Abstract
OBJECTIVE:
To investigate the correlations of magnetic resonance imaging (MRI) manifestations with survivin gene expression in primary hepatocellular carcinoma (HCC).
METHODS:
A total of 84 HCC patients receiving partial hepatectomy in the Surgery Department and Oncology Department in our hospital from April 2011 to May 2014 were recruited. At 1 week before operation, MRI was used to examine the imaging features of liver, a certain size of area was defined and the signal value of each sequence was recorded. HCC and para-carcinoma tissues were collected after operation, and the expression levels of survivin were detected via immunohistochemistry (IHC). All patients were followed up for 30 months after operation, and the Kaplan-Meier survival curve was drawn. The correlations of survivin expression with MRI features and signal parameters of each sequence were analyzed.
RESULTS:
There was no expression of survivin in normal liver tissues. In HCC and para-carcinoma tissues, the nuclei of positive cells showed brown yellow. The positive expression rate of survivin in HCC tissues was 76.19% (64/84), which was significantly higher than that in para-carcinoma tissues (20.81%, 20/84) (
CONCLUSION:
Survivin is significantly up-regulated in HCC tissues and associated with tumor growth and lymph node metastasis. Clinical detection of survivin level combined with MRI examination might be beneficial for clinical diagnosis and treatment of HCC.
Introduction
Primary hepatic carcinoma ranks third in cancer deaths in the world, and it is the second most common malignant tumor causing cancer death in China, seriously threatening the life and health of people [1]. Primary hepatic carcinoma mainly includes three different pathological types, namely hepatocellular carcinoma (HCC), intrahepatic cholangiocarcinoma (ICC), and HCC-ICC mixed type [2]. The three types are quite different in pathogenesis, biological behavior, histological morphology, treatment method, prognosis, etc., among which HCC accounts for more than 85%–90%. Therefore, “hepatic carcinoma” in this study referred to HCC [3]. HCC is characterized by rapid progression, early invasion and metastasis, high recurrence rate, and poor prognosis of patients, whose, however, etiology and pathogenesis have not yet been determined. It is currently believed that HCC is related to cirrhosis, viral hepatitis, aflatoxin and other carcinogenic chemicals and environmental factors [4].
Survivin is a new human gene in inhibitor of apoptosis protein (IAP) family. Recent studies have shown that survivin can directly inhibit the activity of caspase-3 and caspase-7 or bind to caspase-9, and interacts with spindle microtubules to inhibit G2/M phase of cell proliferation [5]. Studies have shown that survivin is associated with a variety of extrinsic and intrinsic activation signaling pathways of tumor cells, such as the induction of upstream p53 mutations and downstream B-cell lymphoma-2 associated X protein (Bax)/Bcl-2 signaling pathway [6]. The positive expression of survivin is detected in germ cells, developing fetal tissues and normal endometrium, and the abnormal expression of survivin is also detected in cancer cells [7]. Some studies suggest that the overexpression of survivin in breast cancer, neuroblastoma, lung cancer, esophageal cancer and colorectal cancer is associated with poor histology and advanced disease, and it indicates the decline in survival rate and/or recurrence time of patients [8].
Magnetic resonance imaging (MRI) is a noninvasive and repeatable examination method with a good resolution of soft tissues. Through different sequences, multi-directional and multi-level scan can be performed, which can clearly display the boundary, morphology, internal structure, involvement and infiltration of the lesion, so as to distinguish normal tissues from lesions [9]. Dynamic enhancement scan can not only show the morphological changes in the tumor, but also reflect the microvascular perfusion, degree of angiogenesis, grading and malignant degree, and evaluate the therapeutic effect and prognosis of tumors [10].
Currently, clinical diagnosis of HCC mainly focuses on serum alpha fetoprotein (AFP) level, hepatitis B virus (HBV) and hepatitis C virus (HCV) infection and imaging data [11]. Research has shown that survivin plays an important role in HCC. However, the level of survivin is rarely detected in HCC patients in the clinical diagnosis of HCC staging and prognosis. In this study, therefore, the survivin level in HCC tissues was detected qualitatively and quantitatively via immunohistochemistry (IHC), and its correlations with the corresponding MRI manifestations were analyzed. This study may help understand the relationship between tumor imaging and molecular biology, and provide new ideas for the clinical diagnosis and prognosis of HCC.
Clinical data of HCC patients enrolled
Clinical data of HCC patients enrolled
Patients
A total of 84 HCC patients receiving partial hepatectomy in the Surgery Department and Oncology Department in our hospital from April 2011 to May 2014 were enrolled in this study. There were 51 males and 33 females with an average age of (52.34
No patients had a history of alcohol abuse. Child-Pugh staging was used for the clinical severity and progression of cirrhosis, and there were 15 cases of grade A, 24 cases of grade B and 45 cases of grade C. When multiple lesions were removed in one patient, the largest lesion tissues and surrounding tissues were selected. Specific clinical data of 84 HCC patients were summarized in Table 1.
Detection of survivin level by IHC
Survivin level was measured by immunohistochemistry staining using IHC kits (Zhongshan Goldenbridge, Guangzhou, China) according to the manufacturers’ instructions. In brief, HCC tissues and corresponding para-carcinoma tissues were collected from patients and fixed in 4% paraformaldehyde, dehydrated and embedded in paraffin followed by being into 4
IHC results were read and scored by an experienced pathologist. Based on the method of Birner [12], IHC score was calculated according to the proportion of positive cells in the visual field and staining intensity of positive cells as follows: no positive cells
MRI examination
MRI examinations were performed using a GE 1.5T (Signa Horizon, GE Healthcare) MR scanner, and all images were obtained on the axial plane of phased array multi-coil. After conventional sagittal, axial and coronal positioning scans using multi-channel phased-array surface coil, fast spin echo (FSE) T1-weighted image (T1WI) (TR 700 ms, TE 10 ms) and fat-suppression T2WI (TR 4 500 ms, TE 85 ms) were adopted in plain scan. In axial and sagittal scan, the layer thickness was 5 mm, the interlayer spacing was 0.5 mm, the matrix size was 384
All patients underwent enhanced MRI. 0.1 mmol gadolinium diethylenetriamine pentaacetic acid (Gd-DTPA) contrast agent (2–4 mml/s) was injected into the antecubital vein and normal saline was also injected simultaneously. After injection of contrast agent, three-phase gradient-recalled echo (GRE) imaging (Tr/TE, 150/1.6. Matrix: 512
Image analysis
Hepatic MRI image data were measured and post-processed in the GE AW 4.2 workstation using Functool II software. The circular region of interest we drawn surrounded the lesion as much as possible, and the signal value of each sequence in this region was independently analyzed by a radiologist with 14 years of abdominal MRI experience. The signal intensity of HCC and its surrounding liver was evaluated subjectively by the physician:
Statistical methods
Data were analyzed using GraphPad Prism statistical software (Version 5.01, GraphPad Software, Santiago, Chile). Chi-square test was performed for analysis of the relationship between survivin expression and MRI imaging data of HCC patients. Overall survival (OS) curves of patients in high/low-expression survivin groups were drawn using Kaplan-Meier method and compared using log-rank test. Pearson correlation analyses were conducted to assess the correlations of survivin expression with MRI parameters.
Differences in survivin protein expressions in normal liver, HCC and para-carcinoma tissues (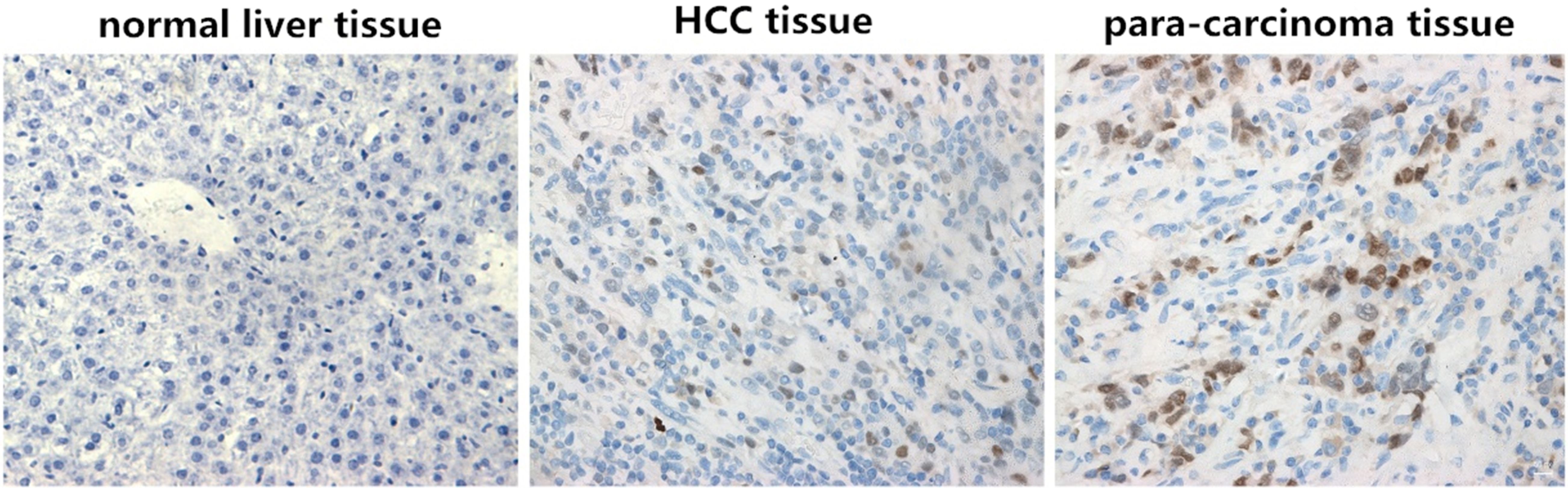
Detection of survivin protein expressions in normal liver, HCC and para-carcinoma tissues via IHC
As shown in Fig. 1, survivin was not expressed in normal liver tissues, but located in the nucleus in tumor tissues and para-carcinoma tissues. The nuclei of positive cells showed brown yellow, but the color depth varied and the color was distributed dispersedly. The positive expression rate of survivin in tumor tissues was 76.19% (64/84), which was significantly higher than that in para-carcinoma tissues (20.81%, 20/84), and the difference was statistically significant (
Differences in survivin protein expressions in normal liver, HCC and para-carcinoma tissues
Differences in survivin protein expressions in normal liver, HCC and para-carcinoma tissues
Kaplan-Meier survival curves were used to analyze the relationship between survivin expression and survival time of HCC patients. As shown in Fig. 2, OS of patients with low expression of survivin was 17.6 months, which was significantly longer than that of patients with high expression of survivin (12.5 months) (
MRI image data of HCC patients
In MRI dynamic enhancement examination after injection of contrast agent, slight enhancement in normal liver tissues started in arterial phase, reached the peak in portal venous phase, and gradually declined in equilibrium phase and delayed phase, but it remained at a higher level of enhancement. The time-signal curve rose slowly. In HCC tissues, obvious enhancement occurred in arterial phase, the enhancement degree was reduced in portal venous phase due to no blood supply of portal vein, and the enhancement degree continued to decline in equilibrium phase, showing a relatively low signal. The time-signal curve showed a “fast ascending-descending type”, which was the MRI enhancement feature of “fast progression and fast extinction” of HCC.
Survivin expression and survival curves of HCC patients.
MRI of HCC.
A 44-year-old male patient with hepatitis B and cirrhosis for many years: T1WI sequence (Fig. 3A) shows nodular abnormal signal shadow in the left medial lobe of liver. T2WI sequence (Fig. 2B) shows high signal in lesions, significant enhancement in arterial phase after dynamic enhancement, and high signal in the center of lesions in portal venous phase and delayed phase (Fig. 3C and D), showing a “fast ascending-descending type”. The circles in figure are the lesion regions of interest. This patient is pathologically confirmed as HCC after surgical resection.
Correlation analysis of survivin expression with MRI image ofpatients
Correlations of signal values of MRI impulse sequence with survivin level
As shown in Table 3, the survivin level had no correlations with the MRI scan shape and edge in the tumor area (
Correlations of survivin expression with MRI parameters of HCC patients
Pearson correlation analyses revealed that the survivin IHC score was not correlated with in-phase T1-weighted GRE, hepatic arterial-phase T1-weighted GRE, portal venous-phase T1-weighted GRE and equilibrium-phase T1-weighted GRE signals (
Discussion
In this study, IHC was used to detect the expressions of survivin in 10 cases of normal liver tissues, 84 cases of HCC tissues and para-carcinoma tissues, in order to evaluate the role of survivin in the development and pathogenesis of HCC. Our results showed that the survivin protein expression was under detection in normal tissues. The positive expression of survivin protein was detected in 64 cases of HCC tissues (76.19%) and 8 cases of para-carcinoma tissues (9.52%), suggesting that survivin might play a role in HCC. Consistent with this, a previous study [13] showed surviving was overexpressed in 55% of patients with HCC. A recent clinical study has shown that the postoperative recurrence rate of patients with survivin mRNA(
At present, although MRI has been widely used in the detection of liver tumors and vascular activity, the identification of benign and malignant tumors, as well as in the prediction of tumor growth [15], the correlations of MRI manifestations of HCC with survivin expression remains poorly understood.
In T1WI of MRI, low, high and equal signals can be shown due to differences in lipid and water content in HCC tissues. This is because water has long T1 and long T2 relaxation time, while lipids have short T1 and longer T2 relaxation time. Increased water content will prolong the T1 relaxation time of tumors, while increased lipids will shorten the T1 relaxation time of tumors [16]. Clinically, about two-thirds of cases have longer T1 relaxation time in MRI, showing a low signal in T1WI. Besides, opposite or equal signals to those in T1 sequence are shown in T2 sequence on the basis of T1 weight.
In this study, during MRI dynamic enhancement examination after injection of contrast agent, slight enhancement in normal liver tissues started in arterial phase, reached a peak in portal venous phase, and gradually declined in equilibrium phase and delayed phase, but it remained at a higher level of enhancement. The time-signal curve rose slowly. In HCC tissues, obvious enhancement occurred in arterial phase, the enhancement degree was reduced in portal venous phase due to no blood supply of portal vein, and the enhancement degree continued to decline in equilibrium phase, showing a relatively low signal. The time-signal curve showed a “fast ascending-descending type”, which was the MRI enhancement feature of “fast progression and fast extinction” of HCC [17].
In this study, the correlations of survivin expressions in 84 cases of HCC tissues with the corresponding MRI imaging data were analyzed. Results showed that the survivin level had no correlations with the MRI scan shape and edge in the tumor area, but it was significantly correlated with tumor diameter, MRI enhancement features and lymphatic metastasis (
In summary, we showed that survivin is significantly up-regulated in HCC tissues and associated with tumor growth and lymph node metastasis. Clinical detection of survivin level combined with MRI examination, especially the opposed-phase T1-weighted GRE and T2-weighted FSE signals, might be beneficial for diagnosis, treatment and prognosis of HCC.
Footnotes
Conflict of interest
None.
