Abstract
BACKGROUND:
Novel biomarkers are needed to predict the effectiveness of the treatment of presurgical neoadjuvant chemotherapy (NAC) in breast cancer (BC).
OBJECTIVE:
This is an exploratory study to assess the impact of 3 cancer-related long non-coding RNAs (lncRNAs) (H19, MALAT1 and GA5) in blood plasma of patients with BC in predicting the response to NAC.
METHODS:
The plasma levels of RNAs were relatively measured by quantitative PCR at baseline, and at the end of the fourth cycle of NAC in patients with locally advanced BC.
RESULTS:
Only H19 was associated with patients’ characteristics, and with the response to NAC. Higher plasma expression of H19 was associated with younger age at diagnosis, triple negative tumors, and Ki-67 index. Patients with a pathological complete response (20%) had lower pre-therapeutic levels of H19 compared with the non-complete responders (relative levels 0.1 vs 0.2, respectively,
CONCLUSION:
Our study reveals that H19, but not MALAT1 and GAS5, may be a useful marker of response to NAC in BC.
Keywords
Introduction
Breast cancer (BC) is currently the most common cancer in women, and second-leading cause of death from cancer. In the last decades, there has been a considerable progress in the treatment of breast cancer [11]. Pre-surgical neoadjuvant systemic therapy (NAC) has been proven to be an effective treatment option for locally advanced BC, and offers additional advantages over adjuvant systemic treatment [24]. First, NAC improves resectability of primary tumors by downstaging and increases the rate of breast-conserving surgery [21]. Second, NAC provides similar outcomes to adjuvant therapies in terms of survival, and disease progression [22]. A further advantage is the possibility of monitoring the therapy effectiveness in the early phases of therapy.
Although some patients achieve complete pathological response (pCR) from NAC, the benefit for a proportion of patients is little or lacking. Currently, there is no effective means of predicting responders from non-responders in NAC setting of breast cancer [1]. Thus, novel biomarkers for predicting the effectiveness of NAC are needed. Several markers have been tested for their potential as predictive markers of response to NAC. Among these, the proliferation marker Ki-67 was found to have predictive value of the response of breast tumors to cytotoxic drugs [17, 15]. In addition, the panel of microRNAs or PD-1 and PD-L1 expressions may be useful as biomarkers to predict treatment responses to NAC in BC [19, 26].
Liquid biopsy is an emerging field of novel cancer biomarkers. This type of biomarkers presents minimally invasive interventions and offer many additional advantages including a repeat testing at different time points [23]. Many studies investigated the relevance of circulating microRNAs in the evaluation of the response to NAC with some individual valuable molecules (for review see reference [14]). Researchers in a study investigated the value of mutated p53 in circulation (ctDNA) wherein pCR was not correlated with ctDNA [4]. Here we conducted an exploratory study to assess the value of cancer-related three long non-coding RNAs (lncRNAs) (MALAT1, GA5 and H19) in the blood plasma of patients with BC during NAC. We questioned whether pre-therapeutic plasma levels or post-therapeutic levels at the end of the fourth cycle of NAC may be useful to predict the response of breast tumors to neoadjuvant chemotherapy. We found H19, a potential biomarker for cancer detection as revealed by a recent meta-analysis [29], as a useful marker of response to NAC in BC.
Patients and methods
Patient cohort
Blood samples were drawn from patients between May 2015, and June 2016. The eligibility criteria were the primary invasive breast cancer of any tumor size and at least one tumor-positive axillary lymph node (Stage II–III) and no contraindication of patients for chemotherapy, and the provision of informed consent. The study was approved by the Institutional Review Board of Oncology Institute.
Pathology prior to neoadjuvant chemotherapy
The core-needle biopsies, taken prior to NAC were routinely processed, and stained using hematoxylin and eosin in the Pathology Unit of Istanbul Medical Faculty, and were evaluated by a senior pathologist. The tumor histology of the patients in the cohort (
Demographic and clinicopathological characteristics of BC patients
Demographic and clinicopathological characteristics of BC patients
Detection of the receptor expression in breast tumor tissues by immunohistochemistry (IHC) and lncRNA quantification in blood plasma of BC patients. IHC was performed on thin sections of breast tumor biopsies. Sections were stained with polyclonal antibodies against ER, PR and HER2. Following detection, slides were counterstained with hematoxylin. Shown are representative breast tissues with no receptor expression (A), ER (B), PR (C) and HER2 expression (D). Plasma levels of lncRNAs were measured from 200 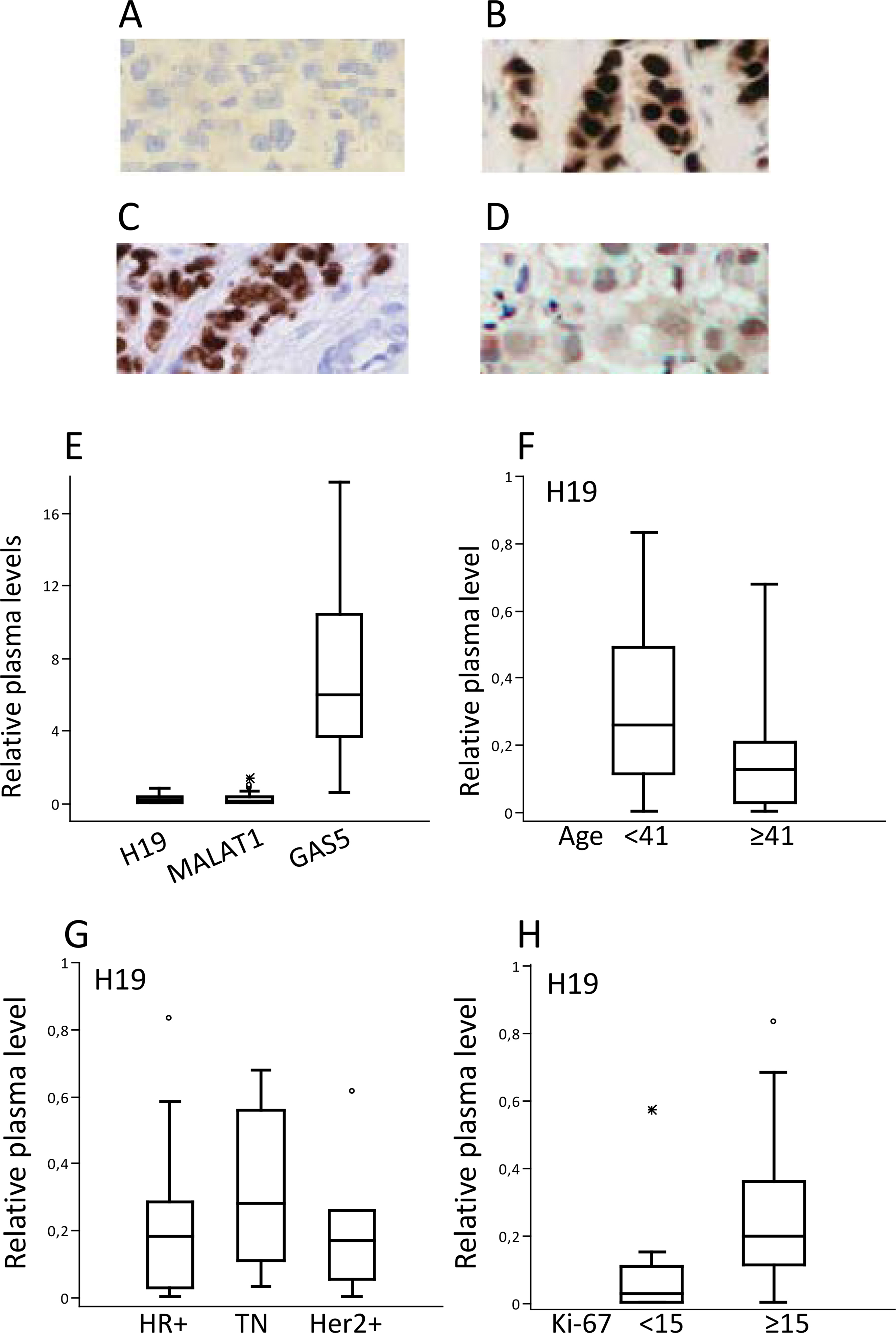
Plasma levels of MALAT1 and GAS5. Shown are pretherapeutic plasma levels of MALAT1 and GAS5 in the BC patients adjusted to the age of patients at diagnosis (A and B, respectively) and in the prognostic subgroups (C and D). Box plots display median, interquartile range (25%–75% percentiles) and lowest and highest levels of the distribution. Symbols * and 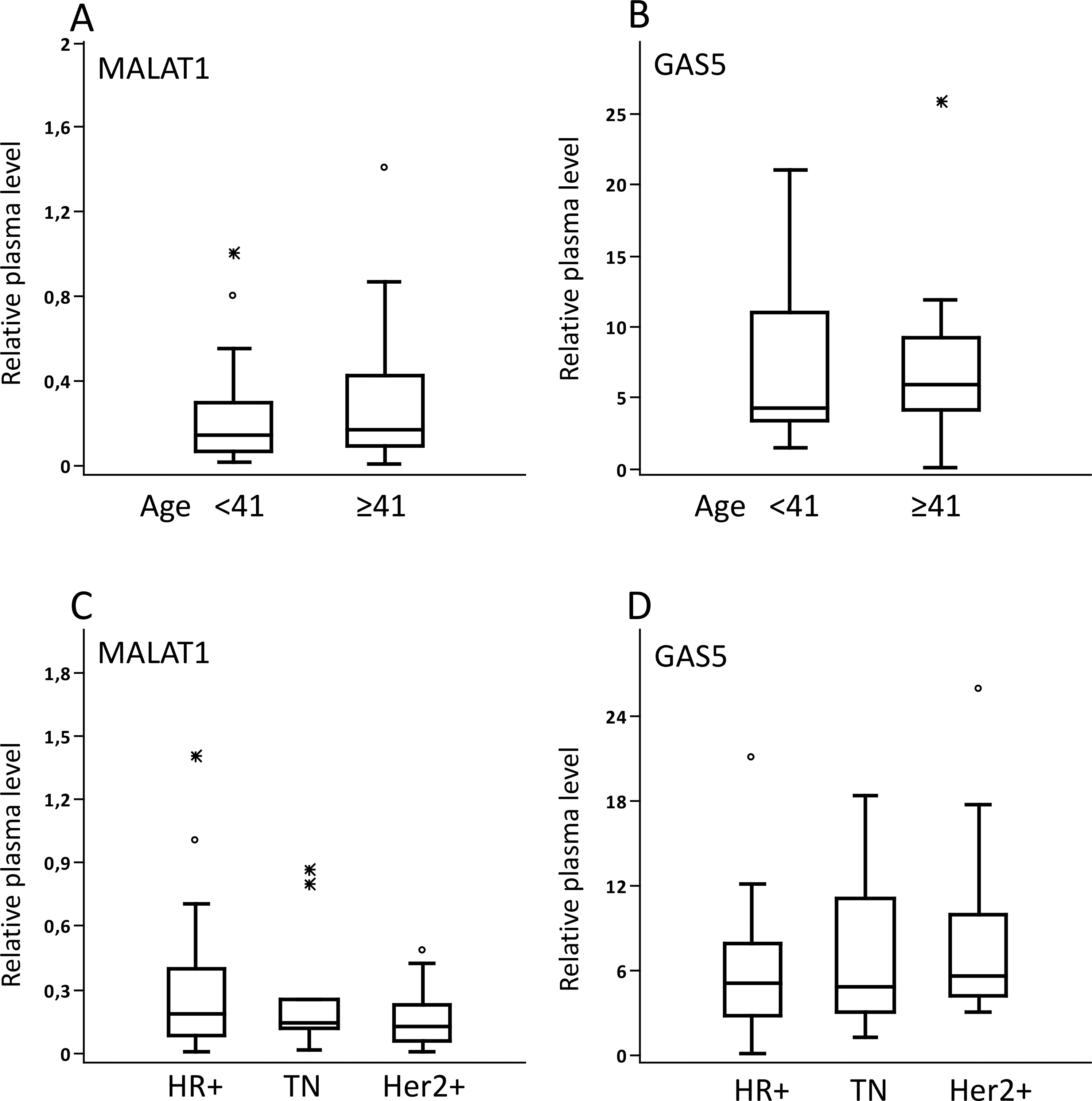
In our study, pathological complete response (pCR) was defined as no residual invasive disease in the breast, and in the lymph nodes. Any remaining positive lymph nodes or residual disease in the breast due to partial tumor response was defined as pathological non-complete response.
lncRNA quantification in blood plasma
Total RNA was extracted from 200
Statistical analyses
The Mann-Whitney test was employed to assess the association of plasma levels of H19, MALAT1 and GAS5 with clinicopathological characteristics. The Wilcoxon signed-rank test was utilized to evaluate the plasma levels of lncRNAs at baseline, and the fourth cycle of neoadjuvant chemotherapy. The correlation between individual lncRNA molecules was determined using the Pearson’s test. Statistical analyses were conducted using the Statistical Package for the Social Sciences (SPSS) version 16.0 (SPSS, Inc., Chicago, IL, USA). Values
Change of plasma levels lncRNA in the BC patients during neoadjuvant chemotherapy. Shown are relative plasma levels of H19 (A), MALAT1 (B) and GAS5 (C) at baseline and at the fourth cycle of neoadjuvant chemotherapy. Box plots display median, interquartile range (25%–75% percentiles) and lowest and highest levels of the distribution. Symbols * and 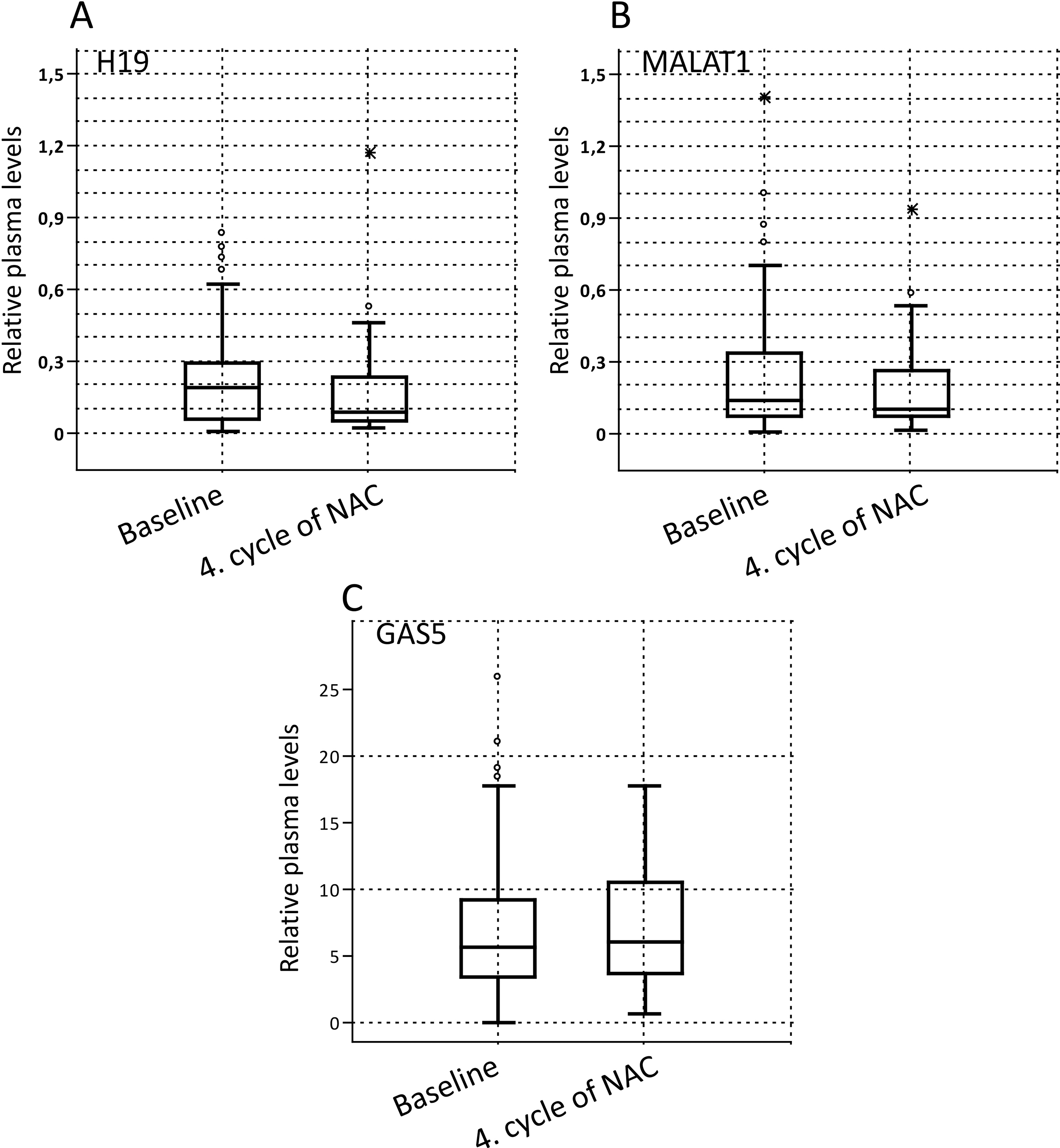
Correlation between MALAT1 and GA55 and dynamics of plasma levels of H19 during therapy. The linear correlation between pretherapeutic levels (A) and levels at the fourth cycle of neoadjuvant chemotherapy (B) of MALAT1 and GAS5. Correlation coefficients at those points were 0.82 and 0.47, respectively. C. plasma levels of H19 at baseline and at the fourth cycle of chemotherapy in individual patients. 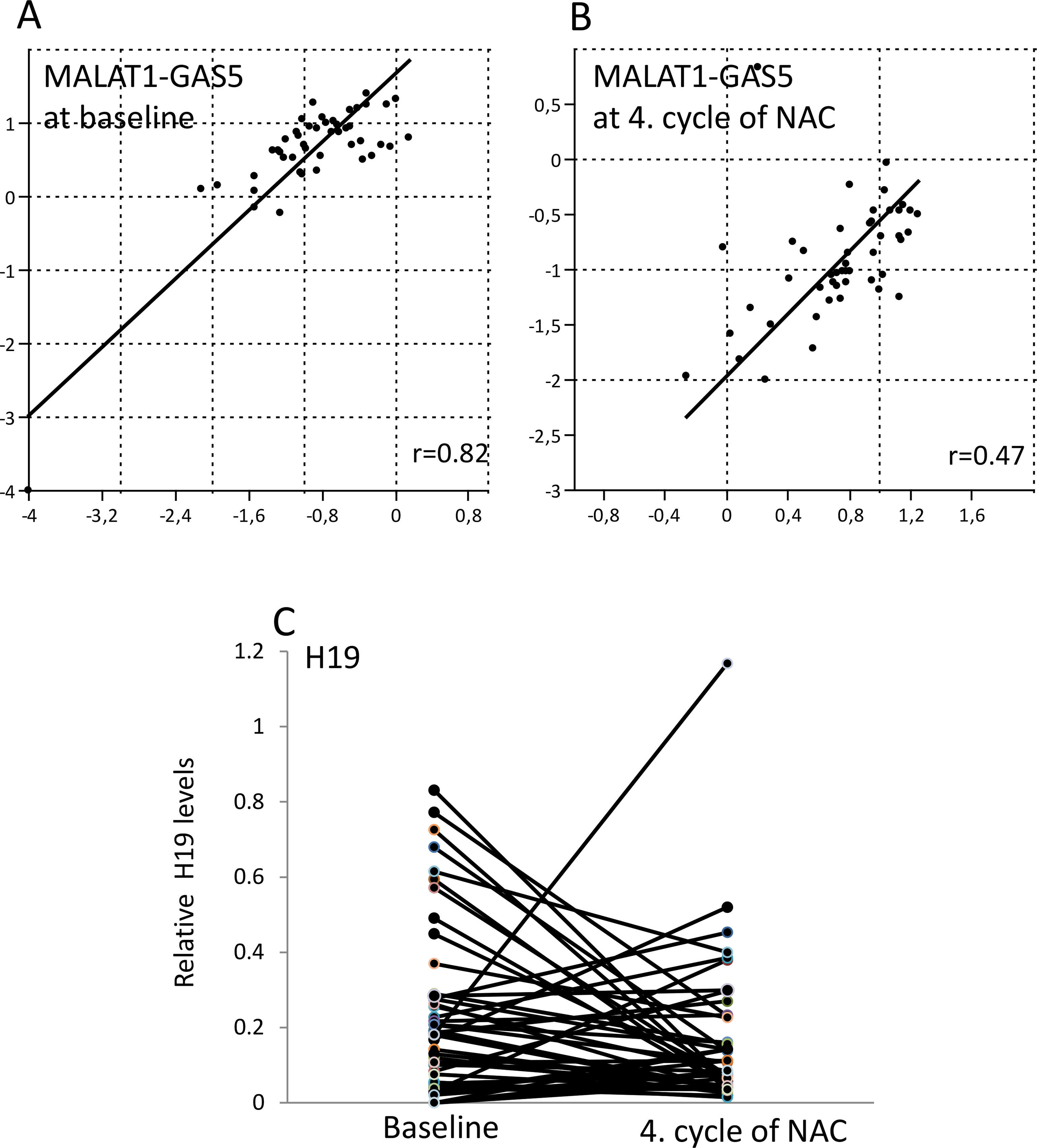
Impact of H19 in pathological response. A. Baseline H19 levels in the patients with a pathological complete response or partial response. B. The ROC curve for H19 expression in respect to the response to cytotoxic therapy, with an AUC value of 69% for differentiating those with complete response from those with a patial response. C. Baseline H19 levels in the patients with a remaining tumors of 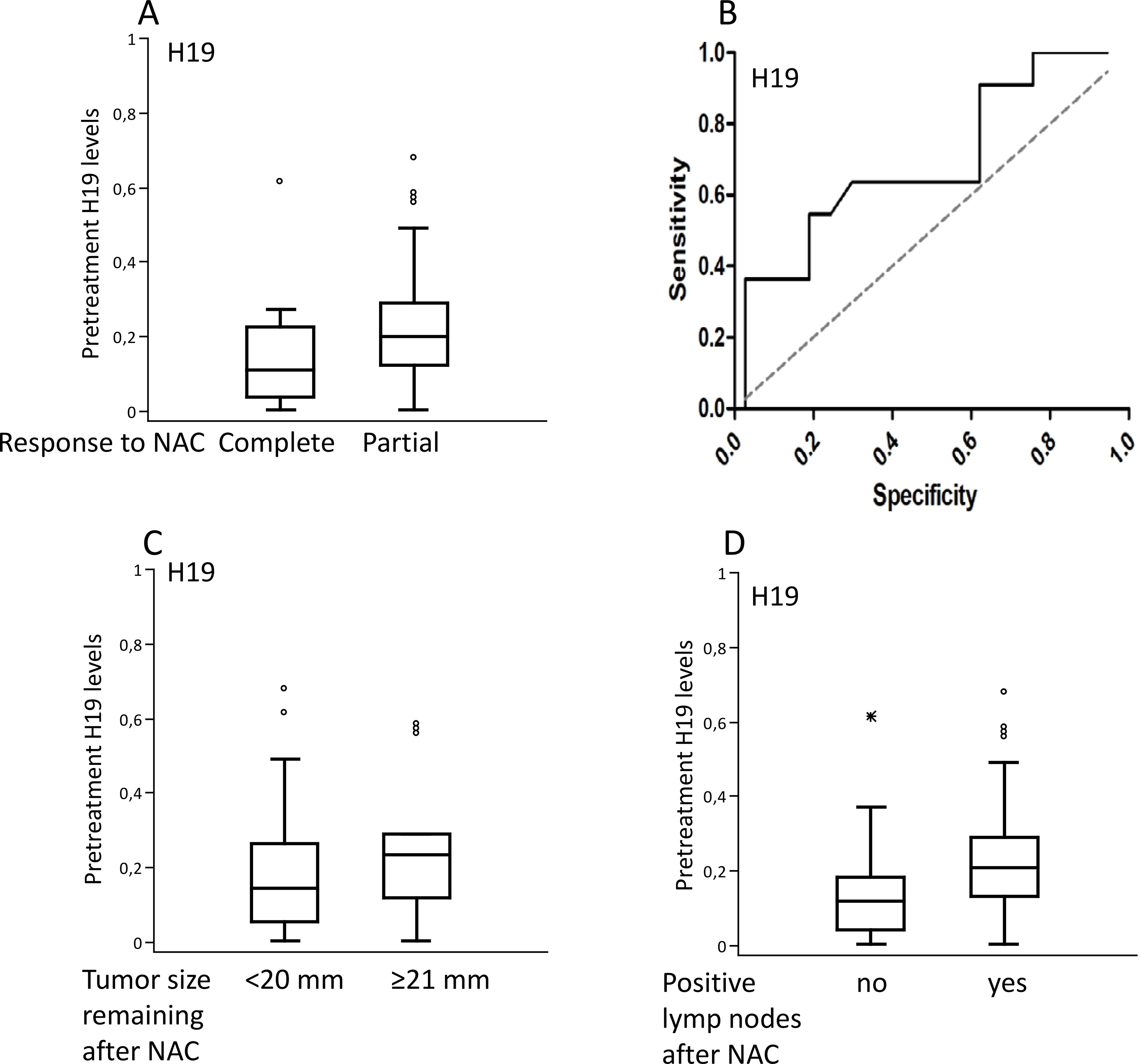
As seen in Fig. 1E, GAS5 was the most abundant molecule in the plasma of the BC patients whereas H19 and MALAT1 had similar plasma levels. We looked at the association of the pretreatment levels of three molecules with demographic and clinicopathological characteristics of the BC patients. Any association to patients’ traits was observed solely for H19. Interestingly, younger patients (
From baseline to fourth cycle of NAC, a notable change of plasma levels was detected for only H19 in all the study group (Fig. 3A). During this period, H19 levels in the complete cohort significantly declined (relative median levels 0.18 vs 0.08, respectively;
Subsequently, we stratified the BC patients in accordance with the response to neoadjuvant chemotherapy. Eleven patients out of 55 (20%) were found to have pCR to NAC (the complete responders). However, the remaining patients had a partial response (the non-complete responders). We compared the plasma levels of lncRNAs between these two groups and found that the patients with a pCR had lower pre-treatment levels of H19 than the levels of H19 in non-complete responders (relative median levels 0.1 vs 0.2,
Discussion
Chemoterapy in the neoadjuvant setting has increasingly been used in patients with early-stage or locally advanced BC. However, the prediction of the response to NAC remains a challenging issue [25]. Biomarkers in tumor tissues or bodily fluids could be useful to identify responders or non-responders. For instance, by repurposing existing microarray data from Gene Expression Omnibus, expression levels of some lncRNAs in breast tumors have been shown to be useful in predicting pCR [5, 18] which indicated that non-coding RNAs may reflect the response of breast tumors to NAC. In the present study we used the circulating H19, MALAT1 and GAS5 in the evaluation of response of breast tumors to NAC. We found that only H19 out of three molecules was associated with patients’ clinical characteristics or with pCR. This suggests that the BC specificity of H19 may be higher than the BC specificity of MALAT1 and GAS5. In line with this, the recent data reveals that H19, an imprinted oncofetal RNA and potential cancer biomarker [29], is involved in BC development [9] as its expression is increased in breast tumors [18], and promotes the proliferation and invasion of BC cells [16, 28]. As a reflection of the overexpression in BC tissues, H19 can be detected in increased quantities in blood plasma of BC patients compared with the blood plasma of the healthy individuals [12, 27]. We found that the pre-therapeutic plasma levels of H19 were higher in patients who were younger at diagnosis, with triple-negative tumors or higher Ki-67 index suggesting that H19 is a marker of aggressive breast cancer. As BC has a more aggressive profile in women below 40 years than in women older than 40 years [13] and triple negative BC is an aggressive subtype of BC [3], H19 might be involved in the development of aggressive BC. The regulation of BC stemness by H19 could be one of the mechanisms for this effect as suggested recently [20], particularly in triple-negative subtype [7]. Similarly, H19 expression was found to be higher in paclitaxel-resistant triple-negative cells than in paclitaxel-sensitive triple-negative cells [10]. Furthermore, recent studies provide evidence that H19 is upregulated in tamoxifen-resistant breast cancer cell lines and tumor tissues and thereby contribute to tamoxifen resistance [7, 13]. These findings suggest that H19 may be associated with aggressive BC.
We demonstrated that patients with lower baseline levels of H19 are more likely to have a complete response to NAC than those with higher pre-therapeutic H19 levels. To our best knowledge, this is the first data indicating an association between circulating H19 and pCR to NAC. This concordance to pCR could not be found when H19 levels at fourth cycle of NAC were employed suggesting the baseline H19 levels are determinants of the response to NAC. We determined a role for H19 also in non-complete responders in addition to complete responders. Interestingly, patients with smaller remaining tumors (
In conclusion, this exploratory study provides evidence that H19, but not MALAT1 and GAS5, may be a marker of response to NAC in breast cancer. The use of H19 in the evaluation of the response to NAC is an additional modality to its potential therapeutic and diagnostic use as currently suggested in several cancers including BC [8]. In this sense, H19 could be combined with the other promising biological markers or with the functional imaging technologies [6] in predicting the response to cytotoxic therapy. Our findings are encouraging and may provide the basis to further explore the role of circulating H19 as a minimally invasive marker in the evaluation of response to NAC in larger studies in BC.
Footnotes
Acknowledgments
The present work was supported by a grant from Istanbul University Scientific Projects Coordination Unit, Istanbul, Turkey (awarded to UG, grant no. TYO-2017-26615).
Conflict of interest
The authors declare that they have no competing interests.
