Abstract
BACKGROUND:
Ovarian cancer (OC) is the fifth most common type of cancer in women worldwide. MiR-27a plays an important role in the development of ovarian cancer. However, the exact function and molecular mechanism of miR-27a in epithelial-mesenchymal transition (EMT) has not been thoroughly elucidated to date.
METHODS:
Quantitative real-time PCR (qRT-PCR) was used to determine the expression of miR-27a and FOXO1 mRNA in ovarian tissues and cells. The function of miR-27a in ovarian cancer was investigated through overexpression and knockdown of miR-27a in vitro. Wound healing and Transwell assays were performed to evaluate the migration and invasive capacity of the cells. A luciferase reporter assay was conducted to confirm the interaction between miR-27a and FOXO1. Western blotting was used to evaluate FOXO1, EMT and Wnt/
RESULTS:
In our study, we found that the mRNA expression level of miR-27a was significantly higher in ovarian cancer tissues and in HO8910 and OV90 cells. Functional experiments showed that miR-27a overexpression potentiated the migration and invasion of HO8910 and OV90 cells, while miR-27a inhibition reduced the cells’ migration and invasion. Moreover, miR-27a upregulated the expression of mesenchymal cell markers and downregulated the expression of epithelial cell markers, which were restored via silencing of miR-27a expression. Subsequently, miR-27a was found to directly target and suppress the expression of FOXO1. Finally, we demonstrated that miR-27a promoted the progression of ovarian cancer cells and induced the process of EMT via the Wnt/
CONCLUSIONS:
Taken together, these results indicate that targeting miR-27a and FOXO1 could represent a strategy for anticancer therapy in ovarian cancer.
Introduction
Ovarian cancer is the second leading cause of death among all gynaecological tumours and is a common female reproductive cancer; it ranks fifth among causes of death for female cancer patients [1, 2, 3]. In 2015, there were 22,500 deaths and 52,100 patients newly diagnosed with OC in China [4]. It is difficult to diagnosis this cancer in early stages due to a lack of significant signs or symptoms. The majority of OC cases are diagnosed at an advanced metastatic stage and have a poor outcome [5]. Thus, improving targeting therapies and studying the mechanisms underlying tumour metastasis are necessary for reducing OC mortality.
Epithelial-mesenchymal transition is defined as a dynamic process in which epithelial cells undergo multiple biochemical changes and assume a mesenchymal phenotype, which has motile and invasive characteristics [2]. Numerous studies have indicated that EMT is related to metastasis, which is defined as a process in which cancer cells spread from the primary tissue to surrounding tissues because the cells lose their cell-cell adhesion ability and gain migratory and invasive capabilities [1, 6, 7]. While undergoing the EMT process, cells upregulate their expression of mesenchymal cell markers (e.g., N-cadherin, vimentin, and fibronectin) and simultaneously downregulate epithelial markers (e.g., E-cadherin,
MiRNAs are approximately 22-nucleotide single-stranded noncoding RNA molecules that are incorporated into the RNA-induced silencing complex (RISC) [9, 10, 11, 12]. Typically, the miRNAs play important roles in cell differentiation, proliferation, apoptosis and EMT. For instance, upregulation of miR-483-5p in human lung adenocarcinoma is correlated with the progression of the tumour via the activation of Wnt/
In the present study, we determined the regulatory role of miR-27a-induced EMT in ovarian cells. Wnt/
Methods and materials
Cell culture
OV90 (a high-grade serous ovarian cancer cell line) and HEK-29 (a human embryonic kidney epithelial cell line) cell lines were purchased from the American Type Culture Collection (ATCC, Manassas, VA, USA). HOSEpiC (a human ovarian surface epithelial cell line) and HO8910 (an ovarian cancer cell line) cell lines were purchased from the Cell Bank of Type Culture Collection of the Chinese Academy of Sciences (Shanghai, China). All cells were authenticated with short tandem repeat (STR) and mycoplasma analysis, which showed that the STR authentication was correct and that they were mycoplasma-negative. The cells were incubated in Dulbecco’s modified essential medium (DMEM; Gibco, Invitrogen) supplemented with 10% heat-inactivated FBS, penicillin (100 U/ml), and streptomycin (100
Tumour tissue specimens
We obtained 37 paired ovarian cancer tissues and adjacent non-tumour tissues from patients who had undergone surgical resection at the Huizhou No. 2 Women’s and Children’s Healthcare Hospital. Patients who had preoperative chemotherapy or radiotherapy and patients who were diagnosed with other malignant diseases were excluded from our experimental group. Informed written consent was obtained from all participants, and ethical approval for the use of ovarian cancer specimens for this study was obtained from the Research Ethics Committee of Huizhou No. 2 Women’s and Children’s Healthcare Hospital.
Cell transfection
The miR-27a mimics and inhibitors (including mimic negative control: miR-NC; inhibitor negative control: anti-NC) and si-FOXO1 were chemically synthesized by Genepharma Corporation (Shanghai, China). OV90 and HOSEpiC cells were seeded in 6-well plates at 3.5
Wound healing assays
Cells were seeded in 6-well plates to form a cell monolayer (near 90% confluence), and a wound was created by scratching the length of the well with a 10
Transwell invasion assay
The upper chamber of the Transwell was coated with Matrigel and then incubated at 37
The primer sets for qRT-PCR
The primer sets for qRT-PCR
cgcagcctgc agcccg.
Total RNA was prepared from tissues and cell lines using a Qiagen RNeasy kit per the manufacturer ’s instructions. The total RNA was treated with DNase I, and 1
MiR-27a is upregulated in ovarian cancer tissues and cell lines. (A) Expression of miR-27a mRNA in ovarian normal tissues and ovarian cancer tissues. (B) The expression of miR-27a mRNA in HO8910 and OV90 cells compared with HOSEpiC cells. U6 was used as an internal reference. Bars represent means 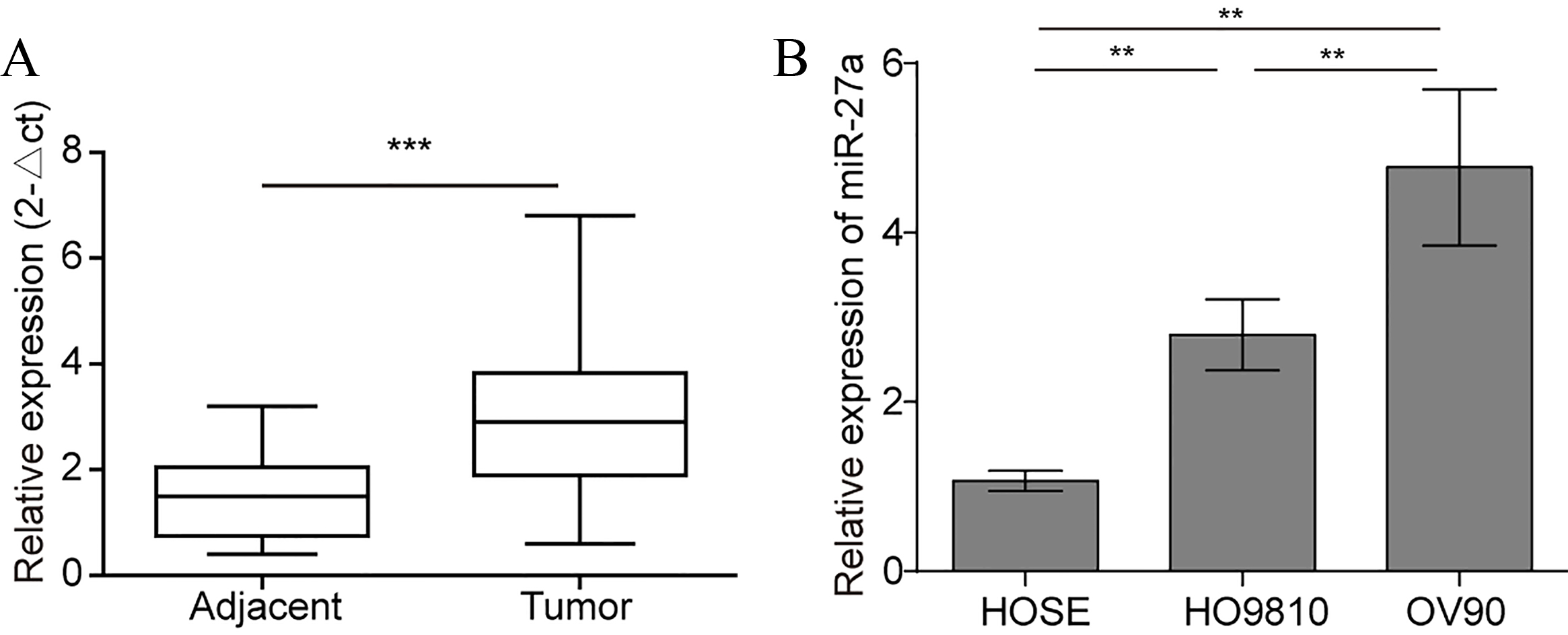
Protein samples were resolved on 12% SDS-PAGE. The separated proteins were electroblotted onto a polyvinylidene fluoride (PVDF) membrane (Millipore, Billerica, MA, USA). The membranes were blocked in blocking buffer (Tris-buffered saline containing 10% non-fat dry milk and 0.1% Tween 20) for 2 h. The membranes were probed with anti-N-cadherin (1:2000, Cell Signaling Technology, Boston, MA, USA), anti-E-cadherin (1:5000, BD Biosciences, San Jose, CA, USA), anti-vimentin (1:1,000, V6630, Sigma-Aldrich), anti-FOXO1 (1:1000, Cell Signaling Technology), and anti-
Targeting prediction analysis
Putative binding genes of miR-27a were analysed by the online bioinformatic tool Targetscan (
Plasmid construction
The luciferase reporter plasmids pGL3-WT-FOXO1 and pGL3-MUT-FOXO1, containing a predicted wild-type or mutant miR-27a binding site, were constructed by PCR amplification from genomic DNA of ovarian cancer cells. Then, the fragment was cloned into the pGL3 basic vector (Promega), which contains a modified coding region of firefly (Photinus pyralis) luciferase. The pRL-TK plasmid (Promega), containing the Renilla luciferase reporter, served as an internal control.
Luciferase reporter assay
To determine the role of miR-27a in FOXO1 3’-UTR promoter activity, 293T cells were pre-cultured on 12-well plates and transfected with 1
Statistical analysis
All experiments were independently repeated three times. All values in the text and figures are expressed as the mean
Overexpression of miR-27a promotes ovarian cancer cell EMT, cell motility, and invasion. (A) qRT-PCR was used to detect the mRNA expression level of miR-27a in HO8910 and OV90 cells. U6 was used as an internal reference. (B) HO8910 and OV90 cells were MOCK-transfected (control) or transfected with miR-27a mimics, miR-NC, and then their migration ability was measured by a wound healing assay. (C and D) The invasion assays of HO8910 and OV90 cells transfected with miR-27a mimics, NC, or controls were performed by using Transwell assays. Magnification, 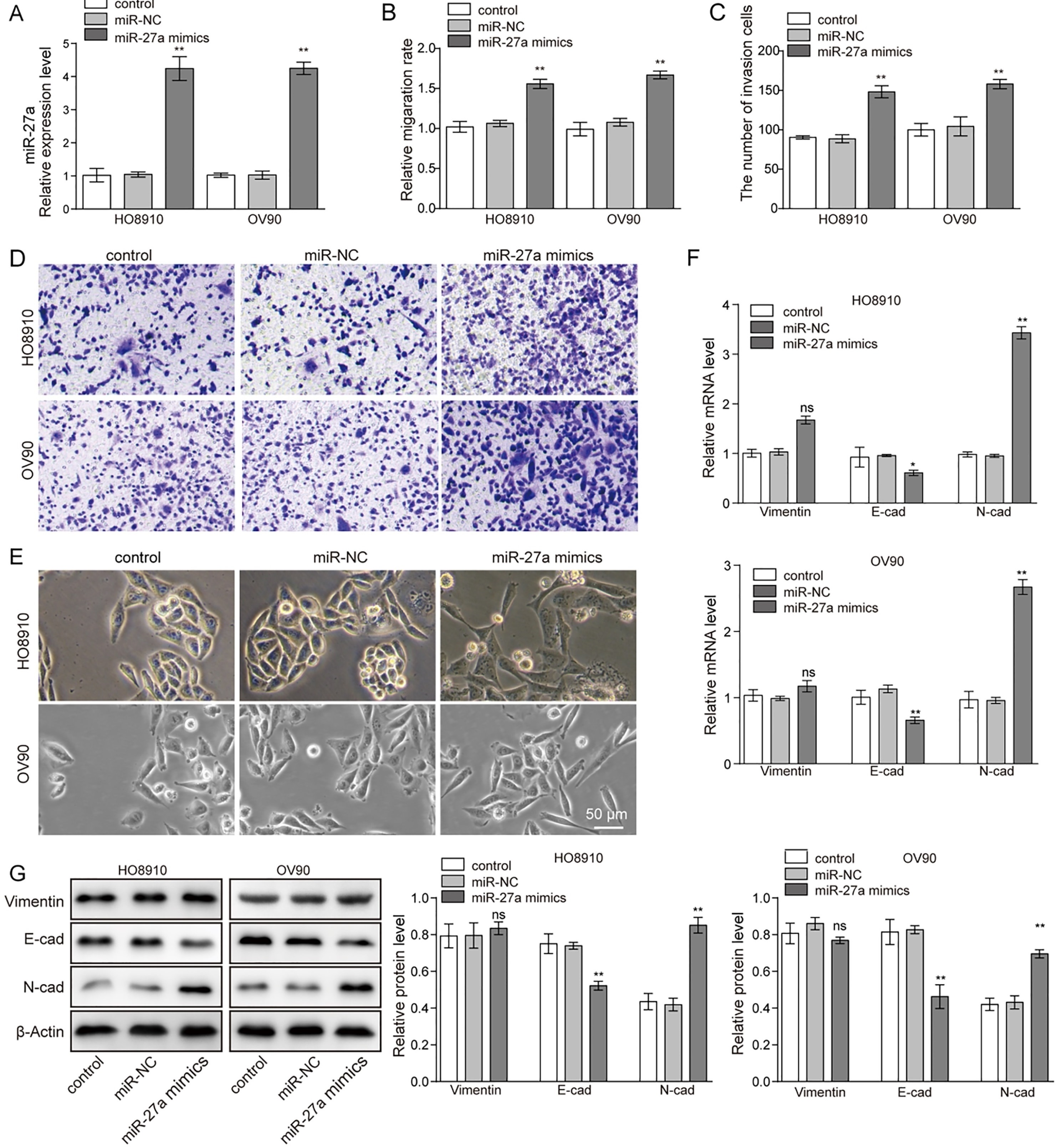
Silencing miR-27a inhibited ovarian cancer cell EMT, cell motility, and invasion. (A) Knockout of miR-27a in HO8910 and OV90 cells followed by measuring the mRNA expression level of miR-27a detected by qRT-PCR. (B) Scratch migration and (C and D) Transwell analyses were used to determine the effect of silencing of miR-27a on the migratory and invasive capability of HO8910 and OV90 cells. *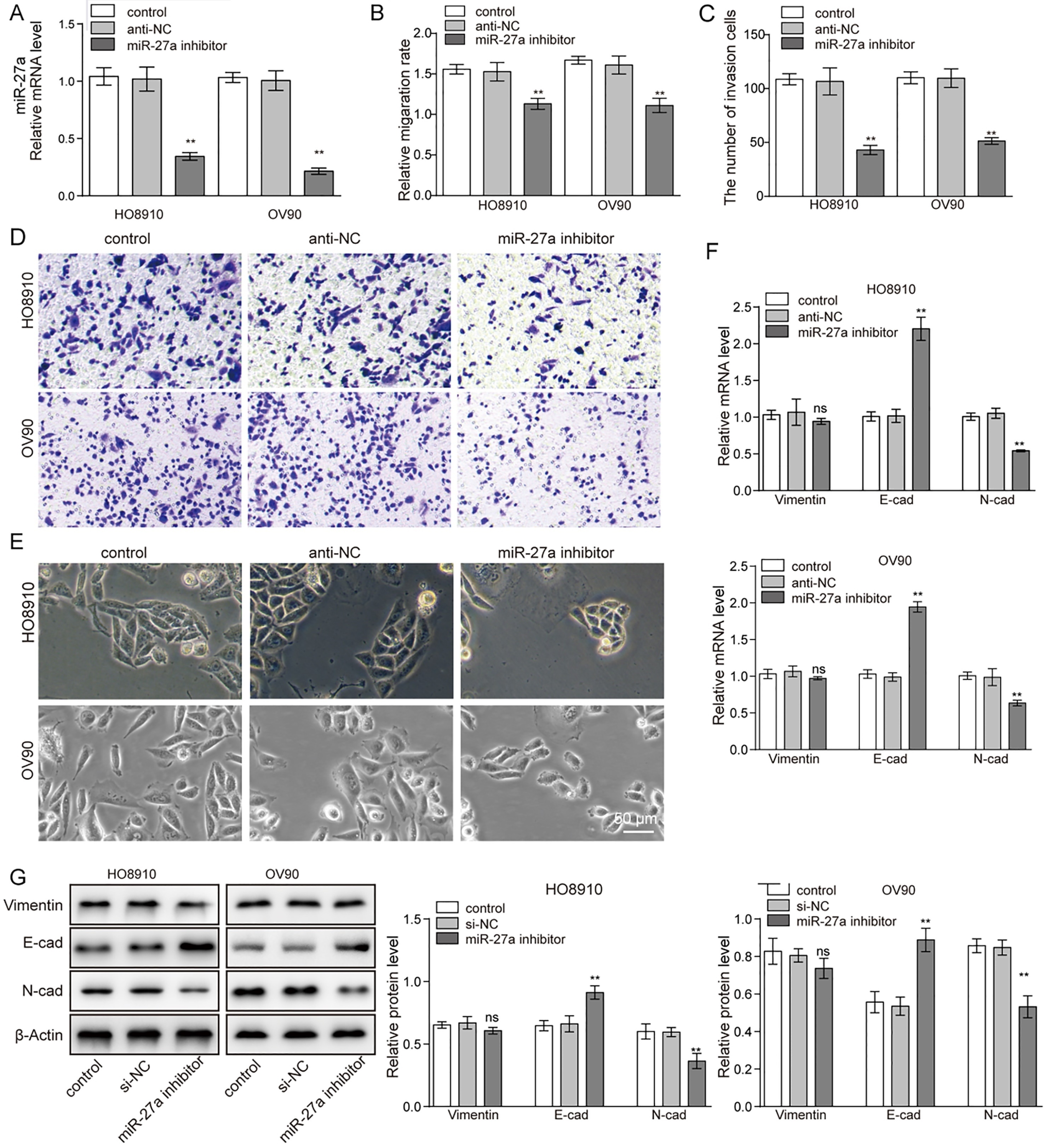
MiR-27a is upregulated in ovarian cancer tissues and cell lines
Numerous studies have shown that high expression of miR-27a is associated with cancer cell proliferation, migration and invasion [14, 17, 23, 24]. The expression level of miR-27a was tested by qRT-PCR. Figure 1A shows that the expression level of miR-27a is dramatically higher in ovarian cancer tissues compared to the corresponding normal tissues. Furthermore, we compared the expression of miR-27a in ovarian cancer cell lines (HO8910 and OV90) with human ovarian surface epithelial (HOSEpiC) cells. We found a 2.6- and 4.5-fold increased expression level of miR-27a in the ovarian cancer cell lines HO8910 and OV90, respectively (Fig. 1B).
MiR-27a promotes the migration and invasive capacity of ovarian cancer cells, and induces EMT activation
To explore the role of miR-27a in ovarian cancer cells, first, the HO8910 and OV90 cells were transfected with miR-27a mimics. The results showed that pretreatment with miR-27a mimics led to a 3.8- and 4.1-fold increased concentration of miR-27a, while there was no significant difference between the control group and the NC group (Fig. 2A). Furthermore, the wound-healing capacity of HO8910 and OV90 cells, which were transfected with miR-27a mimics, was dramatically greater than that of HO8910 and OV90 cells treated with NC (Fig. 2B and Supplement Fig. S1).
We next determined the effect of miR-27a on ovarian cancer cell invasion. Transwell invasion assays showed that the invasive ability of HO8910 and OV90 cells was significantly increased when they overexpressed miR-27a compared with the control group (Fig. 2C and D). As shown in Fig. 2E, miR-27a overexpression resulted in a dramatic transformation towards a mesenchymal cell phenotype, especially in HO8910 cells, which showed an altered cell morphology into a long spindle shape.
To obtain further insights into the EMT-promoting capability of miR-27a in ovarian cancer, E-cadherin, N-cadherin and vimentin were detected using qRT-PCR and western blotting. The results revealed that miR-27a overexpression might significantly increase both the mRNA level and protein level of N-cadherin and vimentin but it decreased E-cadherin compared with the NC cells (Fig. 2F and G). There was no significant difference between the control group and the NC group as shown in Fig. 2A–G. Generally, we could conclude that miR-27a promoted the migration and invasion of ovarian cancer cells. Moreover, their capacity of EMT was obviously increased while overexpressing miR-27a.
Knockdown of miR-27a inhibited cell motility, invasion and EMT of ovarian cancer cells
To further test the function of miR-27a in ovarian cancer cells, an miR-27 inhibitor was transfected into HO8910 and OV90 cells. It resulted in decreases in miR-27a expression by 67% and 79% in HO8910 and OV90 cells, respectively, compared with the expression of miR-27a in the miR-NC group (Fig. 3A). Under the effect of the miR-27a inhibitor, we found that their migration ability was decreased after inhibitor treatment (Fig. 3B and Supplement Fig. S2). As we had supposed, the number of cells invading through the membrane was also significantly decreased compared with the control group (Fig. 3C and D). Knockdown of miR-27a resulted in a significant change in morphology of the OV90 cells, while no significant change was observed for HO8910 (Fig. 3E). To examine the effect of miR-27a on the EMT, the expression of E-cad, N-cad and vimentin were assessed. Consistent with our expectation, the expression of these two genes (N-cad and vimentin) were decreased and only E-cad expression was enhanced following miR-27a inhibitor treatment in both HO8910 and OV90 cells (Fig. 3F). In addition, the western blot results showed decreased expression of N-cad and vimentin proteins in both HO8910 and OV90 cells, and meanwhile the protein level of E-cad was increased (Fig. 3G). Taken together, our findings indicate the miR-27a inhibitor could block the capacity for migration, invasion and EMT in ovarian cancer cells.
MiR-27a directly targets the FOXO1 3’-UTR. (A) Binding sites of miR-27a in the 3’-UTR of FOXO1 are shown. (B) The relative luciferase activity was measured in HEK293 cells co-transfected with the WT or MUT FOXO1 3’-UTR with miR-27a mimics or miR-NC. (C and D) The relative mRNA and protein levels of FOXO1 were detected in cells transfected with or without miR-27a mimics and miR-27a inhibitors. (E and F) The expression level of FOXO1 in the tumour and normal adjacent tissues were determined by qRT-PCR (E) and WB (F). *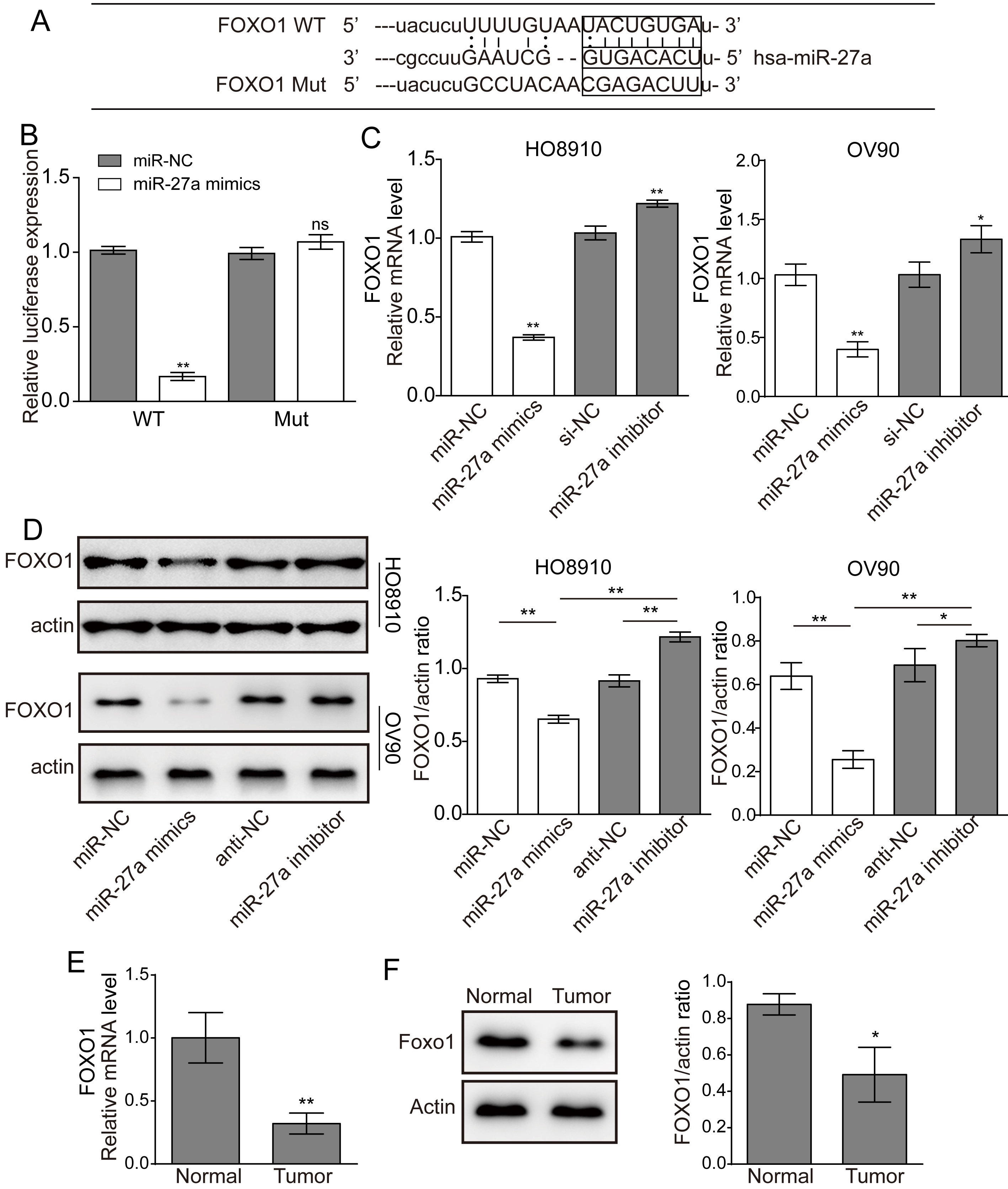
MiR-27a activates the Wnt/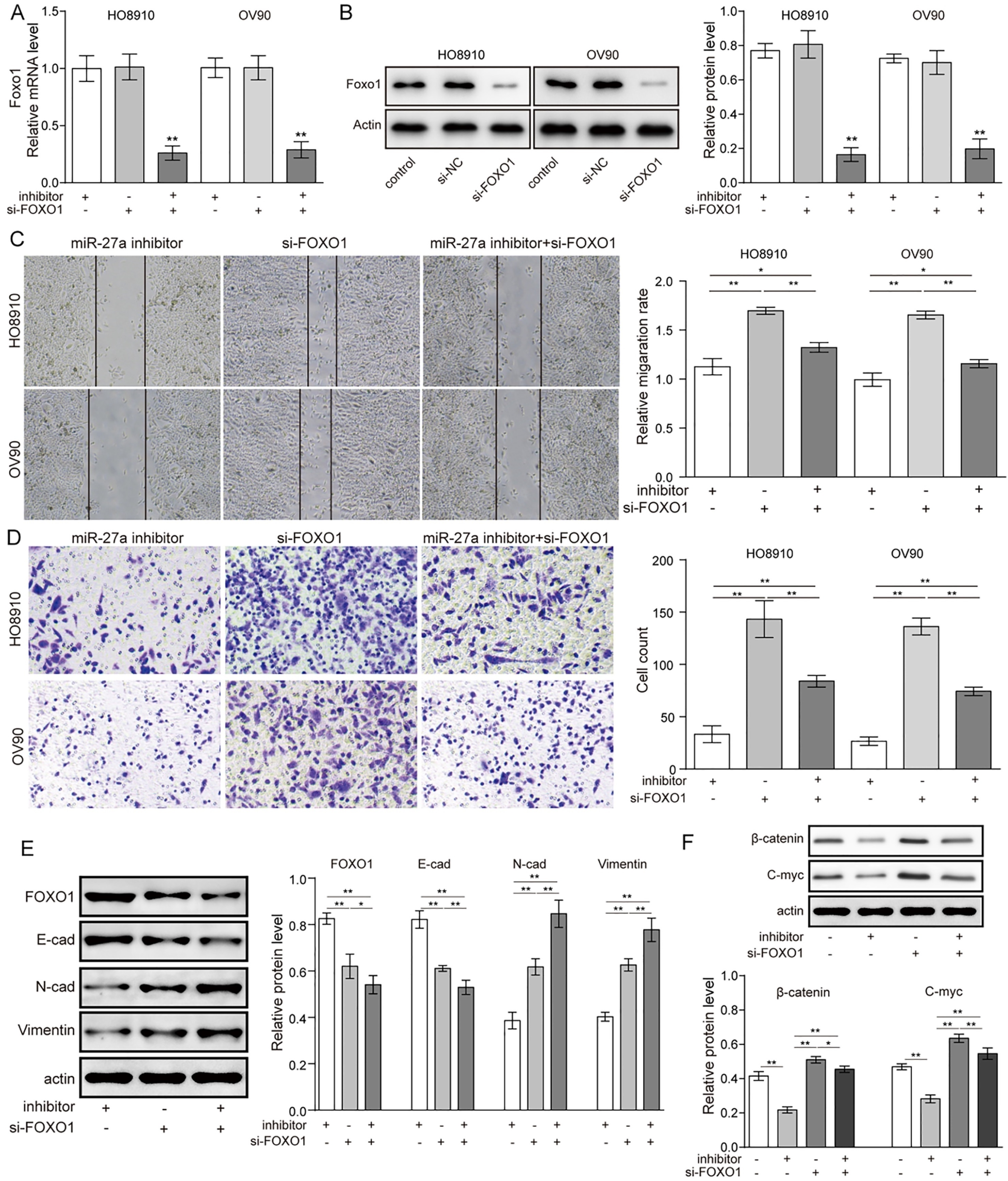
To dissect the molecular mechanism involved in miR-27a regulated metastasis in ovarian cancer cells, the putative binding proteins of miR-27a were analysed by the online bioinformatic tool RNA hybrid. The results showed that the 3’-UTR of FOXO1 carried a binding sequence for miR-27a (Fig. 4A). Then, we constructed luciferase reporters for the FOXO1 gene that contained either a wild-type (WT) 3’-UTR or a mutant (mut) 3’-UTR with mutated sequences in the miR-27a binding site. Cells were cotransfected with miR-27a mimics together with the plasmids pGL3-WT-FOXO1 or pGL3-mut-FOXO1. The FOXO1 3’-UTR activity was measured with a luciferase assay. The results revealed that the wild-type FOXO1 reporter gene (pGL3-WT-FOXO1) activity was clearly suppressed by miR-27a overexpression compared with the miR-NC group. In contrast, the mutant FOXO1 reporter gene (pGL3-mut-FOXO1) was not significantly affecting by miR-27a transfection (Fig. 4B).
We also determined whether miR-27a is involved in regulation of the expression of FOXO1. As shown in Fig. 4C and D, overexpression of miR-27a led to a significantly decreased expression level of FOXO1 mRNA and protein, but their expression increased substantially in cells that were treated with miR-27a inhibitors. The findings for HO8910 cells and OV90 cells were concordant. Moreover, qRT-PCR and western blot analysis shown that the expression level of FOXO1 was decreased in ovarian cancer tissues compared with normal tissues (Fig. 4E and F). Taken together, miR-27a directly targets FOXO1 and inhibits its expression in ovarian cancer cell lines.
MiR-27a activates the Wnt/
-catenin pathway by targeting FOXO1
qRT-PCR and western blotting were performed to investigate whether using si-FOXO1 could inhibit the mRNA and protein level of FOXO1 (Fig. 5A and B). Knockdown of FOXO1 caused a significant increase in cell migration and invasive capacity (Fig. 5C and D). Co-transfection with si-FOXO1 and miR-27a inhibitors restored the effect of miR-27a and the cells’ capacity for cell migration and invasion, compared with cells only treated with miR-27a inhibitors. Similarly, western blot assays showed that knockdown of FOXO1 could decrease the expression of E-cadherin while increasing the levels of N-cadherin and vimentin. Meanwhile, co-treatment with si-FOXO1 and the miR-27a inhibitor led to downregulation of E-cadherin and upregulation of N-cadherin and vimentin, which is completely opposite to the results in the miR-27a inhibitor treatment group (Fig. 5E). It is known that activation of Wnt/
Discussion
Ovarian cancer has become one of the significant issues in recent years. Approximately 6,700 women in the UK are diagnosed with ovarian cancer each year. Therefore, it is necessary to explore the molecular mechanisms underlying ovarian cancer. Many studies have shown that miRNA has an important role in cellular homeostasis by regulating cellular signalling in such processes as proliferation, migration, and invasion, and breakdown of this management can finally lead to carcinogenesis [13, 25, 26, 27]. A growing body of evidence suggests that miR-27a exerts important roles in many cancers. For instance, miR-27a negatively regulates SMAD2 and SMAD4 by binding to the 3’-UTR of SMAD2 and SMAD4 and impairs their functions in lung cancer cells [16]. MiR-27a is highly expressed in GC tissues and cells, and it might promote cell proliferation, migration and invasion by targeting SF-RP1 via activation of the Wnt/
EMT plays an important role in cancer metastasis. EMT occurs under physiological conditions; however, is also a key mechanism required for malignant progression. “Oncogenic EMT” allows tumour cells to acquire invasive properties and to develop metastatic growth characteristics. Moreover, it protects them from hostile conditions during the dissemination process [28]. In this study, we found that miR-27 might be responsible for promoting migration, invasion and EMT, while miR-27a inhibition markedly weakened EMT and the invasive capacity of ovarian cancer cells. All of these findings indicate that miR-27a plays an oncogenic role in OC by potentiating the metastatic behaviours and EMT of OC cells.
FOXO1, belonging to the O subgroup of forkhead transcription factors, is a well-known tumour suppressor and has been found to play an important role in the development and progression of various human cancers [29, 30, 31, 32, 33]. FOXO1 is known to be associated with cancer cell migration, invasion and EMT in vitro as well as tumourigenicity and metastasis in vivo [9, 29, 30, 34, 35]. This study showed that miR-27a targeting the expression of FOXO1. Overexpression of miR-27a significantly decreased the mRNA level of FOXO1. Importantly, knockout of FOXO1 promote the migration, invasion, and EMT of cancer cells, as well as overexpression of miR-27a. These findings indicate that miR-27a could modulate activation of EMT by inhibiting FOXO1. This effect has been supported by previous studies, such as reports that adipose tissue-secreted miR-27a promotes liver cancer by targeting FOXO1 in obese individuals. Chitosan degradation products promote nerve regeneration by stimulating Schwann cell proliferation via the miR-27a/FOXO1 axis [36]. Furthermore, our results showed that si-FOXO1 upregulated the expression of
As an important regulator of EMT, the Wnt signalling pathway plays a crucial role [37, 38, 39]. Several signalling pathways, including Wnt/
In conclusion, the present study showed that miR-27a upregulates the migration, invasion and EMT of ovarian cancer cells. Moreover, miR-27a has an important role in increasing the expression of Wnt/
Supplementary data
The supplementary files are available to download from
sj-docx-1-cbm-10.3233_CBM-181229.docx - Supplemental material
Supplemental material, sj-docx-1-cbm-10.3233_CBM-181229.docx
Footnotes
Acknowledgments
We would like to give our sincere gratitude to the reviewers for their constructive comments.
Conflict of interest
The authors declare that they have no conflict of interest.
