Abstract
BACKGROUND:
For patients with pancreatic cancer, a preoperative assessment of prognosis is crucial to predict cancer recurrence and to prepare a postoperative adjuvant strategy and appropriate patient-counsel.
OBJECTIVE:
We evaluated the prognostic predictive power of complement factor B (CFB) by comparing it to that of other known tumor markers in resected pancreatic cancer patients.
METHODS:
From 2012 to 2013 period, we retrospectively reviewed the plasma CFB levels of 35 pancreatic cancer patients. The patients were divided into two groups according to serologic CFB values. Disease-free survival (DFS) and overall survival (OS) rates were analyzed.
RESULTS:
Based on the cut-off values of plasma CFB, 15 patients were placed in the low CFB group and the other 20 patients were placed in the high CFB group. There was a significant difference in DFS between the two groups (Low CFB vs. High CFB: 36.9 months vs. 13.9 months,
CONCLUSION:
Preoperative plasma CFB can be used to predict the prognosis of resectable pancreatic cancers; it outperforms both CA 19-9 and CEA.
Introduction
Despite advances in the understanding of the biology of pancreatic cancer over the past few decades, pancreatic cancer remains one of the most deadly malignant diseases of the gastrointestinal tract. The overall 5-year survival rate of pancreatic cancer is approximate 6%, and it is the fourth leading cause of cancer-related deaths in the United States [1]. At present, the only potentially curative treatment is surgical resection. However, only 20% of patients have the chance to undergo surgical resection at the time of initial diagnosis due to lack of an early diagnostic marker for pancreatic cancer [2]. Furthermore, even when curative surgery is performed, more than half of patients experience a recurrence within 1 year, and the 5-year survival rate of resected pancreatic cancer is less than 20% [3]. As a result, many pancreatic cancer patients are undergoing pancreatectomy with low oncologic efficacy. Therefore, both early diagnosis and appropriate prediction of the prognostic potential of pancreatic cancer in a preoperative setting are crucial to avoid oncologically unbeneficial pancreatectomies.
Many clinic-pathological factors have been suggested for predicting pancreatic cancer prognosis. Several pathologic characteristics such as lymph node metastasis, perineural invasion, lymphovascular invasion, and differentiation are known to be significant prognostic factors; they are usually confirmed by the pathological examination of surgical specimens after operation [4]. CA 19-9 is one of the serum tumor markers that is used to aid cancer diagnosis, assess prognosis, guide choice of treatment, and monitor progress during and after treatment in pancreatic cancer [5]. It is the only tumor marker approved by the US Food and Drug Administration (FDA) for pancreatic cancer and is generally used before an operation to predict postoperative prognosis [6]. However, several disadvantages of CA 19-9 have been identified. For example, CA 19-9 is expressed in benign diseases and many types of gastrointestinal cancer [7] and cannot be used to distinguish pancreatic cancer from other gastrointestinal cancers [8]. Therefore, there is an urgent need to develop new, efficient pancreatic cancer biomarkers with higher specificity and sensitivity. Research to discover a novel tumor marker for pancreatic cancer that can overcome the limitations of CA 19-9 has suggested several markers [9]. Other studies reported using modified CA 19-9 as adjusted CA 19-9 [10]. However, CA 19-9 and other markers (CEA, etc.) are not widely accepted for general screening due to their low sensitivity and specificity. Furthermore, up to 10 to 15% of the population group lacks the genetic expression of CA 19-9 [11].
Recently, our team identified plasma complement factor B (CFB) as a new pancreatic cancer diagnostic marker [12]. It has a higher specificity than CA19-9 for distinguishing pancreatic cancer from other types of digestive cancers and for discriminating pancreatic cancer patients from non- pancreatic cancer patients. CFB is one of the complement components and a critical component of the alternative pathway amplification loop that is activated by a multitude of infectious agents including various bacteria, viruses, and fungi, in addition to neoplastic cells [13]. A recent report suggested that the complement component may aid tumor growth through immunosuppression and there is increased interest in the relationship between the complement system and cancer [14].
In this study, we attempted to evaluate whether the levels of preoperative plasma CFB can predict oncologic outcomes in resected pancreatic cancer. This potential correlation between preoperative plasma CFB and survival outcome would enhance the diagnostic value of plasma CFB in the treatment of pancreatic cancer.
Patients and methods
Patients’ characteristics
Based on collected medical records covering the January 2012 to December 2013 period, we retrospectively reviewed the plasma CFB levels of 35 pancreatic cancer patients who underwent potentially curative pancreatectomies, excluding locally advanced pancreatic cancer (stage III) and metastatic pancreatic cancer (stage IV). Only patients with solely pancreatic ductal adenocarcinoma by permanent pathologic report were enrolled; those pancreatic cancer patients with combined diseases were excluded.
Measurement of complement factor B and other tumor markers
Fresh blood samples from the pancreatic cancer patients were obtained just before the operation and the plasma CFB levels were measured using the enzyme-linked immunosorbent assay (ELISA) test, as previously described [12]. CA 19-9 levels were measured using the electrochemiluminescent immunoassay (ECLCIA) test (Roche, Manheim, Germany) and the CEA levels were measured using the chemiluminescent immunoassay (CLIA) sandwich test (Beckman Coulter, Brea, USA).
Defining cut-off value of CFB and oncologic correlations
The predictive power of CFB was compared to that of other tumor markers (e.g., CA 19-9 and CEA) and the cut-off value of CFB was calculated. The patients were categorized into two groups (Low CFB vs. High CFB) according to serologic CFB value. The differences in clinic-pathological characteristics and survival outcomes were analyzed for the two groups. Definition of the survival outcomes (DFS and OS) was based on the FDA (Food and Drug Administration) and the survival outcomes were measured from the operation date [15].
Clinicopathologic characteristics
Clinicopathologic characteristics
BMI, Body mass index; PPPD, Pylorus presereving pancreaticoduodenectomy; DPS, Distal pancreatosplenectomy; PNI, perineural invasion; LVI, Lymphovascular invasion; EUS-FNA, Endoscopic ultrasound Fine needle aspiration; NLR, Neutrophil-Lymphocyte ratio; PLR, Platelet-Lymphocyte ratio; CA, Carbohydrate antigen; CFB, Complement factor B.
Linear correlation between CFB and other tumor markers.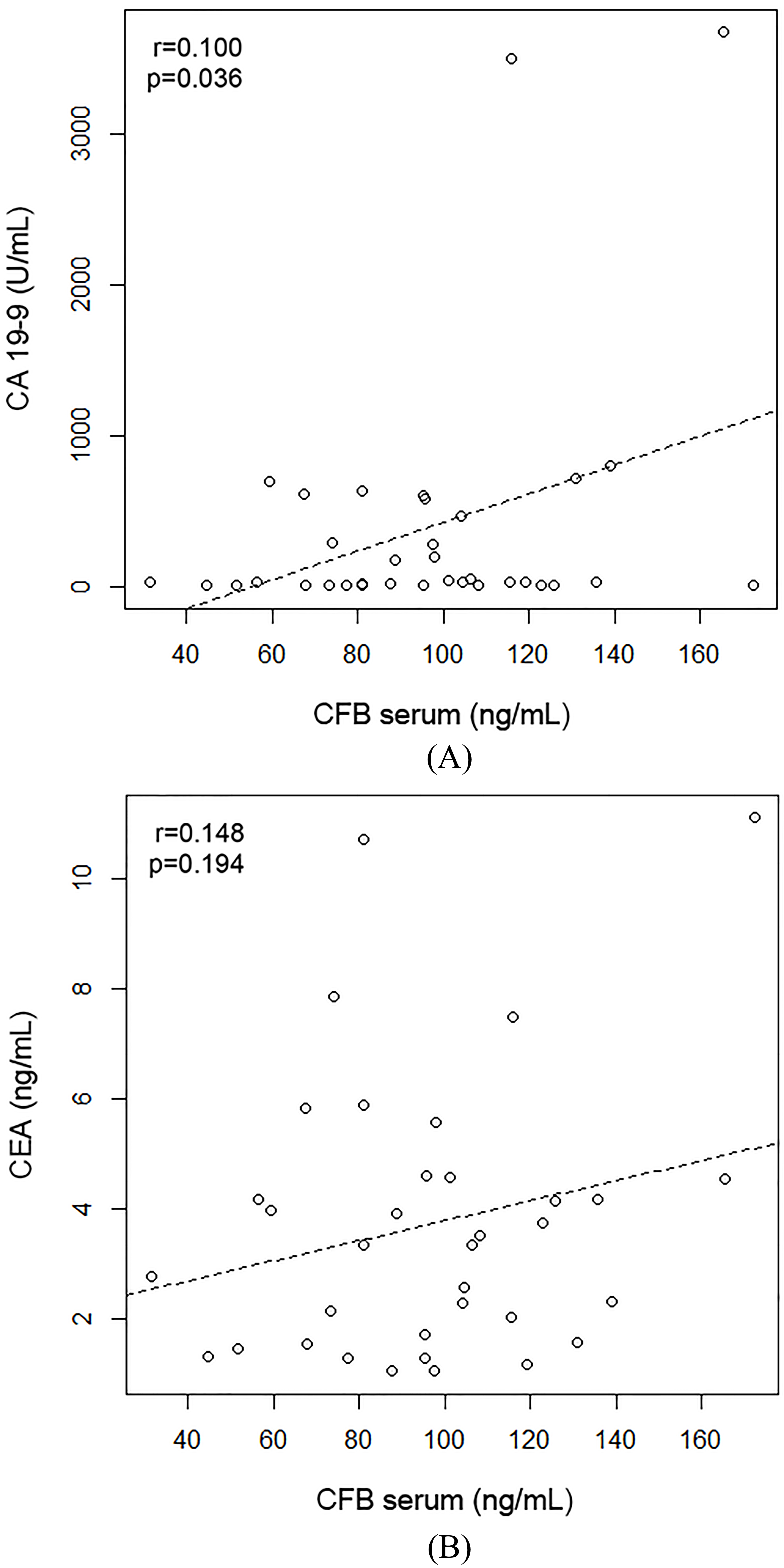
IBM SPSS Statistics version 23.0 (IBM Corp., Somers, NY, USA) was used for all of the statistical analyses. Nominal data were compared with
Results
Patients’ characteristics
The clinicopathologic characteristics of the 35 patients are shown in Table 1. The mean CFB value was 96.4 ng/mL (96.4
ROC curves of specific tumor marker for pancreatic cancer.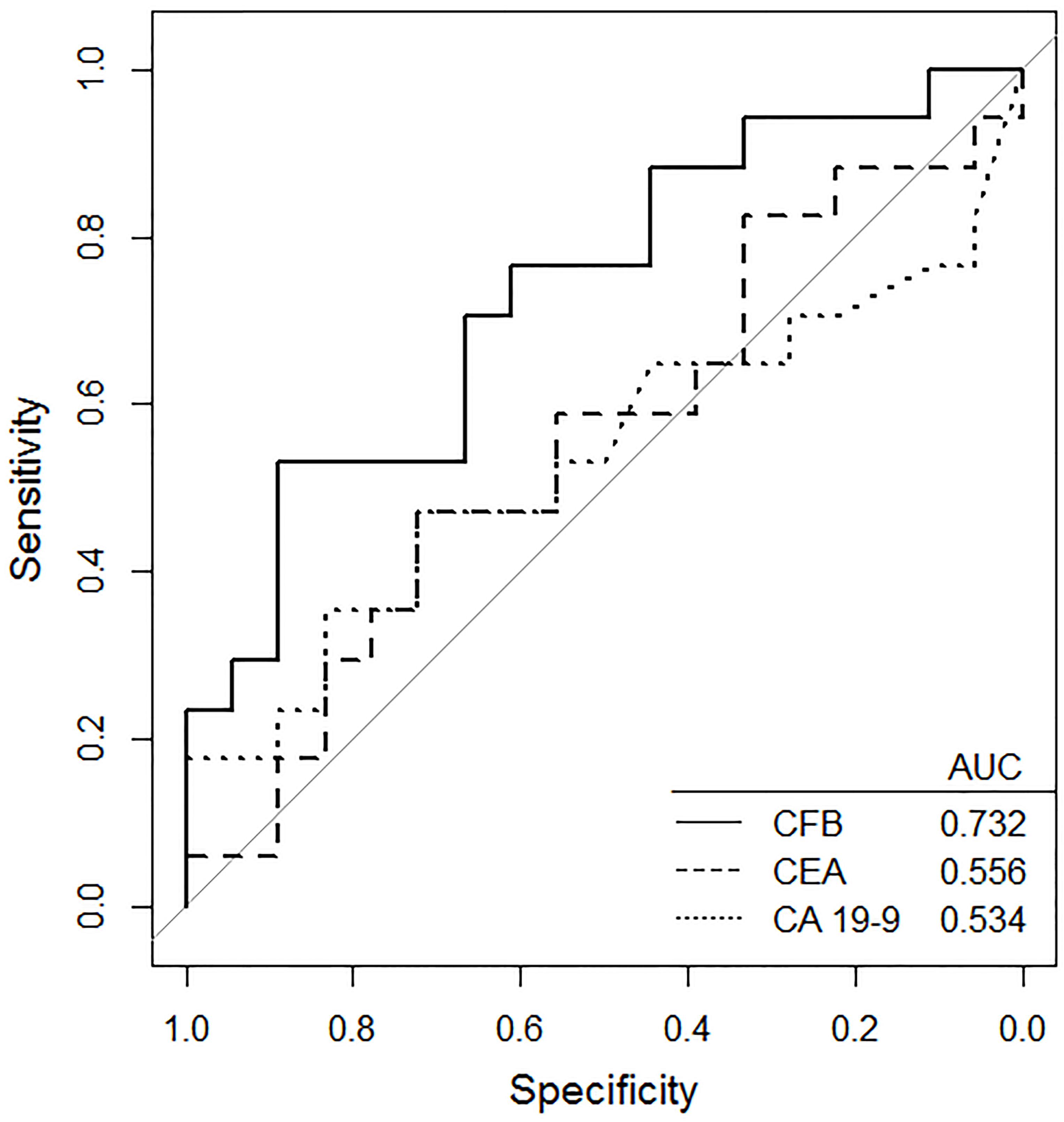
Clinicopathologic characteristics according to CFB level
BMI, Body mass index; PPPD, Pylorus presereving pancreaticoduodenectomy; DPS, Distal pancreatosplenectomy; PNI, perineural invasion; LVI, Lymphovascular invasion; EUS-FNA, Endoscopic ultrasound Fine needle aspiration; NLR, Neutrophil-Lymphocyte ratio; PLR, Platelet-Lymphocyte ratio; CA, Carbohydrate antigen; CFB, Complement factor B.
There was no significant lineal correlation between CFB and the other tumor markers (CFB vs. CA 19-9:
Using Youden’s J statistic, the cut-off value of the preoperative serum CFB was estimated to be 95.0 ng/mL, which may be used to predict early recurrence (recurrence within 1 year of radical pancreatectomy) in resected pancreatic cancer (Sensitivity: 72.2%, Specificity: 70.6%).
Thirty-five patients were divided into two groups according to disease-free time (recurrence within 1 year vs. recurrence after 1 year or no recurrence). In the ROC curve, only CFB showed a significant difference, indicating its prognostic potential for pancreatic cancer (AUC: 0.732,
Survival rate according to serologic CFB level (A) Disease-free survival (DFS) (B) Overall survival (OS).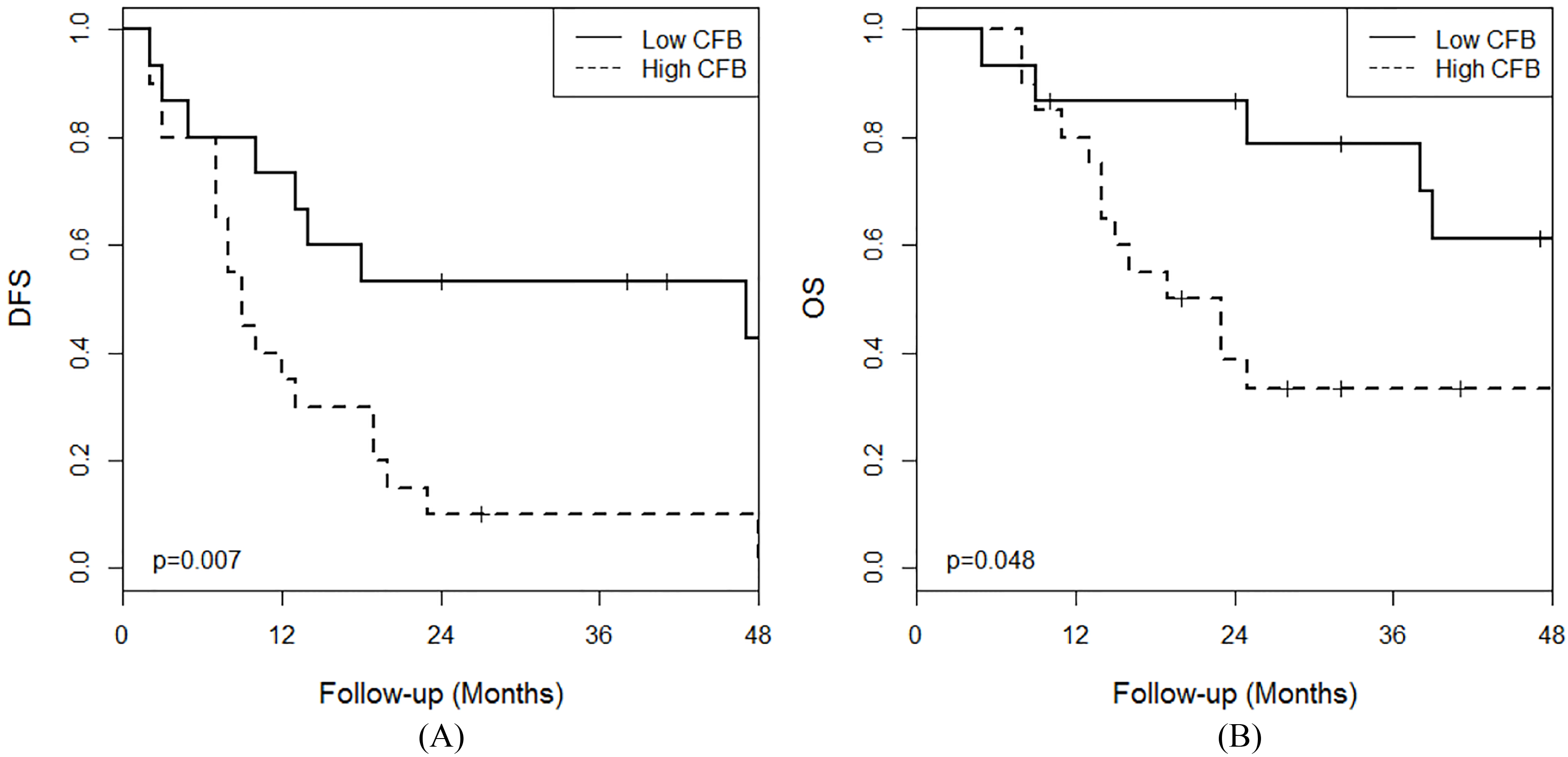
The 35 patients were divided into two group according to the cut-off value of the preoperative serum CFB. Fifteen patients were place in the low CFB group (Serum CFB level
Oncologic outcomes according to CFB level
The median follow-up period of the patients was 39.2 months. There was a significant difference in DFS between the two groups (Low CFB vs. High CFB: 36.9 months vs. 13.9 months,
During the follow-up period, pancreatic cancer recurred in 29 patients: 19 patients (95.0%) belonging to the high CFB group and 10 patients (66.7%) belonging to the low CFB group. However, there was no significant recurrence pattern between the two groups (
Discussion
In general, several requirements are needed for a component to be useful as a tumor marker: 1) appropriateness for the required application; 2) separated value according to different outcomes; and 3) reliability of the value of separation in outcomes [16]. In addition, it should be possible to collect adequate amounts of the specimen from enough patients [17]. Many studies have reported novel tumor markers to facilitate the diagnosis and management of pancreatic cancer. However, most of these tumor markers have not been validated and have not been approved for use by the FDA. In the past several decades, the only such marker has been CA 19-9 [18].
However, CA 19-9 is not recommended for use in the screening or early detection of pancreatic cancer due to its high false positive rate when combined with non-malignant biliary disease or other cancers [19]. For this reason, it has limited usefulness as an aid for diagnosing pancreatic cancer [20]. It is known that an elevated serum CA 19-9 level has a sensitivity of 79% and a specificity of 82% for diagnosing pancreatic cancer in symptomatic patients [21]. Our previous study of CFB as a novel tumor marker for detecting pancreatic cancer revealed that an elevated serum CFB level has a sensitivity of 73.1% and a specificity of 97.9% for diagnosing pancreatic cancer [12]. Due to its high specificity, the false positive rate of CFB was lower than that of CA 19-9 and CFB, indicating its possible value as a novel tumor marker to diagnosis pancreatic cancer.
In fact, CA 19-9 is used in prognosis and for detecting recurrence and monitoring treatment of pancreatic cancer. Many studies have reported that CA 19-9 levels are useful for predicting outcomes and responses to adjuvant chemotherapy [22]. However, due to its Lewis blood group-dependence and the influence of jaundice, CA 19-9 has limitations. CA 19-9 levels are most informative when the serum bilirubin has normalized in Lewis positive genotype patients. Although, Lewis negative genotype occurs in only 5
There have been some reports of the high expression of CFB in pancreatic cancer cells [12]. These findings are consistent with early suggestions of a close correlation between CFB and carcinogenesis, which is promoted by cellular inflammation, and an association with the carcinogenesis of pancreatic cancer [13]. However, the relationship between pancreatic cancer and CFB remains unknown. Moreover, no studies have shown a correlation between the expression of CFB in pancreatic cancer cells and the survival of pancreatic cancer. At a guess, CFB may be expressed not by the pancreatic cell itself but through interaction with other environments; some studies have proposed that CFB may be highly activated by inflammation-promoted pancreatic carcinogenesis [23].
However, the activation of CFB may not be directly affected by the inflammatory response. In our study, there was no significant difference in the rate of EUS-FNA of the two groups. Moreover, there was also no significant difference in the inflammatory factors like NLR and PLR of the two groups. These results may suggest that the pathway of CFB is different from the local or systemic inflammatory pathway.
Recent studies have examined the relationship between pancreatic stellate cells (PSCs) and pancreatic cancer. PSCs are resident cells in the pancreas and have been well established as key players in pancreatic fibrogenesis [24]. In a normal pancreas, PSCs are quiescent, but they become active in response to pancreatic injury or inflammation [25]. Specifically, they induce epithelial-mesenchymal transition (EMT) [26]. EMT is now considered a critical process in cancer progression, and EMT induction in cancer cells results in the acquisition of invasive and metastatic properties and resistance to conventional therapies [27].
Among the factors that induce EMT, signal transducers and activators of the transcription-3 (STAT3) signaling pathway are involved in pancreatic cancer cell invasion and EMT, and the activation of STAT3 signaling pathway is induced by IL-6 [28]. A recent study suggested that CFB is upregulated during IL-6/STAT3 signaling [29]. According to that study, PSCs induced the activation of STAT3 in pancreatic cells and upregulated several genes, of which CFB was the most expressed gene. Thus, although the role of the IL-6/STAT3 pathway in the interaction between PSCs and pancreatic cancer cells remains largely unknown, PSCs might contribute to the increased expression of CFB in pancreatic cancer and the expressed CFB circulated the body, which is one of the complement factors. Therefore, CFB value could be a marker linked to pancreatic cancer.
In our study, there was no correlation between CFB level and TNM stage, especially lymph node metastasis. Today, pancreatic cancer stages are assessed using the TNM staging system, which correlates with OS [30]. Increasing knowledge of cancer pathology and biology has led to suggestions that tumor biological features such as histologic grade or stromal content should be incorporated into staging systems, not only the tumor extension that is the basis of TNM staging [31]. Similar, CFB is associated with extracellular matrix disintegration in pancreatic cancer and this biologic feature could be incorporated into the prognosis of pancreatic cancer [13]. Even though there were no statistical differences, high CFB was noted to show larger tumor size, more frequent incidence of LVI, PNI, lymph, lymph node metastasis, and systemic metastasis in comparative analysis. The sum of the oncologic impact of these biologic properties might influence on long-term oncologic outcome. Therefore, these observations showed that CBF might be unique independent prognostic factor not related to pathological properties.
In addition, CFB was found to be potentially correlated with CA 19-9 (
Although this study found that CFB has the possibility of being a novel tumor marker for pancreatic cancer, there are some limitations. First, this study is a retrospectively designed pilot study showing potential usefulness of preoperative plasma CFB to predict prognosis for resected pancreatic cancer by using a small and heterogeneous sample. To obtain more reliable statistical power, a large and homogeneous sample including even stage III and IV are necessary in near future. In addition, this study focused on the efficacy of the tumor marker as a prognostic factor. To qualify as a novel tumor marker for pancreatic cancer, further studies of the applications of CFB, such as its usefulness in screening, detecting recurrence, and monitoring treatment are required. In summary, plasma CFB it a better predictor of the prognosis for resected pancreatic cancer than CA 19-9 and is a novel pancreatic cancer biomarker. Further studies that use larger samples to examine the CFB pathway for pancreatic cancer are needed.
Footnotes
Acknowledgments
This research was supported by a grant from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health and Welfare, Republic of Korea (grant number: HI16C0257).
