Abstract
BACKGROUND:
Free fatty acid receptor 4 (FFAR4) is associated with the epithelial mesenchymal transition (EMT) and is involved in the progression of several types of cancer. However, the role of FFAR4 in cholangiocarcinoma (CCA) remains unclear.
OBJECTIVE:
The present study evaluated the diagnosis and prognosis of CCA using FFAR4 as a biomarker.
METHODS:
Immunohistochemistry was employed to detect expression of FFAR4 in 98 samples of CCA tissues and adjacent tissues. In addition, expression of E-cadherin, vimentin, Snail-1, CK7 and CK19 in the 98 samples of CCA tissues was detected, and relationships with FFAR4 were analyzed. Correlation between FFAR4 and clinical pathological factors and prognosis was also analyzed.
RESULTS:
FFAR4 was highly expressed in 72.4% (71/98) of CCA tissues and 29.6% (29/98) of adjacent tissues, with a statistically significant difference between the two tissue types (
CONCLUSIONS:
FFAR4 overexpression may mediate the process of CCA EMT. In addition, FFAR4 is promising as a new diagnostic molecule and therapeutic target for CCA.
Introduction
Cholangiocarcinoma (CCA) is an epithelial malignant tumor that originates from different locations on the biliary tree [1]. This tumor type can be divided according to the anatomic location into intrahepatic cholangiocarcinoma (iCCA), hilar cholangiocarcinoma (pCCA) and distal cholangiocarcinoma (dCCA), which have frequencies of 10%–20%, 50% and 30%–40%, respectively [2]. CCA is one of the most aggressive malignant gastrointestinal diseases and has a poor prognosis [1, 2]. Because conventional chemotherapy and radiotherapy have a limited impact on improving long-term survival, radical surgery is currently the only option for CCA patients to be cured or to achieve long-term survival. Unfortunately, due to severe local invasion and distant metastases, many patients when diagnosed with CCA have already missed the best option of radical surgery [3, 4]. Therefore, to improve the early diagnosis of CCA patients and postoperative quality of life, more in-depth studies of the occurrence and progression of CCA are needed.
As an important source of energy and nutrition, free fatty acids (FFAs) act as extracellular signaling molecules, primarily through binding to FFA receptors (FFARs), which belong to a family of G-protein-coupled transmembrane receptors (GPCRs) [5, 6, 7]. Disorders of GPCR signaling and expression have been identified as one of the hallmarks of cancer [8]. Free fatty acid receptor 4 (FFAR4), which is also called GPR120, is a new member of the GPCR family that has attracted considerable attention for its role as a medium-and long-chain FFA receptor in diabetes. Increasing evidence shows that FFAR4 also plays a role in tumorigenesis, migration and metastasis. In contrast to its contributions in diabetes, FFAR4 appears to have opposing functions in cancer biology [9]. The phosphatidylinositol 3-kinase (PI3K)/Akt (protein kinase B) signaling pathway is necessary to activate the FFAR4 signal transduction process and is an important pathway involved in the epithelial mesenchymal transition (EMT) process. Accordingly, this study investigates expression of FFAR4 in human CCA tissues and adjacent tissues; we also analyzed the relationship between FFAR4 expression and that of epithelium markers including E-cadherin, vimentin, snail-1, CK7 and CK19 in CCA tissues. At the same time, we assessed the relationship between FFAR4 expression and clinicopathological characteristics and correlations between FFAR4 expression and prognosis. We found that the mechanism of FFAR4 invasion and metastasis may be related to the EMT. Overall, our results suggest that by enhancing the invasion and metastasis of CCA, FFAR4 is a potential new target for clinical therapeutic drug research and is expected to be an effective marker for the early diagnosis of CCA.
Materials and methods
Samples and clinicopathological data
A total of 98 patients who underwent curative intent surgery for CCA at An Hui Provincial Hospital between January 2007 and January 2015 were enrolled in this study. All patients underwent radical surgical resection, and the pathology was diagnosed as CCA. The tumor tissue and adjacent normal tissue were fixed in formaldehyde and embedded in paraffin. Adjacent normal tissue was defined as bile duct tissue more than 2 cm from the edge of the tumor and confirmed by two experienced pathologists. The study was approved by the research medical ethics committee of An Hui Provincial Hospital. Informed consent was obtained from each patient. In addition, clinicopathological variables, including age, sex, tumor size, tumor location, preoperative serum carbohydrate antigen19-9 (CA19-9) concentrations, histological grade, LNM, PNI and stage of the tumor were collected. The cohort included 51 males and 47 females with an average age of 58.7 years (range 38–78 years). The histological classification of tumor differentiation was based on the WHO classification. The TNM stage was determined in accordance with the seventh edition of the Cancer Staging Manual of the American Cancer Commission (AJCC). The follow-up period was 5–62 months (median 19).
Immunohistochemistry
Sections (4
Statistical analysis
Categorical patient characteristics were compared using the
Differential expression of FFAR4 in CCA tissues and corresponding paracarcinoma tissues (
98)
Differential expression of FFAR4 in CCA tissues and corresponding paracarcinoma tissues (
FFAR4 expression in CCA tissues and paracancerous tissue
To identify expression patterns of FFAR4 in CCA, FFAR4 mRNA expression profiles were obtained from the Ualcan database [13], indicating that FFAR4 was significantly up-regulated in tumors compared with adjacent non-tumor tissues of CCA. Although FFAR4 was often overexpressed in most cancers, down-regulation was observed in some types. Therefore, the role of FFAR4 should be assessed in a cancer-specific manner. Immunohistochemistry was performed to validate and evaluate FFAR4 expression as well as localization in CCA, which was mainly distributed in the cytoplasm of tumor cells (Fig. 1A1 and A2). The results indicate obviously higher (
Correlations of FFAR4 expression with E-cadherin, vimentin, Snail-1, CK7 and CK19 in CCA
Correlations of FFAR4 expression with E-cadherin, vimentin, Snail-1, CK7 and CK19 in CCA
In 98 cases of cholangiocarcinoma, 74 exhibited low expression of E-cadherin; the proportion was 75.5% (Fig. 1D1 and D2). However, in 98 samples of CCA tissue, high expression of CK7, CK19, Snail-1 and vimentin was observed in 60, 68, 66, and 72 cases, accounting for 61.2%, 69.4%, 67.3% and 73.5%, respectively (Fig. 1B1, B2, C1, C2, E1, E2, F1, F2). Expression of FFAR4 in CCA tissues was positively correlated with that of CK7 (
Immunohistochemical staining for FFAR4, CK7, CK19, E-cadherin, Snail-1 and vimentin. FFAR4 is principally localized in cytoplasm of tumor cells; CK7 and CK19 are mainly localized in the bile duct cell membrane and tumor cell membrane; E-cadherin is mainly localized in the bile duct cell membrane; Snail-1 is mainly localized in the nucleus and cytoplasm of tumor cells; vimentin is mainly localized in the cytoplasm of tumor cells. A1: Positive FFAR4 expression in CCA tissue. A2: Negative FFAR4 expression in adjacent tissue. B1: Positive CK7 expression in CCA tissue. B2: Negative CK7 expression in adjacent tissue. C1: Positive CK19 expression in CCA tissue. C2: Negative CK19 expression in adjacent tissue. D1: Negative E-cadherin expression in CCA. D2: Positive E-cadherin expression in adjacent tissue. E1: Positive Snail-1 expression in CCA tissue. E2: Negative Snail-1 expression in adjacent tissue. F1: Positive vimentin expression in CCA tissue. F2: Negative vimentin expression in adjacent tissue. All images were taken at 400 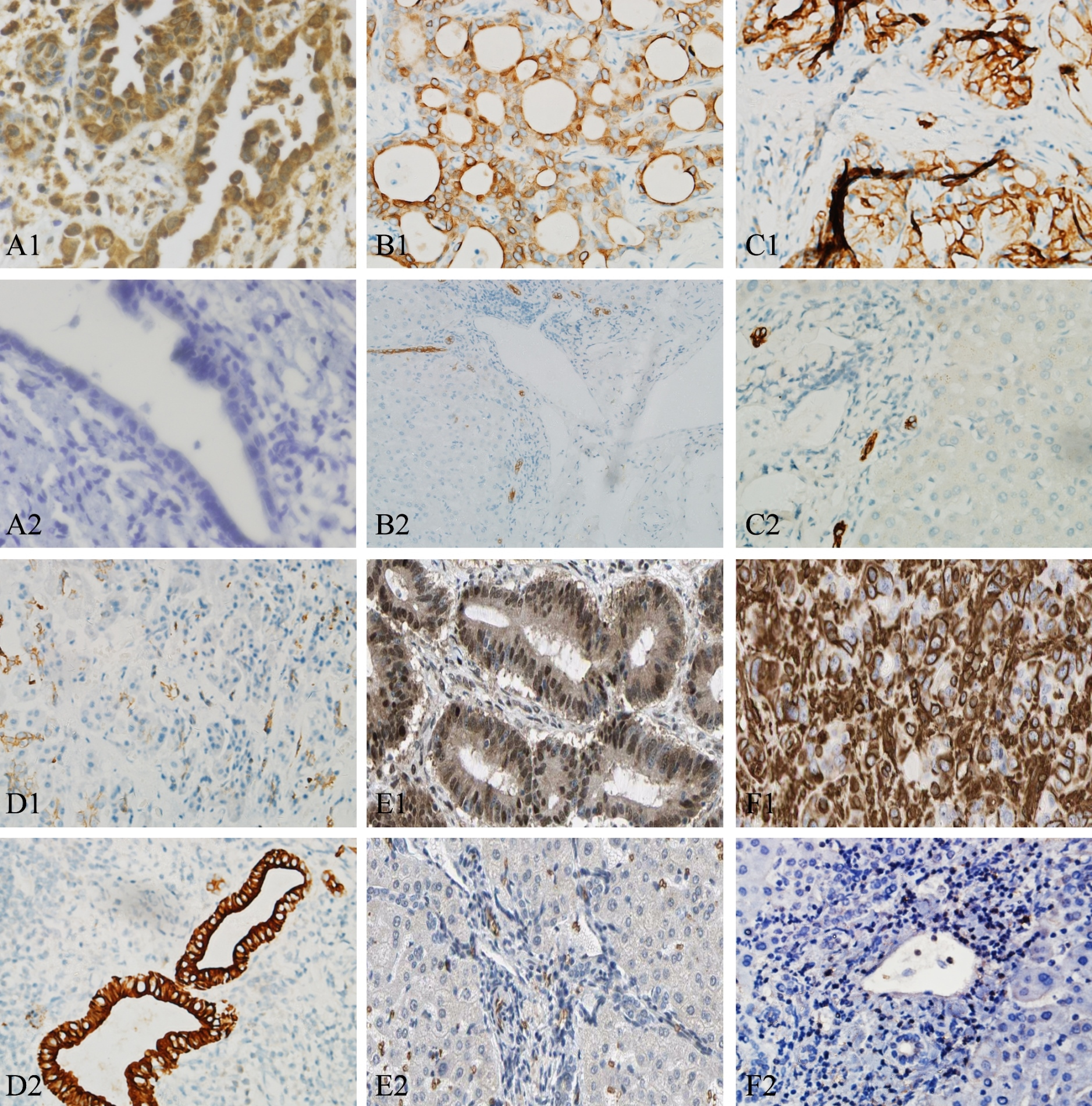
Kaplan-Meier analysis of disease-free survival (DFS) and overall survival (OS) in patients with CCA according to FFAR4 staining. Patients with high FFAR4 expression had a shorter OS and DFS than did those with low FFAR4 expression (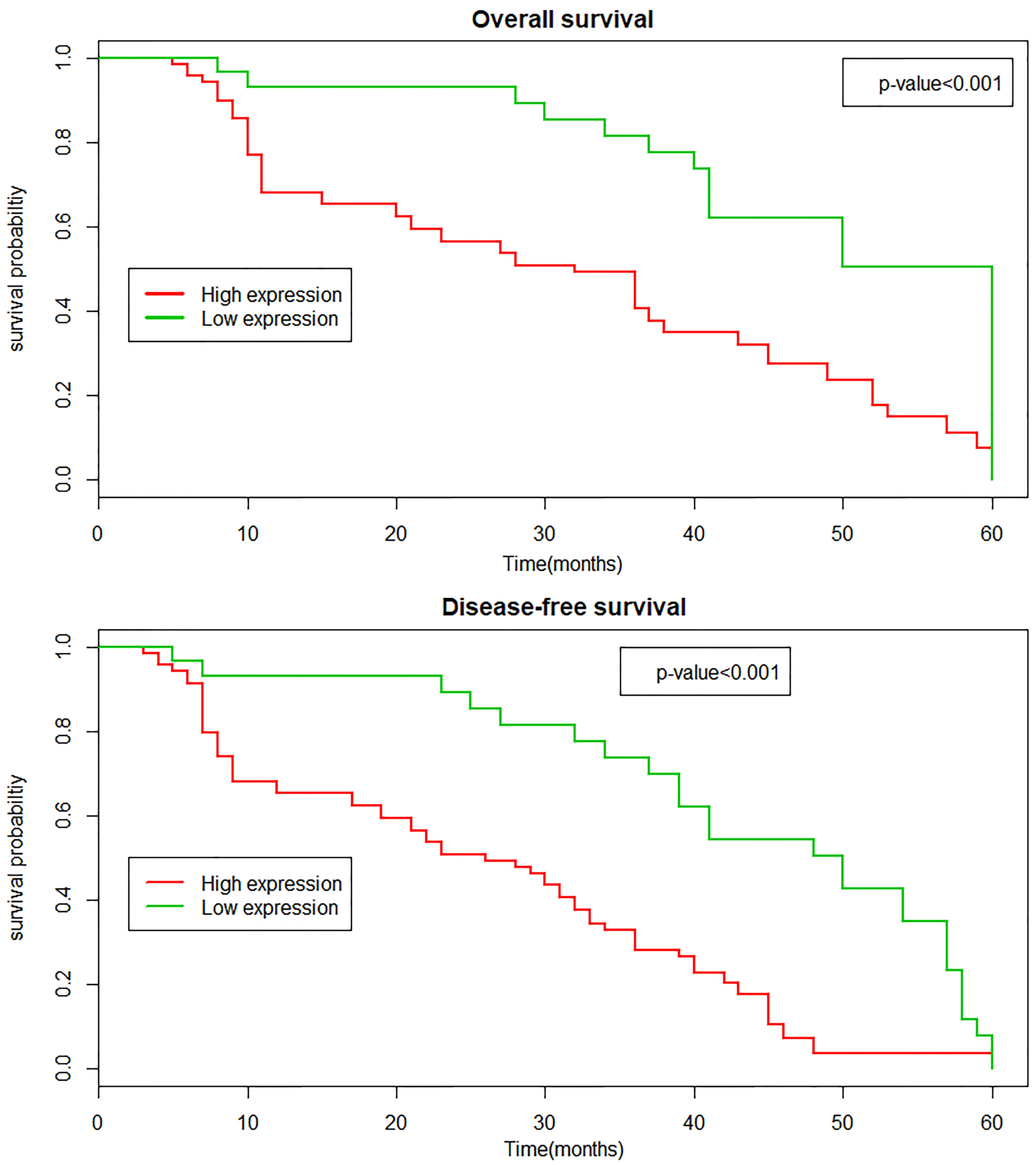
Experimental data show that FFAR4 expression in cholangiocarcinoma was markedly related to histological grade, perineural invasion, LNM, advanced TNM stage and preoperative serum CA19-9 (
Correlations between FFAR4 expression and clinicopathological characteristics in CCA
Correlations between FFAR4 expression and clinicopathological characteristics in CCA
Univariate analysis of factors associated with DFS and OS
Multivariate analysis of factors associated with DFS and OS
Kaplan-Meier survival curves (Fig. 2) were generated to analyze DFS and OS for high and low expression of FFAR4 in cholangiocarcinoma patients. The log-rank test showed that patients with high FFAR4 expression (29.0 months) had a significantly decreased DFS compared to those with low FFAR4 expression (37.0 months;
Univariate analysis suggested that FFAR4 expression, histological grade, LNM, and TNM stage had significant prognostic influence on DFS and OS (Table 4). Multivariate survival analysis revealed FFAR4 expression as an independent predictive factor for DFS [hazard ratio (HR) 0.578; 95% confidence interval (CI) 0.239-0.817;
Discussion
There is increasing evidence that dysregulation of FFAR4 plays an important role in tumorigenesis, migration and invasion. Fukushima et al. [14] found that enhanced FFAR4 expression in pancreatic cancer cells may play a role in stimulating pancreatic cancer cell motility, invasion and tumorigenesis by inhibiting expression of MMP-2. Wu et al. [15] reported that increased FFAR4 expression significantly induced tumor-associated progression of colorectal cancer tissue and cell lines and showed that FFAR4 signaling promotes angiogenic transition and motility in human colorectal cancer (CRC) cells. Moreover, activation of FFAR4 enhances the viability of CRC cells, inducing epithelial-mesenchymal transition. Notably, FFAR4 expression was also significantly increased in paraffin-embedded colorectal cancer tissues derived from patients, correlating with poor pathologic differentiation. Kleemann et al. [16] reported significant expression of the FFAR4 protein in 114 malignant melanoma tissue samples. A study by Serna-Marquez et al. [17] suggested that LA induces the migration and invasion of breast cancer cells through FFAR4 (GPR120)-EGFR and PI3K-/Akt-dependent pathways in the human breast cancer MDA-MB-231b cell line. However, Liu et al. [18] presented evidence that activation of FFAR4 in human prostate cancer cells initiates a pathway that inhibits growth factor-induced signaling and provides a novel mechanism for inhibiting cancer cell proliferation, and Kita et al. [19] observed that stimulation of GPR120 in lung cancer cells led to lower cell motility. These inconsistent findings suggest that FFAR4 may play different roles in different tumors.
Based on the present FFAR4 study, a preliminary analysis of FFAR4 expression in CCA and adjacent normal bile duct tissue was performed to determine whether there is a difference in expression and whether clinically and pathologically related features are associated. To the best of our knowledge, this study is the first to confirm FFAR4 expression in CCA and its clinical significance. FFAR4 is overexpressed in CCA tissues compared to adjacent tissues. Statistical analysis suggested that FFAR4 expression levels were higher in tumors with aggressive clinical and pathological features, such as poor tissue differentiation, neural invasion, lymph node metastasis, TNM staging, and preoperative serum CA19-9 levels.
EMT refers to a specific program of epithelial cell transition to a mesenchymal phenotype. Epithelial cells lose polarity via EMT and connection with the basement membrane. In addition, these cells obtain the capacity for higher invasion, migration, anti-apoptosis and degradation of the extracellular matrix [20]. This is the molecular basis of tumorigenesis, invasion and metastasis. Both E-cadherin, cytokeratin 7, cytokeratin 19, Snail-1 and vimentin are important protein indicators reflecting the process of EMT. The main markers of EMT transformation in tumors are low expression of E-cadherin and the high expression of Snail-1 and vimentin. E-cadherin is a cell adhesion molecule present in the epithelium that accumulates through organization of intracellular catenin and peptide proteins linked to the cytoskeletal actin. In addition, E-cadherin participates in a series of processes, such as cell adhesion, growth and proliferation, and maintains epithelial cell morphology and structure integrity. Snail-1 is a most important marker in the process of primary epithelial tumor cell transformation into mesenchymal cells with migratory ability. In the process of EMT, inhibition of E-cadherin expression by Snail-1 promotes tumor invasion and metastasis [21]. Vimentin, a major intermediate filament protein present in stromal cells, participates in the adhesion, migration, invasion and cell signaling of tumor cells by regulating interaction between cytoskeletal proteins and cell adhesion molecules. Cytokeratin (CK) is also an intermediate filament protein that is mainly expressed in epithelial cells. Due to the high tissue specificity and differentiation specificity of CK, this protein is widely used as a marker for various epithelial and tumor cells. CK7 and CK19 are called the CK of the bile duct because of their specific expression in bile duct epithelial cells [22]. Recent studies have shown that CKs, which are expressed in bile duct cells, undergo certain changes in bile duct injury and bile duct diseases. Bile duct cells and cholangiocarcinoma may additionally express either type of bile duct CK (CK19 or CK7) [23], and abnormal expression of CKs may be related to EMT. In this retrospective study, FFAR4 expression was observed to be negatively correlated with E-cadherin expression in CCA. In contrast, FFAR4 expression was positively correlated with CK7, CK19, Snail-1 and vimentin expression in CCA. These results suggest that FFAR4 overexpression may be closely related to expression of important target proteins in EMT. FFAR4 may also be involved in EMT, affecting the invasion and metastasis of CCA.
In terms of prognosis, our results show that patients with high FFAR4 expression and low FFAR4 expression have significantly shorter survival times. These findings suggest that high FFAR4 expression is associated with poor prognosis in patients with CCA and is promising for use as a valuable prognostic biomarker.
Conclusions
We acknowledge several limitations to this study. This study is a single retrospective study with a relatively small sample size; therefore, expression of FFAR4 in CCA was detected at a superficial level. Overexpression of FFAR4 may be related to the malignancy of CCA. However, the underlying mechanism by which FFAR4 promotes the invasion and metastasis of CCA cells is unclear. It is necessary to expand sample sizes to further confirm our findings and perform in vitro cytology and verification in animal models. In summary, CCA, a malignant tumor with strong invasion and metastasis properties, is usually diagnosed late, which contributes to a poor prognosis. As a newly discovered important G protein-coupled receptor, FFAR4 upregulation may play an important role in the development of CCA. In addition, our study showed that FFAR4 is associated with the EMT process in CCA. Moreover, FFAR4 may be an independent biomarker of poor prognosis of CCA. The mechanism of FFAR4 needs to be further researched, and FFAR4 is a promising new target for the early diagnosis of CCA and a biomarker for clinical drug treatment.
Footnotes
Acknowledgments
This study was supported by the National Natural Science Foundation of China (No 81501354), the Natural Science Foundation of Anhui Province (No 1608085QH197), and the Programs for Science and Technology Development of Anhui Province in 2016 (No 1606c08234).
Conflict of interest
The authors declare that there are no conflicts of interest.
Supplementary data
The supplementary files are available to download from
