Abstract
Protocadherin8 (PCDH8), an integral membrane protein, was reported to be a tumor suppressor involved in tumorigenesis in cancers. We aimed to investigate the expression of PCDH8 and its clinicopathological significance in hypopharyngeal carcinoma. We also examined the possible inactivation mechanism of PCDH8. A total of 80 pairs of hypopharyngeal carcinoma tumor tissues and non-tumor tissues were investigated to examine the immunohistochemical expression of PCDH8. Prognostic value and clinicopathological significance of PCDH8 expression were examined by Kaplan-Meier and log-rank test and Cox’s ones. Ten pairs of tumor tissues and non-tumor tissues were analyzed by RT-PCR and 4 pairs by Western blot respectively. Promoter methylation of PCDH8 was observed in 14 pairs of tumor tissues and non-tumor tissues by methylation-specific PCR (MSP). The expression of PCDH8 in tumor tissues was depressed immunohistochemically when compared with non-tumor tissues and was significantly lower in the advanced pathological stage. Meanwhile, the expression of PCDH8 served as an independent prognostic risk factor for overall survival of hypopharyngeal carcinoma. The mRNA and protein levels of PCDH8 in tumor tissues was also down-regulated than in non-tumor tissues. Moreover, aberrant promoter methylation of PCDH8 occurred frequently in tumor tissues, rather than in non-tumor tissues. For the first time, our study demonstrates the tumor-suppressive function of PCDH8 and its epigenetic inactivation by promoter methylation in hypopharyngeal carcinoma. It suggests that PCDH8 may serve as a useful prognostic biomarker and a potential therapeutic target for hypopharyngeal carcinoma patients.
Introduction
Hypopharynx, also known as laryngopharynx, located behind the throat and on both sides, starting from below the hyoid extension line, ends at the lower edge of the annular cartilage connected to the esophagus. The incidence of hypopharyngeal carcinoma lacks detailed statistical data. Hypopharyngeal carcinoma accounts for about 5% of head and neck cancer, and is predominantly squamous cell carcinoma [19]. Hypopharyngeal carcinoma includes primary hypopharyngeal therioma and adjacent advanced cancer invasion [21]. Because of the special anatomical structure, the pathogenesis of hypopharyngeal carcinoma is concealed, and the exposure of Hypopharynx is difficult. Due to the late occurrence of clinical symptoms, the clinical pathological stage is often advanced (stages III and IV), meanwhile, about 50% of patients accompany with cervical lymph node metastasis. Therefore, the prognosis of hypopharyngeal carcinoma is generally poor [3]. Although advancements have been achieved in treating head and neck cancer with adjuvant chemotherapy, radiotherapy, and targeted molecular therapies, surgical intervention is still the main clinical treatment. According to recent studies, the 5-year survival rate of radiotherapy alone is 12.7%
The protocadherins (PCDHs), a subfamily of the cadherin superfamily, contain 6 extracellular cadherin domains, a transmembrane domain, and different cytoplasmic domains [5, 22]. The PCDHs play a mediating role as regulators of other molecules and cell-cell adhesion [22]. As a member of the PCDHs, PCDH8 is reported to play a significant role in cell adhesion, signal transduction, proliferation, migration, and invasion in nasopharyngeal carcinoma [1], breast cancer [9], renal cell carcinoma [15], gastric cancer [2] and bladder cancer [27]. Meanwhile, the expression of PCDH8 is frequently depressed in these human cancers. However, whether this gene is subject to down-regulated expression in hypopharyngeal carcinoma remains unclear.
As is well-known that epigenetic changes are frequently involved in tumor development and progression, including DNA promoter CPG island (CGI) methylation and histone modifications [16]. Therefore, identification of epigenetic changes involved in the initiation and progression of hypopharyngeal carcinoma may identify novel diagnostic and epigenetic biomarkers and therapeutic targets for early detection of hypopharyngeal carcinoma. According to series of studies, epigenetic mechanism plays a crucial role in tumor development and progression. A numbers of genes, such as Zinc-Finger Protein 545 [25], Jagged1 [28], LATS2 [29] and ZNF382 [26], have already been reported to be frequently silenced by the methylation of DNA promoter, serving as tumor suppressors. However, such epigenetic changes of PCDH8 have not been reported yet in hypopharyngeal carcinoma.
In the present study, we verified the depressed expression and promoter methylation of PCDH8 in isolated hypopharyngeal carcinoma tissues of surgical patients. We also evaluated the relationship between the expression of PCDH8 and clinicopathological features of hypopharyngeal carcinoma. Moreover, we examined the correlation between PCDH8 expression and the patients’ overall survival. On the basis of the research, we found that PCDH8 may be a functional tumor suppressor in the development and progression of hypopharyngeal carcinoma. Furthermore, PCDH8 may serve as a prognostic biomarker for hypopharyngeal carcinoma patients.
Materials and methods
Patients and tissue samples
A total of 80 pairs of tumor tissues and adjacent non-tumor tissues were obtained from 80 patients with hypopharyngeal carcinoma during laryngopharyngectomy at the First Affiliated Hospital of Chongqing Medical University between 2012 and 2017. Each pair of tumor tissue and adjacent tissue was obtained from isolated hypopharyngeal carcinoma tissues resected from the surgery. These tissues were examined by experienced pathologists according to the World Health Organization classification to exclude the possibility of incidental tumors. Informed consent was obtained from each participant before any treatment. Significantly, none of the patients with hypopharyngeal carcinoma received any form of anti-tumor therapy or adjuvant chemotherapy before surgery. This study was approved by the ethics committee of Chongqing Medical University. All tissue samples were flash-frozen in liquid nitrogen at the time of collection and then stored at
Immunohistochemistry
Immunohistochemistry for PCDH8 was performed on 4
The immunohistochemistry results were analyzed by two different experienced pathologists and scored using a double-blind method. Five high-power fields (
Total-RNA extraction and RT-PCR
According to the instructions for the Trizol total RNA extraction kit (TaKaRa, Japan), total RNA was extracted from 10 pairs of tumor tissues and adjacent non-tumor tissues of patients with hypopharyngeal carcinoma mentioned above. The harvested RNA was reverse transcribed into cDNA using PrimeScript RT reagent Kit with gDNA Eraser (TaKaRa, Japan) and then stored at
Protein extraction and Western blotting analysis
Proteins were extracted from tumor tissues and adjacent non-tumor tissues using radio immunoprecipitation assay (RIPA) lysis buffer (Beyotime, Shanghai, China), followed by centrifugation at 12000
DNA extraction and methylation-specific PCR (MSP)
Genomic DNA was extracted from tumor tissues and adjacent non-tumor tissues using GenElute Mammalian Genomic DNA Miniprep Kit (SIGMA- ALORICH, St. Louis, MO, USA). The integrity of DNA was detected by gel electrophoresis. Samples were stored at
(A) Positive immunohistochemistry for PCDH8. The numbers of PCDH8 positive cells were more than 50%. (B) Negative immunohistochemistry for PCDH8. The numbers of PCDH8 positive cells were less than 50%. (C) Slide without primary antibody incubation was selected as negative control. The magnification of all photomicrographs was 
Statistical analyses were performed with SPSS version 22.0 software (SPSS Inc, Chicago, IL, USA). The associations between the expression of PCDH8 and clinicopathological features were evaluated by chi-square test. For overall survival analysis, Kaplan-Meier survival analysis was used and the differences in survival were analyzed using the log-rank test. Univariate and multivariate Cox proportional hazard models were used to evaluate the prognostic effect of PCDH8 expression in hypopharyngeal carcinoma. The difference of PCDH8 methylation status between tumor tissues and non-tumor tissues were evaluated using Fisher’s exact test. Results for the RT-PCR assays were presented as mean
Results
Identification of PCDH8 deletion in immunohistochemistry
According to the Immunohistochemistry score method described previous, the expression of PCDH8 in 80 tumor tissues and 80 adjacent non-tumor tissues was evaluated to be positive or negative (Fig. 1). More importantly, we found that the expression of PCDH8 was significantly down-regulated in tumor tissues (60/80, 75.0%) when compared to the adjacent non-tumor tissues (18/80, 22.5%) (
The immunohistochemical expression of PCDH8 in tumor tissues and non-tumor tissues
The immunohistochemical expression of PCDH8 in tumor tissues and non-tumor tissues
Correlations between expression of PCDH8 and clinicopathological features of hypopharyngeal carcinoma patients
Univariate and multivariate Cox regression analyses of overall survival in hypopharyngeal carcinoma patients
Statistical analyses of the correlation between the expression of PCDH8 and clinicopathological parameters are summarized in Table 2. Obviously, the expression of PCDH8 was down-regulated in advanced pathological stage (T3–T4) compared to early pathological stage (T1–T2) (
Correlations between the expression of PCDH8 and overall survival in patients with hypopharyngeal carcinoma. Patients with PCDH8 positive expression showed significant longer overall survival than those with PCDH8 negative expression (log-rank test, 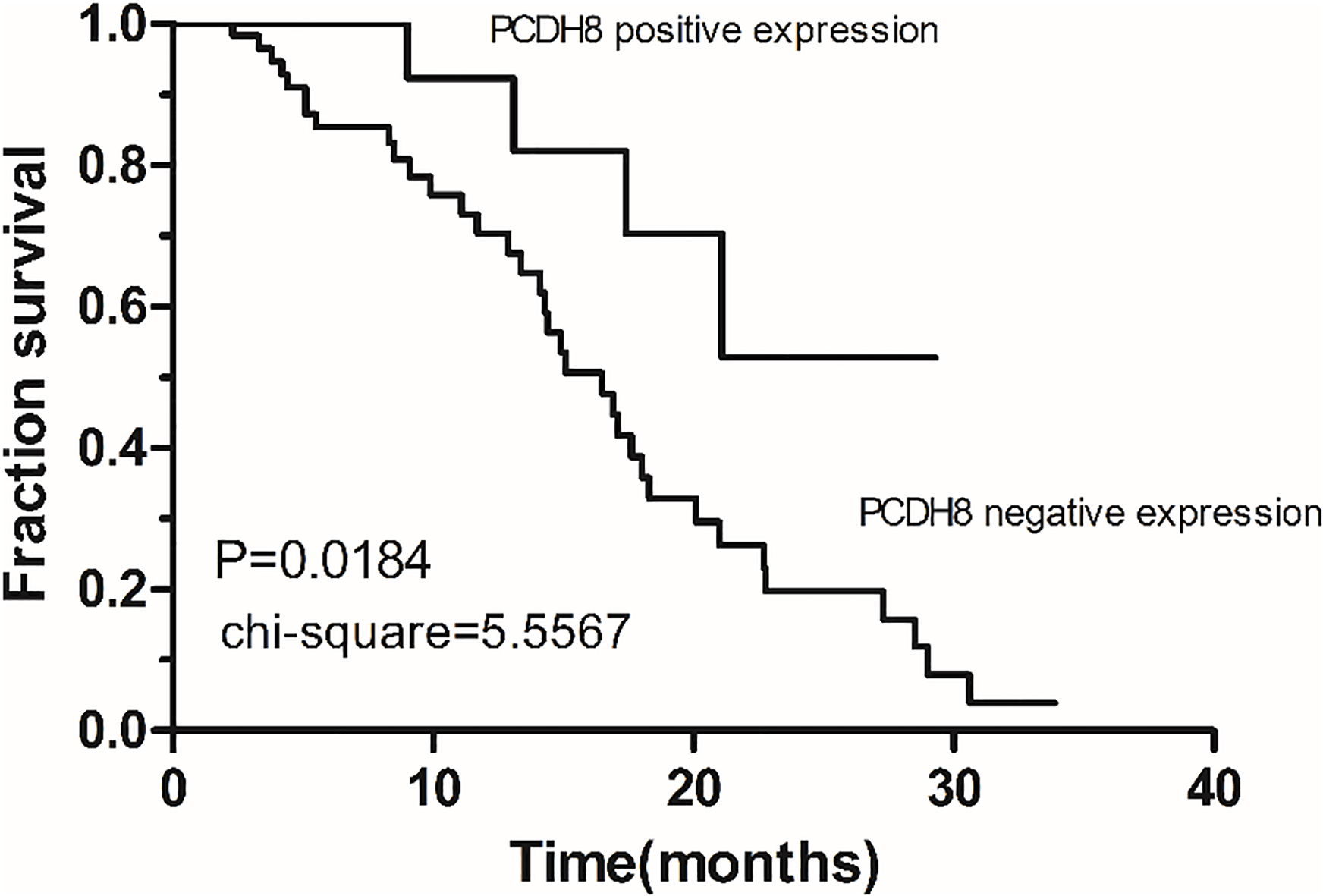
To investigate whether PCDH8 was aberrantly expressed in hypopharyngeal carcinoma, we first examined PCDH8 mRNA level in 10 pairs of hypopharyngeal carcinoma tumor tissues and adjacent non-tumor tissues using real-time PCR (RT-PCR). We found that PCDH8 expression was significantly downregulated in 100% (10/10) of hypopharyngeal carcinoma tumor tissues when compared to the adjacent non-tumor tissues (
The expression level of PCDH20 in tumor tissues and non-tumor tissues was measured by quantitative RT-PCR and Western blot assay, respectively. (a) Expression of PCDH8 mRNA level by real-time RT-PCR. The abscissa indicates the type of tissues. The ordinate indicates the relative mRNA expression levels of PCDH8. The line in the grouped column scatter represented mean values of PCDH8 mRNA (
In the current study, the methylation status of PCDH8 in 14 pairs of hypopharyngeal carcinoma tumor tissues and non-tumor tissues was detected by methylation-specific PCR(MSP). PCDH8 methylation was detected in 7 (50.0%) tumor tissues compared to 1 non-tumor tissue (7.1%) (Fig. 4). Meanwhile, the difference between tumor tissues and non-tumor tissues was statistically significant (
The Methylation of PCDH8 in tumor tissues and non-tumor tissues
The Methylation of PCDH8 in tumor tissues and non-tumor tissues
Representative analysis of methylation of the PCDH8 promoter in hypopharyngeal carcinoma tumor tissues and adjacent non-tumor tissues. Methylation-specific polymerase chain reaction (MSP) results are shown. M: methylated; U: unmethylated; N: non-tumor tissue; T: tumor tissue.
Hypopharyngeal squamous cell carcinoma (HSCC) is one of the most common and aggressive head and neck cancers with poor prognosis and high recurrence [8, 24]. Although advances have been made in surgery, radiotherapy and chemotherapy, the 5-year survival rate of patients with HSCC ranges from 40 to 50%, with a morbidity of 1 case per 100,000 people [6, 12, 23]. It is of great important to identify novel prognostic and predictive markers to understand this multifaceted disease process and to identify which patients need more aggressive treatment after initial curative surgery. Fortunately, PCDH8 has been reported as an effective tumor suppressor involved in tumorigenesis in cancers [1, 2, 9, 15, 27], according to a variety of clinical studies and cytology experiments. Meanwhile, the decreased expression of PCDH8 has also been verified in these cancers above, contributing to its down-regulated tumor-suppressive function. However, whether the expression of PCDH8 is reduced in hypopharyngeal carcinoma and whether this gene can be a potential tumor biomarker and therapeutic target in hypopharyngeal carcinoma remain unclear.
The protocadherins (PCDHs), different from classic cadherins, are not just cell-adhesion proteins involved in homophilic interactions, and their heterophilic interactions with other molecules may be more significant for their various physiological functions [5, 22]. DNA methylation of CpG islands within the promoter region of genes is an alternative mechanism of gene silence to genetic changes and is frequently involved in the development and progression of many types of human cancers. Aberrant promoter methylation of some genes that are normally unmethylated may be used as potential molecular markers for the diagnosis, surveillance, and prognosis in hypopharyngeal carcinoma. As for several members of PCDHs, expression silence or functional inactivation, caused by mutations or promoter methylation, for example, PCDH8 [1, 2, 9, 15, 27], PCDH10 [4, 10, 11], PCDH17 [20], and PCDH20 [7], has been discovered in varies of cancers, resulting in tumor cell growth, invasion, and metastasis. These results are complementary to our data and also hint that epigenetic inactivation of PCDHs in tumors could be widespread indeed. Therefore, Protocadherin8 (PCDH8) may have multiple tumor-suppressive functions, such as the involvement in cell-cell adhesion, signal transduction, and growth control. However, the association between the function of PCDH8 and the prognosis of hypopharyngeal carcinoma have not been reported yet. This is the first study to investigate the prognostic value of PCDH8 expression and methylation in hypopharyngeal carcinoma based on a relatively large number of clinical samples.
In the current study, we found that PCDH8, localized on human chromosome 13q14.3, was frequently downregulated or totally silenced in 75% hypopharyngeal carcinoma tumor tissues (60/80), but widely expressed in non-tumor tissues (62/80) according to Immunohistochemistry. More importantly, we found that depressed PCDH8 expression was significantly correlated with advanced pathologic stage. Meanwhile, PCDH8 was proved to serve as an independent prognostic risk factor for overall survival of hypopharyngeal carcinoma patients according to Kaplan-Meier & log-rank test and Cox’s ones. Whether it implicates an early diagnosis of carcinogenesis should be an interesting question. Further, we found that PCDH8 functions as a tumor suppressor by RT-PCR and Western blotting analysis. Our data clearly demonstrated that mRNA level and protein expression of PCDH8 is frequently decreased in tumor tissues compared to paired non-tumor tissues. These results suggest that PCDH8 plays an important role in the progression of hypopharyngeal carcinoma. Moreover, methylation-specific PCR (MSP) demonstrated that aberrant promoter methylation of PCDH8 occurred frequently in tumor tissues, rather than in non-tumor tissues. This result indicates that PCDH8 methylation is tumor-specific and may be an early event in development and progression of hypopharyngeal carcinoma.
In summary, we demonstrated in this study that PCDH8 can inhibit the development and progression of hypopharyngeal carcinoma, and examined the clinicopathological significance and prognostic value of PCDH8 expression. We also verified the frequent promoter methylation in hypopharyngeal carcinoma in a tumor specific manner as a possible epigenetic inactivation mechanism. Our data strongly support the notion that PCDH8 is a candidate tumor suppressor in hypopharyngeal carcinoma and may serve as a useful prognostic biomarker and a potential therapeutic target for hypopharyngeal carcinoma patients. It would thus be worthwhile to conduct further study to investigate the cause of PCDH8 methylation and to discover how to reverse the methylation status of PCDH8 in human hypopharyngeal tissues. Meanwhile, the functional mechanism of tumor suppressor also remains further intensive study.
Footnotes
Acknowledgments
This study was supported by the Natural Science Foundation of Key Projects of Chongqing China (Grant Number. cstc2012jjB10015). All authors thank Department of Biochemistry and Molecular Biology, Molecular Medicine and Cancer Research, China Center, Chongqing Medical University for technical assistance and encouragement. The authors contributed equally to this work.
Conflict of interest
The authors declare no conflict of interest.
