Abstract
OBJECTIVE:
Colorectal cancer (CRC) is the 3
METHODS:
Quantitative real-time PCR (qRT-PCR) was performed to determine relevant gene expression levels; western blot was performed to determine protein expression levels; CCK-8, colony formation, wound healing and Transwell invasion assays were used to determined CRC cell proliferation, migration and invasion; in vivo tumor growth was assessed in xenograft mice model.
RESULTS:
TP73-AS1 was up-regulated in both CRC tissues and CRC cell lines. Overexpression of TP73-AS1 was associated with metastasis and advanced clinical stages in CRC patients. Overexpression of TP73-AS1 promoted CRC cell growth, proliferation, migration and invasion in vitro; and knockdown of TP73-AS1 significantly inhibited CRC cell growth, proliferation, migration and invasion in vitro as well as tumor growth in vivo. Bioinformatics analysis and luciferase reporter assay indicated that TP73-AS1 could bind directly with miR-194, and TP73-AS1 negatively regulated the expression of miR-194 in CRC cells. Further study indicated that miR-194 negatively regulated the downstream target of transforming growth factor alpha (TGF
CONCLUSIONS:
this study demonstrated that TP73-AS1 regulated CRC progression by acting as a competitive endogenous RNA to sponge miR-194 to modulate the expression of TGF
Introduction
Colorectal cancer (CRC) is the third most common cancer and is one of the leading cause of cancer death worldwide [1]. Up to date, although many achievements have been made in the diagnosis and therapy of CRC, a great majority of the CRC patients are diagnosed at an advanced stage that has missed the optimal time for a radical operation, and CRC still causes significant morbidity and mortality [2, 3, 4]. A growing evidence revealed that CRC carcinogenesis is a complicated biological process involving many genomic mutations and complex biological signaling networking [5, 6, 7]. In this regard, to further understand the molecular mechanisms underlying the development and progression of CRC is critical for us to find novel approach for the management of CRC.
Recently, studies have found that protein-coding genes constitute only small portions of the genome encoding proteins, and most of the human genome is transcribed into noncoding RNAs such as microRNAs (miRNAs) and long non-coding RNAs (lncRNAs) [8]. The lncRNAs are a class of newly discovered non-coding RNAs, and the deregulation of lncRNAs have been implicated in the pathogenesis of various human diseases, particularly in cancer [9, 10]. In the aspect of CRC, lncRNAs have been suggested for their important roles in the CRC diagnosis, therapy, and prognosis. For instance, the lncRNA HOTAIR was found to regulate polycomb-dependent chromatin modification and was associated with poor prognosis in CRC [11]. Takahashi et al., showed that amplification of PVT-1 inhibited CRC cell apoptosis and contributed to poor prognosis in CRC [12]. The lncRNA MALAT1 was shown to promote tumor growth and metastasis in colorectal cancer through binding to SFPQ and releasing oncogene PTBP2 from SFPQ/PTBP2 complex [13]. Recently, lncRNAs were found to function as a competitive endogenous RNA (ceRNA) to sponge miRNAs, which in turn regulated the CRC progression. Huang et al., showed that the novel long intergenic noncoding RNA UCC promoted colorectal cancer progression by sponging miR-143 [14]. The lncRNA FBXA19-AS functioned as an oncogene by sponging miR-203 in CRC [15]. The lncRNA P73 antisense RNA 1T (TP73-AS1) is a newly discovered lncRNA, and TP73-AS1 is the antisense of the coding gene TP73, which is located on human chromosomal band lp36.32 [16]. As far as we know, TP73-AS1 was found to play oncogenic role in various types of cancers including myeloma, esophageal squamous cell carcinoma, glioma, liver cancer, and breast cancer [16, 17, 18, 19, 20, 21]. However, no study has investigated the role of TP73-AS1 in CRC.
Given the oncogenic role of TP73-AS1 in CRC, in the present study, we found that TP73-AS1 was up-regulated in the CRC tissues and the overexpression of TP73-AS1 was associated with metastasis and advanced clinical stage. Further in vitro and in vivo studies also showed that TP73-AS1 had enhanced effects on the CRC cell proliferation, migration and invasion in vitro, and knock-down of TP73-AS1 suppressed tumor growth in vivo. In addition, the mechanistic studies also revealed that TP73-AS1/miR-194/transforming growth factor alpha (TGF
Materials and methods
Clinical samples
The CRC tissues and adjacent normal colon tissues were obtained from 84 CRC patients who underwent surgical resection in The First Affiliated Hospital of Xi’an Medical University from January 2013 to December 2015. None of the patients received any treatment before surgery, and all the clinical samples were confirmed by the pathologists. The clinical and pathological features of the CRC patients were obtained from patients’ history record. This study was approved by the Ethics Committee of The First Affiliated Hospital of Xi’an Medical University and all patients provided informed consent before participating in this study.
Cell lines
The human embryonic kidney (HEK) 293T cells, the normal colon epithelial cell line, NCM460 and CRC cell lines including HT29, SW480 and HCT116 were purchased from the American Type Culture Collection (Manassas, VA, USA). Cells were cultured in DMEM (Sigma, St. Louis, MO, USA) supplemented with 10% fetal bovine serum (FBS; Sigma, St. Louis, MO, USA) and cells were kept in a humidified incubator with 5% CO
Vector construction, miRNAs, siRNAs and cell transfection
The pcDNA3.1 vector and TP73-AS1 overexpression vector (pcDNA3.1-TP73-AS1) were commercially purchased from Genepharma (Shanghai, China). The miRNAs including mimics control, miR-194 mimics, inhibitors control and miR-194 inhibitors, and the siRNAs including control siRNA and TGF
RNA extraction and quantitative real-time PCR (qRT-PCR) analysis
Total RNA from cells or tissues were extracted by using the TRIzol reagent (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions. The All-in-One miRNA qRT-PCR Detection Kit (GeneCopoeia, Rockville, MD, USA) was used to detect miR-194 expression level according to the manufacturer’s instruction. The reverse transcription for TP73-AS1 and TGF
CCK-8 assay
Cell proliferation was detected by the CCK-8 assay (Beyotime, Beijing, China). Briefly, cells were seeded on a 96-well plate and cells were then incubated with CCK-8 solution for 2 h at room temperature according to the manufacturer’s instructions. The cell proliferative index was measured by detecting the absorbance at the wavelength of 450 nm.
Colony formation assay
Cell growth was detected by the colony formation assay. Briefly, cells were trypsinized and re-suspended in DMEM with 10% FBS, and the cells were then seeded in the six-well plates and cultured in a humidified incubator containing 5% CO
Wound healing assay
The cell migration was detected by the wound healing assay. Briefly, cells were trypsinized and seeded in the 6-well plates, and 12 h later, an artificial wound was created by using a sterile 200
Cell invasion assay
The cell invasion was determined by the Transwell cell invasion assay. Briefly, cells were trypsinized and cells in serum-free medium were seeded on the upper chamber with 8
Lentivirus packaging
The retrovirus system (Clontech Laboratories, Mountain View, CA, USA) was used to establish cell lines with stable down-regulation of TP73-AS1. The sh-TP73-AS1 was subcloned into the pLNCX2 plasmids, and GP293 cells were transfected with pLNCX2-sh-TP73-AS1 or pLNCX2 and the package plasmids according to the manufacturer’s instructions. Virus particles were harvested 48 h after transfection. HCT116 cells were infected with virus particles and maintained in DMEM containing G418. After 2 weeks of selection, cells with stable down-regulation of TP73-AS1 or control cells were pooled.
TP73-AS1 is significantly up-regulated in CRC tissues and cell lines. (A) Relative expression of TP73-AS1 in CRC tissues (
The 5-week-old BABL/c nude mice were obtained from the the Animal Center of the Chinese Academy (Shanghai, China). Briefly, 1
Luciferase reporter assay
The fragment from TP73-AS1 containing the predicted miR-194-binding site and the fragment from 3’UTR of TGF
Western blot assay
The protein levels were detected by western blot assay. Total cellular proteins were extracted and separated by using SDS-PAGE, and western blotting was performed in accordance with standard procedures. The primary antibodies including anti-TGF
Effects of TP73-AS1 on CRC colony formation, cell proliferation, migration and invasion. (A) Transfection of pcDNA3.1-TP73-AS1 up-regulated TP73-AS1 expression as measured by qRT-PCR assay in HT29 cells (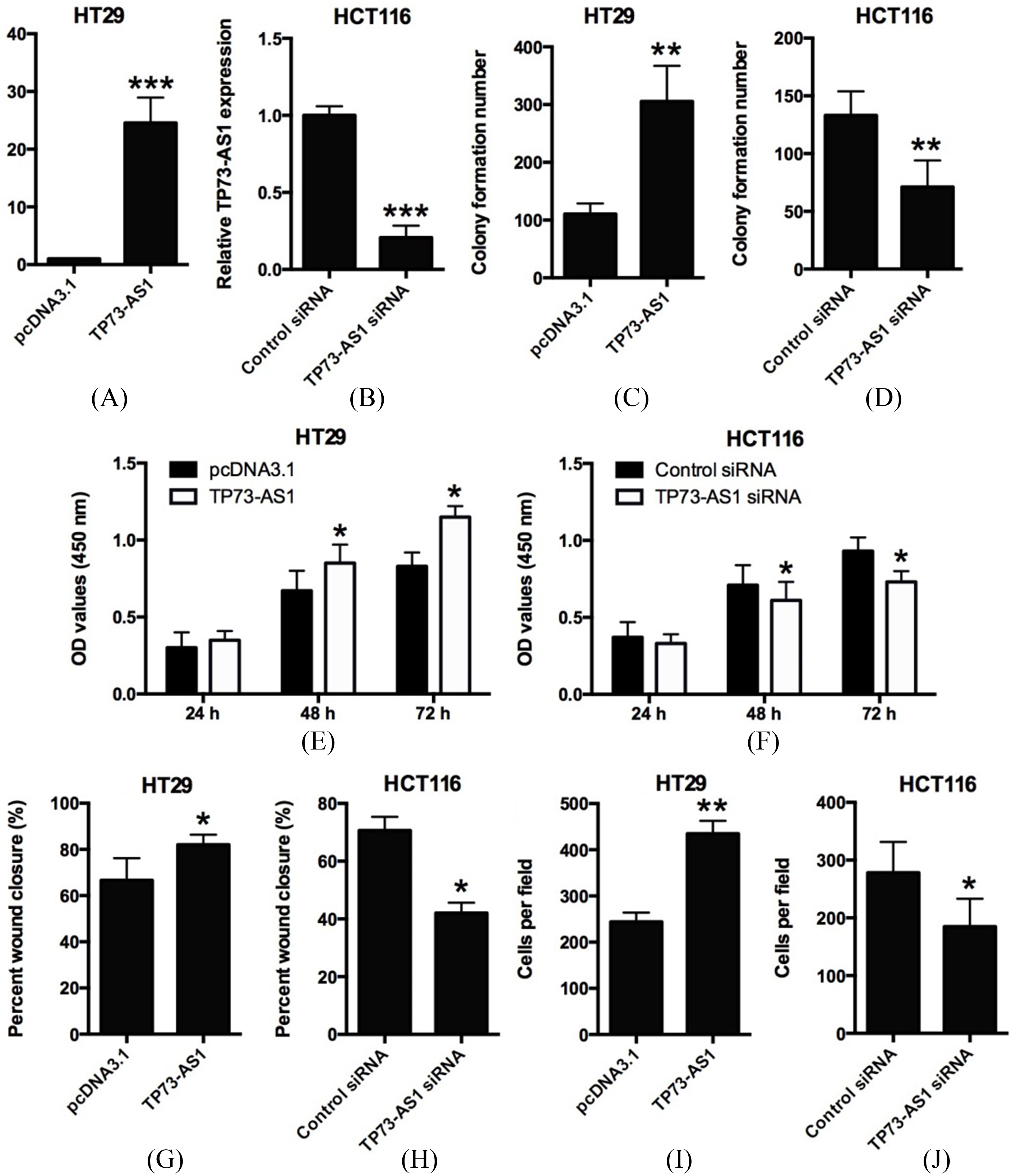
Knockdown of TP73-AS1 inhibits tumor growth in vivo. (A) Knockdown of TP73-AS1 significantly inhibited tumor growth of HCT116 cells in nude mice model. The tumor volume formed by HCT116 sh-TP73-AS1 cells was significantly lower than that of HCT116 sh-NC cells (
The statistical analysis and graph plotting were performed by using the GraphPad Prism software version 6.0. Significant differences between the in vivo and in vitro data were determined by using Student’s t tests or one-way ANOVA. The correlation between TP73-AS1 expression and miR-194 expression in CRC tissues were analyzed by Pearson correlation test. The data are presented as mean
Results
The expression of TP73-AS1 in CRC tissues and cell lines
The expression of TP73-AS1 in CRC tissues and cell lines was detected by qRT-PCR assay. We found that TP73-AS1 was significantly up-regulated in 84 CRC tissues compared with 84 paired adjacent normal tissues (Fig. 1A). To further examine the clinical significance of TP73-AS1 in CRC, the expression of TP73-AS1 was compared among different groups based on clinical features. The expression level of TP73-AS1 in CRC tissues from patients with distant metastasis was significantly higher than that from patients without distant metastasis (Fig. 1B). In addition, the expression level of TP73-AS1 was significantly increased with more advanced clinical stage (Fig. 1C). In addition to the expression of TP73-AS1 in the tissues, TP73-AS1 was up-regulated in CRC cell lines including HT29, SW480 and HCT116 compared with normal colon cell line, NCM460 (Fig. 1D).
Effects of TP73-AS1 on CRC colony formation, cell proliferation, migration and invasion
Based on the expression features of TP73-AS1 in clinical samples and CRC cells lines, we hypothesized that TP73-AS1 might play an oncogenic role in CRC. Firstly, we performed the cell transfection experiments to transiently overexpress the TP73-AS1 by transfecting HT29 cells with TP73-AS1-overexpressing vector (pcDNA3.1-TP73-AS1) (Fig. 2A), and to transiently knockdown TP73-AS1 by transfection HCT116 cells with TP73-AS1 siRNA (Fig. 2B). Then, we conducted colony formation, CCK-8, wound healing and cell invasion assays, and the results showed that ectopic TP73-AS1 overexpression significantly promoted colony formation (Fig. 2C), cell proliferation (Fig. 2E), cell migration (Fig. 2G) and cell invasion (Fig. 2I) in HT29 cells compared with control group (pcDNA3.1 group). On the other hand, knockdown of TP73-AS1 by TP73-AS1 siRNA transfection significantly suppressed colony formation (Fig. 2D), cell proliferation (Fig. 2F), cell migration (Fig. 2H) and cell invasion (Fig. 2J) in HCT116 cells. Collectively, these results demonstrated that TP73-AS1 is a positive regulator of cell proliferation, migration and invasion in CRC cells.
Knockdown of TP73-AS1 inhibits tumorigenesis in vivo
To further examine if TP73-AS1 could regulate tumorigenesis in vivo, cells (HCT116/sh-NC and HCT116/sh-TP73-AS1) were subcutaneously injected into the flank of the nude mice, and the tumor volume was monitored every 7 d for 42 d post inoculation. At the end of the experiment, the mice were killed and the tumors were taken out. The results showed that the tumor volume formed by HCT116/sh-TP73-AS1 cells were markedly less than that formed by HCT116/sh-NC cells (Fig. 3A). The qRT-PCR results confirmed that the expression of TP73-AS1 in tumor tissues from sh-TP73-AS1 group was significantly higher than that from sh-NC group (Fig. 3B).
Effects of TP73-AS1 on miR-194 expression. (A) Schematic representation of the predicted target site for miR-194 in TP73-AS1.(B) Luciferase reporter assay in HEK293T cells co-transfected with reporter plasmid and the indicated miRNAs. Overexpression of miR-194 significantly decreased the luciferase activity in the wild-type reporter plasmid (wild-type pmirGLO-TP73-AS1); knockdown of miR-194 significantly increased the luciferase activity in the wild-type reporter plasmid (
Studies have demonstrated that lncRNAs may involve in the ceRNA regulatory network. By using the online software program (DIANA tools;
Effects of miR-194 on TGF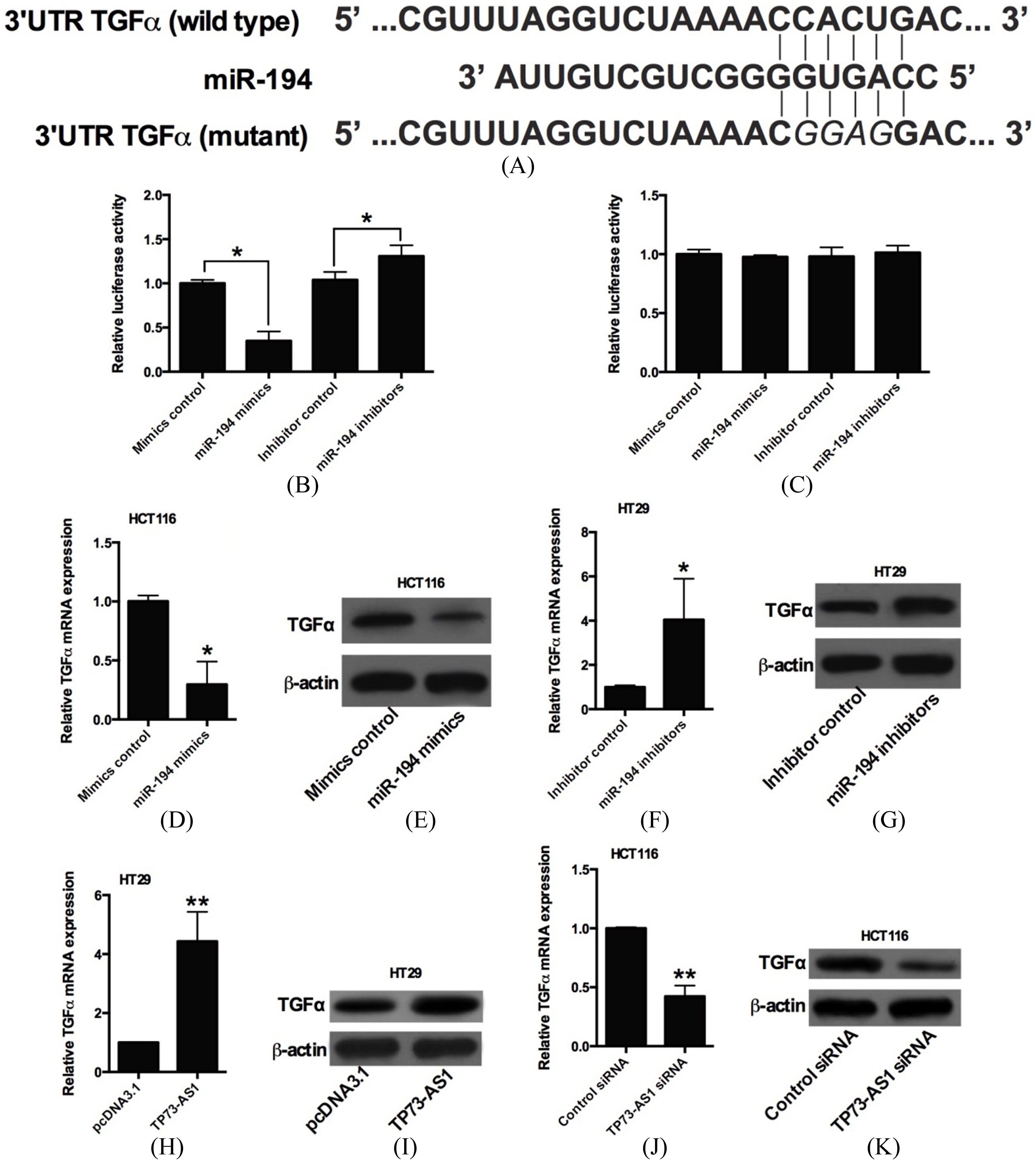
MiR-194 and TGF
Studies have showed that miR-194 repressed gene expression by targeting the 3’UTR, and the TargetScan software (
MiR-194 and TGF
mediated the biological effects of TP73-AS1 in HT29 cells
As shown in Fig. 6A and B, co-transfection with TP73-AS1 and mimics control significantly promoted cell proliferation and invasion compared with co-transfection with pcDNA3.1 and mimics control in HT29 cells; while co-transfection with pcDNA3.1 and miR-194 mimics significantly suppressed cell proliferation and invasion compared with co-transfection with pcDNA3.1 and mimics control in HT29 cells (Fig. 4A and B). Co-transfection with TP73-AS1 and miR-194 mimics significantly reversed the effects of TP73-AS1 overexpression on cell proliferation and cell invasion in HT29 cells (Fig. 6A and B). Consistently, co-transfection with pcDNA3.1 and TGF
Discussion
The lncRNAs are a new class of non-coding RNAs, which play important roles in the pathogenesis of human cancers. Recent studies have suggested the oncogenic role for TP73-AS1 in several types of human cancers [16, 17, 18, 19, 20, 21]. In this study, we for the first time found that TP73-AS1 levels were up-regulated in CRC tissues compared with adjacent normal colon tissues, and overexpression of TP73-AS1 in CRC tissues was positively correlated with metastasis and advanced clinical stage. Functional and mechanistic studies revealed that TP73-AS1 promoted cell growth, cell proliferation, cell migration and invasion by acting as a ceRNA of miR-194 in CRC.
TP73-AS1 was first reported to be down-regulated in oliogdendroglial tumors and its knockdown by siRNA induced chemoresistance in glioma cells [22]. Zang et al., showed that TP73-AS1 was up-regulated in esophageal cancer tissues and were strongly correlated with tumor location and TNM stage in clinical samples, and TP73-AS1 knockdown also suppressed esophageal cancer cell proliferation in vitro and in vivo [19]. In brain glioma, TP73-AS1 was up-regulated in glioma tissues and cell lines and was associated with poorer prognosis in patients with glioma. In addition, TP73-AS1 knockdown also suppressed glioma cell proliferation and invasion via sponging miR-142 and modulating HMGB1/RAGE pathway [20]. Similarly, TP73-AS1 was also up-regulated in hepatocellular carcinoma (HCC) tissues and cell liens, and high expression of TP73-AS1 was correlated with worse clinicopathological features, poorer prognosis and shorter survival [17], and further mechanistic study revealed that TP73-AS1 modulated HCC cell proliferation via miR-220a-depedent HMGB1/RAGE regulation [17]. More importantly, TP73-AS1 also was found to regulate breast cancer progression via interacting with miR-200a [18, 21]. In the present study, we consistently found that TP73-AS1 was up-regulated in CRC tissues and TP73-AS1 overexpression was positively correlated with metastasis and advanced clinical stage. In vitro studies showed that TP73-AS1 promoted CRC cell growth, cell proliferation, cell migration and invasion, and in vivo study also demonstrated that knockdown of TP73-AS1 suppressed tumor growth in the nude mice. These results suggest that TP73-AS1 may play an oncogenic role in CRC.
To further understand the mechanisms of TP73-AS1-mediated CRC progression, we performed the bioinformatics analysis and the results showed that miR-194 was among of these predicted miRNAs, which may have potential interaction with TP73-AS1. In the present study, the physical interaction between TP73-AS1 and miR-194 was confirmed by luciferase reporter assay, and overexpression of TP73-AS1 down-regulated the expression of miR-194, while knockdown of TP73-AS1 increased the expression of miR-194 in CRC cells. In the aspect of clinical samples, miR-194 was found to be down-regulated in CRC tissues; and the expression of TP73-AS1 was inversely correlated with miR-194 expression in CRC tissues. Overexpression of miR-194 suppressed CRC cell proliferation and invasion, and also reversed the effects of TP73-AS1 overexpression in CRC cells. Indeed, miR-194 was found to be frequently down-regulated in CRC [23], and the down-regulation of miR-194 contributed to colorectal carcinogenesis via targeting AKT2 pathway [23]. In addition, studies also revealed that miR-194 suppressed tumor growth by regulating the MAP4K4/c-Jun/MDM2 signaling pathway [24]. Collectively, these data suggest miR-194 as a tumor suppressor in CRC, and TP73-AS1 exerted its oncogenic function possibly via acting as a ceRNA to sponge miR-194 in CRC cells. However, we need to be cautious that TP73-AS1 may also target other miRNAs, which may require further investigation.
As miRNAs regulated the targeted genes by targeting the 3’UTR, we performed bioinformatics analysis to predict the downstream targets if miR-194, and TGF
In summary, we demonstrated the first time that TP73-AS1 was up-regulated in CRC tissues, and TP73-AS1 up-regulated miR-194-targeted gene TGF
Footnotes
Acknowledgments
This study was supported by the Research Program of The First Affiliated Hospital of Xi’an Medical University (Grant No. XYFY13-12).
Conflict of interest
The authors have no conflict of interest to disclose.
