Abstract
BACKGROUND:
Epithelial-mesenchymal transition (EMT) is one of the main events in colorectal cancer(CRC) spread. Snail-1 is a zinc transcription factor that mediates EMT in tumor cells probably by down-regulation of E-cadherin and claudin-1.
OBJECTIVES:
To detect the expression of epithelial markers (claudin-1 and E-cadherin) and mesenchymal markers (snail-1 and vimentin) in primary cancer colon. Also, to select stage II cancer patients of a high risk that can benefit from postoperative adjuvant chemotherapy.
METHODS:
Reverse transcription-polymerase chain reaction (RT-PCR) and immunohistochemical analysis were performed to investigate snail-1, claudin-1, E-cadherin and vimentin expressions at mRNA and protein levels in fresh tissues of cancer colon and normal colonic mucosa. The correlations between the expression of these markers and clinicopathological parameters were performed.
RESULTS:
Normal colonic mucosa revealed complete membranous expression of claudin-1, preserved E-cadherin and negative snail-1 and vimentin expressions. Compared to control, the expression of snail-1 and vimentin mRNA in cancer colon was significantly up-regulated while the expression of claudin-1 and E-cadherin mRNA was significantly down-regulated. These changes were significantly associated with stage and lymph node involvement at both mRNA and protein levels(
CONCLUSION:
There is a clinical significance of snail-1, claudin-1, E-cadherin and vimentin as possible markers for recognizing patients with lymph node involvement, advanced stage and high incidence of tumor recurrence in cancer colon.
Abbreviations
Introduction
Colorectal cancer (CRC) is the 3rd most common malignancy worldwide and the 3rd cause of mortality among cancer patients mainly due to distant metastasis [1]. About 25% of CRC patients presented with stage II. While chemotherapy is beneficial for stage III patients, there is a controversy about its significance for stage II patients. This can be explained by that there is a greater risk for micrometastasis in stage III and targeting this micrometastasis is dependent on chemotherapy. However, recognition of novel biomarkers with different patterns of expression in stages II and III and within stage II patients may be useful in distinguishing stage II patients that possess high liability for recurrence that benefits from adjuvant chemotherapy [2].
Epithelial-mesenchymal transition (EMT) is a biological event that makes cells lose their epithelial criteria and epithelial cells acquire fibroblast-like features with under-expression of cell adhesion molecules including E-cadherin. This is accompanied by loss of cell polarity and acquiring of specific mesenchymal properties. In addition, up-regulation of mesenchymal markers, such as vimentin, occurs [3]. Epithelial tumors undergo EMT to facilitate their loss of cell adhesion in addition to acquiring of migratory and invasive characters [4].
Snail-1 is a zinc-finger transcription factor. It is produced in cells that have developed nearly complete EMT. Snail-1 triggers EMT mainly by downregulation of E-cadherin. In addition, it may have the same effect on claudin-1, as well [5].
Cell adhesion is kept by adherens junctions (AJs) and tight junctions (TJs). AJ components include catenins (
In this study, we evaluated the expressions of snail-1, claudin-1, E-cadherin and vimentin at mRNA and protein levels in colon cancer. Moreover, we aimed to highlight the molecular criteria in the selection of the high-risk group of stage II patients’ candidate for postoperative chemotherapy. Combinations of these biological markers will aid in understanding tumor biology, spread, and recurrence to help in colonic cancer targeted therapy.
Patients and methods
Patient history and tissue preparation
The present study was performed in Pathology, Medical Biochemistry and Molecular Biology, Clinical Oncology, General Surgery and Internal Medicine Departments, Faculty of Medicine, Zagazig University. After written informed consent from participants, fresh tissue samples with normal adjacent tissues were obtained from 50 patients with primary colon cancer admitted to Zagazig University Hospitals from January 2015 to January 2017. Patients with previous radiotherapy on the pelvic region or previous chemotherapy were excluded. In addition, stage II patients that did not receive postoperative chemotherapy only were included in our study for follow-up.
All patients were diagnosed as primary cancer colon by history, clinical examination and investigations (pelvi-abdominal ultrasonography, computed tomography, colonoscopy and tissue biopsy). All patients were prepared for surgery by routine pre-operative investigations (complete blood picture, coagulation profile, liver functions, renal functions, blood sugar and viral markers) and colonic preparation (chemical and mechanical). Normal colonic mucosa specimens adjacent to the carcinoma were enrolled in this study as a control.
Fresh colonic tissues were obtained at surgery time, separated into two parts. One part was immediately kept at
Primer sequence of studied genes
Primer sequence of studied genes
Clinicopathological features, immunohistochemical markers and gene expression of 50 patients with colonic carcinoma
Continuous variables were expressed as mean
Relation between clinicopathological features and claudin-1, E-cadherin, vimentin, snail-1 gene expression in 50 patients with colon cancer
Continuous variables were expressed as mean

Scatter plot with regression line shows (a) Correlation between E-cadherin gene expression and claudin-1 gene expression; (b) Correlation between vimentin gene expression and claudin-1 gene expression; (c) Correlation between vimentin gene expression and E-cadherin gene expression; (d) Correlation between snail-1 gene expression and claudin-1 gene expression; (e) Correlation between snail-1 gene expression and E-cadherin gene expression; (f) Correlation between snail-1 gene expression and vimentin gene expression.

(a) Normal colonic mucosa showing positive snail-1 immunoreactivity only in the stroma but not in the mucosa (Immunoperoxidase stain, X200); (b) Mucoid carcinoma of colon showing positive nuclear stain for snail-1 (Immunoperoxidase stain, X400); (c) High-grade colon cancer showing nuclear and cytoplasmic stain for snail-1 (Immunoperoxidase stain, X400).
Relation between clinicopathological features and immunohistochemical staining for E-cadherin, vimentin, and snail-1 in 50 patients with colon cancer

Normal colonic mucosa showing complete membranous stain for E-cadherin (Immunoperoxidase stain, X200); (b) Colonic adenocarcinoma, well-differentiated showing preserved E-cadherin reaction (Immunoperoxidase stain, X400); (c) Colonic adenocarcinoma, poorly differentiated showing decreased E-cadherin expression (Immunoperoxidase stain, X200).
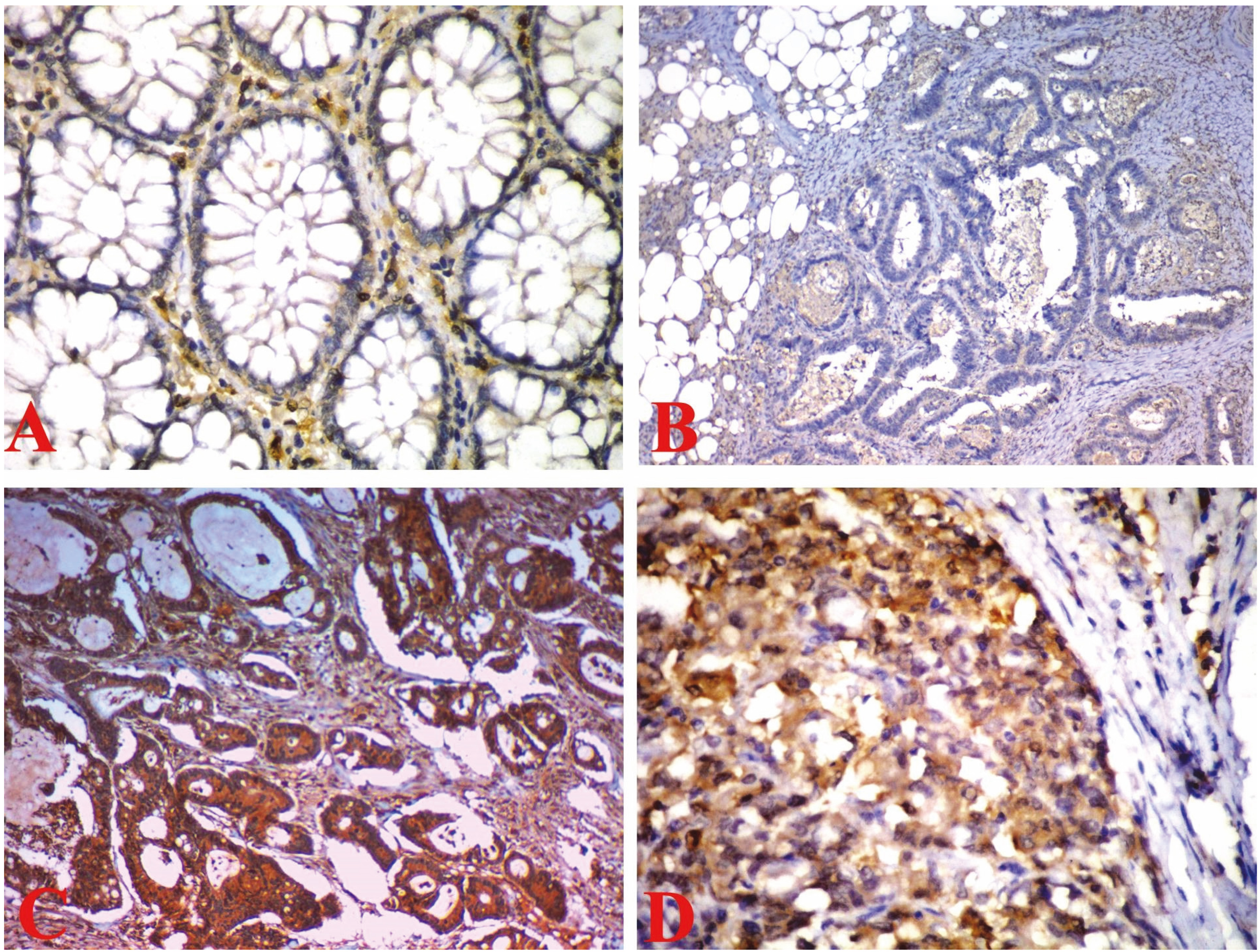
(a) Normal colonic mucosa showing positive vimentin immunoreactivity only in the stroma but not in the mucosa (Immunoperoxidase stain, X400); (b) Colonic adenocarcinoma, G-I showing negative vimentin stain (Immunoperoxidase stain, X200); (c) Colonic adenocarcinoma, G-II showing positive cytoplasmic vimentin stain (Immunoperoxidase stain, X200); (d) Colonic adenocarcinoma, G-III showing positive cytoplasmic vimentin stain (Immunoperoxidase stain, X400).

(a) Normal colonic mucosa showing complete membranous stain for claudin-1 (Immunoperoxidase stain, X200); (b) Colonic adenocarcinoma, G-I showing complete membranous stain for claudin-1 (similar expression) (Immunoperoxidase stain, X400); (c) Colonic adenocarcinoma G-II showing complete membranous stain for claudin-1 (similar expression) (Immunoperoxidase stain, X400); (d) Colonic adenocarcinoma G-III showing cytoplasmic stain for claudin-1 (Immunoperoxidase stain, X400).
Relation between clinicopathological features and immunohistochemical staining for Membranous/Cytoplasmic claudin-1 in 50 patients with colon cancer
Categorical variables were expressed as number(percentage)
Relation between E-cadherin, vimentin, snail and claudin-1 proteins
Categorical variables were expressed as number(percentage), continuous variables were expressed as mean
Relation between immunohistochemistry staining and gene expression
Continuous variables were expressed as mean
Comparison between recurrent and non-recurrent Stage II colon cancer as regard clinicopathological features, immunohistochemistry staining, and gene expression
Categorical variables were expressed as number(percentage); Continuous variables were expressed as mean
Colonic tissues were homogenized with a power homogenizer until disappearance of tissue clumps. Then, total cellular RNA was extracted using IQeasy plus CTB RNA extraction kit (iNtRON Biotechnology, Seongnam, Korea) according to the instructions of the manufacturer. Reverse transcription of one ug of RNA was performed using Power cDNA synthesis Kit (iNtRON Biotechnology, Korea) according to the instructions of the manufacturer. Real-time PCR was performed in 20 uL reactions containing 5 uL of the cDNA, 100 pmol/uL of each primer (0.5 uL each) (Biolegio, The Netherlands), 10 uL of EvaGreen PCR Master mix (Jena Bioscience GmbH, Jena, Germany) and 4 uL PCR-grade water. Real-time PCR was performed using Mx3005P (Stratagene, La Jolla, CA, USA) following that protocol: initial denaturation and polymerase activation at 95
The relative gene expressions for snail-1, claudin-1, E-cadherin and vimentin were calculated by the
Immunohistochemical staining
Four
Assessment of immunohistochemistry
Nuclear snail-1 expression was assessed as follow; first, staining intensity was scored as 0, 1, 2, or 3 (no staining, weak, moderate and strong intensities) respectively. Second, percentage scores, positive nuclei were counted ranged (0–100%) then we calculated total H-score by multiplying the score of intensity by that of the percentage, H score ranged (0–300). An H-score is considered positive if it is more than the median [14].
Claudin-1 membranous stain intensity and extent were evaluated in tumor tissue and the adjacent normal mucosa. The intensity of staining was graded as either weak
E-cadherin was expressed as a membranous stain and evaluated semi-quantitatively as follow; score 3
Vimentin cytoplasmic expression was considered positive whenever
Statistical analysis
All data were analyzed using SPSS 20.0 for windows (SPSS Inc., Chicago, IL, USA) and MedCalc 13 for windows (MedCalc Software bvba, Ostend, Belgium). Independent Student t- and Mann-Whitney U tests were used for normally and non-normally distributed data, respectively. One way ANOVA and Kraskall Wallis H tests were used to compare more than two groups of normally distributed data and non-normally distributed data, respectively. Chi-square test or Fisher’s exact test were used for comparison. Spearman’s rank correlation coefficient was calculated to assess the relationship between gene expressions.
Results
Patient characteristics
This study included 50 patients with cancer colon (mean age
Reverse transcriptase-polymerase chain reaction (RT-PCR) results
In cancer colon tissues, the mean values of relative mRNA expression of snail-1, claudin-1, E-cadherin and vimentin were (0.31
Significant differences in claudin-1, E-cadherin and vimentin mRNA expressions were found with increasing tumor grade (
There were significant positive correlations between E-cadherin and claudin-1 mRNA expressions (
Results of immunohistochemistry
Snail-1 was demonstrated in the nuclei of fibroblasts and macrophages of the stroma but not in normal colonic mucosa. Nuclear expression was detected in 60% of cancer colon cases while combined nuclear and cytoplasmic expression was in 30% of cases. In addition, snail-1 immunoreactivity was up-regulated with increased tumor grade but no statistically significant relationship was found between tumor grade and histological type and snail-1 expression (
Concerning E-cadherin expression, all normal colonic mucosa showed preserved E-cadherin immunoreactivity while 48% cases of cancer colon designated decreased E-cadherin immunoreactivity with a significant difference between the two groups (
With regard to vimentin, normal colonic mucosa expressed vimentin in the stroma only. As regard colon cancer, twelve cases (24%) showed cytoplasmic expression. All positive vimentin cases revealed nodal metastasis. Ten cases that showed positive vimentin were stage IV and two cases were stage III. No statistically significant relationship was found between histological type and vimentin (
With regard to claudin-1, all normal colonic mucosa showed diffuse, strong membranous stain. Eighteen cancer colon cases (36%) showed a similar pattern of stain to the normal adjacent mucosa. A discontinuous membranous stain pattern was interpreted as decreased claudin-1 expression and staining scores ranged from
Cytoplasmic claudin-1 expression was observed in 21 cancer colon cases (42%). In contrast to a membranous expression which decreased with advanced tumor grade, cytoplasmic expression of claudin-1 was significantly increased with tumor grade (
There were significant associations between E-cadherin and claudin-1 protein expressions (
Relation between immunohistochemistry staining and gene expression
Protein expressions of snail-1, claudin-1, E-cadherin and vimentin were consistent with their mRNA expression in cancer colon (Table 7).
Comparison between recurrent and non-recurrent cancer colon cases in stage II
It was found that up-regulation of snail-1 and vimentin mRNA expression and down-regulation of claudin-1 and E-cadherin mRNA expression (not protein expression) were correlated with recurrence of stage II cases (8 cases recurrent versus 14 not recurrent). Two of them were of low pathological risk and were converted to high risk by mRNA expression (Table 8).
Discussion
It takes about 7–12 years for development of invasive carcinoma from the normal colonic epithelium. Many genetic and epigenetic factors are active during that time [18, 19]. Therefore, the development of reliable biomarkers for early detection of progression and metastasis becomes essential. These markers may recognize a special group of high-risk patients who may benefit from chemotherapy.
Concerning snail-1, it is present in the nucleus. After its phosphorylation, it is translocated to the cytoplasm and is subsequently degraded [20]. The extracellular environment has a role in the regulation of its activity such as cellular attachment to the extracellular matrix [21]. Cytosolic snail-1 may help the migration of tumor cells. In addition, it modulates protein levels involved in pre-mRNA processing and location by regulating post-transcriptional modifications [20].
There is a controversy concerning snail-1 expression in cancer. Franci et al. described the presence of protein in carcinoma cells only [22], while Zhu et al. found it in normal epithelium [23]. This could be explained by that snail-1 might act by the maintenance of a balance between cell proliferation and apoptosis [5]. However, both studies were similar in the subcellular distribution of snail-1. Its shift to the cytoplasm with both cytoplasmic and nuclear or only cytoplasmic localization has been confirmed.
In our study, the snail-1 protein was only detected in the stromal fibroblast and macrophages of normal colonic mucosa and a significant up-regulation of snail-1 mRNA expression in cancer colon compared to control was observed. In addition, combined nuclear and cytoplasmic snail-1 immunoreactivity was detected nearly in one-third of cancer colon cases similar to the study performed by Bezdekova et al. [5].
Moreover, significant associations between snail-1 mRNA and protein up-regulation and lymph node involvement were observed. These results are supported by the previous studies by Fan et al. and Kroepil et al. [24, 25].
Concerning claudin-1, its altered expression can affect tumor advancement indirectly by attenuating tight junction properties or directly by its effect on signaling pathways [26]. Growth factors EGF and TGF-
In our study, decreased expression of claudin-1 mRNA and protein (at the membranous level) in cancer colon compared to normal mucosa was observed. There was a significant association between down-regulated claudin-1 mRNA and protein levels and increasing tumor grades, lymph node involvement, and advancing tumor stage. Moreover, we found that decreased claudin-1 expression was a predictor of recurrence, especially in stage II. These results were in line with some previous studies [27, 28, 29, 30, 31, 32]. However, others reported claudin-1 up-regulation in cancer colon [33, 34, 35, 36, 37].
In our work claudin-1 showed significant cytoplasmic over-expression with increasing tumor grade, lymph node involvement, and tumor stage. These results are consistent with Bezdekova et al. and Dhawan et al. [5, 33]. In addition, cytoplasmic claudin-1 expression was observed in 42% of cancer colon cases compared to 50% found by Bezdekova et al. in CRC cases [5]. They stated that a cytoplasmic claudin-1 mislocalization is a key event in the induction of EMT, as it leads to increase cell movement and subsequent tumor spread [5].
It has been reported that protein kinase A and C were important for the regulation and cytoplasmic localization of claudin-1 which is accompanied with loss of tumor suppressor gene (APC) function and nuclear
E-cadherin is a transmembrane cell adhesion glycoprotein. Epithelial cells of the colorectal crypt normally express it at cell borders. Decrease to loss of E-cadherin (expression or function) is associated with loss of epithelial differentiation and subsequent distant metastasis. Its loss is considered to be the main step of EMT as its loss reduces cell-cell contact leading to impaired intercellular signaling, but not to direct tumor transformation. It mediates Ca2
In our study, decreased expression of E-cadherin mRNA and protein in cancer colon compared to control was observed. There was a significant association between E-cadherin mRNA and protein under expression and increasing tumor grades, lymph node involvement, and advancing tumor stage. These results are hand in hand with Ersöz et al. and Süren et al. [29, 30]. On the other hand, Bezdekova et al. did not find any differences in E-cadherin expression between tumor and normal epithelium. They explained that by the ability of snail-1 to interact with catenin and enhancing Wnt-dependent transcription directly [41].
Concerning vimentin, it plays a role in cell migration, attachment and signaling and presents in stromal fibroblasts [42]. It binds to and stabilizes the phosphorylated form of the MAP kinase ERK that affects cell signaling and cell-cycle control. In addition, it regulates factors involved in EMT and facilitates transport of kinases into the nucleus. Furthermore, it indirectly regulates gene expression by its effect on nuclear shape and architecture [43].
In our study, increased expression of vimentin in cancer colon compared to control was observed at the level of mRNA, not at the protein level. In addition, overexpression of vimentin mRNA and protein was associated with advanced tumor stage and lymph node involvement. This can be explained by the inability of normal colonic mucosa to express vimentin but advanced stages of cancer colon express it, so it imparts an induction of an epithelial-to-mesenchymal phenotype in stages III and IV cancer colon cells. These results are going hand to hand with Qi et al. [44].
Moreover, there was a significant negative correlation between snail-1and E-cadherin mRNA and protein expressions. This result is similar to those confirmed by previous studies that stated that snail-1 induces EMT mainly by down-regulation of E-cadherin directly by attachment to E-boxes in the E-cadherin promoter [45, 46, 47, 48, 49]. On the other hand, Kroepil et al. did not find a significant correlation between snail-1 and E-cadherin in CRC. E-cadherin loss could be explained by the effect of the Wnt-signaling cascade on EMT. Wnt signaling and snail-1 may be interconnected at multiple levels [50].
In addition, a negative correlation between snail-1and claudin-1 mRNA and protein expressions was observed in this study in agreement with the previous study that showed that snail-1 may downregulate the expression of claudin-1 [51].
Moreover, there were significant negative correlations between vimentin and E-cadherin mRNA and protein expressions and between vimentin and claudin-1 mRNA and protein expressions. These correlations are similar to those confirmed by Qi et al. who stated that EMT involved up-regulation of the mesenchymal markers as vimentin and down-regulation of E-cadherin [44].
Segregation of stage II colon cancer that is at a higher risk for distant spread would help in the better selection of candidate cases for adjuvant chemotherapy. In this study, based on cut-off values of snail-1, claudin-1, E-cadherin and vimentin genes (
Conclusion
Over-expression of the mesenchymal markers (snail-1 and vimentin) and underexpression of the epithelial markers (claudin-1 and E-cadherin) at both mRNA and protein levels were observed in colon cancer tissues and these changes were significantly associated with lymph node involvement and advanced tumor stage. The expression level of snail-1, claudin-1, E-cadherin and vimentin genes can be used to stratify stage II colon cancer into high-risk and low-risk groups. Subcellular cytoplasmic localizations of snail-1 and claudin-1 may have a role in changes of cellular features and increase metastatic behavior. A targeted therapy of cancer colon can be developed to inhibit tumor growth and metastasis depending on molecular bases.
Footnotes
Conflict of interest
None.
