Abstract
OBJECTIVE:
This study aimed to evaluate predictive value of 14 pro-angiogenic miRNAs for cardiotoxicity induced by epirubicin/cyclophosphamide follow by docetaxel (EC-D) in breast cancer (BC) patients.
METHODS:
Three hundred and sixty-three BC patients receiving EC-D neoadjuvant chemotherapy were consecutively enrolled in this prospective cohort study. Peripheral blood sample was obtained from each patient, and plasma was separated. The expressions of 14 pro-angiogenic miRNAs, cardiac troponin I (cTnI) and N-terminal pro brain natriuretic peptide (NT-proBNP) were evaluated. Left ventricular ejection fraction (LVEF) level at C0, the end of 4 cycles of EC chemotherapy (C4), the end of 4 cycles of docetaxel treatment (C8), 3rd months (M3), 6th months (M6), 9th months (M9) and 12th months (M12) after surgery were assessed.
RESULTS:
LVEF decreased at C4, C8, M3, M6, M9 and M12 compared with C0, and the total cardiotoxicity incidence was 5.2%. Additionally, the levels of let-7f, miR-17-5p, miR-20a, miR-126, miR-210 and miR-378 were reduced in cardiotoxicity patients. Multivariate logistic regression revealed that miR-17-5p and miR-20a were independently predictive factors for less cardiotoxicity. Receiver operating characteristics (ROC) curve displayed a satisfactory predictive value for lower cardiotoxicity risk with area under curve (AUC) of 0.842 of the combination of the miR-17-5p and miR-20a expressions. In addition, let-7f,miR-126, miR-210 and miR-378 levels negatively correlated with cTnI expression, and let-7f and miR-130a expressions reversely correlated with NT-proBNP level.
CONLUSIONS:
miR-17-5p and miR-20a could be served as biomarkers for lower cardiotoxicity induced by EC-D neoadjuvant chemotherapy in BC patients.
Introduction
Breast cancer (BC), the most common diagnosed cancer among females with roughly 1,700,000 new cases and 520,000 deaths in 2012 worldwide, accounts for 15% new female cancer patients and displays an increasing incidence in China in 2015, demonstrating a correlation with lifestyle change that results in the elevated prevalence of obesity and inactivity [1, 2, 3, 4]. For the purpose of down staging BC for surgeries or to achieve breast conserving, neoadjuvant therapy rises as a great importance in clinical practice [5, 6]. Unfortunately, cardiotoxicity induced by chemotherapeutics, such as the anthracycline-based drugs, largely limits the use of neoadjuvant chemotherapy in BC patients [7, 8].
MicroRNA (miRNA), a category of small non-coding RNAs consists of roughly 19–23 nucleotides, regulates cells differentiation, proliferation, migration and apoptosis through mediating target genes and proteins [9, 10, 11, 12]. Moreover, the modulating function of miRNAs in several cardiac pathological processes have been validated to some extent, such as angiogenesis, myocardial fibrosis and cardiac remodeling [13, 14, 15, 16]. Angiogenesis has long been considered as a crucial process in cardiac diseases and is also a therapeutic target through promoting angiogenesis and improving cardiac tissue perfusion [17, 18, 19]. Accumulating studies report that there are miRNAs proven to be pro-angiogenic, however, the correlations of those pro-angiogenic miRNAs with cardiotoxicity induced by neoadjuvant chemotherapy in BC patients are still obscure [20].
Therefore, the aim of our study was to evaluate predictive value of 14 pro-angiogenic miRNAs for cardiotoxicity induced by epirubicin/cyclophosphamide follow by docetaxel (EC-D) in BC patients.
Materials and methods
Patients
In this prospective cohort study, we consecutively enrolled 365 BC patients who received EC-D neoadjuvant chemotherapy between Jan 2014 and Dec 2016 in The Second People’s Hospital of Liaocheng. Inclusion criteria were: 1) Diagnosed as primary BC confirmed by clinical, imaging and pathology examination. 2) Aged 18–75 years old. 3) Left ventricular ejection fraction (LVEF)
This study was performed under Institutional Review Board approvals from The Second People’s Hospital of Liaocheng and conducted according to Declaration of Helsinki. Written informed consents were obtained from all patients.
Treatment
In this present study, treatments of all BC patients were not intervened. Based on disease condition ands patients’ willing, patients received EC-D neoadjuvant chemotherapy. Conventional neoadjuvant chemotherapy regimen was as follows: epirubicin, 100 mg/m
Candidate miRNAs selection
Through analyzing the previous articles, the 14 pro-angiogenic miRNAs were selected from two studies, including a review and a research article [20, 21].
Samples collection
Ten ml peripheral blood sample was obtained from each patient before neoadjuvant chemotherapy (Cycle 0, C0), and plasma was separated by centrifugation from each sample. Plasma pro-angiogenic microRNAs expressions were determined by quantitative polymerase chain reaction (qPCR), the level of cardiac troponin I (cTnI) was assessed by immunofluorescence and the level of N-terminal pro brain natriuretic peptide (NT-proBNP) was evaluated by electrochemiluminescence immunoassay.
Primers information in this study
Primers information in this study
LVEF was assessed by echocardiography at C0, the end of 4 cycles of EC chemotherapy (C4), the end of 4 cycles of docetaxel treatment (C8) and 3rd months (M3), 6th months (M6), 9th months (M9) and 12th months (M12) after surgery according to modified Simpson method [22]. Cardiotoxicity was defined as the LVEF declined by 10% from baseline to below 53% (the lower limit of normal) [23], heart failure, acute coronary artery syndrome or fatal arrhythmia [24].
Study flow.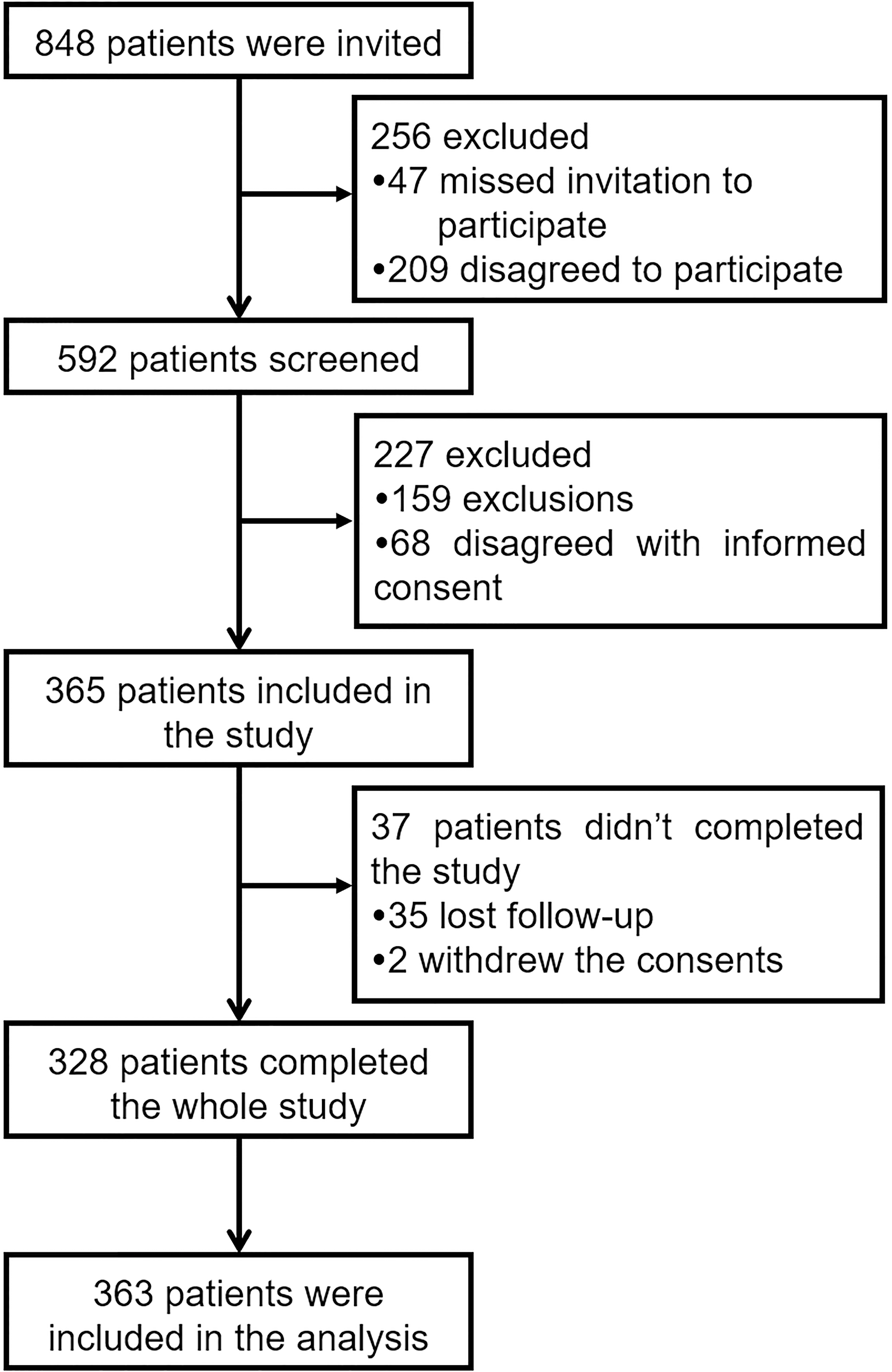
Baseline characteristics of BC patients (C0)
Data were presented as mean
The expression of 14 pro-angiogenic miRNAs were detected by qPCR assay. Firstly, total RNA from 200
Detection of cTnI and NT-proBNP
The cTnI level was evaluated by One Step Test for CK-MB/cTnI/Myo (Colloidal Gold) (Getein Biotech, Inc., China) with catalog number Getein1100. The expression of NT-proBNP was assessed by the Cobas
Baseline relative expressions of pro-angiogenic miRNAs (C0)
Baseline relative expressions of pro-angiogenic miRNAs (C0)
Data were presented as median (1/4–3/4 quantiles).
The SPSS 22.0 software (IBM Corp., Ltd, USA) and Graph Pad 6.0 (GraphPad Software Inc, USA) were used for statistical analysis in our study. Data was presented as mean
Results
Study flow
The study flow was displayed in Fig. 1, which presented that there were 848 patients who were invited at the initiation of our study and 256 patients were excluded due to missed invitation (
Baseline characteristics of BC patients
As listed in Table 2, the mean age of total BC patients was 45.38
Correlation of pro-angiogenic miRNAs with cTnI level. let-7f (B), miR-126 (J), miR-210 (L) and miR-378 (N) expressions were negatively correlated with cTnI level, however, other pro-angiogenic miRNAs were not correlated with cTnI expression (A, C–I, K and M). Spearman test was used to assess the correlations of the expressions of 14 pro-angiogenic miRNAs with cTnI level. 
Correlation of pro-angiogenic miRNAs with NT-proBNP level. The levels of let-7f (B) and miR-130a (K) were reversely correlated with the level of NT-proBNP, while no association of the other pro-angiogenic miRNAs expressions with NT-proBNP level was discovered (A, C–J and L–N). Spearman test was used to assess the correlations of the expressions of 14 pro-angiogenic miRNAs wiht NT-proBNP level. 
The expressions of 14 pro-angiogenic miRNAs at baseline detected by qPCR were presented in Table 3, which displayed that the expressions of let-7b, let-7f, miR-17-5p, miR-17-3p, miR-18a, miR-19a, miR-19b-1, miR-20a, miR-92a, miR-126, miR-130a, miR-210, miR-296, miR-378 were 0.998 (0.485–1.460), 1.226 (0.602–2.192), 0.884 (0.542–1.345), 1.054 (0.544–1.812), 1.340 (0.646–2.298), 1.871 (0.901–3.295), 1.309 (0.578–2.038), 1.415 (0.643–2.399), 1.316 (0.679–1.992), 1.457 (0.712–2.392), 2.789 (1.383–4.678), 1.175 (0.550–1.955), 0.736 (0.384–1.378) and 0.878 (0.443–1.456), respectively.
Correlations of 14 pro-angiogenic miRNAs with the levels of cardiotoxicity biomarkers
The levels of let-7f (
As presented in Fig. 3, the expression of let-7f (
Cardiotoxicity incidence among BC patients received EC-D neoadjuvant chemotherapy
As shown in Fig. 4, LVEF level obviously decreased at C4 (
There were 19 (5.2%) patients presented as LVEF level declined by 10% from baseline to below 53% throughout the whole follow up duration, while, no patient had heart failure, acute coronary syndrome or life threatening arrhythmias (Table 4). Therefore, the total cardiotoxicity incidence was 5.2%.
LVEF levels post EC-D neoadjuvant chemotherapy. LVEF level was declined at C4 and C8 compared with C0, although it presented an increasing trend from C8 to M9 and a decreasing trend from M9 to M12, the LVEF levels at M3, M6, M9 and M12 were still decreased compared with C0. Comparison between two groups was determined by t test. 
14 pro-angiogenic miRNAs expressions in cardiotoxicity and non-cardiotoxicity patients. let-7f (B), miR-17-5p (C), miR-20a (H), miR-126 (J), miR-210 (L) and miR-378 (N), cTnI (O) and NT-proBNP (P) expressions decreased in cardiotoxicity patients, but the expressions of other pro-angiogenic miRNAs between non-cardiotoxicity patients and cardiotoxicity patients were of no difference (A, D–G, I, K and M). Comparison between two groups was determined by Wilcoxon rank sum test. 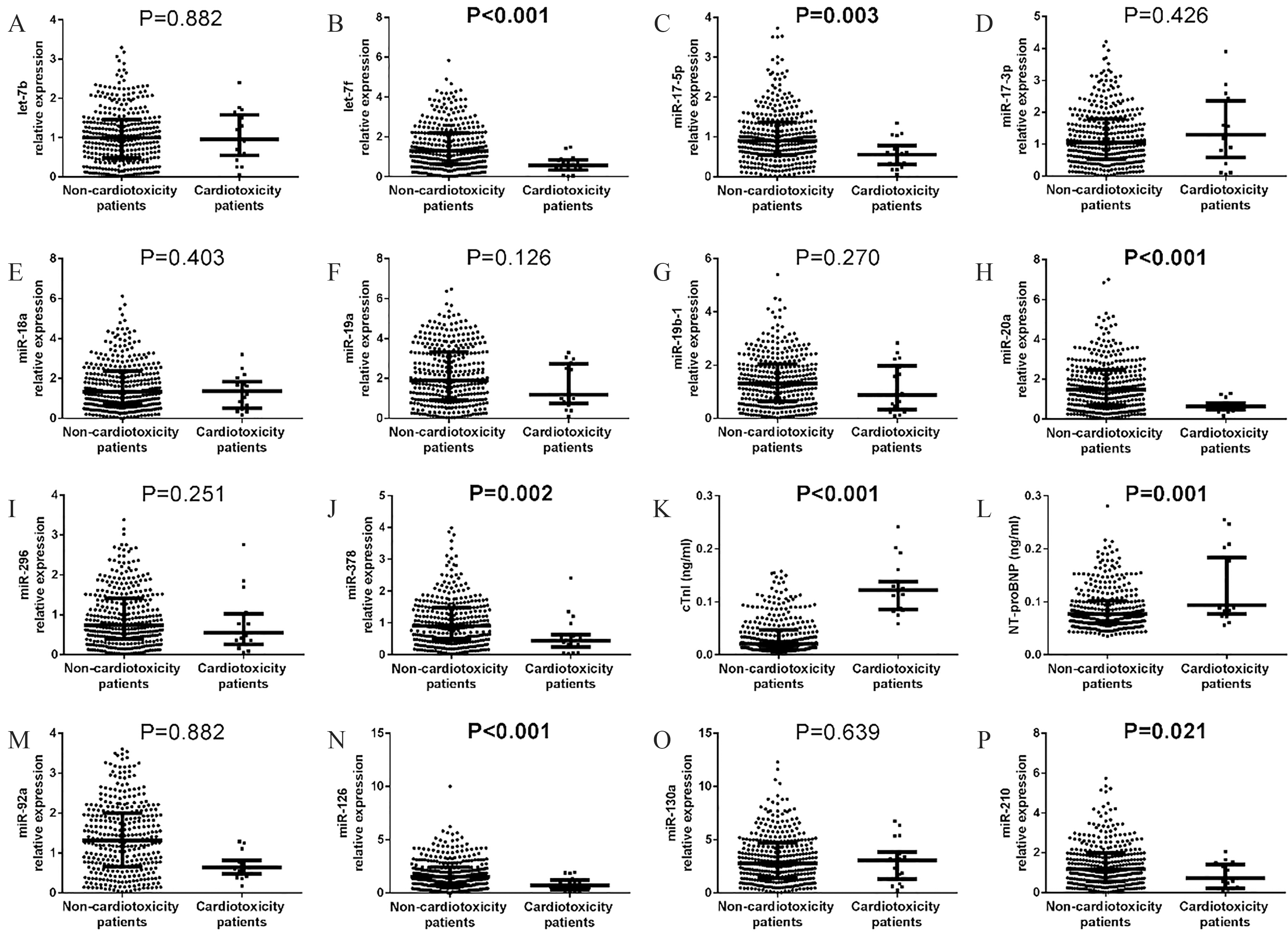
The levels of let-7f (
As listed in Table 5, univariate logistic regression analysis revealed that let-7f (
Predicting value of the combination of miR-17-5p and miR-20a for cardiotoxicity risk
For the purpose of further evaluating the value of independent predictive pro-angiogenic miRNAs for predicting the cardiotoxicity risk in BC patients receiving EC-D neoadjuvant chemotherapy, ROC curve was performed. As shown in Fig. 6, combining the expressions of miR-17-5p and miR-20a had a satisfying predictive value for low cardiotoxicity risk with area under curve (AUC) of 0.842 (95% CI: 0.778–0.906) and a sensitivity of 78.9% as well as a specificity of 80.8% at best cut off. Additionally, the AUC for cTnI and NT-proBNP were 0.940 (95% CI: 0.908–0.972) and 0.718 (95% CI: 0.602–0.833), respectively.
Assessment of cardiotoxicity
Assessment of cardiotoxicity
Data was presented as count (%). LVEF, left ventricular ejection fraction.
ROC curve analysis of independent pro-angiogenic miRNAs for predicting cardiotoxicity risk. ROC curve analysis showed that combining miR-17-5p and miR-20a expressions had a good predictive value for cardiotoxicity risk in BC patients receiving EC-D neoadjuvant chemotherapy. The formula for calculating the joint prediction probability was 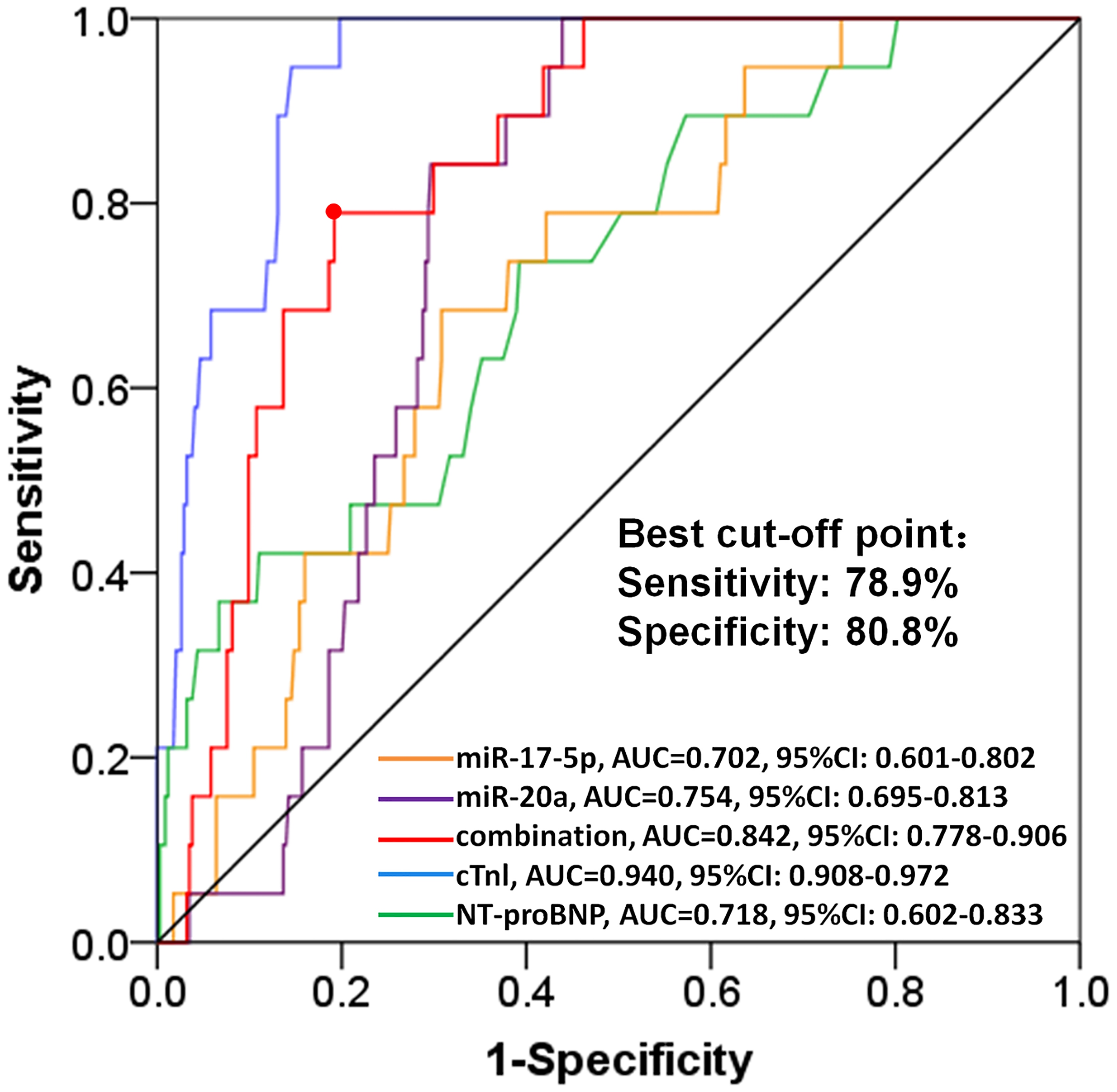
Logistic regression analysis of pro-angiogenic miRNAs in predicting cardiotoxicity risk
Data were presented as
As listed in Supplementary Table S3, complicating with dyslipidemia (
Discussion
In our study, we discovered that: (1) the LVEF kept declining in BC patients receiving EC-D neoadjuvant chemotherapy and the cardiotoxicity incidence was 5.2%; (2) let-7f, miR-126, miR-210 and miR-378 expressions negatively associated with cTnI level, and let-7f as well as miR-130a were negatively correlated with the level of NT-proBNP; (3) multivariate logistic regression revealed that let-17-5p and miR-20a were independently associated with less cardiotoxicity, and ROC curve displayed good predictive value of combining miR-17-5p and miR-20a for lower cardiotoxicity risk.
Anthracyclines are basically used for the adjuvant chemotherapy of BC patients, nonetheless, they can induce cardiotoxicity through multiple approaches. It is reported that anthracyclines increases the oxygen radical that leads to lipid peroxidation, which inhibits mitochondrial respiration, promotes autophagy and apoptosis and destroys sarcomere structures of the cardia muscle cells [25, 26, 27]. Meanwhile, experiments also show that anthracyclines interacts with topoisomerase 2 (Top2)-
In order to better eliminate cardiotoxicity of anthracycline based neoadjuvant chemotherapy for BC patients, increasing studies aim to explore the biomarker for predicting the cardiotoxicity, such as cTnI and NT-proBNP. cTnI is a sensitive biomarker indicating cardiac damage, and is suggestive for numerous diseases such as acute coronary syndrome, heart failure and myocarditis [38, 39]. And NT-proBNP, a prohormone for brain natriuretic peptide representing increased ventricular filling pressure and myocardium stress, is also a sensitive biomarker for cardiac disease and is especially unique for heart failure [40, 41, 42, 43]. miRNAs, as emerging candidates serving as biomarker for cardiotoxicity, are drawing increasing attention among clinicians and researchers. A previous study reports that miR-140-5p exacerbates cardiotoxicity caused by the use of doxorubicin through enhancing myocardial oxidative stress via targeting Nrf2 and Sirt2 [44]. A recent study elucidates that miR-34a-5p promotes cardiotoxicity induced by Adriamycin through targeting Sirt1/p66shc pathway [45]. Another study illustrates that miR-320a enhances cardiotoxicity in patients receiving Adriamycin via targeting vascular endothelial growth factor (VEGF) signaling pathway [46]. Additionally, a clinical study discovers that circulating miR-1 expression could be served as a biomarker for cardiotoxicity induced by Adriamycin with a higher predictive value compared with cTn1 [47]. However, to our best knowledge, there is still no report about the predictive value of pro-angiogenic miRNAs for cardiotoxicity induced by EC-D neoadjuvant chemotherapy, and the effect of those miRNAs in the treatment of EC-D for BC patients is not clear either. Our study discovered for the first time that let-7f, miR-126, miR-210 and miR-378 expressions negatively associated with cTnI level, and let-7f as well as miR-130a were negatively correlated with NT-proBNP level. In addition, multivariate logistic regression displayed that miR-17-5p and miR-20a expressions were independently associated with less cardiotoxicity, and the combination of miR-17-5p and miR-20a disclosed a good predictive value for lower cardiotoxicity risk according to ROC curve analysis. The probable explanations of our results might be: (1) pro-angiogenic miRNAs diminish cardiotoxicity by promoting angiogenesis, regulating myocardial fibrosis, improving tissue perfusion and accelerating tissue recovering [48, 49, 50, 51]; (2) those pro-angiogenic miRNAs act similarly to miR-140-5p, miR-34a-5p and miR-320a in regulating the injury caused by Adriamycin, however this theory remains to be clarified by experimental studies in the future [44, 45, 46].
There were still several limitations in our study: (1) there was selection bias due to this was a single center study with the enrolled patients barely from the north China; (2) we only evaluated the expressions of 14 pro-angiogenic miRNAs before neoadjuvant chemotherapy but not at other periods; (3) the detail mechanism of those independent predictive pro-angiogenic miRNAs in regulating the process of cardiotoxicity induced by EC-D neoadjuvant chemotherapy still need to be investigated by further experimental studies; (4) our results showed that treatment combined with trastuzumab positively correlated with cardiotoxicity incidence, which caused bias in our study.
In conclusion, miR-17-5p and miR-20a could be served as biomarkers for lower cardiotoxicity induced by EC-D neoadjuvant chemotherapy in BC patients.
Footnotes
Conflict of interest
The authors have no financial conflicts of interest.
Supplementary data
Reverse transcript primers information in this study RT, reverse transcript.
Gene
RT primer (5’-
let-7b
CTCAACTGGTGTCGTGGAGTCGGCAATTCAG
TTGAGAACCACAC
let-7f
CTCAACTGGTGTCGTGGAGTCGGCAATTCAG
TTGAGAACTATAC
miR-17-5p
CTCAACTGGTGTCGTGGAGTCGGCAATTCAG
TTGAGCTACCTGC
miR-17-3p
CTCAACTGGTGTCGTGGAGTCGGCAATTCAG
TTGAGCTACAAGT
miR-18a
CTCAACTGGTGTCGTGGAGTCGGCAATTCAG
TTGAGCTATCTGC
miR-19a
CTCAACTGGTGTCGTGGAGTCGGCAATTCAG
TTGAGTGTAGTGC
miR-19b-1
CTCAACTGGTGTCGTGGAGTCGGCAATTCAG
TTGAGGCTGGATG
miR-20a
CTCAACTGGTGTCGTGGAGTCGGCAATTCAG
TTGAGCTACCTGC
miR-92a
CTCAACTGGTGTCGTGGAGTCGGCAATTCAG
TTGAGAGCATTGC
miR-126
CTCAACTGGTGTCGTGGAGTCGGCAATTCAG
TTGAGCGCGTACC
miR-130a
CTCAACTGGTGTCGTGGAGTCGGCAATTCAG
TTGAGAGTAGCAC
miR-210
CTCAACTGGTGTCGTGGAGTCGGCAATTCAG
TTGAGCAGTGTGC
miR-296
CTCAACTGGTGTCGTGGAGTCGGCAATTCAG
TTGAGACAGGATT
miR-378
CTCAACTGGTGTCGTGGAGTCGGCAATTCAG
TTGAGACACAGGA
LVEF at each visit Data were presented as mean
Time point
LVEF (%)
C0
67.15
4.42
C4
64.24
5.53
C8
62.74
6.32
M3
62.99
6.59
M6
64.44
6.72
M9
65.25
7.29
M12
64.61
7.53
Correlation of baseline characteristics with the occurrence of cardiotoxicity Data were presented as count (%) or median (25
Characteristics
Cardiotoxicity
Non-cardiotoxicity
(
(
value
Age
0.780
11 (5.5)
189 (94.5)
8 (4.8)
157 (95.2)
BMI
0.208
6 (8.1)
68 (91.9)
13 (4.5)
278 (95.5)
Smoke
0.492
Yes
6 (6.6)
85 (93.4)
No
13 (4.7)
261 (95.3)
Hypertension
0.208
Yes
6 (8.1)
68 (91.9)
No
13 (4.5)
278 (95.5)
Diabetes mellitus
0.898
Yes
1 (5.9)
16 (94.1)
No
18 (5.2)
330 (94.8)
Dyslipidemia
Yes
9 (13.4)
58 (86.6)
No
10 (3.4)
288 (96.6)
Hyperuricemia
Yes
8 (11.8)
60 (88.2)
No
11 (3.7)
286 (96.3)
Chromic kidney disease
0.069
Yes
2 (16.7)
10 (83.3)
No
17 (4.8)
336 (95.2)
Higher ECOG performance
0.888
0
15 (5.2)
274 (94.8)
1
4 (5.6)
68 (94.4)
2
0 (0.0)
4 (100.0)
HER2
Positive
8 (10.4)
69 (89.6)
Negative
11 (3.8)
277 (96.2)
LVEF (%)
65.0 (63.0–68.0)
67 (64.0–71.0)
0.169
cTnI (ng/ml)
0.122 (0.086–0.138)
0.021 (0.010–0.047)
NT-proBNP
0.094 (0.077–0.184)
0.077 (0.060–0.102)
(ng/ml)
Combined with trastuzumab
Yes
7 (10.6)
59 (89.4)
No
12 (4.0)
287 (96.0)
