Abstract
BACKGROUND:
The Indian subcontinent has the highest incidence and prevalence of oral squamous cell carcinoma (OSCC). Inflammation and apoptosis are two emerging hall marks of cancer that might play a significant role in tumorigenesis and metastasis. Concurrent expression of proinflammatory cytokine (IL-1
OBJECTIVES:
The present study aimed to evaluate the immunohistochemical expression of IL-1
METHODS:
A retrospective study was conducted utilizing 40 formalin fixed paraffin embedded histologically diagnosed cases of OSCC comprising of 20 metastatic OSCC and 20 non-metastatic OSCC.
RESULTS:
Increased expression of IL-1
CONCLUSION:
Over expression of IL-1
Keywords
Introduction
Pie-chart showing the demographic details of OSCC patients considered for the study. A. Age distribution. B. Gender distribution. C. Tobacco status distribution. D. Staging distribution. E. Site distribution. F. Nodal status distribution.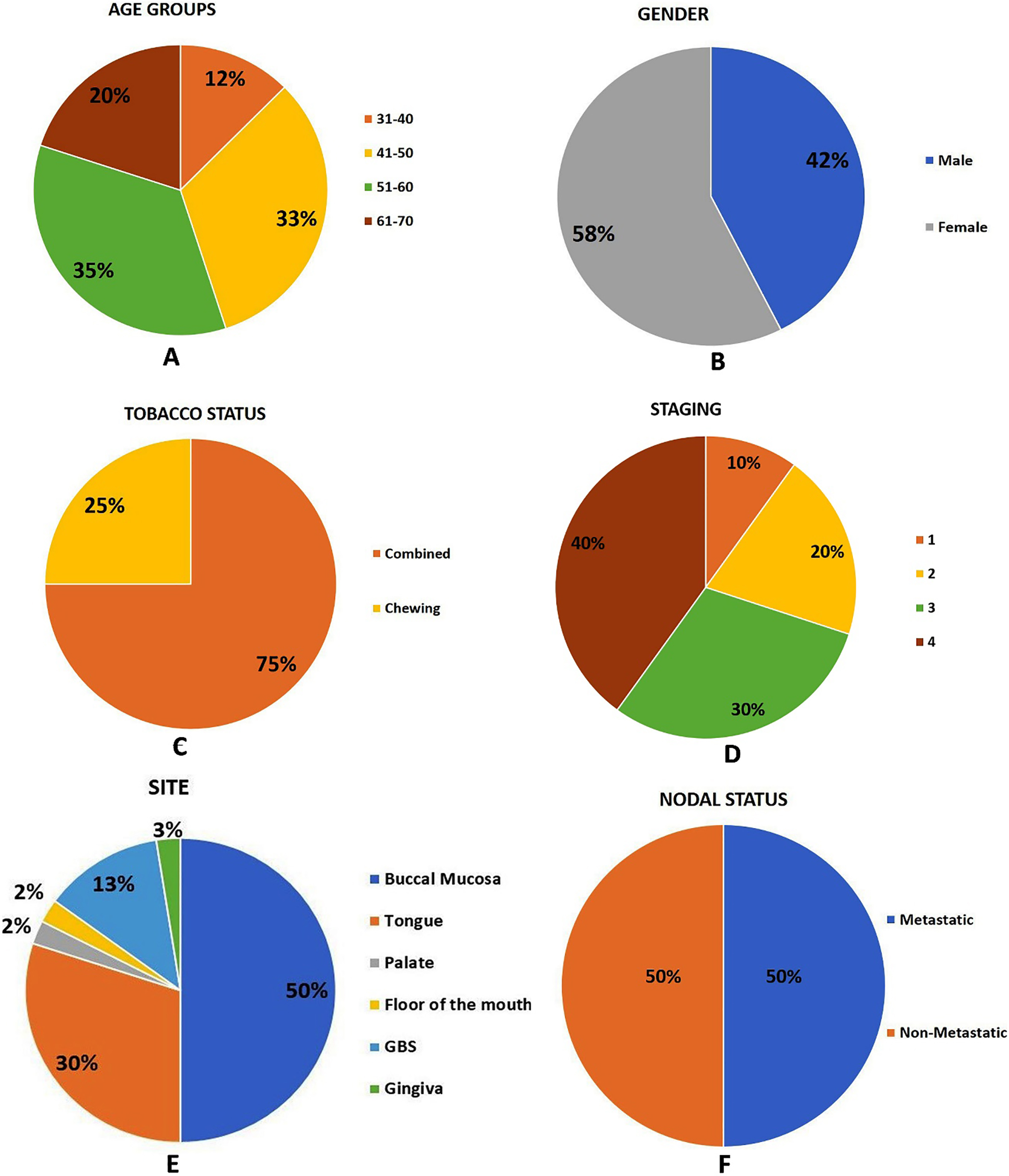
Despite numerous advancements over the years , annually oral cancer accounts for an estimated 275,000 cases globally. India accounts for about 30% of all new cases annually. OSCC has a survival rate
The link between apoptotic proteins such as caspases and cytokines remain a sparsely researched area in oral oncology. The role of caspases in regulating inflammation resulting in malignancy still remains unclear [7, 8, 9]. Despite the fact that caspases act mainly within the cell, the production and activity of caspases is triggered and modulated by external signals via cytokines thus, making these two entities “Inter-connected” in biological processes. It is proposed that cytokines are produced in response to inflammation and serve as an important link between inflammation and cancer through intrinsic and extrinsic effects [10, 11, 12, 13, 14]. Pro-inflammatory cytokine called Interleukin 1 family (IL-1) has been associated with poor prognosis and survival of patients with gastric and colorectal adenocarcinoma [15]. The tumorigenic potential of pro-inflammatory cytokine IL-1
The correlation and interplay between apoptosis and inflammation is not yet investigated in OSCC. Hence the present study aimed to evaluate the expression of IL-1
Ethical clearance for conducting the study was obtained from ethics committee of M.S. Ramaiah University of Applied Sciences (Ref: UECHT/2016-18/PGDT/03).
Selection of cases
Data were collected using a pre-formatted data collection form. For each case, the corresponding information on age, gender, tobacco status, tumour size, nodal status, metastases (TNM) and follow-up data of five years were collected from clinical records Fig. 1. The clinico-pathological features were classified according to American Joint Committee on Cancer (AJCC) designated staging by TNM classification system. The following criteria was applied for selection of cases. Histopathologically diagnosed incisional biopsies of primary OSCC (metastatic and non-metastatic) were considered having complete clinical data record.
Recurrent cases of OSCC were excluded. Slovin’s formula was applied to the current study to determine sample size, which is expressed as
Tissue specimens
A total of 40 formalin fixed paraffin embedded (FFPE) histologically diagnosed cases (20 metastatic and 20 non-metastatic OSCC) were retrieved from archives of the Oral Pathology and Microbiology Department, Faculty of Dental Sciences, M.S. Ramaiah University of Applied Sciences, Bangalore. Normal healthy oral mucosa (NOM,
Tissue processing and immunohistochemistry
Deparaffinizing and rehydrating
FFPE tissue specimens measuring 4
Antigen retrieval and enzyme blocking
All slides and antigen retrieval buffer, pH 6.0 (Pathnsitu Cat# PS007, Hyderabad, India) were held in coplin jars in a water bath set to 92
Photomicrographs of control tissue used for standardization A. Section of kidney stained with IL-1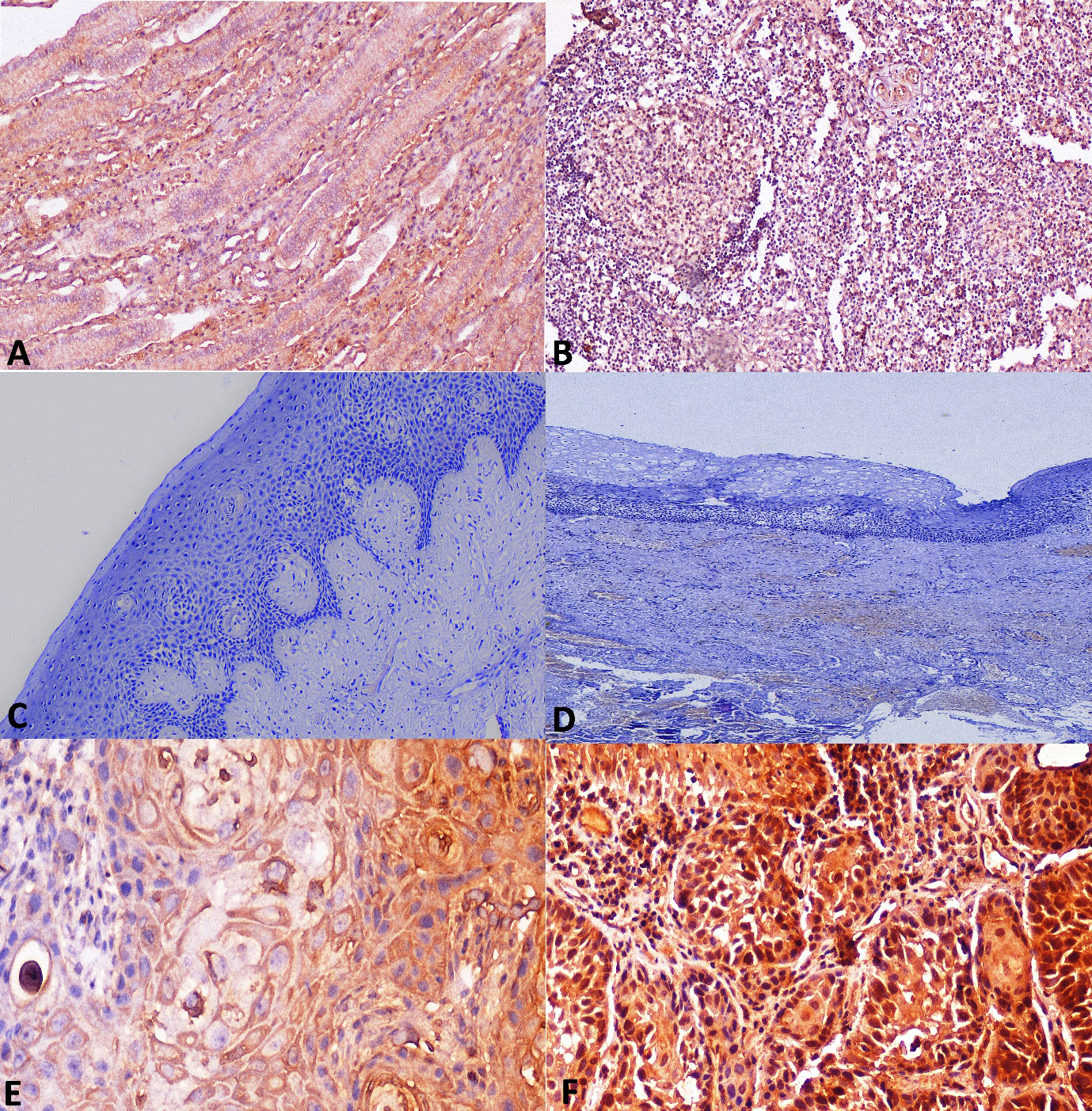
The sections were incubated with anti-mouse monoclonal primary antibody (IL-1
Photomicrographs of OSCC A. Section of non-metastatic OSCC tissue showing invading tumor islands (H&E stain, x100). B. Non-metastatic OSCC stained with antibody to IL-1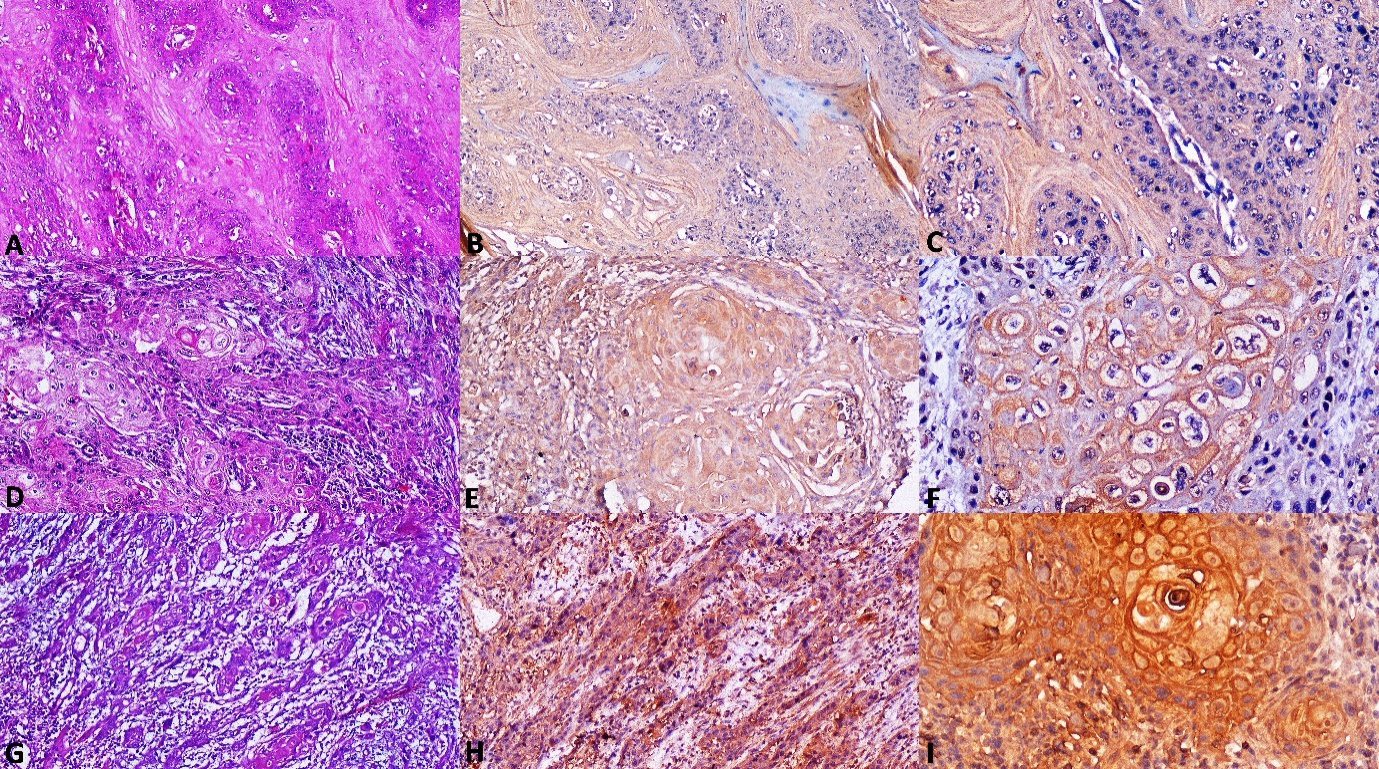
Immunohistochemical results were semi quantitatively evaluated in a blinded pattern by two observers (PS and DA), at a magnification of x100 and x400 using Olympus Optical Microscope BX53F2, Tokyo, Japan. Any discrepancy in the scores was solved by a group consultation with third observer (SVS) utilizing penta-head Olympus Optical Microscope U-MDOB3, Tokyo, Japan. The photomicrographs were captured with a Jenoptik Progres Gryphax Arktur USB 3.0 microscope camera, Jena, Germany. For both Caspase-3 and IL-1
Photomicrographs of OSCC A. Section of non-metastatic OSCC tissue showing invading tumor islands with dysplasia (H&E stain, x100). B. Non-metastatic OSCC stained with antibody to Caspase-3 showing low cytoplasmic expression (IHC, x100). C. Caspase-3 showing low cytoplasmic expression in non-metastatic OSCC at higher magnification (IHC, x200). D. Section of Metastatic OSCC tissue showing invading tumor islands with keratin pearls (H&E stain, x100). E. Metastatic OSCC stained with antibody to Caspase-3 showing high expression (IHC, x100). F Caspase-3 showing high expression in metastatic OSCC at higher magnification (IHC, x200). G. Section of Metastatic OSCC tissue showing invading tumor strands and cords (H&E stain, x100). H. Metastatic OSCC stained with antibody to Caspase-3 showing high expression (IHC, x100). I. Caspase-3 showing high expression in metastatic OSCC at higher magnification (IHC, x200).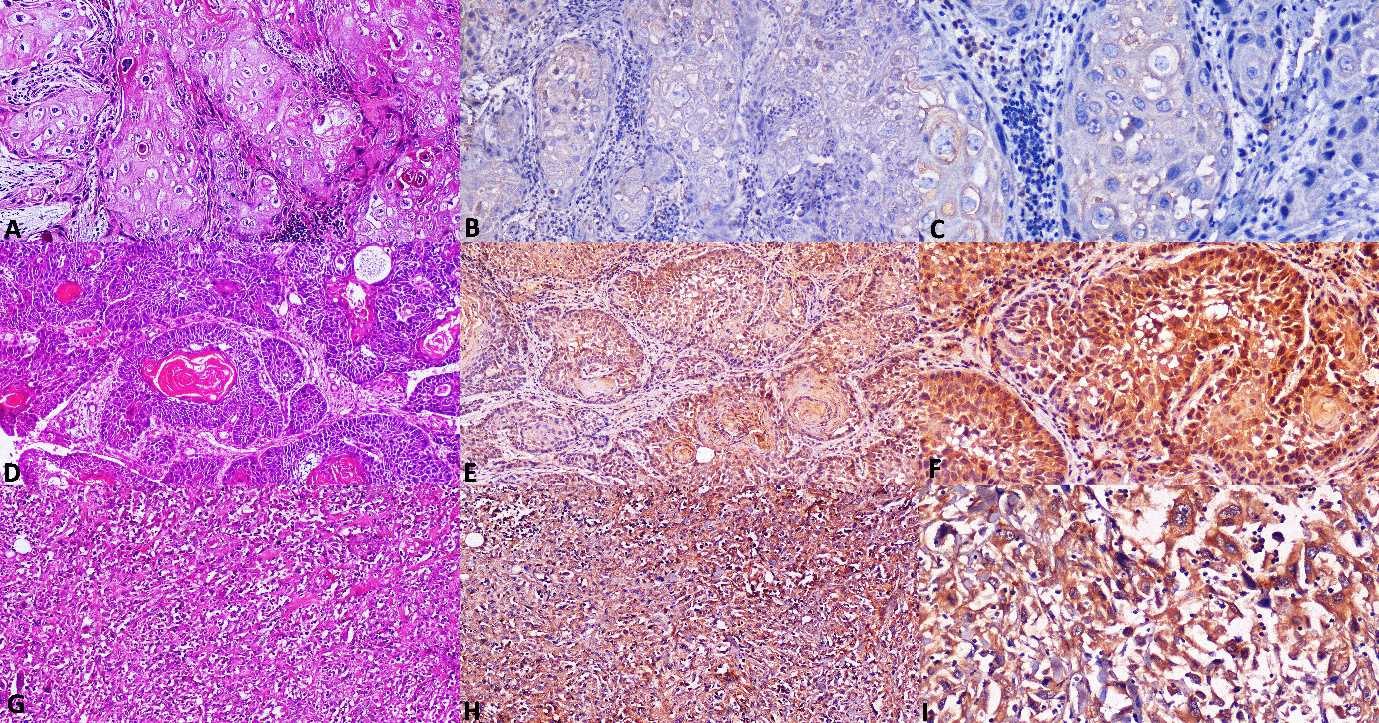
Following meticulous standardization and obtaining of negative background staining using kidney and tonsil tissue (Fig. 2A and B), the IHC procedure was initiated. NOM taken as healthy controls were immunonegative for IL-1
IL-1
expression in non-metastatic and metastatic OSCC
Brown cytoplasmic immunostaining was observed in tumour cells of OSCC positive control tissue for IL-1
Photomicrographs of metastatic OSCC A. Section of metastatic OSCC tissue showing tumor islands and muscle invasion (H&E stain, x100). B. Metastatic OSCC stained with antibody to IL-1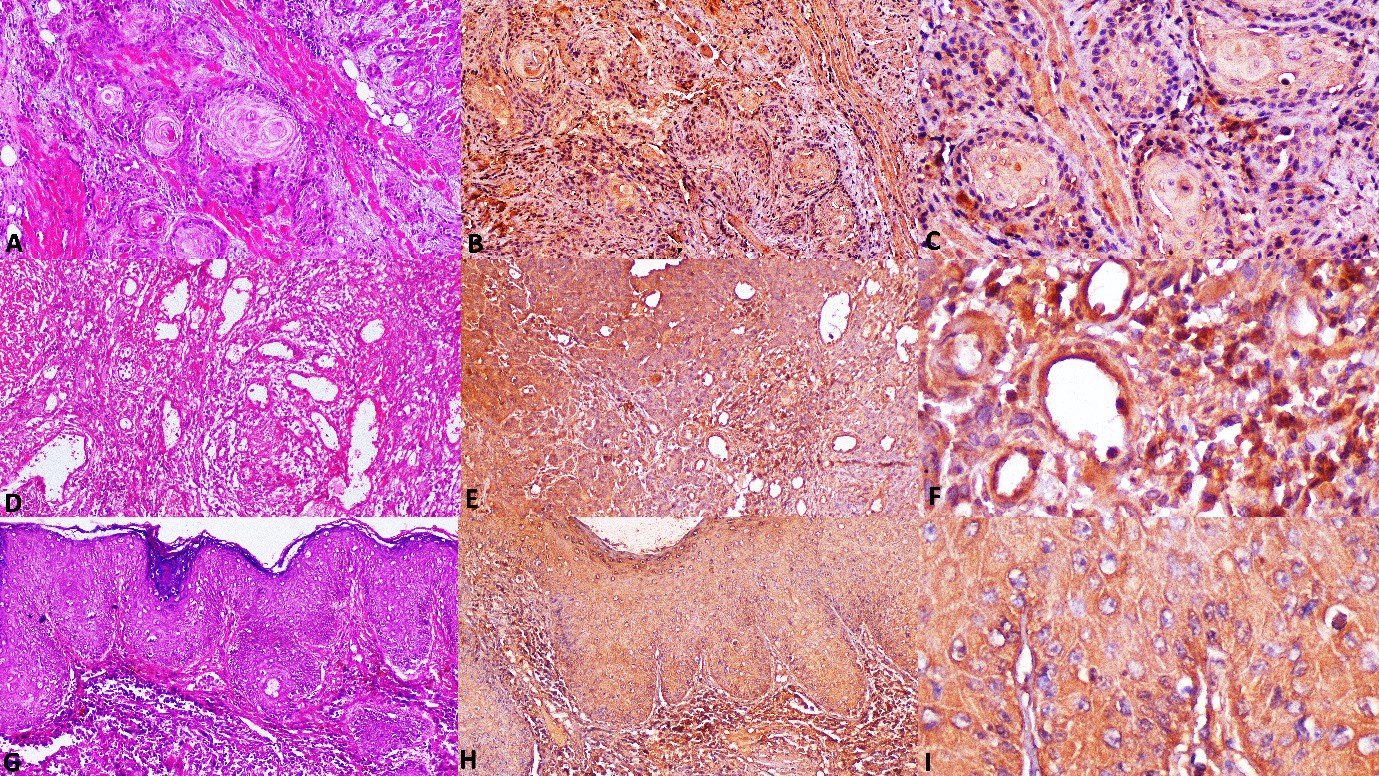
Comparison of IL-1
Mann-Whitney U Test,
Brown cytoplasmic immunostaining was observed in tumour cells of OSCC positive control tissue for Caspase-3. Among 20 non-metastatic OSCC cases, all 20 (100%) cases showed a positive cytoplasmic immunoreactivity for Caspase-3, of which 8 (40%) cases showed a high score, 6 (30%) cases showed intermediate score and 6 (30%) cases showed low score (Fig. 4A–C). In case of metastatic OSCC, 18 (90%) cases showed high score (Fig. 4D–I), and 2 (10%) cases showed intermediate score. None of the metastatic OSCC cases showed low or negative immunoreactivity for Caspase-3. When Caspase-3 expression was compared between non-metastatic and metastatic OSCC a statistically significant result was obtained using Mann-Whitney U Test (Table 2).
Comparison of Caspase-3 expression in non-metastatic and metastatic groups
Comparison of Caspase-3 expression in non-metastatic and metastatic groups
Mann-Whitney U Test,
When analysis of coefficient of correlation was executed, expression of IL-1
Correlation of IL-1
and Caspase-3 expression in non-metastatic and metastatic Oral Squamous cell carcinoma
Correlation of IL-1
Correlation of IL-1
Chi square test,
Photomicrographs of metastatic OSCC with inverse expression of markers A. Section of metastatic OSCC tissue showing dysplastic features (H&E stain, x100). B. Metastatic OSCC showing negative immuno reaction for antibody to IL-1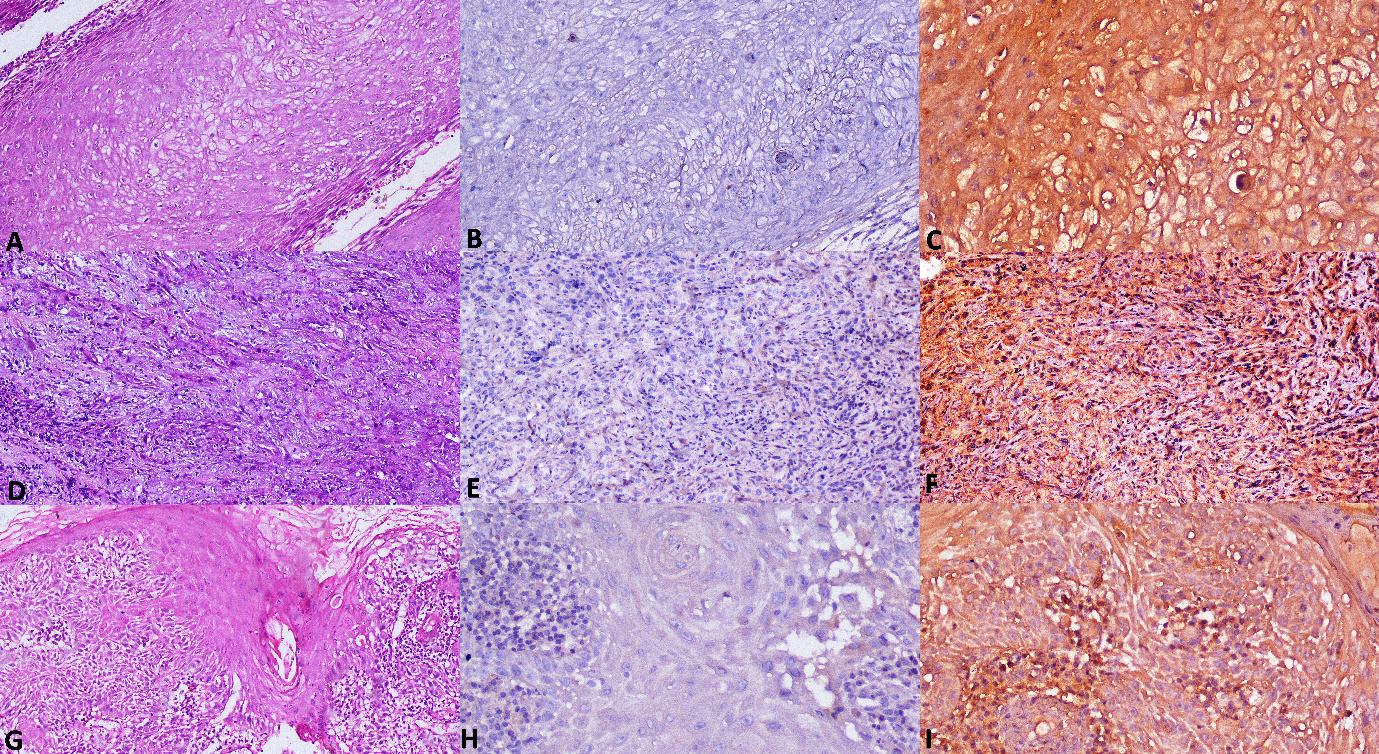
When IL-1
Caspase-3 expression and clinicopathological correlation
When Caspase-3 expression was correlated with clinicopathological parameters statistically significant result was obtained in case of nodal involvement (
Correlation of Caspase-3 expression with clinicopathologic parameters of OSCC
Correlation of Caspase-3 expression with clinicopathologic parameters of OSCC
Chi square test,
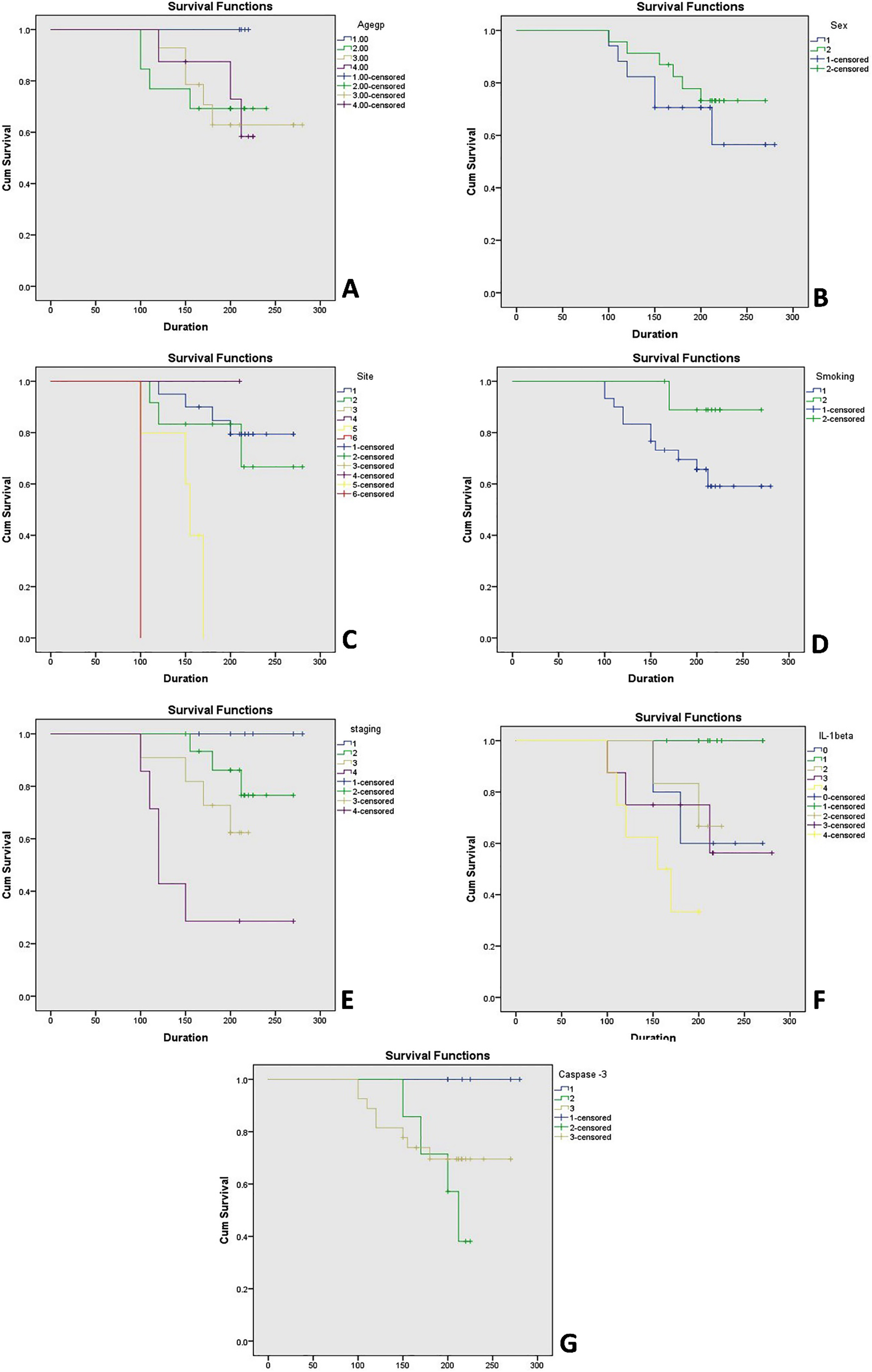
Comparison of clinicopathological parameters (age, gender, site, tobacco status and staging) with cumulative survival was done by univariate analysis using Log-Rank (Mantel-Cox) test (Table 6) and Kaplan-Meier plots (Fig. 7). Correlation of site with the survival rate in OSCC patients showed a statistically significant result for tongue carcinoma (
Comparison of clinical variables/Clinicopathological Parameters with the cumulative survival using univariate analysis [Log-Rank (Mantel-Cox) test]
Comparison of clinical variables/Clinicopathological Parameters with the cumulative survival using univariate analysis [Log-Rank (Mantel-Cox) test]
Footnotes
Acknowledgments
The authors thank Dr. Vijayalakshmi Kotrashetti, Professor and Head, Department of Oral Pathology and Microbiology and Dr. Kishore Bhat, Professor, Department of Microbiology, Maratha Mandal’s NGH Institute of Dental Sciences and Research Centre for assisting with the laboratory procedures. The authors thank Dr. Suvi Kanchan for performing statistical analysis.