Abstract
BACKGROUND:
Laryngeal squamous cell carcinoma (LSCC) constitutes the third most frequent head and neck cancer. Several tissue biomarkers have been studied for their prognostic significance in LSCC.
OBJECTIVE:
To investigate the prognostic significance of BCL2L12, a new member of the BCL2 family, in primary LSCC along with well-examined biomarkers such as BCL2 and BAX.
METHODS:
Cancerous tissue specimens of patients with primary LSCC were collected during 2005 and 2012 as pretreatment tissue biopsy. The specimens were immunohistochemically evaluated for the protein expression of BCL2L12, BCL2 and BAX. Kaplan-Meier survival curves and Cox proportional hazard regression models were performed to evaluate prognosis.
RESULTS:
In the study cohort of 78 patients with primary LSCC, Kaplan-Meier survival curves demonstrated that advanced-stage LSCC patients with BCL2L12-positive tumors had significantly higher OS time in comparison with advanced-stage LSCC patients with BCL2L12-negative tumors (
CONCLUSIONS:
BCL2L12 protein expression could be used as a favorable prognostic tissue biomarker in patients with primary advanced-stage LSCC. On the contrary, BCL2 and BAX did not correlate with prognosis in patients with primary LSCC.
Introduction
Head and neck squamous cell carcinoma (HNSCC) constitutes the eighth most commonly diagnosed cancer, accounting for approximately 6% of all cancer cases worldwide [1]. Laryngeal squamous cell carcinoma (LSCC) was the most frequent among subtypes of HNSCC until the beginning of the 21st century [2].
Several tissue and serum biomarkers have been studied for their prognostic significance in LSCC using distinct techniques, including immunohistochemistry [3], polymerase chain reaction (PCR) and real-time PCR [4]. For instance, Foteinou et al. observed that low mRNA expression levels of kallikrein-related peptidase 4 (KLK4) predicted shorter disease-free survival (DFS) in LSCC [4]. Additionally, Kumar et al concluded that tumor expression of P53 and BCL-xL might be correlated with successful laryngeal preservation in patients with laryngeal cancer treated with chemotherapy (CTH) and radiotherapy (RT) [5]. Also, Zhang et al reported that LSCC patients with positive cyclin D1 protein expression had significant higher median survival time than LSCC patients with negative cyclin D1 protein expression [6].
Data are controversial about correlation of BCL2 family genes expression and prognosis in LSCC. Jackel et al. reported that positive BCL2 protein expression predicted shorter overall survival (OS) [7], while others supported that positive [8] or high [9]BCL2 protein expression were associated with better OS. In other studies, BCL2 protein expression was not correlated with prognosis in early-stage LSCC [10] or LSCC of any stage [11]. Interestingly, high BCL2 protein expression was associated with resistance to radiotherapy in LSCC [3, 12]. On the contrary, data are consistent that BAX protein expression does not predict DFS or OS in LSCC [8, 7].
The BCL2L12 gene, a member of the BCL2 family, was discovered and cloned in 2001 [13]. The prognostic significance of BCL2L12 mRNA expression has already been assessed in several cancer types [14, 15, 16]. Fendri et al. observed that BCL2L12 mRNA expression was a significant predictor of unfavorable prognosis in patients with nasopharyngeal carcinoma (NPC) [17]. Moreover, Geomela et al. observed that BCL2L12 mRNA expression was lower in advanced-stage LSCC. The authors suggested that BCL2L12 mRNA expression might serve as potential prognostic biomarkers in LSCC [18].
To enhance the knowledge of this subject, we investigated the prognostic value of BCL2L12 expression in patients with primary LSCC. We also examined the association of BCL2 and BAX protein expression (as well-examined biomarkers) with prognosis in the same cohort of primary LSCC.
Methods
Collection of tissue specimens
Prospectively collected cancerous laryngeal tissue specimens were obtained from patients with LSCC as pretreatment tissue biopsy, at the first Department of Otorhinolaryngology, Hippocration Hospital, Medical University of Athens, National and Kapodistrian University of Athens. All tissue specimens were examined by a pathologist of the Hippocration Hospital, who confirmed the malignancy. The specimens were collected between 2005 and 2012. The study was approved by the Institutional Ethics Committee of the National and Kapodistrian University of Athens, in accordance with the ethic standards of the 1964 Declaration of Helsinki and its latter amendments. Inclusion criteria included patients with a primary LSCC. Exclusion criteria included patients with recurrence, patients with metastatic disease at time of diagnosis and patients who refused treatment.
All tissue specimens were accompanied by detailed data such as age at diagnosis, TNM stage (VII Edition), primary tumor site (T-status), nodal status (N), metastasis (M), alcohol consumption and smoking (pack-years index). Laryngeal cancer site (glottic, supraglottic or subglottic area), multiple primary cancer sites, type of treatment, radiotherapy, chemotherapy and type of surgery were also recorded. Follow-up information was available for all patients and included disease-free survival (DFS) status, overall survival (OS) status (deceased or alive) along with the dates of the events, region of metastatic disease and cause of death. TNM stage, T-status and nodal status were analyzed independently to detect possible subgroups, as previously observed [19].
Immunohistochemistry
Paraffin sections 5
Immunohistochemical evaluation
Sections were evaluated under a light microscope. Ten fields at x200 were randomly selected and examined by two other experienced pathologists of the Department of Pathology, independently. In case of disagreement, the final score was determined by general agreement. All markers were separately evaluated in the cancerous cell population, the tumor-adjacent dysplastic epithelium and the stroma. The percentage of immunopositive cells (below 30% was characterized as weakly stained, between 31% and 69% as moderately stained, and above 70% as strongly stained) and a three-scale intensity scheme (staining intensity: negative
Statistical analysis
Descriptive statistics analysis was initially per- formed. Associations between staining positivity of BCL2L12, BCL2 and BAX and clinicopathological variables of LSCC patients were analyzed using the chi-square (
Clinicopathological characteristics of 78 LSCC patients
Clinicopathological characteristics of 78 LSCC patients
Primary treatment of 78 LSCC patients
Associations between BCL2L12 protein status and categorical clinicopathological variables of 78 LSCC patients
Clinicopathological characteristics of the study cohort
The study included 78 cancerous laryngeal tissue specimens. The mean age of the patients was 61.76
Patients’ primary treatment is presented in Table 2. All tumors were resected with clear margins either with transoral surgery with CO
During the follow-up period, the mean DFS time was 71.37
BCL2L12 protein expression in LSCC tissue specimens and its association with patients’ clinicopathological variants
BCL2L12 protein expression was categorized as positive or negative, as described in Material and methods. Thus, 44/78 (56.4%) LSCCs were classified as BCL2L12-positive and 34/78 (43.6%) as BCL2L12-negative (Table 3). Primary tumor site was found to be significant correlated to BCL2L12 protein expression status. LSCCs of high T-status were more frequently BCL2L12-negative than LSCCs of low T-status (
Associations between BCL2 and BAX protein status and categorical clinicopathological variables of 78 LSCC patients
Associations between BCL2 and BAX protein status and categorical clinicopathological variables of 78 LSCC patients
BCL2 protein expression was categorized as high or low, as described in Material and methods. Thus, 30/78 (38.5%) LSCCs were classified as BCL2-high and 48/78 (61.5%) as BCL2-low (Table 4). No significant associations were observed between BCL2 protein expression and T-status (
BAX protein expression was also categorized as high or low, as described in Material and methods (2.3). Thus, 19/78 (24.4%) LSCCs were classified as BAX-high and 59/78 (75.6%) as BAX-low (Table 4). T-status and TNM stage were found to be significantly correlated to BAX protein expression status. LSCCs of high T-status and high TNM stage were more frequently BAX-low than LSCCs of low T-status (
The role of BCL2L12 protein as a prognostic factor of LSCC patients’ survival
In order to evaluate BCL2L12 protein expression in terms of predicting survival outcome, we performed Kaplan-Meier survival analysis. In Kaplan-Meier survival curves with regard to DFS time, no significant difference was observed between LSCC patients with BCL2L12-positive tumors and LSCC patients with BCL2L12-negative tumors (
Kaplan-Meier survival curves for overall survival (OS) of LSCC patients with BCL2L12-positive tumors and LSCC patients with BCL2L12-negative tumors. 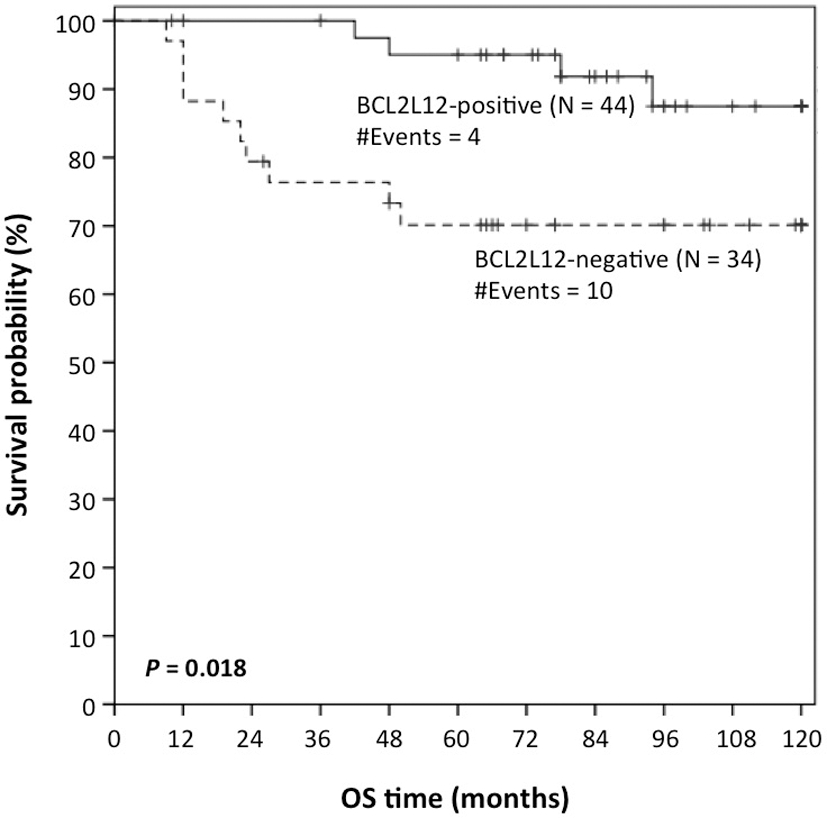
Kaplan-Meier survival curves for overall survival (OS) of advanced-stage LSCC patients with BCL2L12-positive tumors and advanced-stage LSCC patients with BCL2L12-negative tumors. 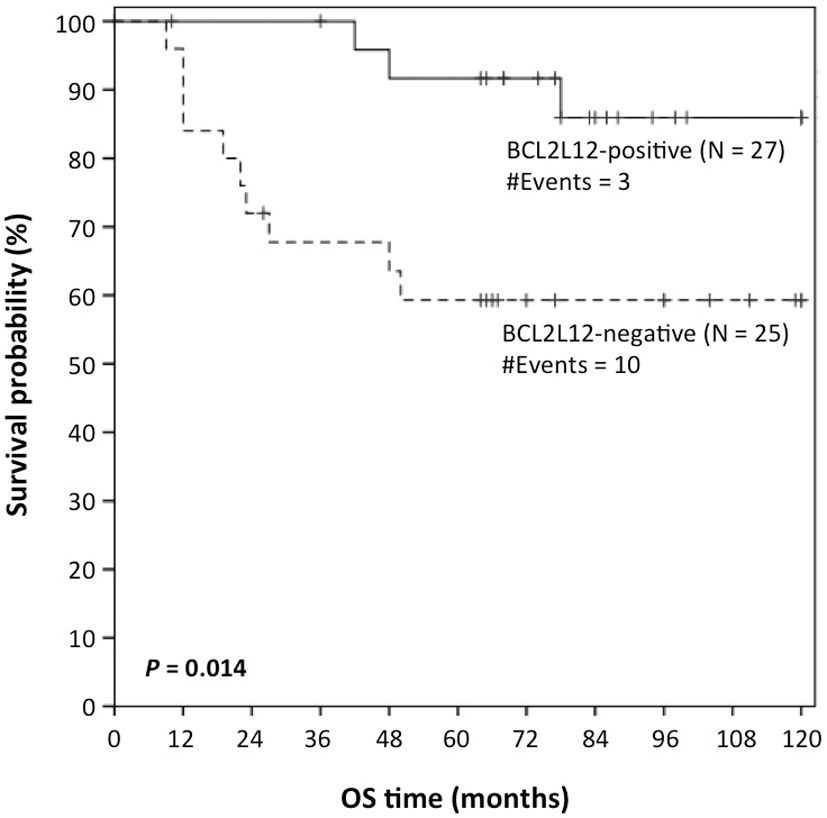
BCL2L12 protein expression, BCL2, BAX protein expression and survival of 78 LSCC patients
We also used Kaplan-Meier survival curves to further evaluate BCL2L12 protein expression in terms of predicting OS in early-stage (TNM stage I/II) and advanced-stage (TNM stage III/IV) LSCC patients. With regard to OS time, no significant difference was observed between early-stage LSCC patients with BCL2L12-positive tumors and early-stage LSCC patients with BCL2L12-negative tumors (
In Cox univariate survival analysis (Table 5), LSCC patients with BCL2L12-positive tumors had significantly lower risk of death compared to LSCC patients with BCL2L12-negative tumors (hazard ratio [HR]
In order to evaluate BCL2 and BAX protein expression in terms of predicting survival outcomes, we also performed Kaplan-Meier survival analysis. In Kaplan-Meier survival curves with regard to DFS and OS time, no significant difference was observed between LSCC patients with BCL2-high tumors (or BAX-high tumors) and LSCC patients with BCL2-low tumors (or BAX-low tumors) (
In Cox univariate survival analysis (Table 5) with regard to survival probabilities, no significant difference was observed between LSCC patients with BCL2-high tumors (or BAX-high tumors) and LSCC patients with BCL2-low tumors (or BAX-low tumors) (
Discussion
Recently, Geomela et al. observed that BCL2L12 mRNA expression was lower in advanced-stage LSCC in a cohort of patients with HNSCC. The authors suggested that BCL2L12 mRNA expression might represent a potential prognostic biomarker in LSCC [18]. Thus, the authors of the current study performed a review of the literature in Pubmed Database using combinations of the following keywords: ‘BCL2L12’, ‘larynx’, ‘laryngeal’, ‘cancer’, ‘survival’ and ‘prognosis’. Only Geomela’s study [18] matched our search terms. To enhance the knowledge of this subject, we examined the prognostic value of BCL2L12 expression in patients with primary LSCC. To further investigate the prognostic significance of BCL2 and BAX, we also examined the association of BCL2 and BAX protein expression with prognosis in primary LSCC.
The evaluation was performed with immunohistochemical analysis. Immunohistochemistry is an optimal technique to assess protein expression and it is easily and routinely used in clinical practice. The use of a semi-quantitative score for the evaluation has been described in several studies [21, 20]. Alternatively, a quantitative real-time PCR methodology, aiming at the determination of mRNA expression levels, could be used for the same purpose, as recommended by multiple studies [17, 18, 22, 23].
Our data demonstrated that BCL2L12 protein was significantly more frequently expressed in primary LSCC of low T-status than in primary LSCC of high T-status. These data indicated that positive BCL2L12 protein expression in primary LSCC could be correlated with better prognosis. In accordance with the aforementioned results, we observed in the COX univariate regression analysis that positive BCL2L12 protein expression in primary advanced-stage LSCC predicted a lower risk of death. This implied that positive BCL2L12 protein expression could be used as a favorable prognostic tissue biomarker of higher OS in advanced-stage primary LSCC. Accordingly, Kaplan-Meier survival analysis demonstrated that advanced-stage LSCC patients with BCL2L12-positive tumors had significantly higher OS time in comparison with advanced-stage LSCC patients with BCL2L12-negative tumors. As expected, early-stage LSCC patients had significantly higher OS time in comparison with ad- vanced-stage LSCC patients. Interestingly, positive BCL2L12 protein expression did not predict lower risk of recurrence. Similarly, in Kaplan-Meier survival curves, LSCC patients with BCL2L12-positive tumors had not significantly lower DFS-time in comparison with LSCC patients with BCL2L12-negative tumors.
BCL2L12, a novel member of the BCL2 apoptosis-related family, is involved in apoptosis and several malignancies. Data imply that BCL2L12 mRNA expression may be considered as favorable biomarker in gastric [14], colon [15] and breast cancer [24]. Our results suggest that BCL2L12 protein expression could be also used as a favorable prognostic tissue biomarker in patients with primary advanced-stage LSCC. Undoubtedly, further studies are needed to examine the potential application of BCL2L12 protein expression in clinical practice as a favorable prognostic tissue biomarker in primary advanced-stage LSCC and to elucidate the biochemical pathways in which BCL2L12 is involved.
Furthermore, our data demonstrated that high or low BCL2 and BAX protein expression did not predict recurrence or death. Also, high or low BCL2 and BAX protein expression did not significantly correlate with DFS and OS time. These results are in accordance with previously published studies investigating BCL2 [25] and BAX [8] expression in LSCC. The difference between BCL2L12 and BCL2 prognostic role in LSCC could be attributed to the fact that BCL2 is merely antiapoptotic, whereas BCL2L12 has been shown to act as a double-edge sword, either facilitating or impeding apoptosis, depending on the cell type and the expressed BCL2L12 splice variants and protein isoforms [26, 27].
Our study group consisted of 78 LSCC patients. The clinicopathological characteristics of our study group resembled characteristics of study groups described in larger studies with patients with laryngeal cancer [28, 29]. Thus, our smaller study group could be considered representative of laryngeal cancer patients. Additionally, treatment strategies correlated with international standards [30, 31, 32, 33, 34, 35]. Nevertheless, our preference for total laryngectomy in the advanced-stage LSCC could be criticized, since Jenckel et al. implied that aim of every treatment decision should be larynx preservation and preservation of laryngeal function [36]. However, the following studies supported our preference. Richard et al. noticed a significant better 2-year survival in T3-LSCC patients treated with surgery and adjuvant RT in comparison with T3-LSCC patients treated with concurrent CRTH alone (
In conclusion, positive protein expression of BCL2 L12 gene, a member of the BCL2-family, predicted significantly higher OS time and lower risk of death in patients with primary advanced-stage LSCC. Our results imply that BCL2L12 protein expression could be used as a favorable prognostic tissue biomarker in patients with primary advanced-stage LSCC. Further studies are needed to examine its use in the clinical practice. On the contrary, BCL2 and BAX protein expression did not correlate with prognosis in primary LSCC. Our data suggest that BCL2 and BAX cannot be used as potential prognostic tissue biomarkers in patients with primary LSCC.
Footnotes
Conflict of interest
The authors declare that they have no competing interests.
