Abstract
BACKGROUND:
Circulating microRNAs (miRNAs) have been implicated as novel biomarkers for various types of cancers. The aim of the study is to identify serum miRNAs with potential in detecting gastric cardia adenocarcinoma (GCA).
METHODS:
A three-phase study was designed with 102 GCA patients and 84 cancer-free controls. In the screening phase (3 GCA pools vs. 1 normal control (NC) pool), a total of 35 miRNAs were identified using quantitative reverse transcription polymerase chain reaction (qRT-PCR) based Exiqon panel. Subsequently, these miRNAs were further assessed by qRT-PCR in the training phase (30 GCAs vs. 30 NCs) and testing phase (72 GCAs vs. 54 NCs). Finally, the expression levels of the identified miRNAs were assessed in GCA tissues and exosomes.
RESULTS:
Five up-regulated miRNAs (miR-200a-3p, miR-296-5p, miR-132-3p, miR-485-3p and miR-22-5p) were identified in serum of the GCA patients compared with NCs. The areas under the receiver operating characteristic curve (AUCs) of the five-miRNA panel were 0.766 and 0.724 for the training and testing phases, respectively. In addition, miR-200a-3p, miR-296-5p, miR-485-3p and miR-22-5p were significantly up-regulated in GCA tissues. However, none of the miRNAs in the exosomes showed different expression between GCA patients and NCs.
CONCLUSIONS:
We identified a five-miRNA panel in peripheral serum samples as a non-invasive biomarker in detection of GCA.
Introduction
Gastric cancer, the second leading cause of cancer death worldwide [1], has two main sites: cardia (proximal, gastroesophageal junction) and non-cardia (fundus, body, distal and lesser or greater curvature). Exactly, gastric cardia cancer occurs in the 1 cm proximal and 2 cm distal region of the esophagogastric junction according to the AEG classification proposed by Siewert [2]. Gastric cardia adenocarcinoma (GCA) is the main histological type of gastric cardia cancer. Recent studies have suggested that GCA has distinct epidemiological, histopathological, molecular biological, characteristics which distinguished this tumor from the adenocarcinomas of distal stomach [3, 4, 5, 6]. In China, GCA is one of the prevalent fatal malignancies and shares much similar geographic distributions with esophageal squamous cell carcinoma (ESCC). Over the past few decades, GCA becomes one of the most rapidly growing malignant tumors in western countries [7], which increased by approximately fivefold to six-fold [8, 9], whereas the incidence of non-cardia gastric cancer and esophageal carcinoma remained stable or decreased during this period. In China, the incidence rate of GCA is about 50/100,000, and the local area (Henan province) was as high as 190/10,000 [10]. Diagnosis is often delayed for many patients due to the lack of specific symptoms. The patients with advanced stages have no chance of surgery, thus leading to a poor 5-year survival rate, approximately 25% [11]. Currently, endoscopic mucosal biopsy is the gold standard for the diagnosis of GCA. For its expensive price and invasiveness, it is difficult to be popularized in China. Moreover, the existing tumor markers, such as CEA, CA724 and CA199, have no sufficient sensitivity and specificity for diagnosis of GCA [12]. Accordingly, it is urgent for us to find novel and reliable non-invasive biomarkers for GCA detection as a critical step towards early intervention and reducing mortality.
MicroRNAs (miRNAs) are short non-coding RNAs (20-22nt) that can regulate a wide range of biological processes, including cell proliferation, apoptosis, aging, differentiation and cell-cycle control, by repressing translation or promoting degradation of target mRNAs [13]. Generally, a single miRNA can regulate multiple target genes, and miRNAs may serve as either tumor suppressors or oncogenes in various cancers [14]. Accumulating research demonstrated that the miRNA expression profiles could distinguish cancer patients from normal individuals [15, 16, 17]. Meanwhile, circulating miRNAs could be stably detected in peripheral blood [18]. These findings showed that circulating miRNAs could act as excellent non-invasive biomarkers for the detection and diagnosis of various types of cancers. Most studies have explored the miRNA expression data in non-cardia gastric cancer, or several studies related with gastric cancer [19] included some patients with gastric cardia adenocarcinoma [20], but few focused on GCA. Liu et al. found that significant downregulation and proximal promoter methylation of miR-203a and miR-203b in GCA tissues [21], which focused on the mechanism of miRNA in GCA. Gao et al. [10] performed miRNA profiles with GCA tissues, which was invasively obtained by endoscope or surgery. However, no study comprehensively conducted circulating miRNA profiles in GCA. Thus, there is an urgent need for the detection of circulating miRNA in GCA.
In present study, a three-phase study was designed to identify serum miRNA-panel as an effective biomarker for diagnosis of GCA. Firstly, the serum miRNA expression profile of GCA patients were screened using quantitative reverse transcription polymerase chain reaction (qRT-PCR) based miRCURY LNA microRNA Array. The candidate miRNAs were further evaluated in the training and testing phases by qRT-PCR. Moreover, the identified miRNAs were assessed in the GCA tissues and exosomes.
Materials and methods
Study design, patients and samples
All of the 102 GCA cases and 84 normal controls (NCs) were recruited from First Affiliated Hospital of Nanjing Medical University between 2014 and 2015. The anatomical sites of the GCA are in 1 cm proximal and 2 cm distal region of the esophagogastric junction. All of the GCA patients were newly diagnosed and confirmed by histopathology, who were not treated with chemoradiotherapy before blood or tissue collection. Blood samples were collected by SST Advance tubes (Becton, Dickinson and Company) and separated within 6 hours in order to prevent the contamination by cellular nucleic acids. The serum samples were stored at
Our study was designed for three phases. In the screening phase, 30 GCA serum samples and 10 NCs were randomly selected and pooled as 3 GCA samples and 1 NC sample (10 samples were pooled as 1 pool sample). According to the manufacturer’s protocol, a total of 25 ng RNA isolated from each pooled serum sample was reverse transcribed to cDNA by using the miRCURY Locked Nucleic Acid (LNA
Our study was approved by Institutional Review Boards of the First Affiliated Hospital of Nanjing Medical University, and each subject signed an informed consent.
The study was conducted according to the approved guidelines by Hospital Ethics Committee.
Exosomes isolation
According to the manufacturer’s instructions, 200 ul serum of each sample was incubated by using ExoQuick Exosome Precipitation Solution (System Biosciences, Mountain View, Calif) for 30–60 min at 2–8
RNA extraction
MirVana PARIS Kit (Ambion, Austin, TX, USA) was used to extract RNA from 200
Clinical characteristics of 102 GCA patients and 84 normal controls
Clinical characteristics of 102 GCA patients and 84 normal controls
MiRNA was amplified by using the specific primers of reverse transcription (RT) and polymerase chain reaction (PCR) from Bulge-Loop
The expression of miRNAs in serum samples and exosomes was determined using the 2
Statistical analysis
The software geNorm and the comparative Ct method were applied to evaluate the stability of candidate reference miRNA expression based on expression level both in cases and controls. The lower stability value (
All the statistical analyses were performed using SPSS software (version 22.0, IBM, USA). Mann-Whitney test was used to compare the differential miRNAs expression in different groups. Demographic and clinical characteristics among groups were analyzed by the
Overview of the experiment design. GCA: gastric cardia adenocarcinoma; NC: normal control.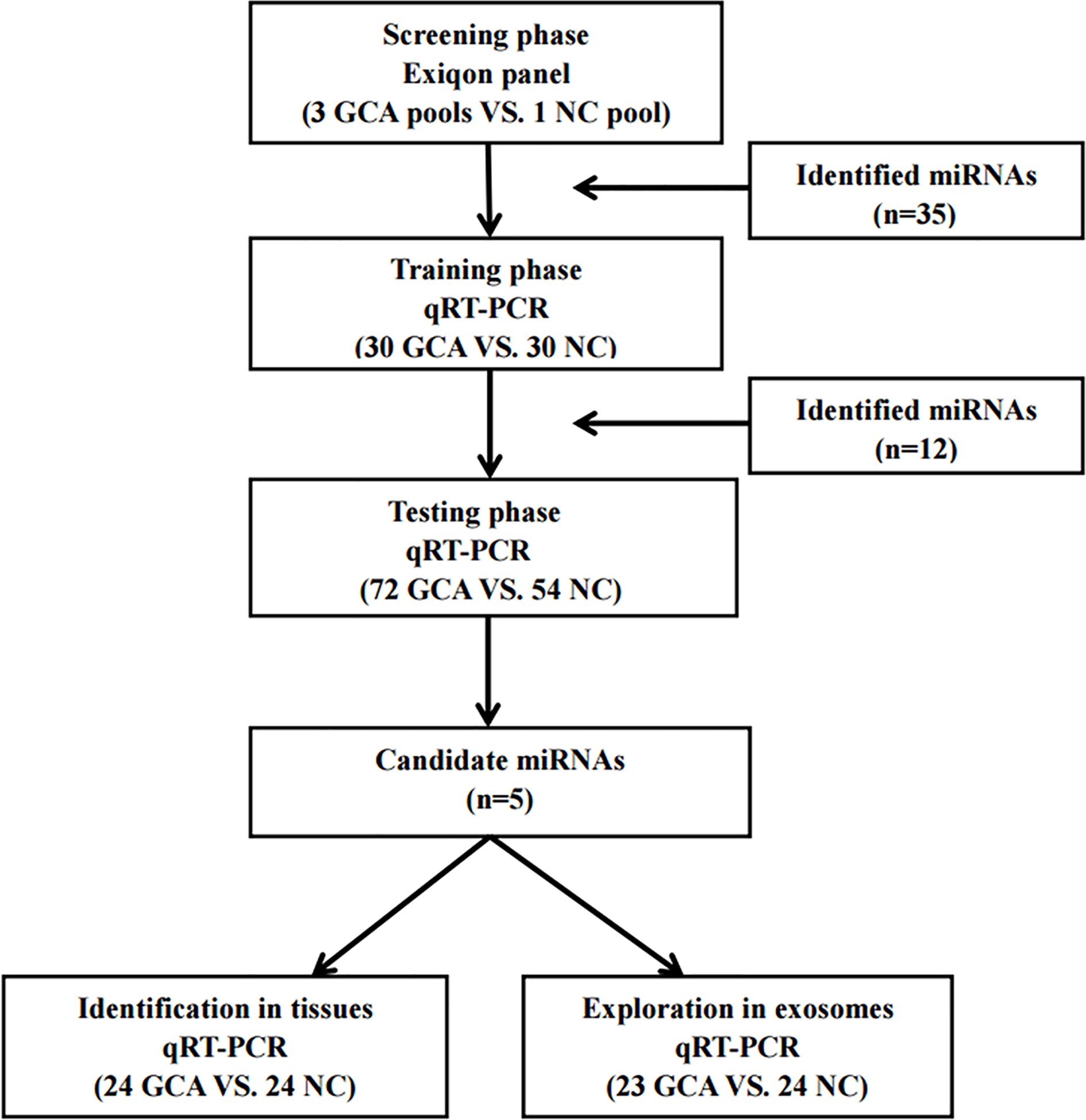
Expression levels of the five miRNAs in the serum of 102 GCA patients and 84 controls (in the training and testing phases). Y axis was presented as log10 (concentration; fmol/L). A: miR-200a-3p; B: miR-296-5p; C: miR-132-3p; D: miR-485-3p; E: miR-22-5p; T: tumor; N: normal controls. Horizontal line: mean with 95% CI.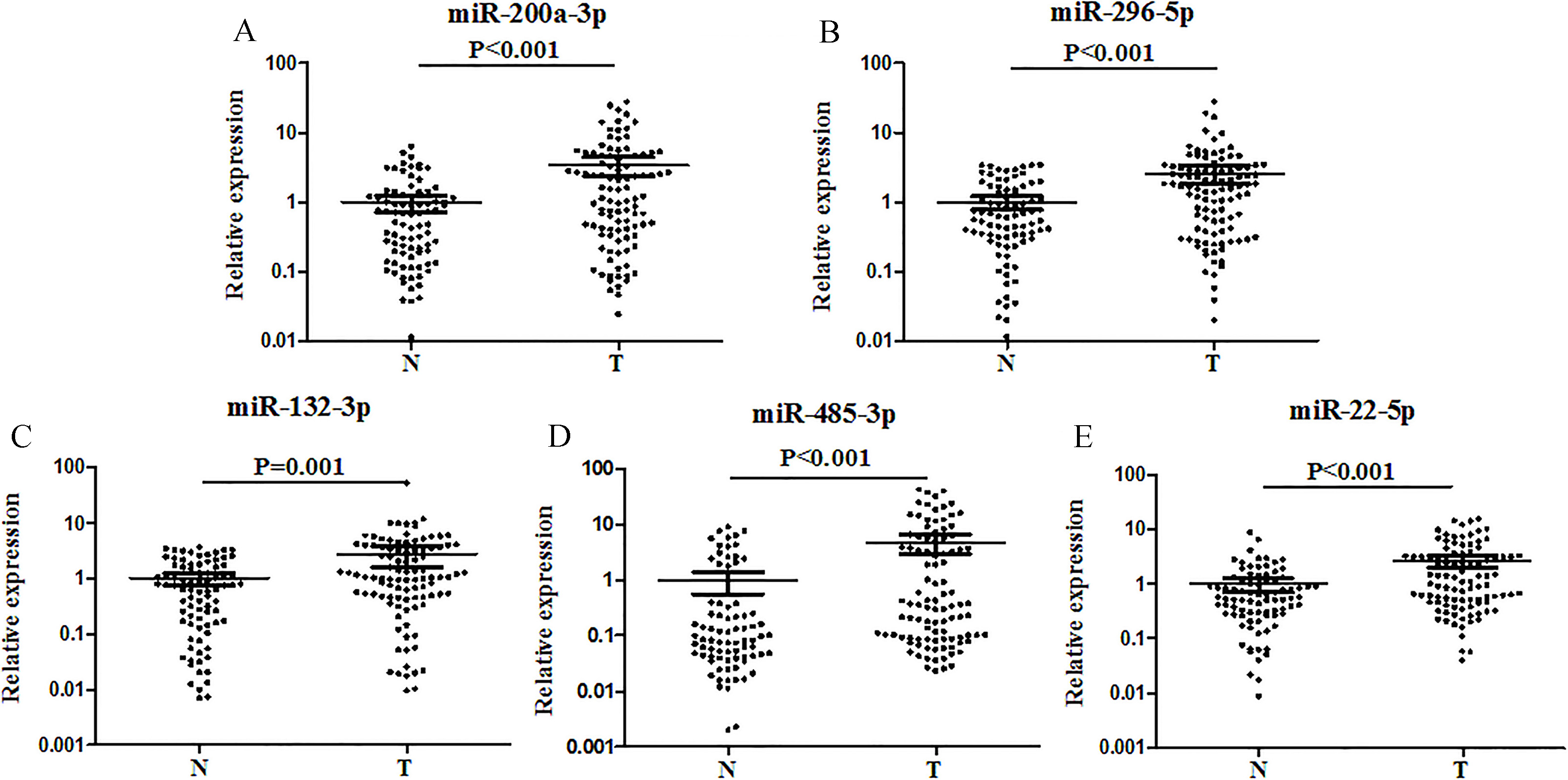
Receiver-operating characteristic (ROC) curve analyses of the five-miRNA signature to discriminate GCA patients form normal controls. A: the combined two phases of training and testing phases (102 GCA vs. 84 NCs); B: training phase (30 GCA vs. 30 NCs); C: testing phase (72 GCA vs. 54 NCs). AUC: areas under the curve.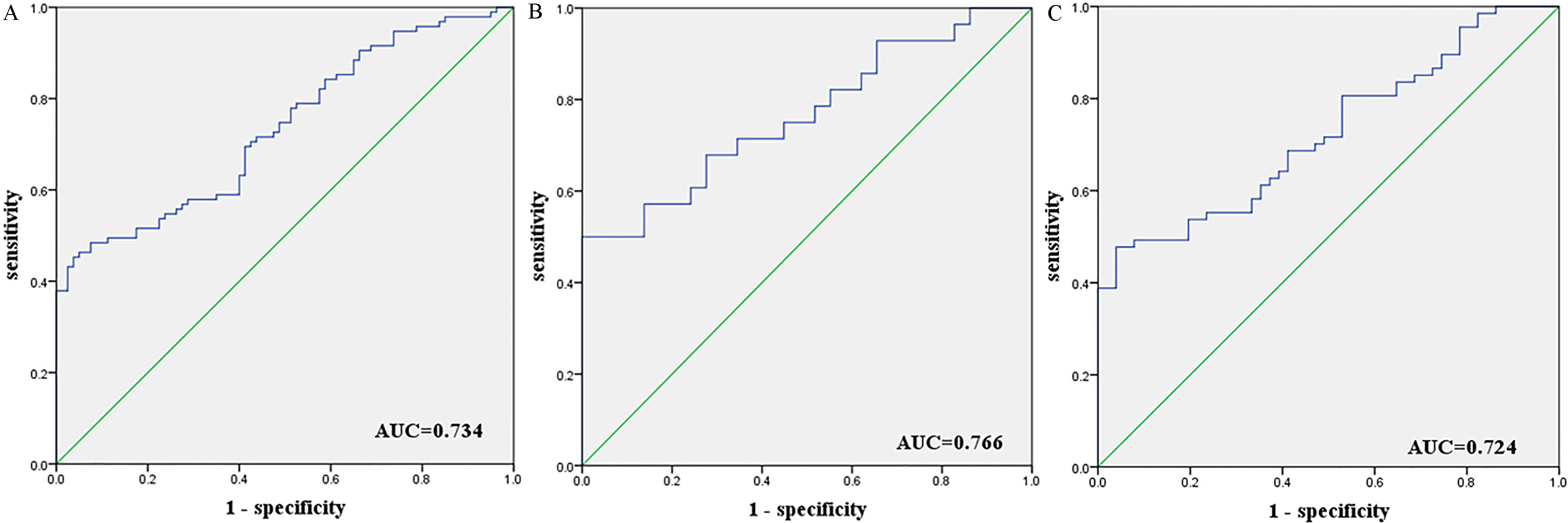
Expression of the five miRNAs in the tumor tissues of 24 GCA patients and 24 NCs. miR-200a-3p, miR-296-5p, miR-485-3p, miR-22-5p were significantly up-regulated in GCA tissues by qRT-PCR. A: miR-200a-3p; B: miR-296-5p; C: miR-485-3p; D: miR-22-5p; E: miR-132-3p; T: tumor; N: normal controls. Horizontal line: mean with 95% CI.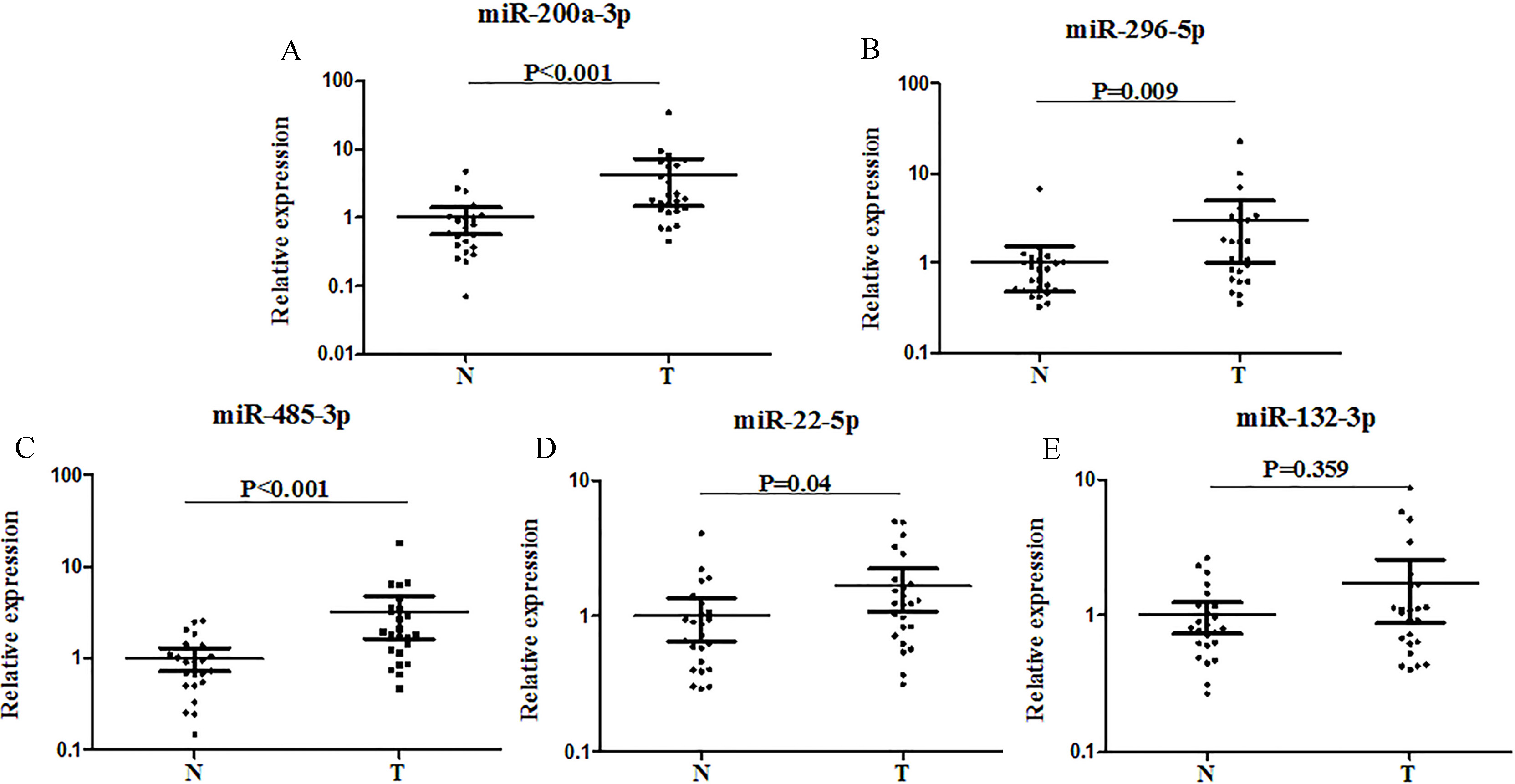
Expression of the five miRNAs in the serum exosomes of 23 GCA patients and 24 NCs. A: miR-200a-3p; B: miR-296-5p; C: miR-132-3p; D: miR-485-3p; E: miR-22-5p; T: tumor; N: normal controls. Horizontal line: mean with 95% CI.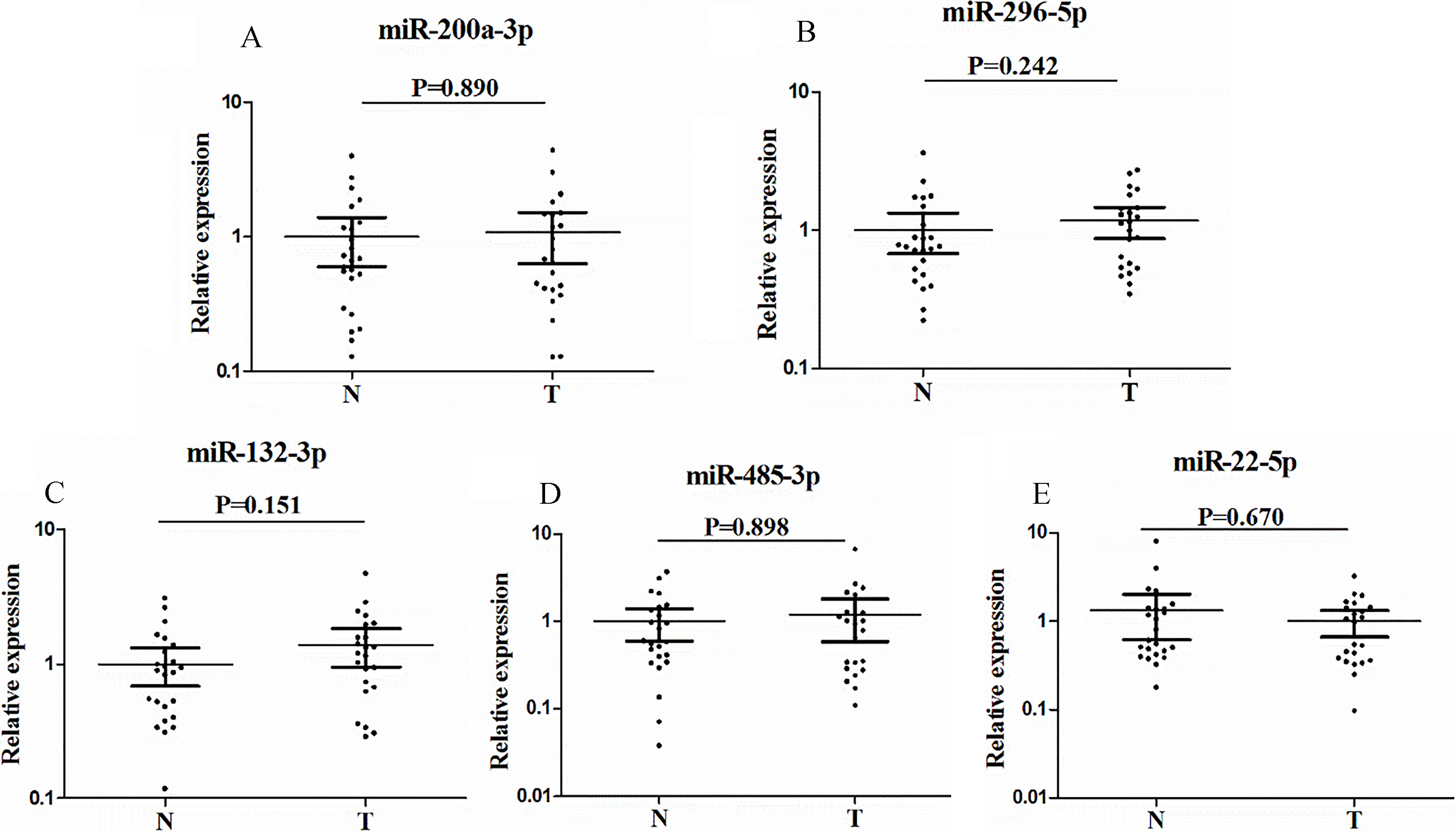
Characteristics of the subjects
A total of 186 subjects, including 102 GCA patients and 84 NCs, were enrolled in our study to assess the dysregulated miRNAs in serum of GCA patients. The clinical and pathology characteristics of the subjects were listed in Table 1. All of the subjects were divided into two phases after the screening phase: the training phase and the testing phase (The flow chat of the study was shown in Fig. 1). There was no significant difference in age or gender between GCA patients and NCs in each phase.
MiRNAs screening
To identify candidate miRNAs in peripheral serum of GCA patients, a total of 179 human miRNAs were initially analyzed by the Exiqon miRCURY-Ready-to-Use-PCR-Human-panel-I+II-V1.M based on the qRT-PCR platform in 3 GCA and 1 NC pooled serum samples (10 subjects for each pool). By qRT-PCR, each miRNA was assayed twice on 384-well plates. Only the ones that had a Ct value less than 37 and 5 lower than negative control were included in the further analysis. Eventually, 35 miRNAs showed at least a 1.5-fold altered expression (Table S1) in all 3 GCA pooled samples compared to the NC pooled sample. These miRNAs were chosen to further validation.
Evaluation of candidate miRNAs in serum by qRT-PCR
To select a stable endogenous reference, geNorm software was used for evaluation of the stability of candidate reference miRNA (miR-16-5p [23], miR-425 [24], miR-103a-3p [25] and miR-191-5p [26]). As shown in Fig. S1, miR-103a-3p and miR-191-5p were the most stable miRNAs with lower
The candidate miRNAs selected in the screening phase were further examined by using qRT-PCR between 30 GCAs and 30 NCs. Twelve miRNAs, in the training phase, with mean fold change
Diagnostic value of the candidate miRNAs
To evaluate the diagnostic value of the 5 miRNAs, the data from training and testing phases were combined. The AUC were 0.658, 0.663, 0.642, 0.692 and 0.674 for miR-200a-3p, miR-296-5p, miR-132-3p, miR-485-3p and miR-22-5p, respectively (Fig. S2). The AUC was 0.734 (95% CI: 0.662–0.807; Fig. 3A), when we combined the 5 miRNAs together as a panel, which showed higher accuracy than individual miRNA in discriminating GCA cases from NCs. Meanwhile, we also assessed the five-miRNA panel in the training and testing phases, and the AUCs were 0.766 (95% CI: 0.642–0.890; Fig. 3B) and 0.724 (95% CI: 0.634–0.814; Fig. 3C), respectively.
To understand the association of the five serum miRNAs, we assessed different TNM stages for all 102 patients. However, there was no different expression of each miRNA in GCA patients with stage III
The candidate miRNAs in tissue samples and serum exosomes
To validate the consistency of the miRNAs in serum and tissue of GCA patients, the expression levels of the five miRNAs were examined in additional 24 tissue samples. As shown in Fig. 4, the expression of miR-200a-3p, miR-296-5p, miR-485-3p, and miR-22-5p was significantly higher (
For further research, the five miRNAs in serum exosomes were investigated in additional 23 serum samples to explore the potential form of the candidate miRNAs. When compared to controls, none of the five miRNAs (miR-200a-3p, miR-296-5p, miR-132-3p, miR-485-3p and miR-22-5p) from GCA patients showed significant difference in the serum exosomes (Fig. 5).
Bioinformatics analysis of candidate miRNAs
To decipher the potential function of the candidate miRNAs, DIANA-TarBasev7.0 was utilized to identify the target genes of each candidate miRNAs. We put the five identified miRNAs into the DIANA-miRPath v3.0, a pathway analysis web-server for miRNA targets, to investigate the pathways with Kyoto Encyclopedia of Genes and Genomes (KEGG) analysis and Gene Ontology (GO) analysis (Table S3). KEGG analysis showed that TGF-beta signaling pathway seemed to be regulated by miR-200a-3p and miR-132-3p. GO analysis demonstrated miR-296-5p, miR-132-3p and miR-22-5p were related to TRIF-dependent toll-like receptor signaling pathway.
Discussion
The incidence of GCA gradually increased over the last 40 years, in contrast to the decreasing incidence of gastric cancer worldwide. Moreover, GCA may carry a worse long-term survival. Deans and Lehmann et al. demonstrated that GCA had distinct tumor characteristics and biological behaviors compared with non-cardia gastric adenocarcinoma [27, 28]. These findings suggested that cardia cancer is a distinct disease from gastric cancer, which may require different diagnosis and treatment strategies. Unfortunately, there is currently lack of noninvasive methods for the diagnosis of GCA. MiRNAs are known to play important roles in cancers. Majority of the studies to date have investigated the diagnostic value of peripheral circulating miRNAs in non-cardia gastric cancer samples.
In the present study, we focused on the GCA, aiming to discover a panel of serum miRNAs which might have potential value in detecting GCA. A total of 102 GCA patients were classified as the type II Siewert tumor [10, 29], the true cardia cancer, locating in 1 cm proximal and 2 cm distal region of the esophagogastric junction. A three-phase study was designed to identify a panel of serum miRNAs as an effective biomarker. In the screening phase, Exiqon miRNA qPCR panels, which showed better sensitivity and linearity than TaqMan platform while less abundant miRNAs were measured [30], was used to analyze differential expression profiling of serum miRNAs in 3 GCA and 1 NC pooled samples. Normalization is a necessary step for reliable qRT-PCR assays. The critical step in the following validation phases was to choose proper endogenous reference miRNAs. MiR-16-5p is relatively stable in the circulation and has been applied as endogenous reference in many studies [23]. The other three miRNAs, such as miR-425 [24], miR-103a-3p [25] and miR-191-5p [26] were also used as reference miRNAs in some studies. In our study, identified by the geNorm algorithm, miR-103a-3p revealed lower
Among the five identified miRNAs, miR-200a-3p is a member of miR-200 family (miR-200a, -200b, -200c, -141, -429). The miR-200 family were highly expressed within epithelial cells and acted as master regulators by targeting zinc finger-box bind homeobox 1 (ZEB1) and 2 (ZEB2) to maintain epithelial integrity [31]. Chang et al. [32] showed that miR-200a-3p was significantly down-regulated in gastric cancer tissues compared with normal tissues. It could act as tumor suppressor by targeting ZEB 1/ZEB 2, through the Wnt/
The diagnostic value of the peripheral miRNA identified in this study has also been evaluated in some other cancers. Up-regulated miR-200a-3p could have diagnostic utility in various cancers, including ovarian cancer [47], pancreatic cancer [48] and breast cancer [49]. Overexpressed miR-296-5p could be a diagnostic biomarker in esophageal squamous cell carcinoma [50]. Down-regulated plasma miR-132-3p was found in malignant mesothelioma patients [51]. In a word, the specificity of peripheral miRNA as biomarker is necessary to be further studied.
Circulating miRNAs were believed to be derived from tumor tissues and were released into the bloodstream by passively leaked or actively transported [52]. As they were extremely stable even can against RNase degradation [18], circulating miRNAs were demonstrated contained in exosomes [53], or protected by the formation of a protein-miRNA complex [54]. Herein, we assessed the expression of the five miRNAs in GCA tissues. The results showed that all the five miRNAs were up-regulated in the GCA tissue samples compared to normal controls, while the miR-200a-3p, miR-296-5p, miR-485-3p and miR-22-5p yielded statistical significance. Our findings confirmed the theory of the origin of circulating miRNAs. Then, we further explored exosomal miRNA in serum samples of GCA patients to study the potential form of the five serum miRNAs. It turned out that none of the five miRNAs (miR-200a-3p, miR-296-5p, miR-132-3p, miR-485-3p and miR-22-5p) from GCA patients showed significant difference in the serum exosomes compared to controls. Arroyo et al. [54] showed that potentially 90% of miRNAs studied copurified with the ribonucleoprotein complex (Ago2 ribonucleoprotein complex), but a minority of specific miRNAs associated predominantly with vesicles. The theory may explain our results. The five miRNAs remained stable in the blood might form Ago2-miRNA complex, rather than via the package of exosomes.
There were some limitations in our research. Firstly, the study was single-center and the samples of serum and tissue were not large enough, which may result in possible bias in the outcomes. Secondly, we used the KEGG analysis and GO analysis to evaluate the relationships between candidate miRNAs and pathways, but not through experiments. Thus, future studies are warranted to explore the function of the miRNAs in GCA. In addition, our study focused on GCA, which arose in the boundary between esophagus and stomach. The relationship and specificity of the miRNAs in esophagus cancer and gastric cancer should also be explored in the future.
In summary, we identified a five-miRNA signature, which could function as a non-invasive biomarker in the detection of GCA, and it may contribute to the popularity of cancer screening. Our work comprehensively explored the miRNA in peripheral serum of GCA, which might serve as a basis for further functional studies.
Footnotes
Acknowledgments
This work was supported by the National Natural Science Foundation of China [Grant number: 81672400; 81370516; 81702364]; the Natural Science Foundation of Jiangsu Province [Grant number: BK20171085], Natural Science Foundation of Education Committee of Jiangsu Province (No. 15KJB320001) and Six Talent Peaks project in Jiangsu Province (No. WSN-030).
Conflict of interest
None.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-181258.
