Abstract
Bladder cancer is frequently occurred in urinary system and has complicated pathogenesis factors including both genetics and environmental factors that have not been fully illustrated. Hypoxia can further induce tumor progression. ROCK2 has abnormal expression in various tumors but its expression or functional role in bladder cancer have not been illustrated. In vitro cultured bladder cancer cell line T24 was randomly assigned into control group, hypoxia group (prepared under hypoxic culture), and ROCK2 siRNA group (transfected with ROCK2 siRNA after hypoxia treatment). Real-time PCR and Western bot measured ROCK2 expression. MTT assay tested cell proliferation, and cell migration was quantified. Cell apoptosis was measured by caspase3 activity assay kit and Transwell chamber measured cell migration. Western blot quantified expressional change of HIF-1
Introduction
Bladder cancer (BC) is the ninth common malignant tumor worldwide. As one frequently occurred cancer in urinary system, the incidence of BC is rapidly increasing by years [1, 2]. BC mainly derives on bladder mucosa, and is the most popular tumor in urinal-reproductive system in China and second higher incidence in Western countries only next to prostate cancer [3]. BC has high incidence and the highest mortality rate in urinary-reproductive system. Having relatively high recurrent rate, BC is susceptible to invasion and metastasis [4, 5]. BC can occur in all age groups, with higher incidence in aged population and male specific bias [6]. Currently major treatment approaches for BC include surgery, chemotherapy and radiotherapy. However, due to high frequency of metastasis and recurrence, BC has unfavorable prognosis with lower 5-year survival rate [7]. BC has complicated pathogenic factors including genetic influences and environmental factors as smoking and occupation can affect tumorigenesis. However, the pathogenesis mechanism of BC has not been fully illustrated [8].
Primer sequences
Primer sequences
During tumor occurrence and progression process, a tumor specific microenvironment can be formed with oxygen deficient or hypoxia conditions as the result of glucose catabolism, oxygen depletion and carbonate metabolism [9]. Under hypoxia conditions, tumor cell can facilitate angiogenesis via glycolysis pathway, and has shift of energy metabolism, or even causing generation of drug resistant cells, leading to enhanced malignancy of tumors [10, 11]. Rho related coil formation protein kinase (ROCK) is one Rho associated kinase and is one important protein in Rho/ROCK signal pathway [12]. ROCK has two similar subtypes ROCK1 and ROCK2 [13]. ROCK2 participates in various cellular pathology and physiology evens, and in onset and progression of multiple diseases [14]. ROCK2 showed up-regulation in various tumors including liver cancer, breast carcinoma and prostate cancer [15, 16]. However, the expression or related functional role of ROCK2 in BC under hypoxia conditions have not been fully illustrated.
Major reagent and equipment
BC cell line T24 (HTB-4™) was purchased from ATCC cell bank (US). DMEM culture medium, penicillin-streptomycin dual antibiotics were purchased from Hyclone (US). Fetal bovine serum (FBS), DMSO and MTT powder were purchased from Gibco (US). Trypsin-EDTA digestion buffer was purchased from Sigma (US). PVDF membrane was purchased from Pall Life Sciences (US). Western blot reagents were purchased from Beyotime Biotech (China). ECL reagent was purchased from Amersham Biosciences (US). Rabbit anti-human HIF-1
Grouping of BC cell line T24
BC cell line T24 kept in liquid nitrogen was resuscitated for passage incubation. Cells were randomly assigned into three groups: control group; hypoxia group that was prepared under hypoxia conditions, and ROCK2 siRNA group that was transfected with ROCK2 siRNA after hypoxia model preparation.
Hypoxia model preparation and liposome transfection of ROCK2 siRNA into BC cell T24
Cells were prepared for hypoxia model under 37
Real-time PCR for ROCK2 expression in BC cell line T24
Trizol reagent was used to extract mRNA from all groups of BC cell line T24. DNA reverse transcription was performed following manual instruction of test kit. Primers were designed using PrimerpRemier6.0 based on target gene sequence, and were synthesized by Invitrogen Biotech (US) as in Table 1. Real-time PCR was measured on target genes under the following conditions: 56
Western blot measuring ROCK2, HIF-1
, E-cadherin, Wnt4 and
-catenin protein expression
Cellular proteins were extracted from all groups of BC cells. In brief, RIPA lysis buffer containing proteinase inhibitor was added into cells for 30 min lysis on ice. Cells were ruptured by ultrasound (5 s, 4 times), and cell lysate was centrifuged at 10000 g for 15 min under 4
MTT assay for cell proliferation effect
T24 cells at log-growth phase were collected and were inoculated into 96-well plate using DMEM medium containing 10% FBS at 5
Caspase3 activity assay
Caspase3 activity in all groups was measured following the manual instruction of test kit. In brief, cells were digested by trypsin and were centrifuged at 600 g for 5 min at 4
Transwell chamber assay
Following the manual instruction, serum-free DMEM medium was switched. After 24 h later, the bottom and upper phase of the membrane were coated with 50 mg/L Matrigel dilution (1:5) for 4
Cell migration assay
Cell migration was measured from all groups using scratch assay. In brief, 5
Statistical processing
All data were presented as mean±standard deviation (SD). Student t-test was employed to compare means between two groups. SPSS 11.5 software was used for statistical analysis. Analysis of variance (AVNOA) was used to compare difference among groups. A statistical significance was defined when
ROCK2 expression in BC cell line T24 under hypoxia culture. (A) Real-time PCR for ROCK2 mRNA expression in BC cell line T24 under hypoxia culture. (B) Western blot for ROCK2 protein expression in BC cell line T24 with hypoxia culture. (C) Analysis for the expression for ROCK2 protein expression in hypoxia T24 cells. *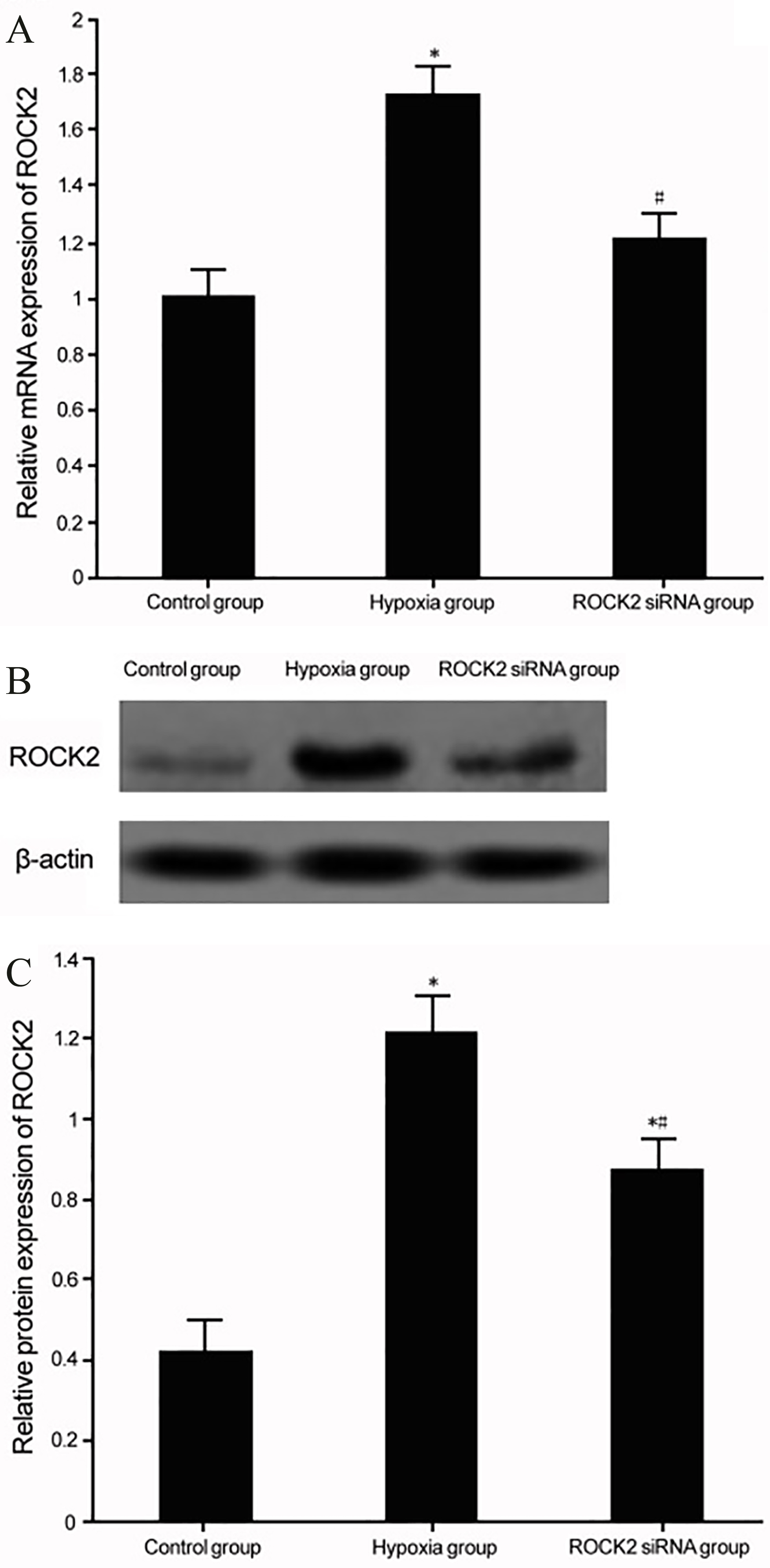
ROCK2 expression in BC cell line T24 under hypoxia conditions
Real-time PCR and Western blot were used to measure ROCK2 expression in BC cell line T24 under hypoxia conditions. We found elevated expression of ROCK2 in T24 cells upon hypoxia challenge (
Effects of ROCK2 modulation on BC cell line T24 proliferation under hypoxia. *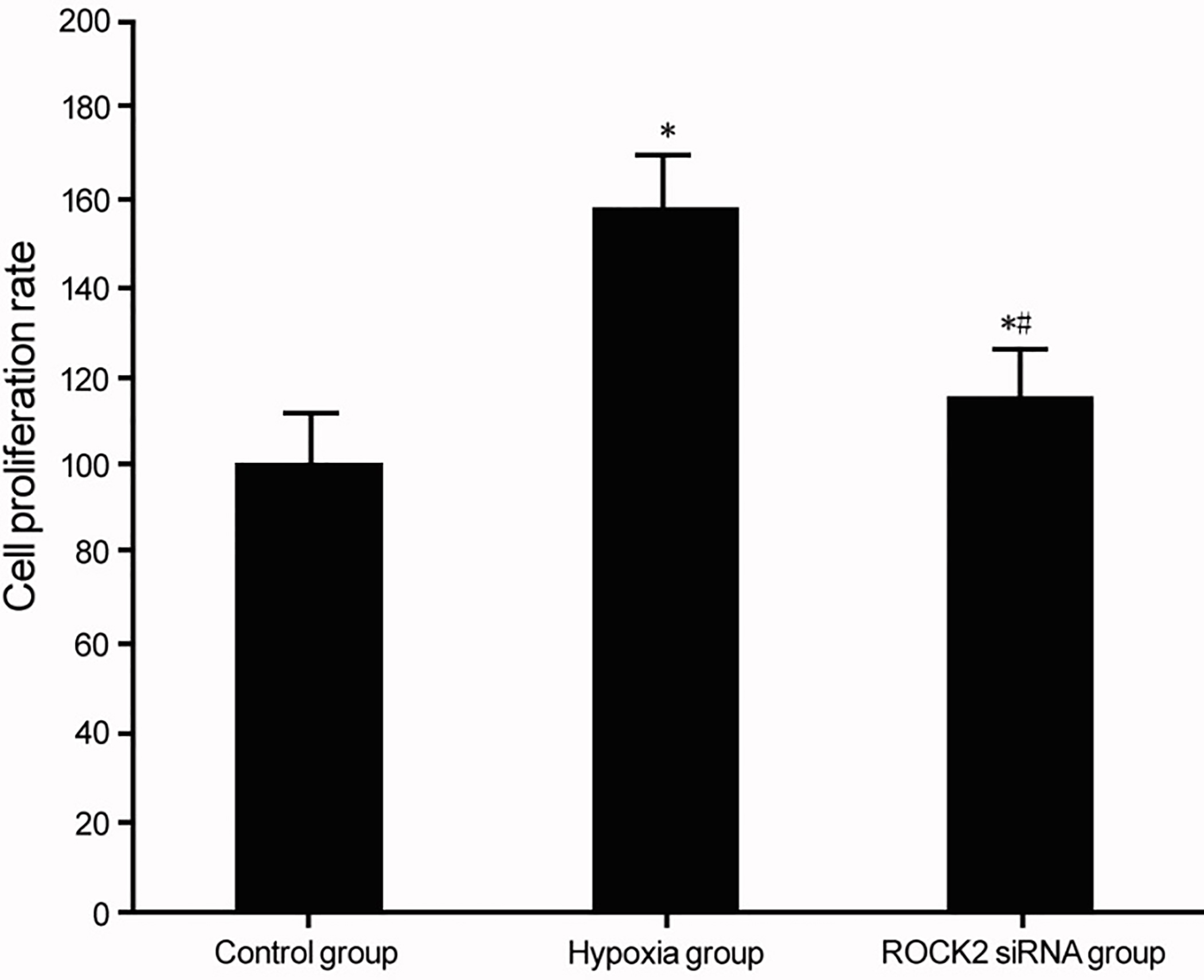
MTT assay was used to measure the effect of ROCK2 on the proliferation of BC cell line T24 under hypoxia conditions. Results showed significantly enhanced proliferation of BC cell line T24 under hypoxia conditions (
Regulation of ROCK2 and BC cell line T24 migration under hypoxia conditions. (A) Effects of ROCK2 regulation on the migration of BC cell T24 under hypoxia conditions. (B) Analysis for the effects of ROCK2 modulation on T24 cell migration after hypoxia. *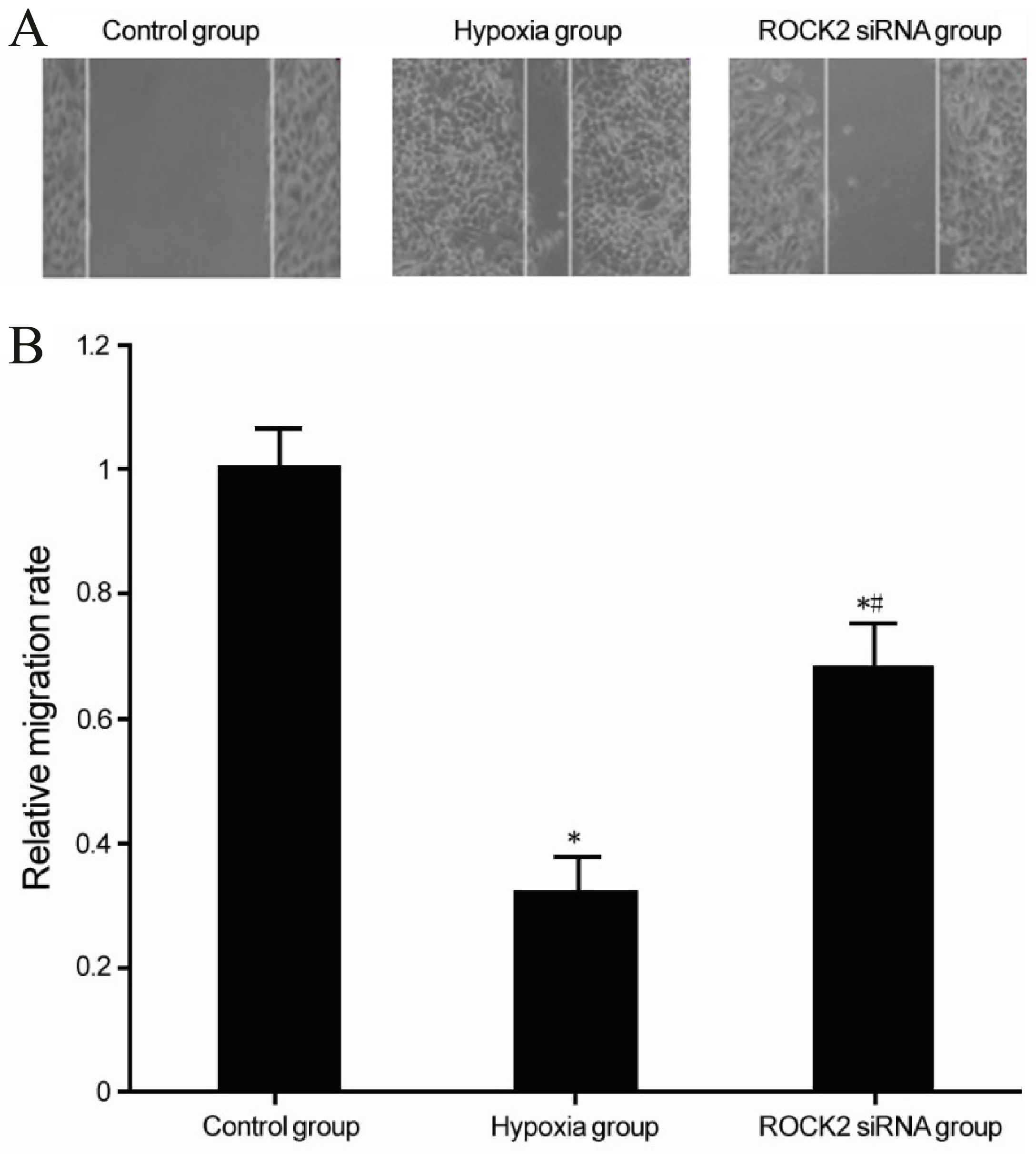
ROCK2 modulation and T24 cell invasion under hypoxia conditions. (A) Effects of ROCK2 regulation on the invasion of BC cell line T24 under hypoxia culture. (B) Analysis for the effect of ROCK2 modulation on the invasion of T24 cells under hypoxia. *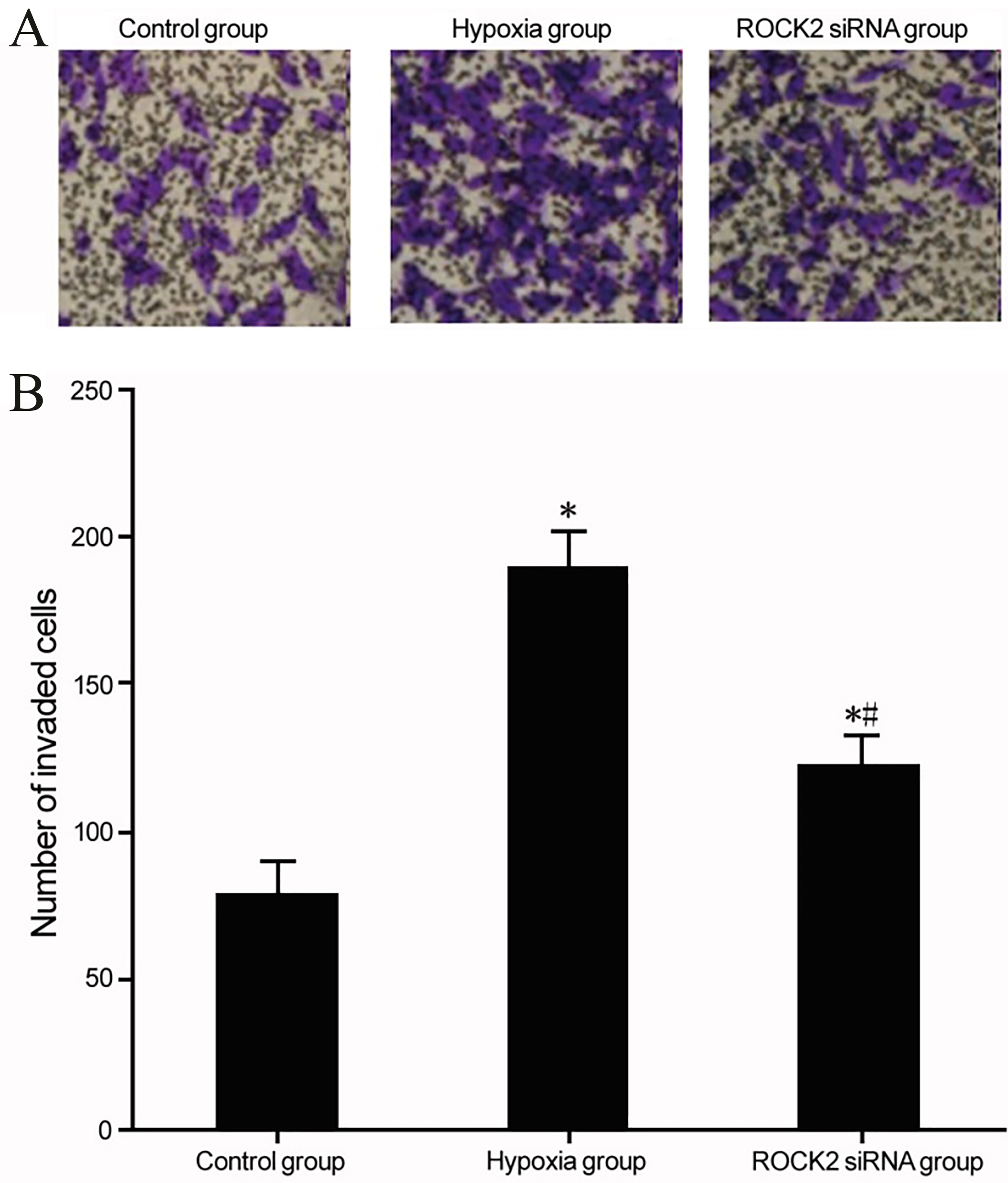
Modulation of ROCK2 and Caspase 3 activity in BC cell line T24 under hypoxia conditions. *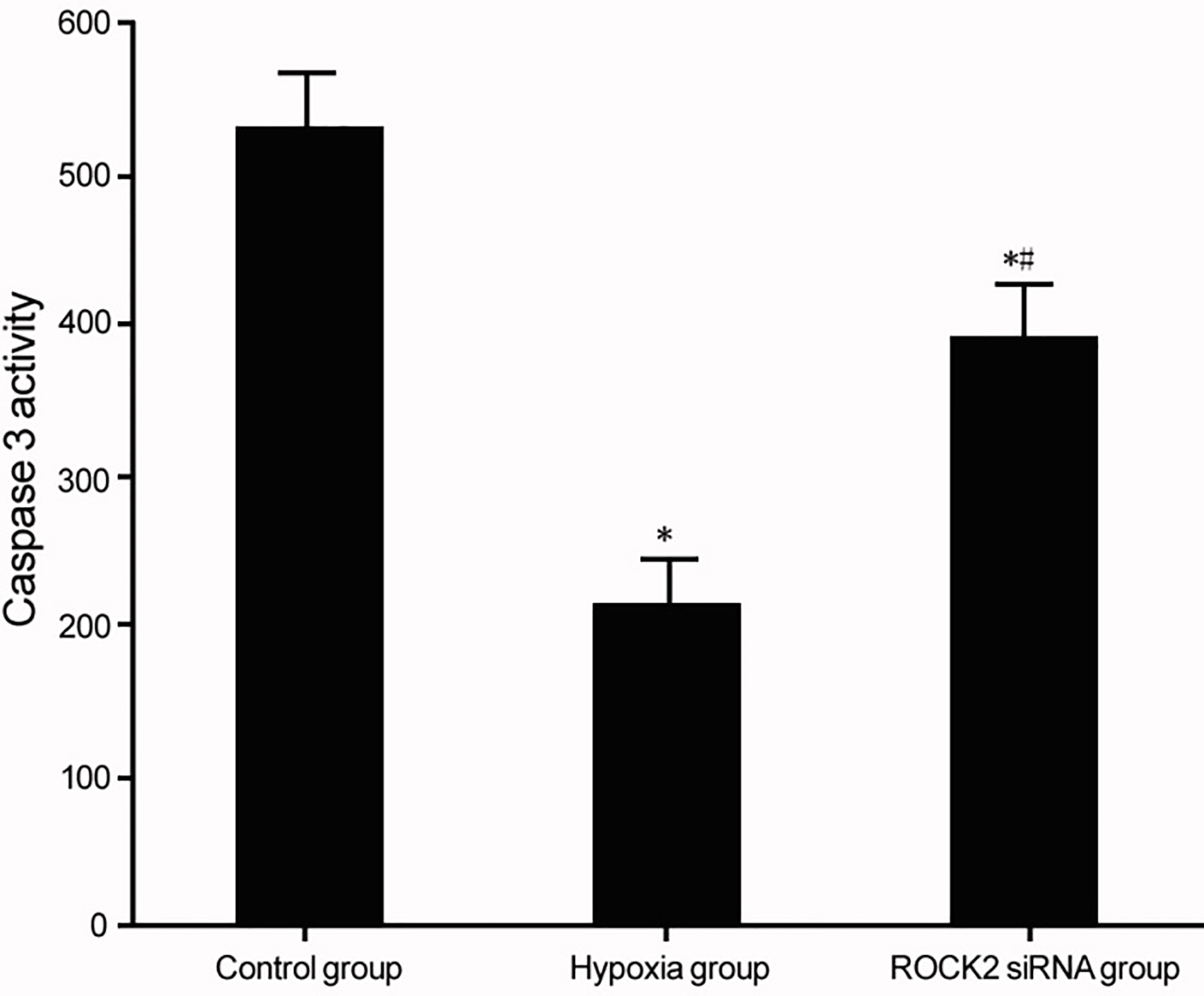
The effect of ROCK2 modulation on HIF-1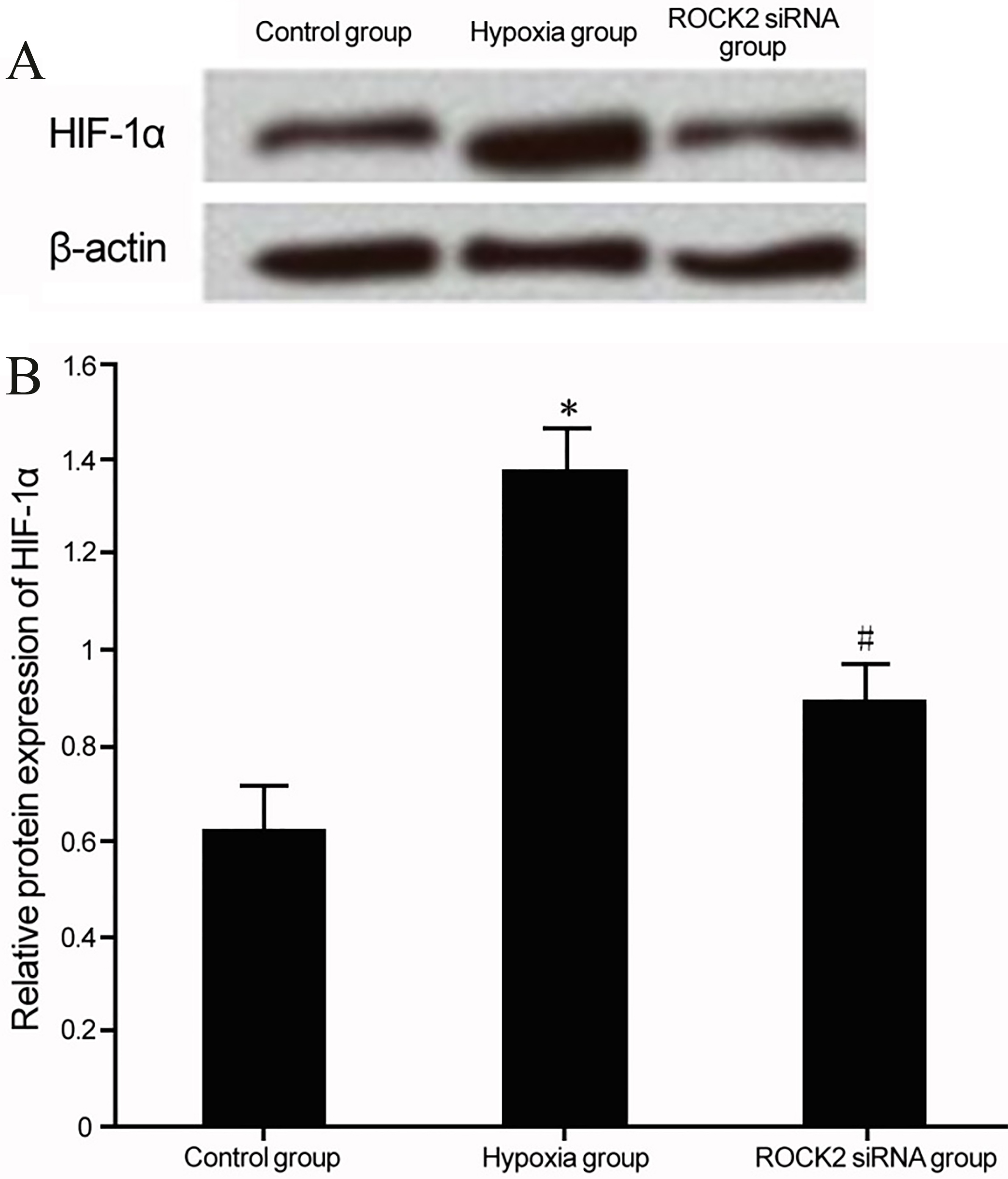
Under hypoxia conditions, BC cell line T24 had significantly enhanced cell migration (
The modulation of ROCK2 on T24 cell invasion
Under hypoxia condition, BC cell line T24 showed significantly enhanced invasion (
Effects of ROCK2 modulation on Caspase3 activity of BC cell line T24 under hypoxia
Caspase 3 activity assay was used to measure the effect of ROCK2 modulation on apoptotic activities of BC cell line T24 under hypoxia culture. Under hypoxia, T24 cells showed reduced Caspase 3 activity (
Regulation of ROCK2 and HIF-1
expression of T24 cells under hypoxia culture
Under hypoxia culture, BC cell line T24 presented elevated HIF-1
Modulation of ROCK2 and E-cadherin expression in BC cell line T24 under hypoxia. (A) Western blot for the effect of ROCK2 regulation on E-cadherin expression in BC cell line T24 under hypoxia culture. (B) Analysis for the effect of ROCK2 modulation on E-cadherin expression in BC cell line T24. *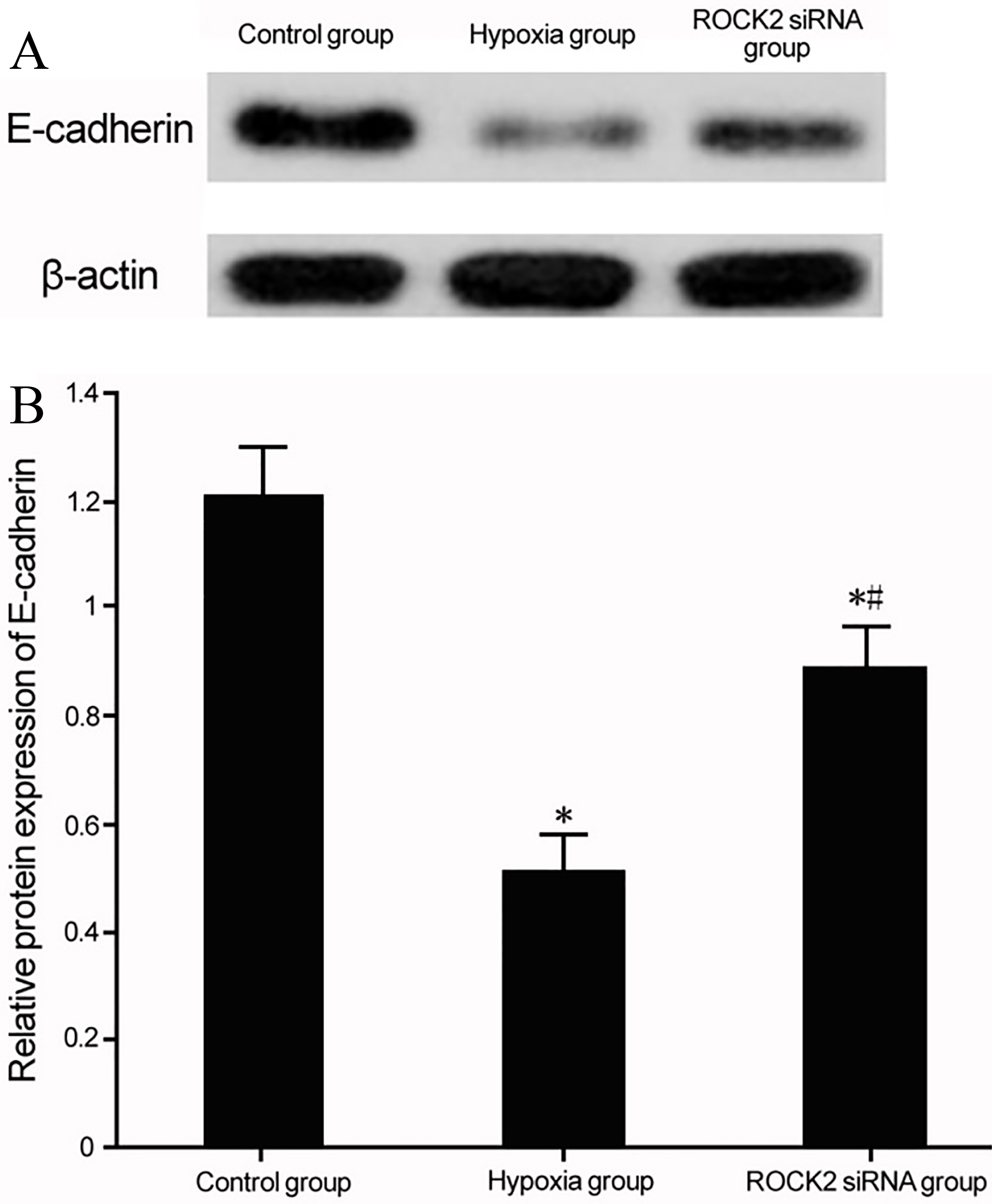
Under hypoxia conditions, BC cell line T24 presented decreased E-cadherin expression (
The effect of ROCK2 modulation on Wnt signal pathway expression in BC cell line T24 under hypoxia culture. (A) Western blot for the effect of ROCK2 modulation on Wnt4 and 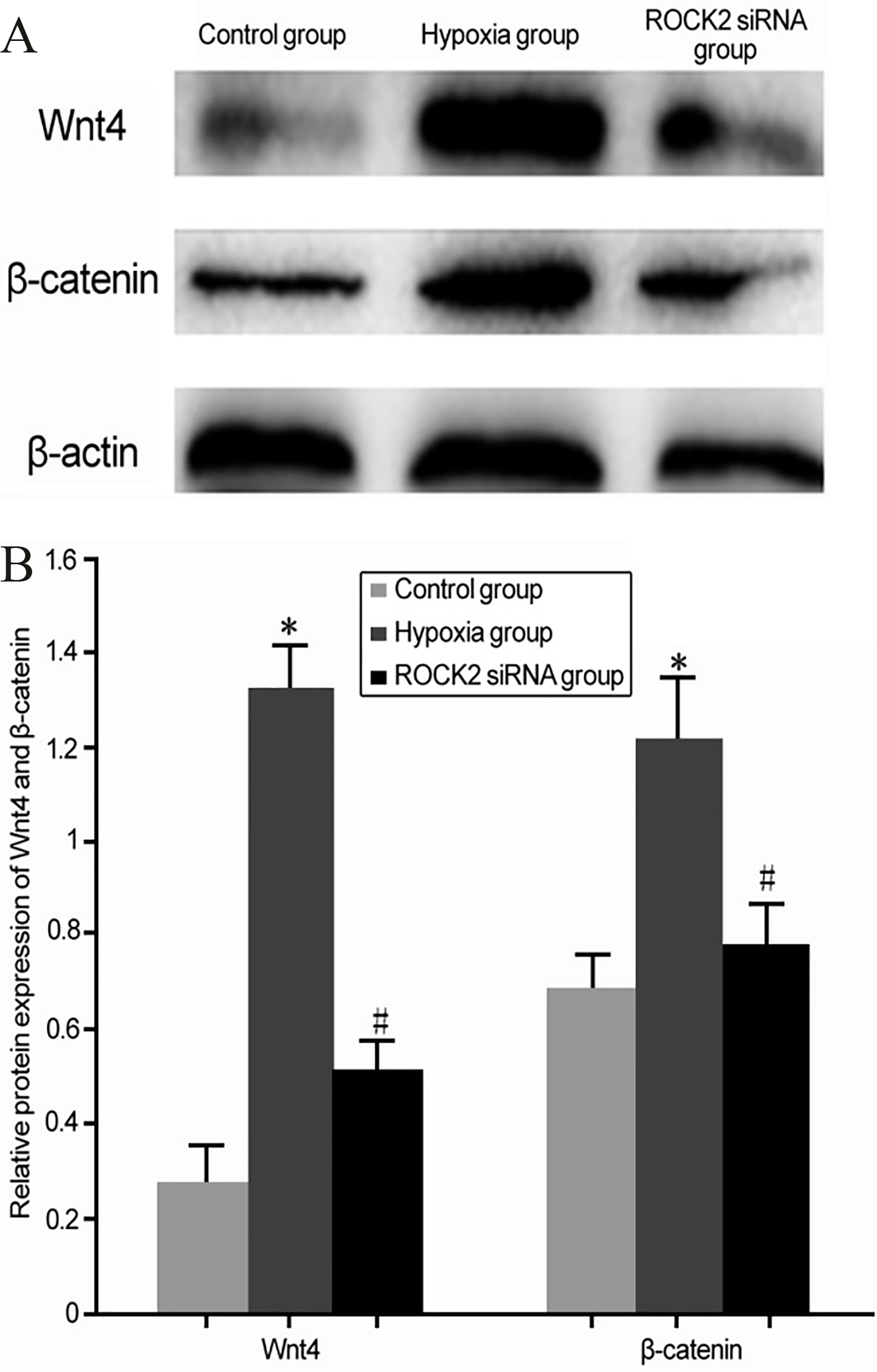
Western blot was used to analyze the effect of ROCK2 on Wnt signal pathway in BC cell line T24 under hypoxia culture. Under hypoxia culture, T24 cells presented enhanced expression of Wnt signal pathway proteins including Wnt4 and
Discussion
BC has complicated pathogenesis mechanism involving multiple factors such as genetics, environment and diet habit [7, 8]. Due to atypical symptom of BC at early stage, patients frequently cannot achieve timely diagnosis [17]. The metastasis of BC is the critical factor affecting the patient prognosis. The whole process of BC involves multiple stages, factors and cascades under the regulation of multiple genes. EMT plays important roles in BC metastasis [18]. Hypoxia is one common marker for malignant tumor growth as it can promote tumor cell survival, and maintain continuous division of tumor cells under hypoxia conditions [19]. This study demonstrated that under hypoxia conditions, BC cells presented ROCK2 up-regulation, leading to enhanced tumor cell proliferation, lower apoptosis and suppressed cell migration or invasion potency. EMT process is closely correlated with down-regulation of deficient of cell adhesion molecules E-cadherin [20]. This study demonstrated down-regulation of E-cadherin in BC cells under hypoxia challenge, indicating that hypoxia culture could facilitate EMT occurrence of BC.
The down-regulation of ROCK2 in BC cells under hypoxia conditions could suppress tumor cell proliferation, facilitate Caspase3 activity, leading to enhanced tumor cell apoptosis, further suppressing cell migration and invasion. On the other hand, ROCK2 down-regulation could facilitate E-cadherin expression of BC cells under hypoxia culture, indicating that ROCK2 modulation could improve hypoxia induced BC tumor cell occurrence or progression. Further studies about its related mechanisms found that modulation of ROCK2 could suppress the expression of hypoxia induced factor HIF-1
Conclusion
Under hypoxia conditions, down-regulation of ROCK2 can improve hypoxia stress via modulating Wnt signal pathway, to facilitate BC cell apoptosis, inhibit BC cell proliferation, migration, invasion or EMT formation.
Footnotes
Conflict of interest
None.
