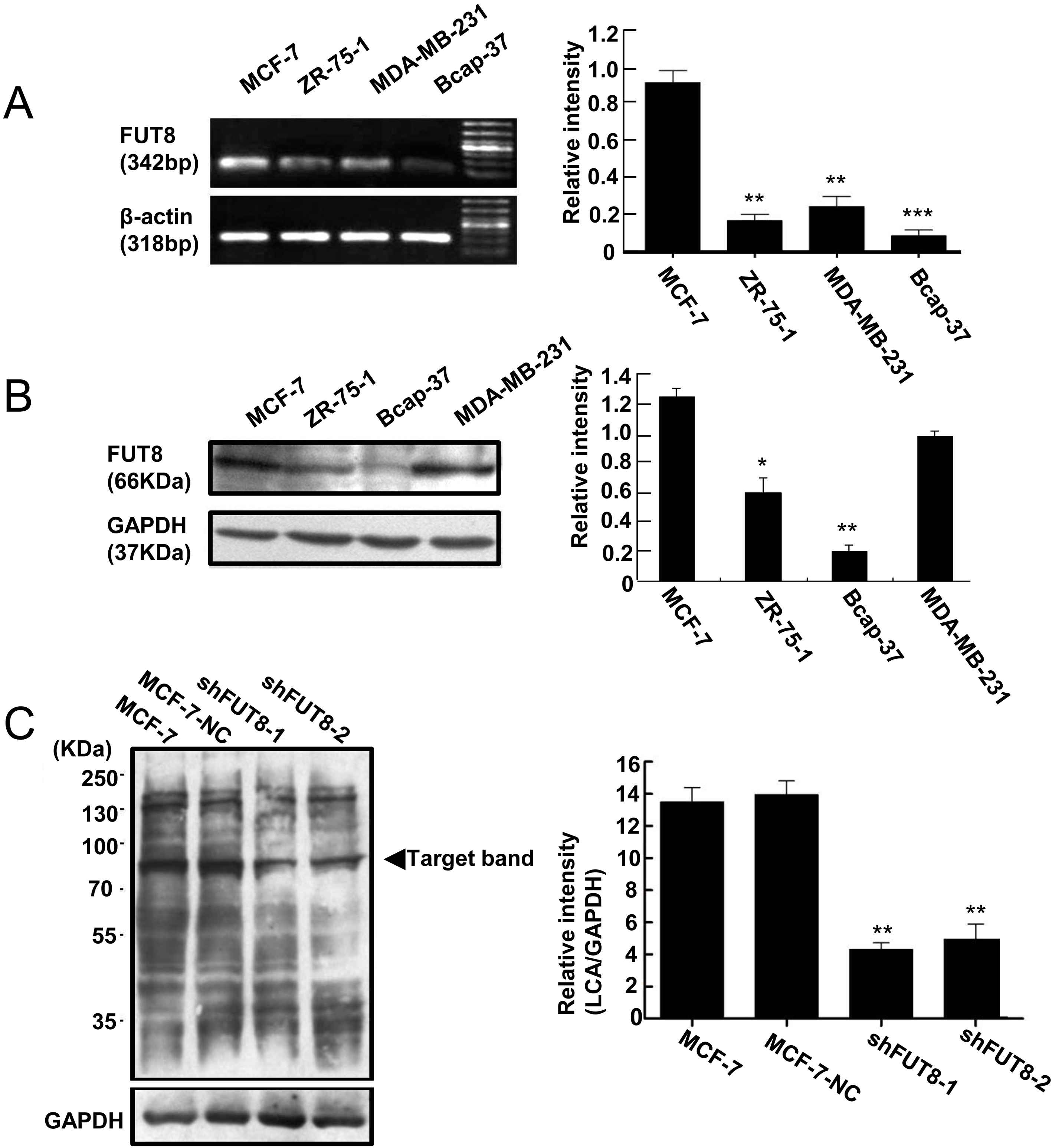
Our previous work revealed that FUT8 expression in shFUT8-1 and shFUT8-2 cells is efficiently downregulated when compared with MCF-7-NC and MCF-7 cells, and shFUT8-2 cells express lower FUT8 levels than shFUT8-1 cells [12].
To validate the effects of FUT8 interference, core fucosylation levels were detected by lectin blots. The results showed that non-carriers transfection had no influence on the core fucosylation level in MCF-7-NC cells, because the core fucosylation level in MCF-7-NC was similar to the core fucosylation level in MCF-7. The shFUT8-1 and shFUT8-2 cells displayed much lower core fucosylation levels, which indicated lentiviral RNA interference vectors decreased core fucosylation levels through inhibiting FUT8 expression (Fig. 1C).
FUT8 deficiency suppressed the adhesion ability of MCF-7 cells
A non-specific cell adhesion assay was applied to investigate the influence of FUT8 expression on MCF-7 cell adhesion. As is shown in Fig. 2A, the absorbance of OD 570 nm in MCF-7-NC cells were 0.83
FUT8 deficiency inhibits the adhesion, invasion and migration of MCF-7 cells. (A) Cell adhesion evaluation of MCF-7 cells after FUT8 interference via cell adhesion assay. (B) The images and quantitative analysis of invaded MCF-7-NC, shFUT8-1 and shFUT8-2 cells by transwell assay. (Scale bars 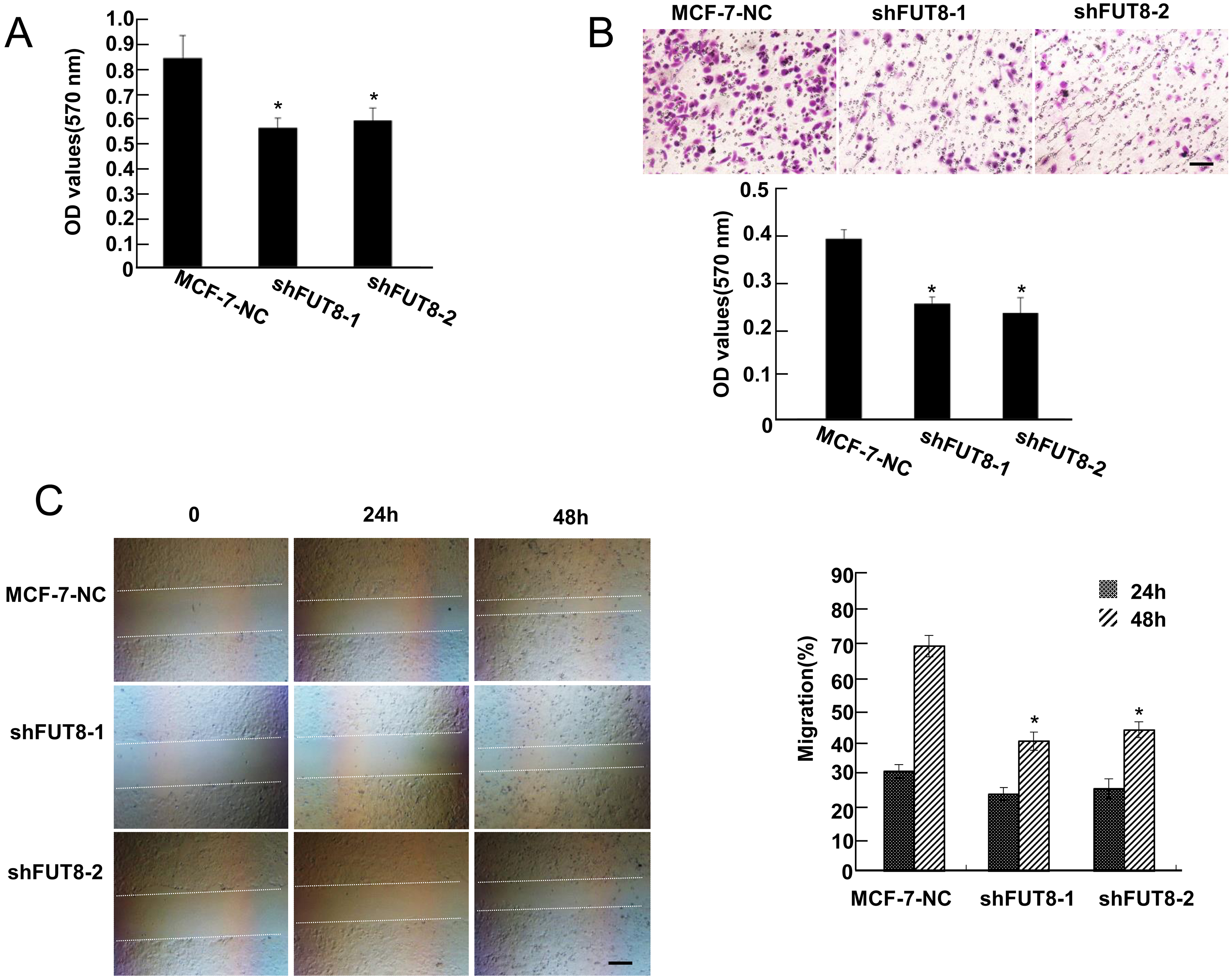
To investigate the effect of FUT8 expression on invasion ability of human breast cancer cells, the transwell assay was carried out. As presented in Fig. 2B, MCF-7-NC cells exerted high invasion ability, while FUT8 deficiency effectively decreased the numbers of MCF-7 cells migrating through the transwell membrane to the bottom chamber. Thus, FUT8 deficient MCF-7 cells exerted weaker invasion potential than MCF-7-NC cells.
FUT8 deficiency suppressed the migration of MCF-7 cells
The migratory capabilities of MCF-7-NC, shFUT8-1 and shFUT8-2 cells from one side of the wound to the other side were investigated by wound healing assay. Compared with MCF-7-NC cells, the migration rate (%) of shFUT8-1 and shFUT8-2 cells changed little in 24 h (Fig. 2C). The migration rate (%) of shFUT8-1 and shFUT8-2 cells decreased significantly by 48 hours, showing that MCF-7 cell migration is inhibited in the absence of FUT8.
FUT8 deficiency suppressed the phosphorylation of FAK
FAK activation by E-cadherin stimulates the potential of migrating cells, so the phosphorylation of FAK directly impacts the migration of cancer cells [33]. The Western blot results in Fig. 3A show that the phosphorylation of FAK was enormously inhibited by downregulating the expression of FUT8 in shFUT8-1 and shFUT8-2 cells, compared with MCF-7-NC cells.
(A) The phosphorylation levels of FAK in MCF-7-NC, shFUT8-1 and shFUT8-2 cells, analyzed by Western blot. (B) The protein expression and core fucosylation of integrin-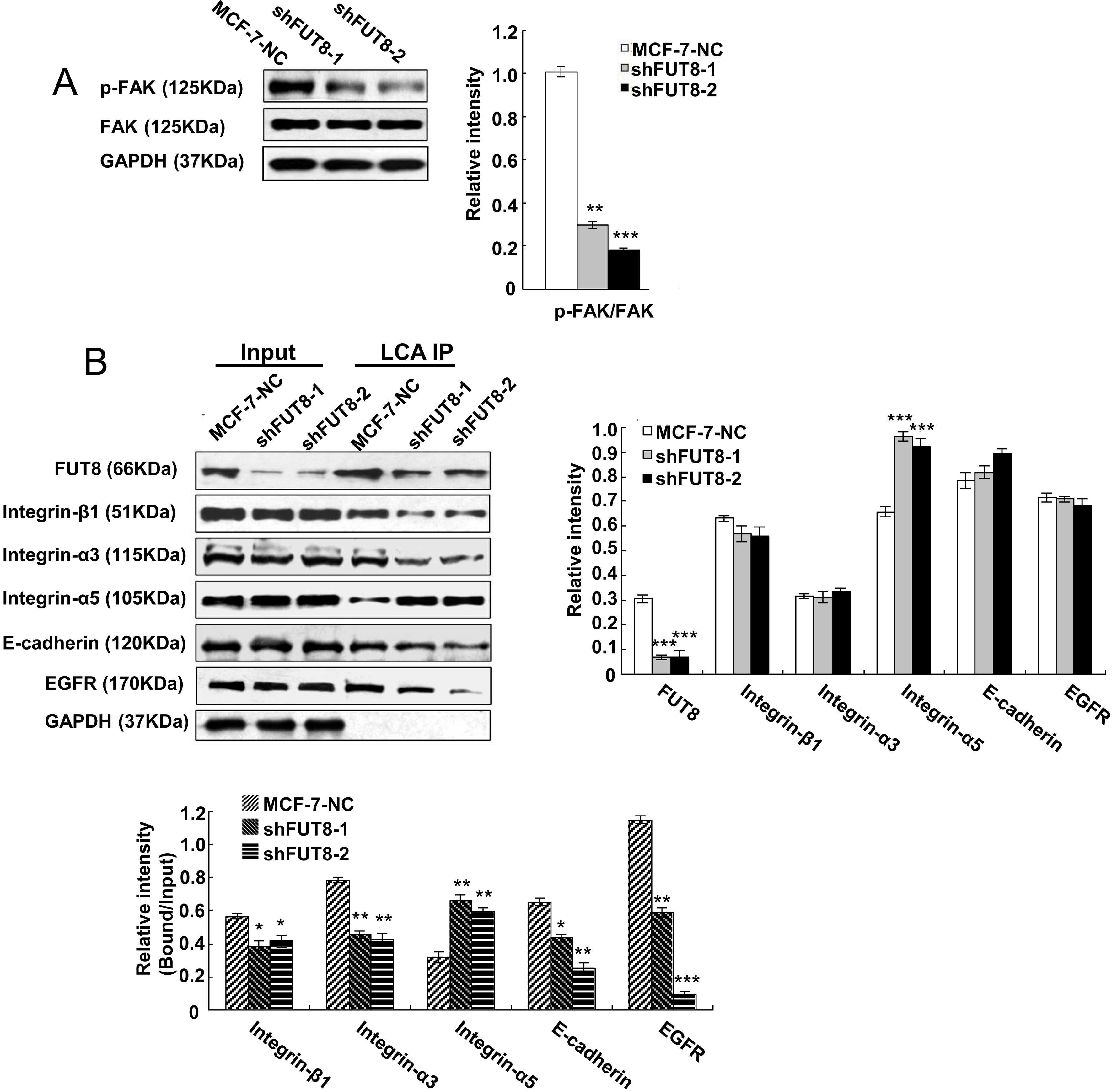
The Western blot results in Fig. 3B showed that the absence of FUT8 had no obvious influence on the expressions of integrin-
FUT8 deficiency suppressed nuclear
-catenin accumulation
(A) The total 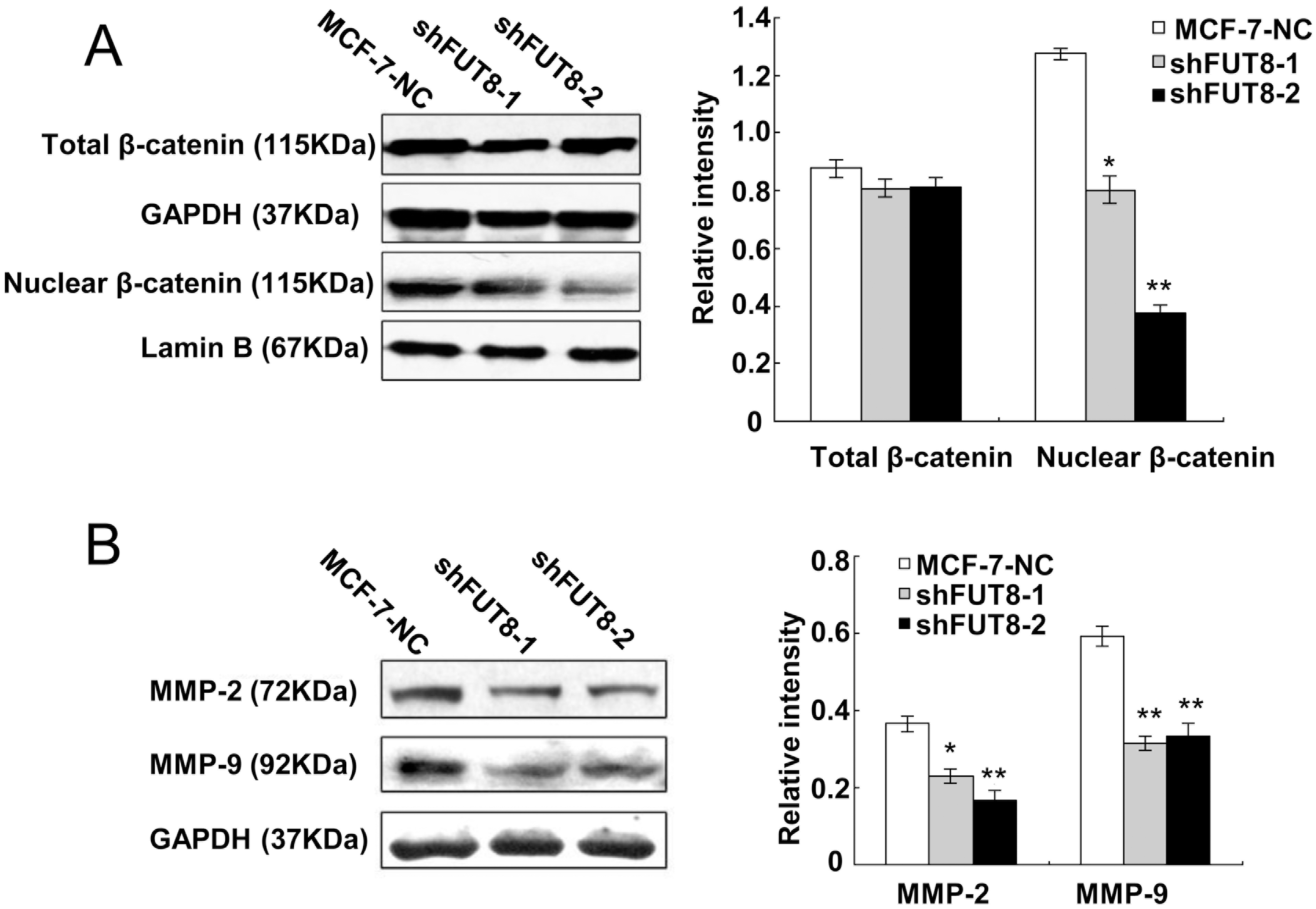
MMPs have close associations with cancer cell invasion and migration, and the deduction of MMP-2 and MMP-9 has been reported to inhibit cancer cell invasion. In our results, shFUT8-1 and shFUT8-2 cells had decreased expression of MMP-2 and MMP-9 compared with MCF-7-NC cells (Fig. 4B). The decreased expression levels of MMP-9 in shFUT8-1 and shFUT8-2 cells were comparable, while shFUT8-2 cells showed a higher reduction in MMP-2 expression than shFUT8-1 cells.
Discussion
Glycoproteins have been considered for the past decade as specific biomarkers for diagnosis and prognostic evaluation of cancers [5]. FUT8 is the only enzyme able to catalyze
E-cadherin is the marker of epithelial cells and its decreased expression reduces the amount of epithelial cell junctions, increases epithelial-mesenchymal transition and promotes cells acquiring the capabilities of migration and invasion [17]. In our results, the expression of E-cadherin changed little in shFUT8-1 and shFUT8-2 cells, compared with MCF-7-NC cells, which is in agreement with the former observation that FUT8 expression has no significant influence on E-cadherin expression in miR-10 overexpressed MCF10A cells [18]. Therefore, the suppressed migration and invasion by interfering FUT8 was achieved without inhibiting EMT in MCF-7 cells. Core fucosylation of E-cadherin can regulate nuclear
Core fucosylated integrin-
The studies of FAK and endocytosed integrins revealed that FAK regulates integrin activation, sustains integrin active conformation and promotes its recycling into new adhesions, with the help of focal adhesions [25]. The recycling of activated integrins happens in the initiating stage of migrating cells. Therefore, the activation of FAK promotes migration potential. The phosphorylation of FAK can be achieved by core fucosylation of E-cadherin through regulating the SRC/FAK pathway, so core fucosylation of E-cadherin decides the fate of cell migration by FAK/integrin pathway [26, 27]. In our results, insufficient FUT8 expression inhibited core fucosylation of E-cadherin and the downstream FAK/integrin pathway. Next, the reduced phosphorylation of FAK repressed core fucosylation of integrins and reduced the adhesion ability of human breast cancer cells.
The biological activities, such as potentiating cancer malignancy, of epidermal growth factor receptors (EGFR) are also regulated by FUT8-catalyzed core fucosylation [28]. The core fucosylation of EGFR and its downstream signals induced by EGF are inhibited in FUT8
Metalloproteinases (MMPs) play pivotal roles in cancer metastasis, invasion and angiogenesis through changing the microenvironment of tumor cells [29, 30, 31]. Among the members of MMP family, MMP-2 exerts tumor promoting effects and its genetic polymorphism increases the rate of breast, lung, colorectal and gastric cancer incidence [32, 33, 34, 35]. MMP-9 is also vital in tumor incidence and metastasis, and its genetic polymorphism increases the risk of bladder cancer and tumor malignancy [36, 37]. In our result, FUT8 interference suppressed the expressions of MMP-2 and MMP-9, which indicated that reducing FUT8 expression can inhibit the deterioration of tumor microenvironment catalyzed by MMP-2 and MMP-9.
In conclusion, FUT8 deficient cells were utilized to investigate the influence of FUT8 expression on human breast cancer cells migration capability. Our results revealed that FUT8 deficiency represses human breast cancer cell migration through three mechanisms. Firstly, FUT8 deficiency inhibited FAK/ integrin pathway by suppressing core fucosylation of E-cadherin; Secondly, FUT8 deficiency reduced nuclear
Footnotes
Acknowledgments
This work was supported by the Scientific Research Startup Foundation for Postdoctoral of Heilongjiang Province (No. LBH-Q17178) and National Natural Science Foundation of China (No. 81802834, 81202084). University Nursing Program for Young Scholars with Creative Talents in Heilongjiang Province (UNPYSCT-2018035) and Key Program from Qiqihar Medical University of China (No. QY2014Z-01).
Conflict of interest
The authors declare no conflict of interests.
