Abstract
BACKGROUND:
Although up-regulation of EIF3B correlates with poor prognosis in carcinomas, the role of EIF3B in non-small cell lung cancer (NSCLC) is rarely known.
OBJECTIVE:
We aimed to investigate correlation of EIF3B with clinicopathological features and prognosis in NSCLC patients, and clarify its effect on cells proliferation and apoptosis.
METHODS:
Two hundred and eleven NSCLC patients underwent surgery were retrospectively reviewed. Tumor tissue and paired adjacent tissue were obtained. EIF3B expression was detected by immunohistochemistry, qPCR and western blot. EIF3B inhibitor, blank inhibitor, blank mimic and EIF3B mimic plasmids were transfected to A-549 cells. Cells proliferation and apoptosis were measured by CCK-8 and AV/PI. All processes were repeated for validation in PC9 cells.
RESULTS:
EIF3B expression increased in tumor tissue compared to paired adjacent tissue, and positively correlated with tumor size, lymph node metastasis and TNM stage. K-M curves revealed patients with EIF3B high expression had shorter DFS and OS, and its high expression independently predicted unfavorable DFS and OS. In vitro, EIF3B expression increased in NSCLC cells compared to normal cells. EIF3B increased cells proliferation but inhibited cells apoptosis.
CONCLUSIONS:
EIF3B overexpression correlates with advanced disease conditions and poor prognosis, and it promotes cells proliferation while inhibits apoptosis in NSCLC.
Introduction
Non-small cell lung cancer (NSCLC), accounting for over 85% of lung cancer, is one of the most frequently diagnosed malignancies and the leading cause of cancer related death worldwide [1, 2, 3]. Due to lack of obvious clinical symptoms, most patients are diagnosed as NSCLC at an advanced disease stage, contributing to worse prognosis, or even death [2, 4, 5]. Although imaging technology, targeted therapies, immunotherapies and individual treatment strategies have been greatly improved, the prognosis of NSCLC is still not satisfied since the 5-year survival rate is just nearly 15% [6]. Therefore, exploring new and convincing biomarkers for prognosis and clarifying novel underlying mechanisms of tumer progression of NSCLC are necessary.
Eukaryotic initiation factors (eIFs), a series of proteins or protein complexes, are essential in the rate-limiting initiation phase of eukaryotic translation, which play critical roles in the progress and prognosis of several carcinomas [7, 8, 9, 10]. As a member of the eIFs family, Eukaryotic translation initiation factor 3 subunit B (EIF3B) is highly expressed in several carcinomas, including bladder, breast, and prostate cancers [10, 11, 12]. Despite of the fact that up-regulation of EIF3B is correlated with poor prognosis in many carcinomas, the role of EIF3B in NSCLC is rarely known [11, 13, 14]. Therefore, our purpose was to investigate the correlation of EIF3B expression with clinicopathological features and prognosis in NSCLC patients, as well as clarify the effect of EIF3B on the cells proliferation and apoptosis in NSCLC cells.
Methods
Participants
Two hundred and eleven NSCLC patients underwent surgery at The Central Hospital of Wuhan from Jan 2009 to Dec 2012 were retrospectively reviewed in this study. The inclusion criteria were as followed: (1) Diagnosed as primary NSCLC based on clinical and pathological confirmation. (2) Age above 18 years. (3) Completed information of tumor features was available to retrieve. (4) Regularly follow-up patients. (5) Formaldehyde fixed, paraffin embedded tumor tissue sample and paired adjacent tissue sample were available to obtain. The exclusion criteria were as follows: (1) Received neoadjuvant therapy before surgery. (2) History of other solid tumors or hematologic malignancy.
Ethics approval
This present study was approved by the Ethics Committee of The Central Hospital of Wuhan, and was conducted under the Statement of Helsinki. All the participants or their guardians signed the informed consents or orally agreed with the informed consents by telephone with tape recording.
Data collection, follow up and survival calculation
Baseline information including age, gender, pathological grade, tumor size, lymph node metastasis and TNM stage were reviewed and collected by Electronic Medical Record System of The Central Hospital of Wuhan in this study. TNM stage was classified according to 7
Immunohistochemistry (IHC) assay for EIF3B expression in tissues
Formaldehyde fixed, paraffin embedded tumor tissue sample and paired adjacent tissue sample were obtained from the Specimen Repository of The Central Hospital of Wuhan. After being deparaffinized and rehydrated, antigen retrieval was conducted in tissue sections in Tris-Ethylene Diamine Tetraacetic Acid (EDTA). Then endogenous peroxidase was block by H
As to the expression of EIF3B assessment, the positive cells were observed and counted under a light microscopy by two specialists without knowing any information about patients. In each slide, 100 cells in 5 high-power fields (HPF,
Cells culture
NSCLC cell lines including A-549, NCI-H1650, NCI-H1299 and PC9 as well as normal lung epithelial cells (DEAS-2B) were purchased from Shanghai Institutes for Biological Science (Shanghai, China) or obtained from Shanghai QeeJen Bio-tech Institution (Shanghai, China). A-549 cells were cultured in RPMI F12K medium (Gibco, USA) supplemented with 10% (v/v) fetal bovine serum (FBS) (Gibco, USA) and antibiotics (100 units/ml penicillin and 100
Detection of EIF3B expression in NSCLC cell lines
Total RNA was extracted from NSCLC cell lines (A-549, NCI-H1650, NCI-H1299 and PC9) and normal lung epithelial cells (DEAS-2B) by TRIzol Reagents (Invitrogen, USA). Quantitative polymerase chain reaction (qPCR) assay and Western Blot assay were performed to detect the mRNA and protein expressions of EIF3B respectively.
EIF3B plasmids transfection and subsequent assays in A-549 cells
EIF3B inhibitor plasmid, blank inhibitor plasmid, blank mimic plasmid and EIF3B mimic plasmid were designed and constructed by a Shanghai Gemma company (Shanghai, China). These plasmids were transfected into A-549 cells as EIF3B (-), NC (-), NC (+) and EIF3B (+) groups. qPCR assay and Western Blot assay were performed to detect the mRNA and protein expressions of EIF3B at 24 h respectively. counting kit-8 (CCK-8) (Abcam, USA) assay was performed to determine cells proliferation at 0 h, 24 h, 48 h and 72 h. Annexin V (AV) apoptosis detection kit with propidium iodide (PI) (Invitrogen, USA) was used to detect cells apoptosis rate at 72 h.
Validation in PC9 cells
In order to further validate the effect of EIF3B on cells proliferation and apoptosis in NSCLC, additional experiments in PC9 cells were carried out. EIF3B inhibitor plasmid, blank inhibitor plasmid, blank mimic plasmid and EIF3B mimic plasmid were transfected into PC5 cells, which were classified as EIF3B (-), NC (-), NC (+) and EIF3B (+) groups respectively. After transfection at 0 h, 24 h, 48 h and 72 h, CCK-8 assay (Abcam, USA) for evaluation of cells proliferation was carried out. At transfection at 72 h, AV-PI (Invitrogen, USA) for cells apoptosis rate was performed.
qPCR assay
The mRNA level of EIF3B was detected by qPCR as follows: According to the manufacturer’s instructions, total RNA samples were extracted with TRIzol reagent (Invitrogen, USA). Total RNA was quantified by OD 260 (Takara, Japan). Then, 1
Western blot assay
Cells were lysed with radioimmunoprecipitation lysis buffer (1% protease inhibitor cocktail, 1% phenylmethanesulfonyl fluoride), and clarified by centrifugation. Subsequently, the protein concentration was detected by the BCA
CCK-8 assay
Ten
AV/PI assay
After being digested by pancreatin and washed by phosphate buffer solution (PBS), cells were suspended in 100
Statistics
Statistical analysis was performed using SPSS Software 21.0 (IBM, USA) and GraphPad Prism 5.01 (GraphPad, USA). Data were mainly presented as mean
Characteristics of NSCLC patients
Characteristics of NSCLC patients
Data were presented as mean
Comparison of EIF3B expression between tumor tissue and paired adjacent tissue
Data were presented as count (percentage). Comparison was determined by McNemar test.
EIF3B expression in tumor tissue and paired adjacent tissue. A: Example of EIF3B high expression in tumor tissue. B: Example of EIF3B low expression in paired adjacent tissue. EIF3B, eukaryotic translation initiation factor 3 subunit B.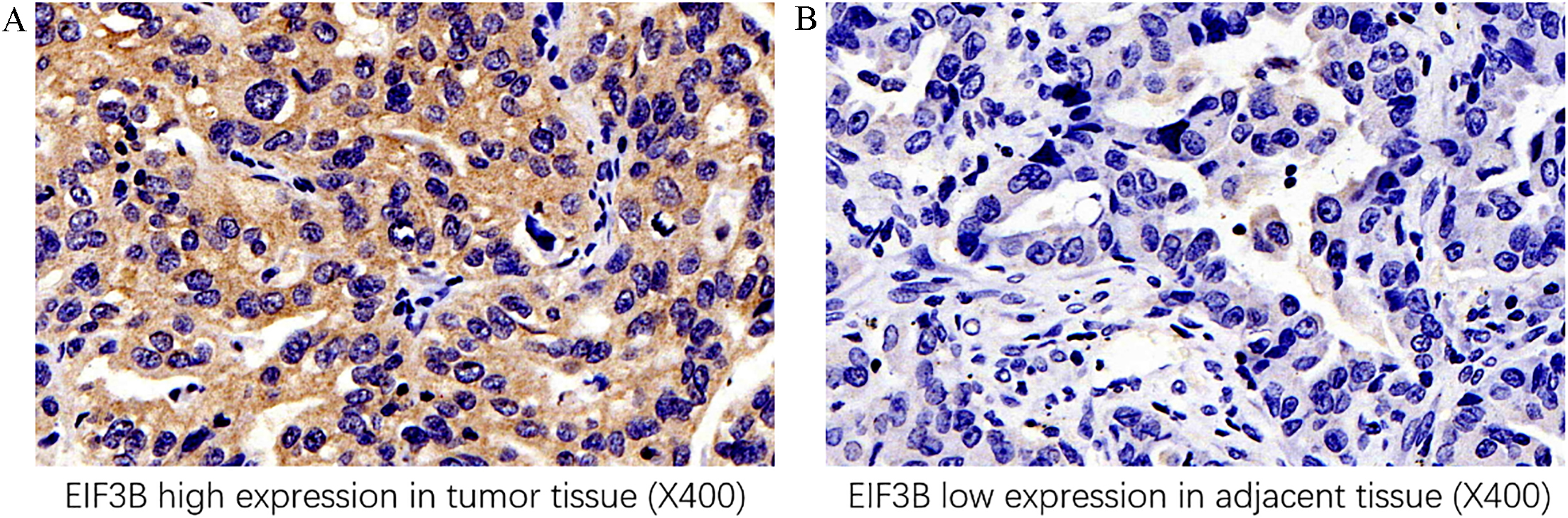
Baseline characteristics
As shown in Table 1, the mean age of 211 NSCLC patients was 61.31
EIF3B expression in tumor tissue and paired adjacent tissue
EIF3B expression was detected by IHC assay, which disclosed that EIF3B expression was higher in tumor tissue (Fig. 1A) compared to paired adjacent tissue (Fig. 1B) (
Correlation of EIF3B expression with clinicopathological features of NSCLC patients
Correlation of EIF3B expression with clinicopathological features of NSCLC patients
Data were presented as count (percentage). Comparison was determined by Chi-square test.
EIF3B expression was positively correlated with tumor size (
Correlations of EIF3B expression with DFS and OS in NSCLC patients
K-M curves revealed that NSCLC patients with EIF3B high expression presented with shorter DFS (
Factors affecting DFS in NSCLC patients by Cox’s analysis
Univariate Cox’s proportional hazards regression was employed to assess factors influencing DFS in NSCLC patients (Table 4), which disclosed that EIF3B high expression (
Cox’s analysis of factors affecting DFS
Cox’s analysis of factors affecting DFS
Data were presented as P value, HR and 95%CI. Baseline factors affecting DFS were determined by univariate and multivariate Cox’s proportional hazards regression model analysis.
Correlation of EIF3B expression with DFS and OS in NSCLC patients. A: Shorter DFS was observed in EIF3B high expression patients. B: Worse OS was presented in patients with EIF3B high expression. Kaplan-Meier curves and log-rank test were performed for comparison of DFS and OS. 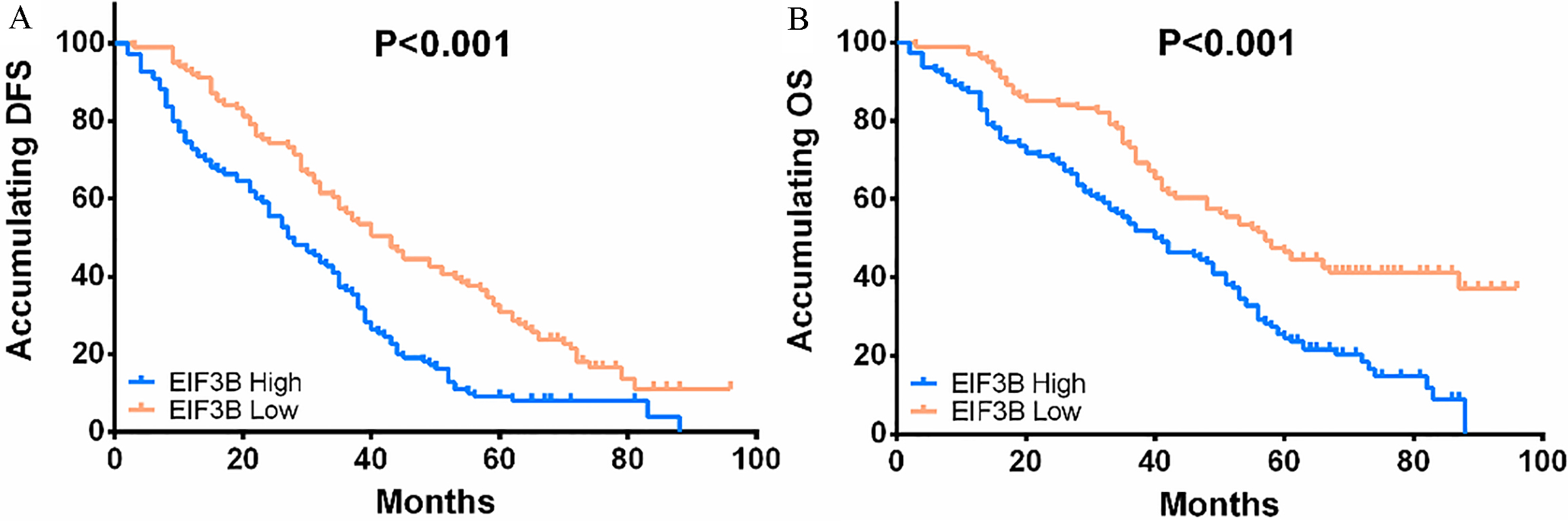
EIF3B expression in NSCLC cell lines. A: mRNA level of elF3B was increased in NSCLC cell lines (including A-549, NCI-H1299, NCI-H1650 and PC9 cells) compared to normal lung epithelial cell line DEAS-2B, and the most significant elevation was in A-549 cells. B: Protein level of elF3B was upregulated in NSCLC cell lines including A-549, NCI-H1299, NCI-H1650 and PC9 cells compared to normal lung epithelial cell line DEAS-2B, and most significantly increased in A549 cells. Comparison between two groups was determined by t test. 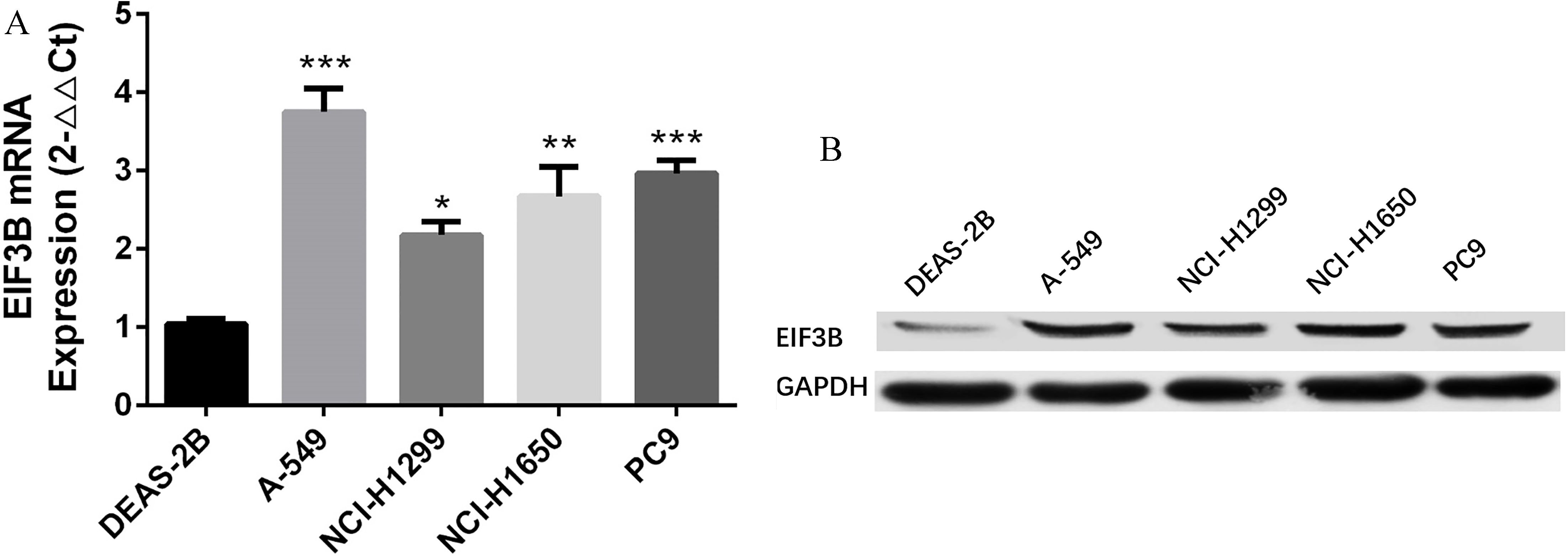
Cox’s analysis of factors affecting OS
Data were presented as
EIF3B promoted proliferation and inhibited apoptosis of A-549 cells. A: mRNA level of EIF3B was downregulated in EIF3B (-) group and upregulated in EIF3B (+) group compared to corresponding NC groups. B: Protein level of EIF3B was lower in EIF3B (-) group and higher in EIF3B (+) group than corresponding NC groups. C: EIF3B promoted cells proliferation of A549 cells. D and E: EIF3B inhibited cells apoptosis of A549 cells. Comparison between two groups was determined by t test. 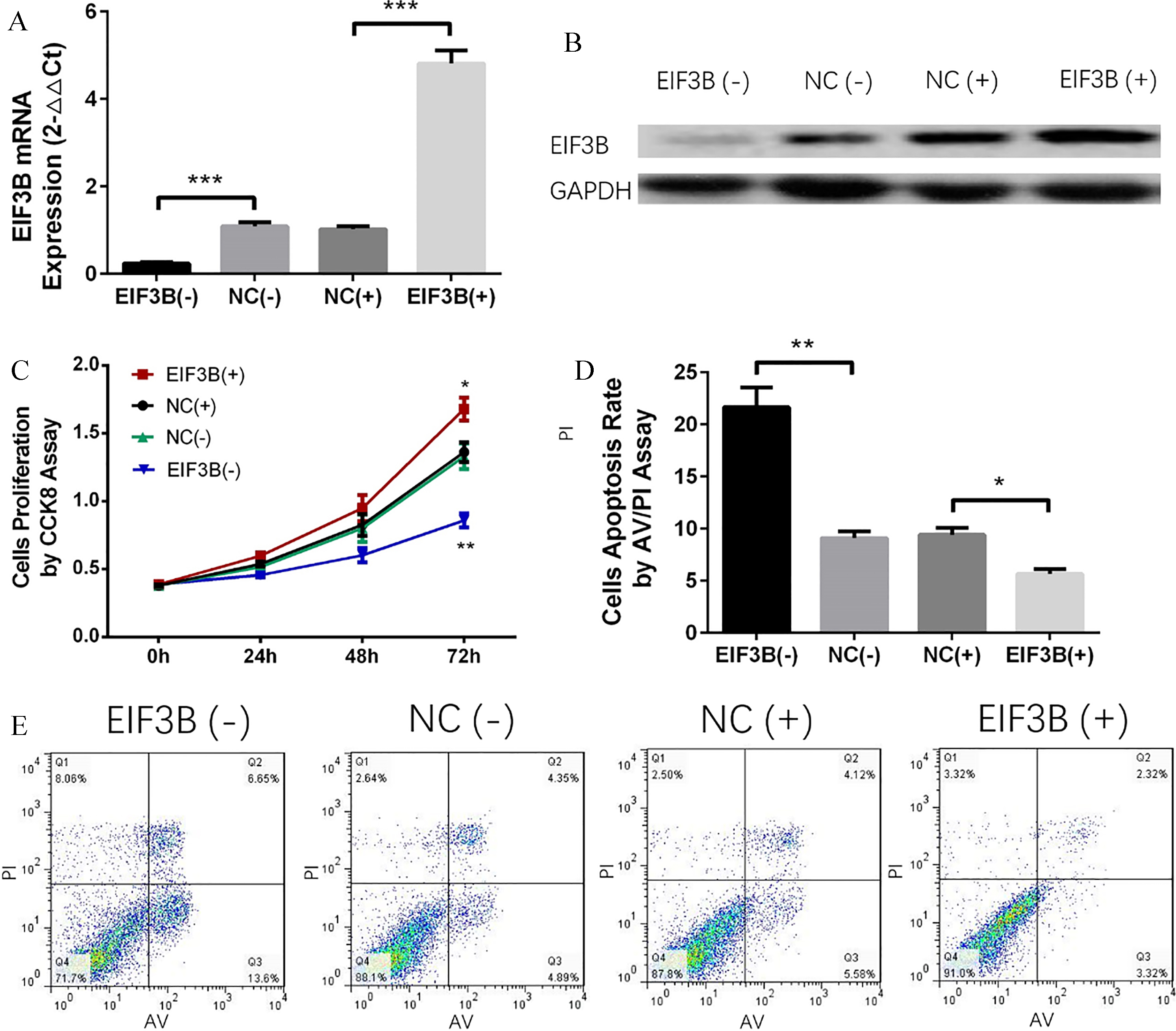
As shown in Table 5, EIF3B high expression (
EIF3B expression in NSCLC cell lines
In order to investigate the effect of ElF3B on NSCLC cells, in vitro experiments were further performed. Compared to normal lung epithelial cells DEAS-2B, both mRNA (Fig. 3A) and protein expressions (Fig. 3B) of EIF3B were increased in NSCLC cell lines including A-549, NCI-H1299, NCI-H1650 and PC9, and was most significantly elevated in A-549 cells. Therefore, A-549 cell line was chosen for the following in vitro experiments.
EIF3B induced cells proliferation and suppressed cells apoptosis of PC9 cells. A: mRNA level of EIF3B was decreased in EIF3B (-) group and increased in EIF3B (+) group compared to corresponding NC groups. B: Protein level of EIF3B was decreased in EIF3B (-) group and increased in EIF3B (+) group than corresponding NC groups. C: EIF3B promoted cells proliferation of PC9 cells. D-E: EIF3B inhibited cells apoptosis of PC9 cells. Comparison between two groups was determined by t test. 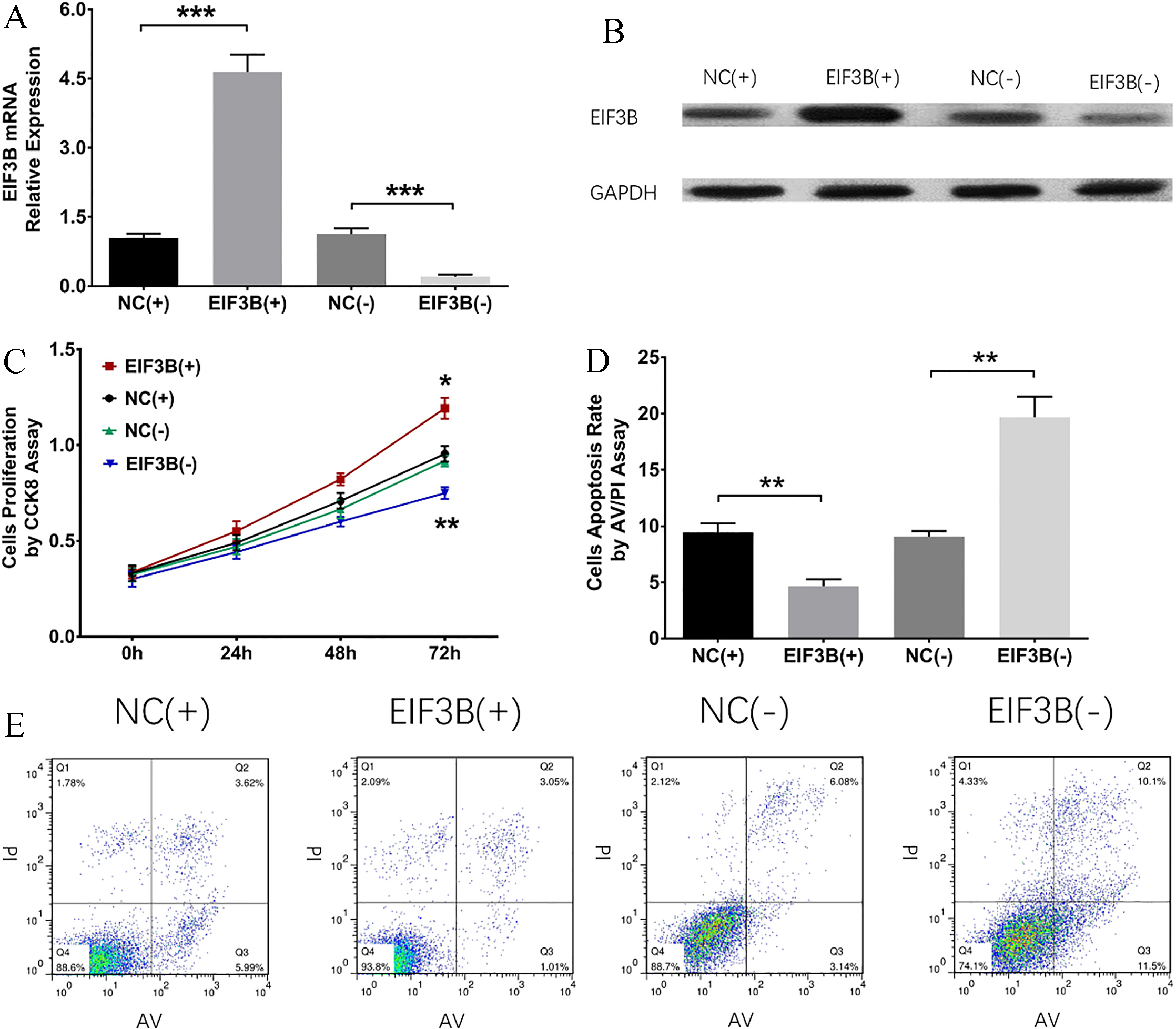
Both mRNA and protein expressions of EIF3B were decreased in EIF3B (-) group compared to NC (-) group and increased in EIF3B (+) group compared to NC (+) group, indicating the transfection was successful (Fig. 4A and B). At 72 h, cells proliferation was promoted in EIF3B (+) group compared to NC (+) group, while it was suppressed in EIF3B (-) group compared to NC (-) group (Fig. 4C), suggesting that EIF3B promoted proliferation in A-549 cells. Cells apoptosis rate was increased in EIF3B (-) group compared to NC (-) group, while it was decreased in EIF3B (+) group compared to NC (+) group at 72 h after transfection (Fig. 4D and E), suggesting that EIF3B inhibited apoptosis in A-549 cells.
Validation for the effect of EIF3B on cells proliferation and cells apoptosis in PC9 cells
To further verify the efficacy of EIF3B on cells proliferation and apoptosis in NSCLC, additional experiments were repeated in another NSCLC cells line PC9 cells. As shown in Fig. 5, mRNA and protein expressions of EIF3B were evaluated in EIF3B (+) group compared to NC (+) group and reduced in EIF3B (-) group compared to NC (-) group, suggesting the transfection was successful (Fig. 5A and B). Cells proliferation was increased in EIF3B (+) group compared to NC (+) group and was decreased in EIF3B (-) group compared to NC (-) group at 72 h (Fig. 5C), indicating that EIF3B promoted proliferation in PC9 cells. For cells apoptosis rate, it was decreased in EIF3B (+) group compared to NC (+) group but increased in EIF3B (-) group compared to NC (-) group at 72 h after transfection (Fig. 5D and E), which indicated that EIF3B suppressed cells apoptosis in PC9 cells.
Discussion
In this study, we observed that: (1) EIF3B expression was higher in tumor tissue than that in adjacent tissue, and it was positively associated with tumor size, lymph node metastasis and TNM stage. Moreover, EIF3B overexpression correlated with worse DFS and OS independently in NSCLC patients. (2) EIF3B promoted proliferation and inhibited apoptosis in NSCLC cells.
EIFs families, which participate in the eukaryotic translation via regulating interaction between ribosome and mRNA, are propo15sed as new therapeutic targets for carcinomas due to their functions in promoting tumorigenesis [14, 16, 17]. An interesting experiment indicates that down-regulation of eukaryotic translation initiation factor 5A2 (EIF5A2) suppresses cells growth in ovarian cancer cell line UACC-1598 [18]. Also, eukaryotic translation initiation factor 3 subunit D (EIF3D) plays a crucial role in promoting cells proliferation via affecting G2/M phase arrest in NSCLC [19]. Additionally, eukaryotic initiation translation factor 2
EIF3B, a common member of EIFs family, has been observed to be overexpressed in many cancers including esophageal squamous cell carcinoma, clear cell renal cell carcinoma, and prostate cancers [11, 14, 17]. In addition, up-regulation of EIF3B is found to be positively correlated with tumor depth, lymph node metastasis and TNM stage in patients with esophageal squamous cell carcinoma [17]. Also, EIF3B high expression is associated with an aggressive tumor phenotype in patients with clear cell renal cell carcinoma [14]. Furthermore, EIF3B expression positively correlates with tumor grade in patients with bladder and prostate cancers [11]. Therefore, these previous studies suggest that EIF3B overexpression associates with advanced disease conditions in patients with several cancers. In line with those previous data, we found that EIF3B expression was higher in tumor tissue than that in adjacent tissue, and it was positively correlated with tumor size, lymph node metastasis and TNM stage in NSCLC patients. The possible reasons are as follows: EIF3B has effect on promotion of cells proliferation, facilitation of cells migration or inhibition of cells apoptosis via modulating various genes expressions or pathways activations (including decreasing integrin
As to the predictive value of EIF3B on the prognosis of cancers, some studies have been performed. For instance, esophageal squamous cell carcinoma patients with high EIF3B expression present with poor DFS and OS [17]. For clear cell renal cell carcinoma, patients with elF3B overexpression also illuminate shorter OS [14]. Additionally, up-regulation of EIF3B correlates with poor disease-specific survival in patients with bladder cancer [11]. Even though those previous studies indicate that EIF3B acts as a novel biomarker for worse prognosis in some cancers, the effect of EIF3B on the prognosis of NSCLC is still unclear. In the present study, we observed that EIF3B overexpression independently correlated with worse DFS and OS in NSCLC patients. The possible reasons were as follows: EIF3B could influence expressions of some proteins or activation of pathways (such as reducing apoptosis inhibitory protein BCL2 expression or activating the pro-survival Akt pathway) to induce cells proliferation and inhibit cells apoptosis, thereby devoting to advanced disease conditions and finally leading to poor prognosis in NSCLC patients [14, 21, 22]. Moreover, elF3B might promote drug resistance via mediating some pathways, including activating the Akt pathway, thereby decreasing treatment efficacy and resulting in poor prognosis in NSCLC patients [21].
In order to investigate the function of EIF3B in cancers and clarify the underlying mechanisms, some in vitro experiments have been performed. For example, eIF3b contributes to osteosarcoma cells proliferation by regulating tumor necrosis factor receptor superfamily member 21 expression [13]. Also, depletion of EIF3B inhibits cells growth, prevents cells migration, hinders cells cycle, as well as disrupts actin cytoskeleton and focal adhesions by decreasing the expression of integrin
In conclusion, EIF3B overexpression correlates with advanced disease conditions and poor prognosis, and it promotes cells proliferation while inhibits cells apoptosis in NSCLC.
