Abstract
Circular RNAs (circRNAs) have gained attention for their involvement in carcinogenesis, but its functional effects in breast cancer (BC) remains largely unclear. In this study, we aimed to explore the expressing pattern, clinical significance and potential function of a newly identified circRNA, hsa_circ_001569, in BC. RT-PCR was performed to detect the expression of hsa_circ_001569 in both BC tissues and cell lines. The associations between hsa_circ_001569 expression and clinicopathological features and prognosis in BC patients were statistically analyzed. Next, we investigated the effects of hsa_circ_001569 on the proliferation, apoptosis, migration and invasion in BC cells lines. The effects of abnormal hsa_circ_001569 expression on EMT pathway and PI3K/AKT pathway were determined using Western blot. We found that hsa_circ_001569 expression was significantly up-regulated in both BC tissues and cell lines. Overexpression of hsa_circ_001569 was associated with Lymph node metastasis, advanced clinical stage and shorter overall survival. Multivariate assay confirmed that hsa_circ_001569 expression was an independent prognostic factor for 5-year overall survival. Furthermore, functional investigations revealed that knockdown of hsa_circ_001569 significantly suppressed the growth and metastatic potentials of BC cells. Besides, molecular mechanistic study revealed that depression of hsa_circ_001569 impeded the activation of PI3K-AKT signaling in BC cells. Our results indicated that hsa_circ_001569 upregulation was associated with BC lymph-node metastasis, clinical stage, and poor prognosis. Hsa_circ_001569 might contribute to progression of BC by modulating PI3K-AKT pathway.
Introduction
Breast cancer is the most common malignant cancer type among women worldwide and a serious disease threatening the health of women [1]. The burden of this disease is increasing in China, with more than one hundred and ninety thousand people being diagnosed and one hundred and fifteen thousand people dying of the disease each year [2, 3]. Although BC has been studied extensively for decades, the potential mechanism of BC progression remains largely unclear [4]. With the development of methods in early detection and adjuvant treatment, BC patients diagnosed at early stages have achieved favorable prognosis [5, 6]. However, most patients diagnosed at an advanced stage were incurable with a 5-year survival rate of 20–40% [7]. The poor prognosis is due to the emergence of metastasis to different organs [8]. Therefore, various studies have been conducted to investigate the molecular abnormalities associated with BC metastasis.
Circular RNAs (circRNAs) are a class of noncoding RNA characterized by covalently closed circular loop with lacking capability of protein coding [9]. Previously, circRNAs are considered to be “genomic transcripts junks”. However, more and more evidences indicate that circRNAs can regulate gene expression at the transcriptional or posttranscriptional level, and thereby are involved in the modulation of various biological progression, such as proliferation, cells differentiation and apoptosis [10, 11]. Recently, the development of sequencing technology makes it easy to screen dysregulated circRNAs [12]. On the other hand, the regulating effects of circRNAs in tumor progression attracts increasing attention because of their functional roles in tumor proliferation and metastasis [13, 14]. These previous findings highlighted the important potential of circRNAs as novel biomarkers and therapeutic targets for diverse tumors, including BC. However, unlike other noncoding RNAs, miRNA and long noncoding RNA (lncRNA), only a few circRNAs were identified and functionally characterized [15]. Therefore, certifying new circRNAs which served as essential roles in progression of cancer is still urgently needed. Besides, phosphoinositide 3-kinase (PI3K)/protein kinase (Akt) signaling played critical roles in multiple aspects of malignant cancer behaviors [16]. The abnormal activation of PI3K/Akt signaling was relevant with the progression of most cancers, including BC [17]. Although miRNAs and lncRNAs had influence on PI3K/Akt signaling, whether circRNAs were able to modulate PI3K/Akt signaling in BC still needed to be elucidated [18].
Hsa_circ_001569, located on chromosome 16q13.1 on the plus strand, is a newly identified circRNA. Previously, up-regulation of hsa_circ_001569 has been reported in gastric cancer and colorectal cancer and its tumor-promotive roles have also been confirmed [19, 20]. However, the expression pattern and functional effects of hsa_circ_001569 in BC have not been investigated. In this paper, we aimed to investigate hsa_circ_001569 as prognostic biomarker in BC and its role in tumor progression.
Materials and methods
Patients and tissue samples
This study included 145 patients who undergone breast cancer associated surgeries at The Affiliated Huaian No. 1 People’s Hospital of Nanjing Medical University between May 2008 and April 2010. All the patients did not receive antitumor treatment before surgery. The clinicopathological characteristics of patients were summarized in Table 1. All collected tissues were stored at
Cell lines and cell transfection
One normal human breast epithelial cell line, MCF-10A, and four human breast cancer cell lines including MCF-7, MDA-MB-468, MDAMB-231 and MDA-MB-453, were purchased from Beijing Zhongyuan Biotechnology Co., Ltd. (Chaoyang, Beijing, China). The RPMI-1640 medium (BioDee Biotech, Haidian, Beijing, China) containing 10% FBS was applied to culture these cells at 37
Primer sequences used for RT-PCR in this study
Primer sequences used for RT-PCR in this study
Cell transfection was conducted using a GenMute siRNA transfection kit (SignaGen, Jinan, Shandong, China). Briefly, the MCF-7 or MDA-MB-231 cells were firstly placed into plates, and continued to be cultured for 24 h. Then, proper amount of small interfering RNAs (siRNAs) was added into the GenMute siRNA transfection reagent, and the mixture was added into the cells after 15 min. After 5–8 h, the medium was changed and the cells were continued to be cultured for further use. The siRNAs against has_circ_001569 (siRNA#1 and siRNA#2) or negative control siRNAs (si-NC) were purchased from Genomeditech Co., Ltd. (Pudong, Shanghai, China), and these siRNAs were chemically modified with 2’-OMe, which could effectively enhance the stability of these siRNAs.
The correlations between hsa_circ_001569 expression and clinicopathological factors of BC patients
Total RNA from breast cancer tissues or cells was isolated using TRIzol regent (Aladdin, Fengxian, Shanghai, China), following being reversely transcribed to cDNA using EpiNext Hi-Fi cDNA Synthesis kit (AmyJet, Wuhan, Hubei, China). The qRT-PCR assays for has_circ_001569 examination were conducted by the use of Bestar SYBR RT qPCR kit (XinghanBio, Xuhui, Shanghai, China). The relative expression of has_circ_001569 was calculated using 2
Western blot assay
Summary of univariate and multivariate Cox regression analyses of overall survival duration
Summary of univariate and multivariate Cox regression analyses of overall survival duration
The has_circ_001569 siRNAs transfected MCF-7 and MDA-MB-231 cells were lysed using an AmyJet cell protein extraction kit (Wuhan, Hubei, China). Subsequently, the cellular lysates from each sample were subjected to 8–10% SDS-PAGE, following being transferred to PVDF membranes, and sequentially examined by corresponding primary antibodies using ECL reagent (Yeason, Pudong, Shanghai, China). The primary antibodies used in this study were as follows: anti-N-cadherin antibody (BOSTER, Wuhan, Hubei, China), anti-vimentin antibody (Abcam, Pudong, Shanghai, China), anti-p-PI3K antibody (Cell Signaling Technology, Beverly, MA, USA), anti-p-AKT antibody (Cell Signaling Technology, Beverly, MA, USA), anti-PI3K antibody (Abcam, Pudong, Shanghai, China), anti-AKT antibody (Protein Tech Group, Wuhan, Hubei, China), anti-GAPDH antibody (Protein Tech Group, Wuhan, Hubei, China).
Briefly, MCF-7 and MDA-MB-231 cells were separately transfected with has_circ_001569 siRNAs or negative control siRNAs. Subsequently, the treated cells were seeded in 96-well culture plates (at 1
Colony formation assay
In brief, MCF-7 and MDA-MB-231 cells were firstly separately transfected with has_circ_001569 siRNAs or negative control siRNAs. Then, the 6-well plates were added with 500 siRNAs treated MCF-7 or MDA-MB-231 cells. After culturing for 2 weeks, 95% methanol was applied to fix the cell colonies, and crystal violet (0.1%; Aladdin, Fengxian, Shanghai, China) was used to stain the colonies. The stained cell colonies were observed and counted using a microscope (Yongke Optical Instrument, Jinshan, Shanghai, China).
Cell apoptosis analysis
The apoptotic rates of MCF-7 and MDA-MB-231 cells after transfection of has_circ_001569 siRNAs were measured using an Annexin V flow cytometry apoptosis assay kit (JiaMay Biotech, Chaoyang, Beijing, China) on a Quanteon flow cytometer (ACEABio, Hangzhou, Zhejiang, China). In short, the cells were collected and resuspended in the mixture of annexin V-FITC and PI in the dark. After 10–15 min, the cells were washed with PBS and sequentially subjected to the flow cytometer to analyze the cell apoptosis.
Hsa_circ_001569 was up-regulated in breast cancer tissues and cells. (A) The expression levels of hsa_circ_001569 in 145 paired BC tissues and adjacent non-tumor breast tissue by RT-PCR. (B) hsa_circ_001569 expression was significantly higher in BC patients with a higher pathological stage. (C) qRT-PCR analysis of hsa_circ_001569 expression in MCF-7, MDA-MB-468, MDAMB-231, MDA-MB-453 cells and MCF-10A. (D) Kaplan–Meier postoperative survival curve for patients with BC and hsa_circ_001569 expression. *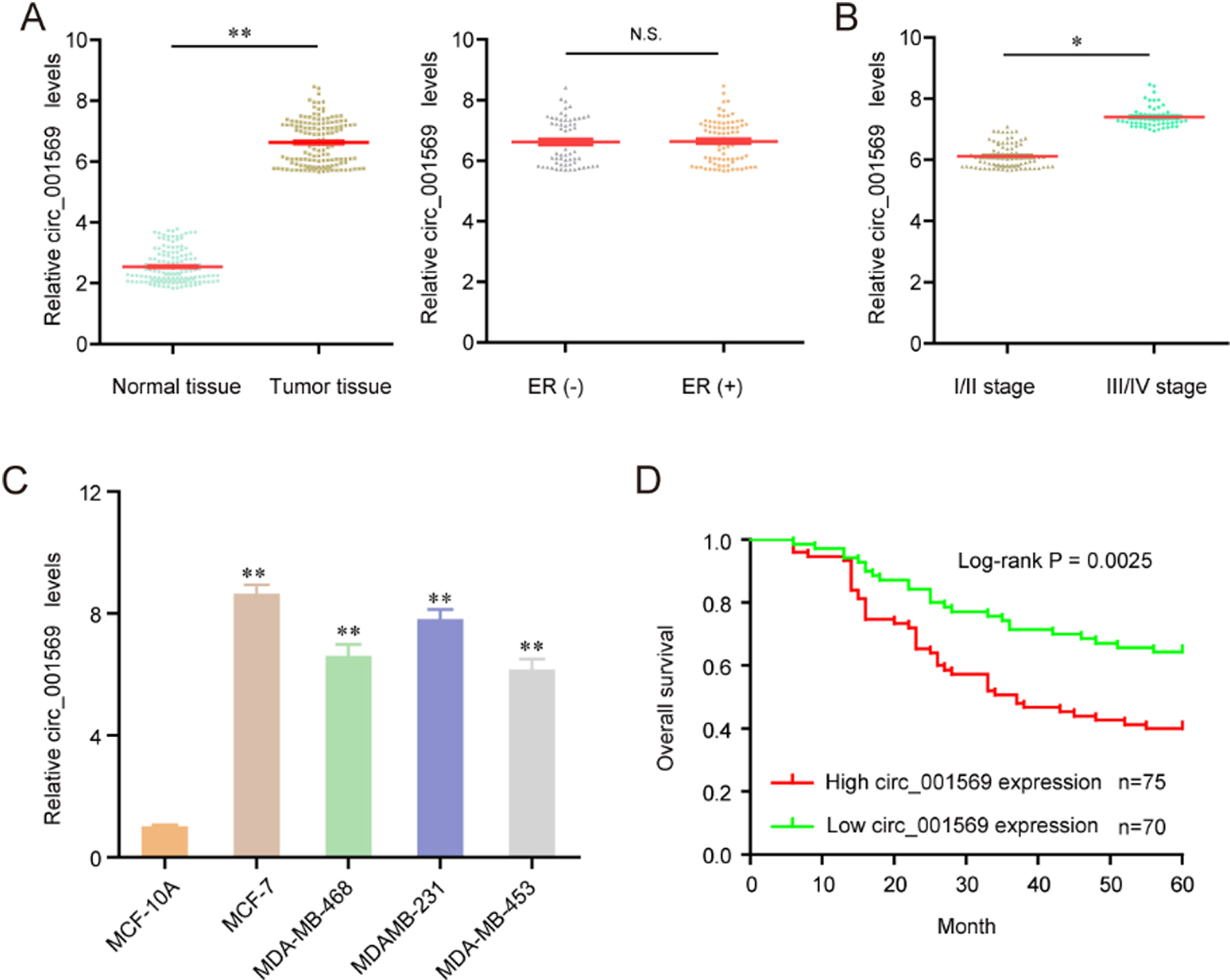
After 24 h of has_circ_001569 siRNAs treatment, MCF-7 and MDA-MB-231 cells were cultured in 12-well plates until the cell confluence reached to about 100%. Subsequently, the wounded areas were generated by scraping the cells using a 200-
Knockdown of hsa_circ_001569 suppressed the proliferation and induced the apoptosis of MCF-7 and MDA-MB-231 cells. (A) Targeted siRNAs (siRNA#1 and siRNA#2) efficiently inhibited the expression levels of hsa_circ_001569 in the MCF-7 and MDA-MB-231 cells. (B) Hsa_circ_001569 inhibition depressed cell proliferation determined by CCK-8 assay. (C and D) The number of MCF-7 and MDA-MB-231 cell colonies was decreased after transfection of hsa_circ_001569 siRNAs. (E) The cell apoptosis was determined by flow cytometry and silencing of hsa_circ_001569 elevated the apoptotic rates of MCF-7 and MDA-MB-231 cells. *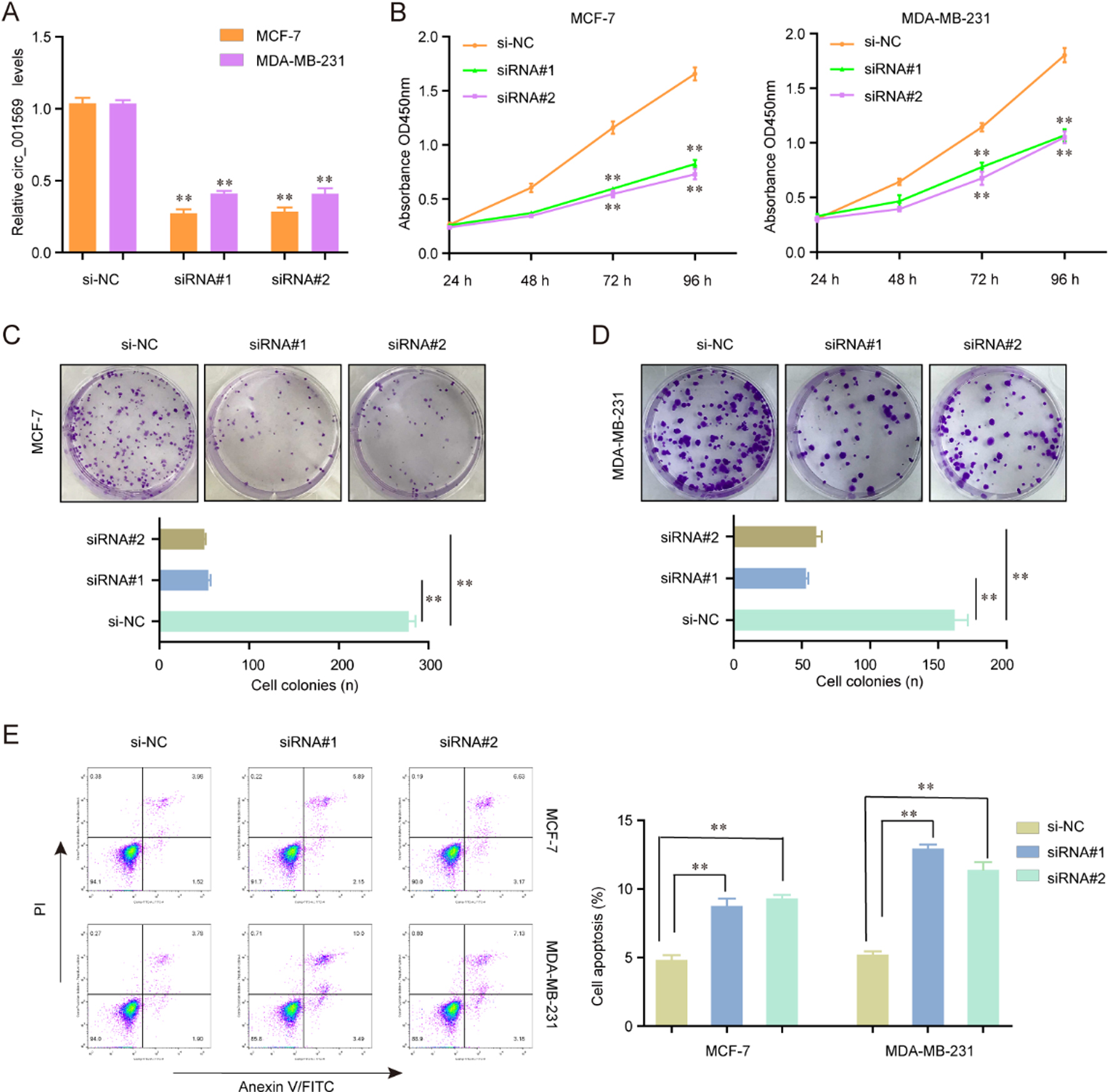
The has_circ_001569 siRNAs transfected MCF-7 and MDA-MB-231 cells (2.5
Statistical analysis
We applied SPSS program (version 19.0; SPSS, Chicago, IL, USA) for statistical analysis. Two-tailed Student’s t-test was utilized for comparisons between two groups, while One-way ANOVA was employed for multiple comparisons. The Kaplan-Meier method with the log-rank test was conducted for overall survival analysis. The Cox proportional hazards regression model was employed for univariate and multivariate analyses to estimate the prognostic factors for survival prediction. A
Suppression of hsa_circ_001569 impeded the invasion and migration of MCF-7 and MDA-MB-231 cells. (A and B) Silencing of hsa_circ_001569 reduced the migratory capacity of MCF-7 and MDA-MB-231 cells. (C and D) The invaded cell number of MCF-7 and MDA-MB-231 cells was decreased after transfection of hsa_circ_001569 siRNAs. (E and F) Depression of hsa_circ_001569 reduced the N-cadherin and vimentin protein levels. *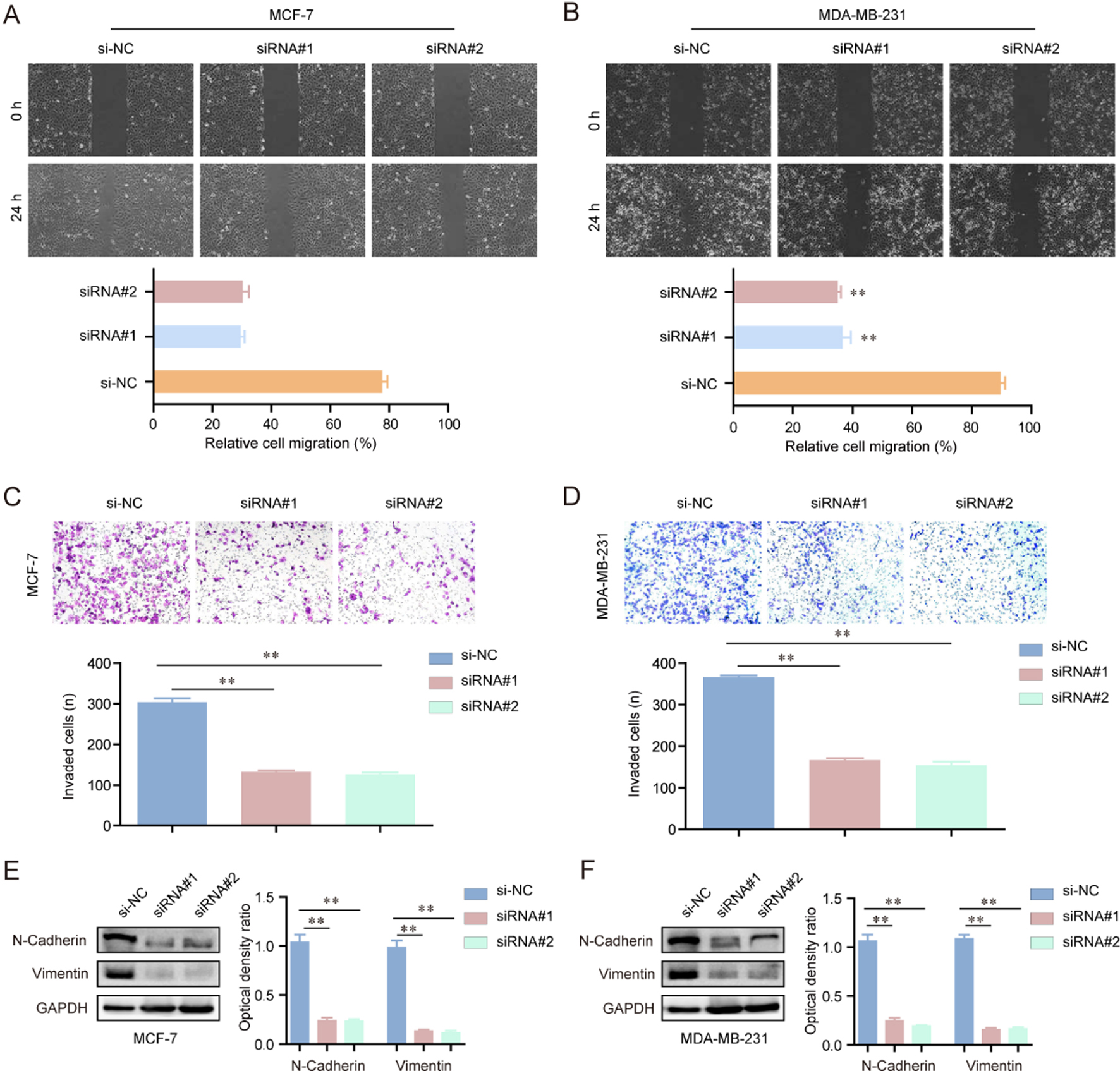
The inhibitory effects of hsa_circ_001569 knockdown on the activation of PI3K/AKT signaling in MCF-7 and MDA-MB-231 cells. (A) Western blot assay detected the protein levels of p-PI3K, PI3K, p-AKT and AKT in MCF-7 cells. Knockdown of hsa_circ_001569 reduced the expression of p-PI3K and p-AKT, but not PI3K and AKT. The optical density of the protein bands was also analyzed by Image J software (NIH, Bethesda, ML, USA). (B) The protein levels of p-PI3K, PI3K, p-AKT and AKT in MDA-MB-231 cells were also analyzed. *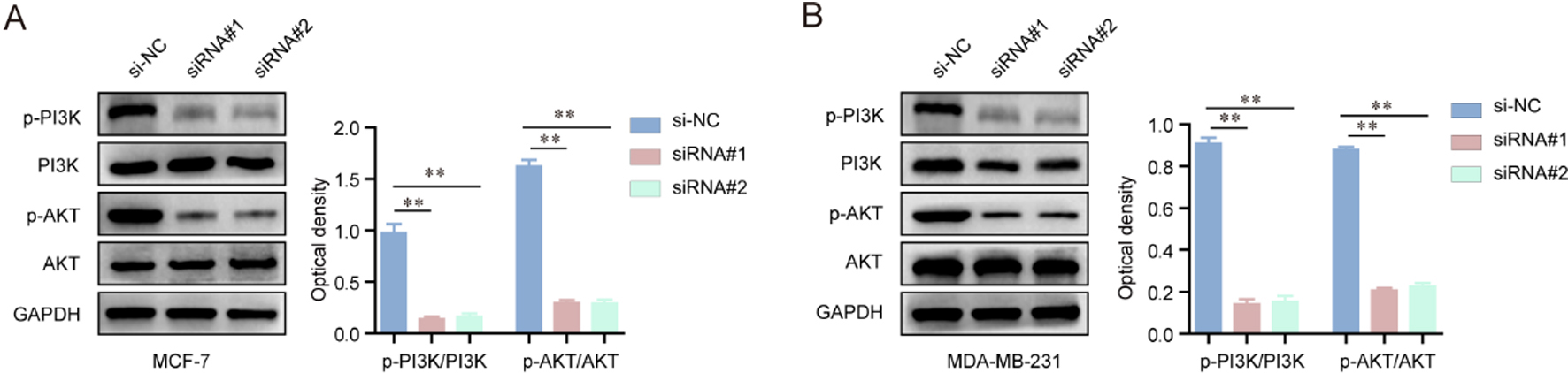
Up-regulation of hsa_circ_001569 predicted poor prognosis in BC
In order to explore whether hsa_circ_001569 was abnormally expressed in BC, we performed RT-PCR to detect its expression in BC tissues and cell lines. As shown in Fig. 1A, we found that hsa_circ_001569 expression was significantly up-regulated in BC tissues compared to matched normal breast tissues (
Then, the relationship between hsa_circ_001569 expression and clinicopathologic features was evaluated. All BC patients were divided into a high expression group (
Depression of hsa_circ_001569 suppressed cellular proliferation and induced apoptosis of BC cells
To investigate the biological functions of hsa_circ_ 001569 in vitro, firstly, we synthesized the siRNAs targeting hsa_circ_001569 (siRNA#1 and siRNA#2), and sequentially transfected them into MCF-7 and MDA-MB-231 cells. Thereafter, qRT-PCR assays were carried out and the results suggested that the hsa_circ_ 001569 expressing levels were significantly inhibited in MCF-7 and MDA-MB-231 cells when they were transfected with hsa_circ_001569 siRNAs (Fig. 2A). In addition, the cellular growth was remarkably depressed in MCF-7 and MDA-MB-231 cells after transfection of hsa_circ_001569 siRNAs with the examination of CCK-8 assays (Fig. 2B). According to the data of colony formation assays, we discovered that there were more MCF-7 or MDA-MB-231 cell colonies formed in the hsa_circ_001569 silencing groups when compared with the controls (Fig. 2C and D). Moreover, we next employed flow cytometry to assess the effects of hsa_circ_001569 on cell apoptosis. As the data shown in Fig. 2E, we found that the hsa_circ_001569 siRNAs transfected MCF-7 and MDA-MB-231 cells presented decreased proliferative rates than the negative control siRNAs (si-NC) transfected cells, implying that knockdown of hsa_circ_001569 promoted apoptosis of MCF-7 and MDA-MB-231 cells. Collectively, these data indicated that hsa_circ_001569 played crucial roles in regulating the proliferation and apoptosis of BC cells.
Knockdown of hsa_circ_001569 inhibited the migration and invasion of BC cells and influenced EMT markers expression
In order to explore the effects of hsa_circ_001569 knockdown on the metastatic potentials of BC cells, wound healing and transwell invasion assays were carried out using MCF-7 and MDA-MB-231 cells. As the results presented in Fig. 3A and B, inhibition of hsa_circ_001569 impeded the migratory rates of MCF-7 and MDA-MB-231 cells in contrast with controls, suggesting that hsa_circ_001569 could enhance the migratory ability of BC cells. Furthermore, transwell invasion assays confirmed that repressing the expression of hsa_circ_001569 in MCF-7 and MDA-MB-231 cells notably reduced cellular invasiveness (Fig. 3C and D). Besides, we further conducted western blot assays to evaluate the changes of epithelial-mesenchymal transition makers (N-cadherin, vimentin) in BC cells after transfecting hsa_circ_001569 siRNAs. The results demonstrated that inhibition of hsa_circ_001569 markedly reduced the expression of the two mesenchymal markers, N-cadherin and vimentin, in MCF-7 and MDA-MB-231 cells (Fig. 3E and F). Overall, these data indicated a promoting role of hsa_circ_001569 in BC cell migration and invasion.
Silence of hsa_circ_001569 impeded the activation of PI3K-AKT signaling in BC cells
We next aimed to elucidate the potential molecular mechanisms by which hsa_circ_001569 contributed to the phenotypes of BC cells. Since accumulating studies had validated that PI3K-AKT signaling exhibited extensively regulatory roles in the development and progression of multiple types of cancers, we next conducted western blot assays to assess the effects of hsa_circ_001569 knockdown on the activity of PI3K-AKT signaling. Therefore, hsa_circ_001569 siRNAs were firstly transfected into MCF-7 and MDA-MB-231 cells, and subsequently the protein levels of molecules involved in PI3K-AKT signaling were detected. The results demonstrated that the protein levels of phosphorylated PI3K (p-PI3K) and phosphorylated AKT (p-AKT) were markedly reduced in MCF-7 and MDA-MB-231 cells after transfection of hsa_circ_001569 siRNAs, while there were no changes of PI3K and AKT expression in these cells (Fig. 4A and B). Therefore, these results indicated that knockdown of hsa_circ_001569 depressed the activation of PI3K-AKT signaling in BC cells.
Discussion
BC has become a major killer across the globe. Poor prognosis of BC patients encourages us to discover novel biomarkers for diagnosis and prediction of prognosis to improve the clinical survival rates. Recently, it was reported that circRNAs were abnormally expressed and associated with clinical prognosis of BC patients, suggesting that circRNAs might be novel tumor biomarkers [21, 22]. In this study, our attention focused on a newly identified circRNA, hsa_circ_001569; we firstly found that the expressing levels of hsa_circ_001569 was significantly up-regulated in both clinical BC tissues and cell lines, which was consistent with the expression trend of hsa_circ_001569 in gastric cancer and colorectal cancer. Then, we explored its clinical significance in BC patients, finding that high hsa_circ_001569 expression was associated with lymph node metastasis and clinical stage, suggesting that it might be involved in the regulation of BC metastasis. Moreover, the results of Kaplan-Meier assays indicated that hsa_circ_001569 overexpression was conversely associated with patient’s overall survival. Further multivariate analyses confirmed that high expression of hsa_circ_001569 was a poor independent prognostic factor for patients with BC, which highlighted the great potential of hsa_circ_001569 as a novel biomarker.
Recently, hsa_circ_001569 was reported to play functional roles in gastric cancer and colorectal cancer. Xie et al. reported that hsa_circ_001569 was highly expressed in colorectal cancer and its overexpression promoted the proliferation and invasion of colorectal cancer cells by targeting miR-145 [20]. Shen et al. showed that the expressing levels of hsa_circ_001569 was up-regulated in gastric cancer. Functional investigations indicated that knockdown of hsa_circ_001569 dramatically decreased cellular viability and accelerated cell apoptosis by modulating miR-145 in gastric cancer [19]. These results revealed that hsa_circ_001569 might act as a positive regulator in the progression of tumors. However, its function in BC remains unknown. In this study, CCK-8 assay, colony formation assay, cell apoptosis analysis, wound healing assay, and transwell assays were performed to investigate the functions of hsa_circ_001569 in the BC cells (MCF-7/MDAMB-231). Our results showed that knockdown of hsa_circ_001569 suppressed BC cells proliferation, migration and invasion, and promoted apoptosis. In addition, in order to explore the mechanism of metastasis, we performed Western blot to explore the effects of hsa_circ_001569 on EMT pathway, finding that its knockdown inhibited the progress of EMT. Overall, our results suggested that hsa_circ_001569 may be a promising novel target for BC therapy.
Some key signaling pathways, such as Wnt/
There are several limitations in this study. Firstly, because of the small number of patients involved in this study, more studies on a great number of patients are necessary to further confirm our results. Secondly, all patients are Asian, thereby assumptions may not translate to Caucasian or African ethnicities. Finally, Further mechanism by which hsa_circ_001569 modulated PI3K-AKT pathway needed to be further studied.
In summary, our study firstly provided evidence that hsa_circ_001569 upregulation was closely associated with BC lymph-node metastasis and clinical stage, and may be an independent risk factor for poor prognosis. Additionally, our results indicated that hsa_circ_001569 may contribute to progression of BC by modulating PI3K-AKT pathway. These results suggest that hsa_circ_001569 may be a target for BC therapy in the future.
Footnotes
Conflict of interest
All authors declare that they have no conflict of interest.
