Abstract
AIMS:
To evaluate the prognostic and clinicopathological features of glioma with BRMS1L expression.
METHODS:
Total 120 glioma samples were obtained as discovery cohort. CGGA, GSE and TCGA datasets were obtained as validation sets. Furthermore, Kaplan-Meier survival and multivariate Cox analysis were used to evaluate the survival distributions. Moreover, the functional role of BRMS1L was also analyzed by transwell assay.
RESULTS:
In the discovery cohort, decreased BRMS1L expression was significantly associated with high-grade glioma as well as the higher mortality in survival analysis (log-rank test,
CONCLUSION:
BRMS1L plays as an anti-oncogene in GBM and indicates a new potential therapeutic target.
Keywords
Introduction
Glioma is the most common type of primary brain tumor, patients with gliomas have an invariable fatal outcome and poor prognosis [1, 2, 3, 13]. According to immunophenotypical similarity to a cell of putative origin, gliomas are subgroupped into astrocytoma, oligodendroglioma and oligoastrocytoma [9]. World Health Organization (WHO) grades II, III and IV gliomas are malignant, diffuse, and incurable [2]. Patients with grade IV gliomas (glioblastomas; GBM) suffers average survival barely exceeding 16 months even with the most aggressive therapies [5]. The GBM cells is characterized by uncontrolled growth, migration and spread throughout the brain tissue [6]. Even though increasing studies reveal underlying mechanisms of the malignant properties of glioma cells and explore reasonable therapeutic targets, the existing clinical prediction and treatment efficacy are still not satisfactory [5].
(A) BRMS1L expression is positively correlated with tumor grade. This result is validated by GSE 16011 microarray database (B), CGGA RNAsequencing database (C), and TCGA RNAsequencing database (D). A single spot is the BRMS1L expression value of an individual patient. Lines in the middle are the mean expression value. Error bars represent standard deviation (SD). * 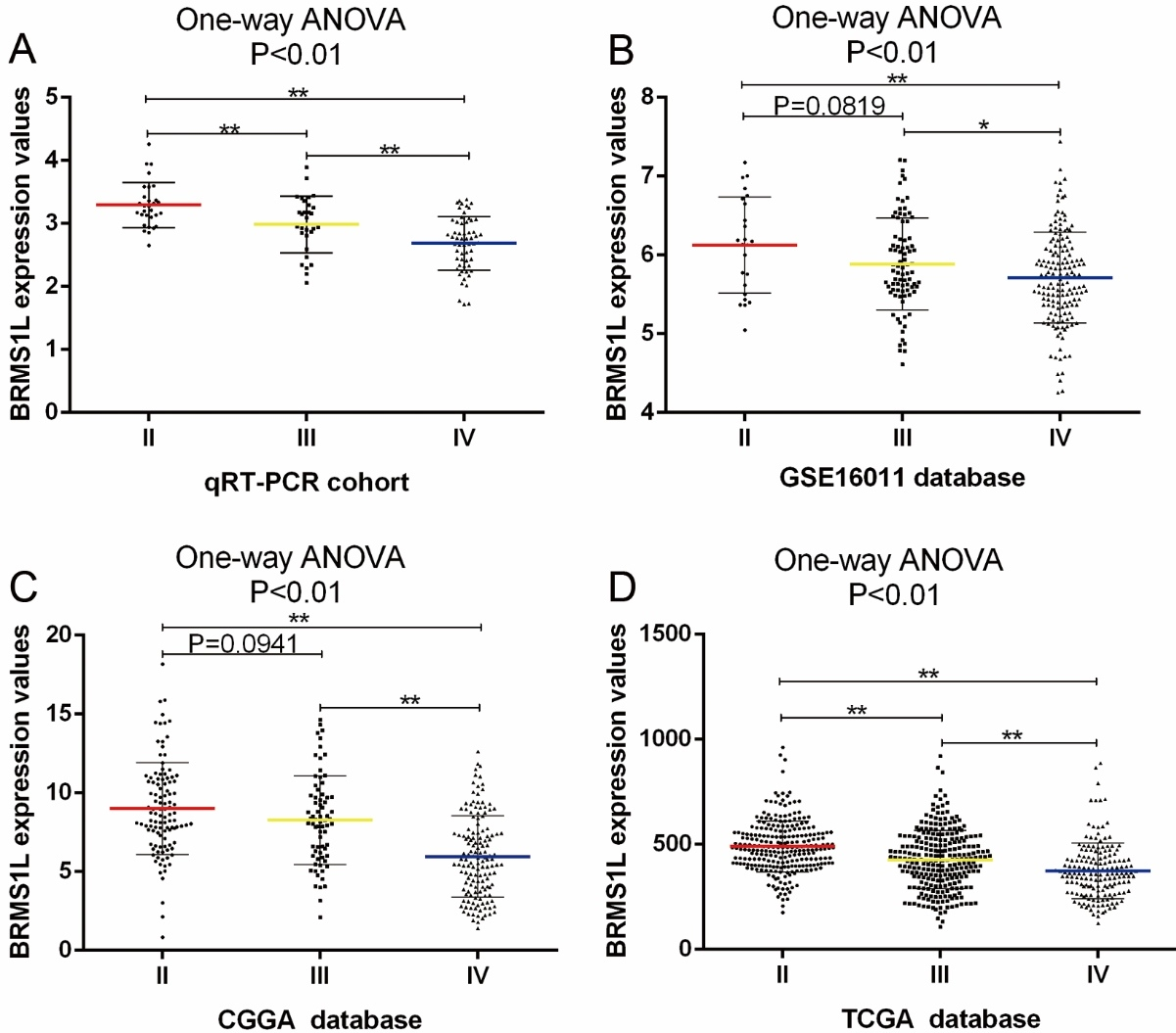
BRMS1L (breast cancer metastasis suppressor 1 like, BRMS1-like) is involved in Sin3A–histone deacetylase (HDAC) co-repressor complex that blocks target gene transcription [4]. Through recruitment of HDAC1 and deacetylation of histone H3K9 at the promoter, BRMS1L modulates epigenetic silencing of Wnt signaling receptor and inhibits breast cancer metastasis [4]. Downregulated BRMS1L expression also contributes to epithelial-mesenchymal transition and invasion in breast cancer [15]. Clinically, reduced BRMS1L in breast cancer is found to be correlated with metastasis and poor survival, also validating BRMS1L is an important breast cancer metastasis suppressor [4]. Meanwhile, the treatment of human lung cancer resulting in downregulated BRMS1L expression also indicates a possible anti-oncogene in lung cancer [8]. Recently, a Glioma Associated Protein-Protein Interaction Network analysis predicts the potential involvement of BRMS1L in the pathogenesis of glioma [14]. However, the explicit role of BRMS1L in glioma malignant phenotype and prognosis prediction needs to be assured.
In the present study, we demonstrated that BRMS1L expression was inversely correlated with glioma grade. Forced BRMS1L expression inhibited aggressive invasion of GBM cells. Loss of BRMS1L in GBM predicted a poor survival by our discovery cohort, GSE16011 and TCGA cohort. Targeting BRMS1L is promising to a novel therapeutic target in GBM treatment.
Glioma samples and patients
Total 120 glioma samples (30 grade II, 30 grade III, and 60 grade IV) were confirmed by pathological diagnosis according to the 2016 WHO classification. After resection, all samples were immediately frozen in liquid nitrogen until RNA extraction. Extent of resection was graded as total or subtotal resection by using MRIs obtained within 72 h after surgical resection by two independent radiologists. Overall survival time (OS) was calculated from the date of histological diagnosis until death or the last follow-up. This study was approved by the institutional review boards of Nanjing Medical University, and written informed consent was obtained from all patients.
The prognostic value of BRMS1L in GBM patients. Low BRMS1L expression confers a poor survival in 60 GBM patients (A), GSE16011 database (B), CGGA database (C), and TCGA database. High group, patients with higher BRMS1L level than the median value or equal to the median one. Low group, patients with lower BRMS1L value than the median one.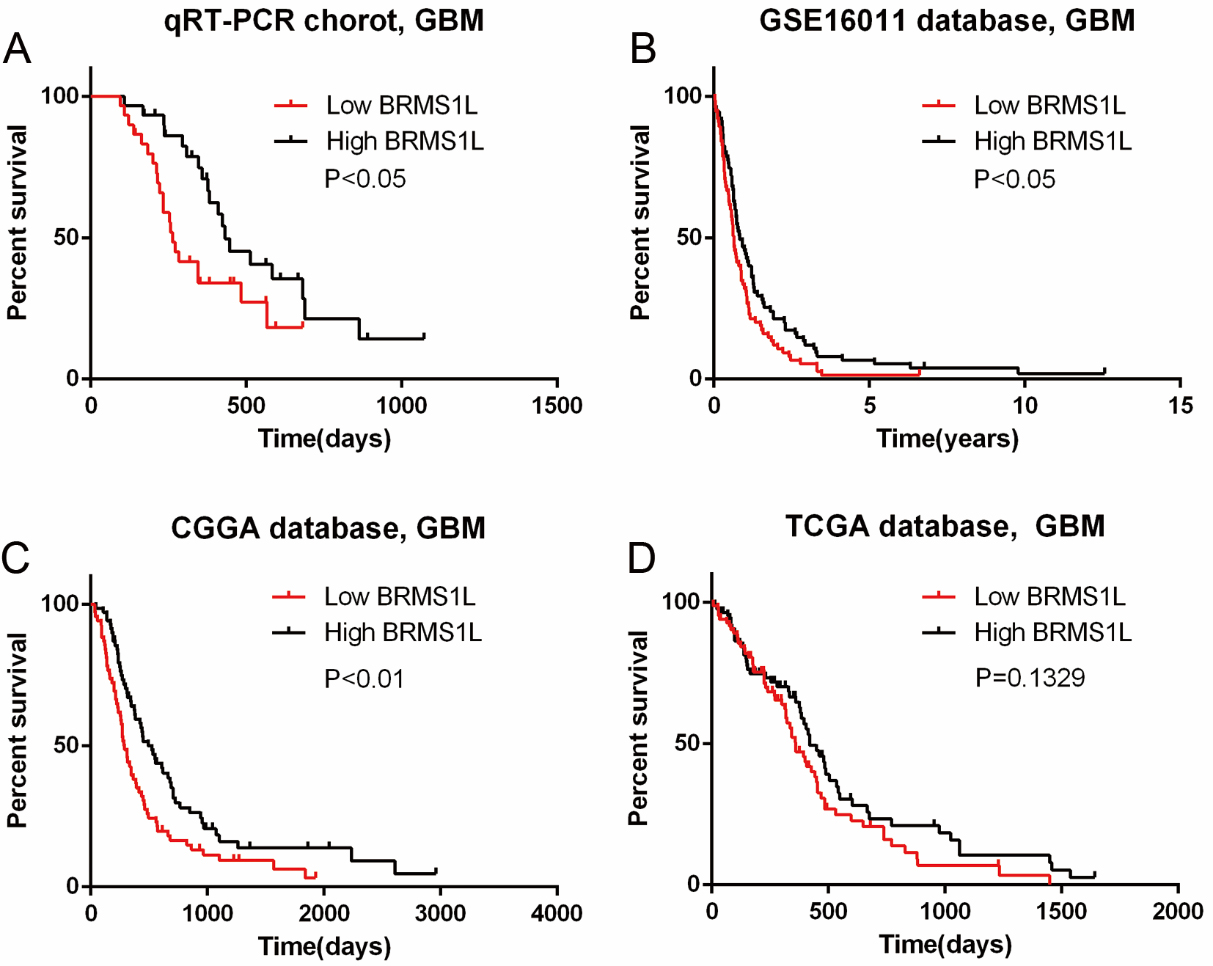
Total RNA was extracted from glioma samples by using TRIzol reagent (Invitrogen). RNA was quantified by absorbance reading at A260/A280
BRMS1L expression analysis in the validation datasets
Chinese Glioma Genoma Atlas (CGGA) RNA sequencing database (
Cell line, cell culture and transfection
U87 and U251 cell lines were purchased from the Chinese Academy of Sciences Cell Bank; cells were maintained in a 37
Invasion assay
Transwell assay was applied to assess cells’ invasion ability, 1
BRMS1L and other clinicopathological factors affecting 60 GBM patients’ overall survival
BRMS1L and other clinicopathological factors affecting 60 GBM patients’ overall survival
HR, hazard ratio.
Seventy-two hours after transfection, total protein was collected using RIPA and quantified by BCA method (KeyGen). Thirty micrograms of protein were subjected to immunoblot analysis with rabbit anti-BRMS1L antibody (Abcam, 1:1000), followed by HRP-conjugated mouse anti-rabbit secondary antibodies. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as control.
Statistical analysis
Statistical analysis was performed on SPSS 16.0 and GraphPad Prism 5.0 statistical software. Student’s t-test and one-way ANOVA test were used to investigate the significance of difference. Kaplan–Meier survival analysis was applied to analyze the survival distributions, and the log-rank test was employed to assess the statistical significance between stratified survival groups. Univariate and multivariate Cox regression analysis were used to evaluate prognostic value of BRMS1L in GBM patients. Patient whose BRMS1L expression was lower than the median value was defined as low expression, while higher than the median value was classified into high expression group. A two-sided
BRMS1L is a tumor anti-oncogene in glioma. (A and B) BRMS1L protein is elevated by BRMS1L over-expression vectors both in U87 and U251 glioma cells. (C and D) Transwell assay shows that both U87 and U251 cells’ invasion ability are significantly inhibited by ectopic of BRMS1L expression. NC means negative control vectors treatment group, Vector means BRMS1L over-expression vectors treatment group.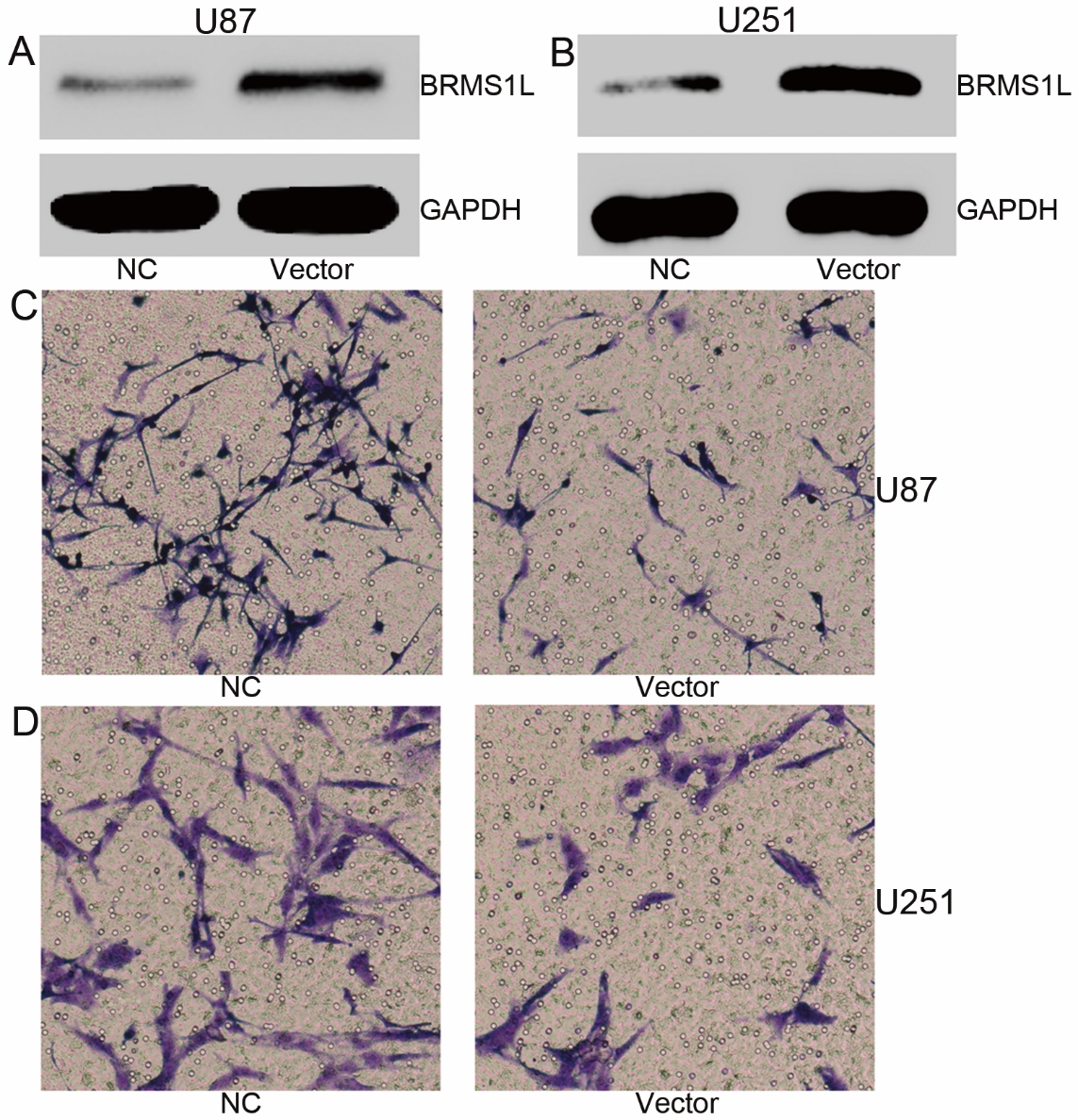
BRMS1L expression is associated with glioma grade
To investigate BRMS1L mRNA expression pattern in glioma, 120 glioma samples (30 grade II, 30 grade III, and 60 grade IV) were analyzed by qRT-PCR, we found that BRMS1L level was negatively correlated with tumor grade (
Reduced BRMS1L expression predicts poor survival in GBM patients
To explore the association of BRMS1L expression level with prognosis of GBM patients, Kaplan-Meier survival curve analysis with a log-rank test of the above 60 grade IV GBM patients was employed, as shown in Fig. 2, GBM patients with lower BRMS1L expression (median survival is 263 days) had a worse overall survival time by contrast to those with higher BRMS1L expression (median survival is 432 days) (
BRMS1L is an independent prognostic biomarker for GBM patients
To further reveal the prognostic value of BRMS1L in GBM patients, a univariate Cox regression analysis was used. As shown in Table 1, lower BRMS1L expression was a risk factor for GBM patients. Additionally, other factors including ender age (
BRMS1L suppresses glioma cell invasion
To assess the functional role of BRMS1L in GBM, gain-of-function assay was done by transfecting BRMS1L overexpression vectors into U87 and U251 glioma cell lines to elevate BRMS1L expression (Fig. 3A and B), and we found that ectopic of BRMS1L suppressed glioma cells’ invasive ability both in U87 and U251 glioma cells (Fig. 3C and D).
Discussion
In spite of some minor improvements in progression-free survival (PFS) of GBM by the combination of temozolomide (TMZ) and other targeted treatments, no satisfactory overall survival has been accomplished [12]. Thus, it is an urgent need that we advance glioma profiles understanding and identify novel genetic alterations of glioma, especially in primary and recurrent GBM. This could contribute to a deeper understanding of the characteristics of GBM, and imply potential targets for personalized therapeutic strategies [7, 11]. Aberrant BRMS1L expression plays a crucial role in carcinogenesis of breast cancer [4]. A previous study have also identified the potential role of loss BRMS1L in the pathogenesis of glioma [14]. Thus, we hypothesized that BRMS1L loss might be associated with glioma. To test this hypothesis, we analyzed the expression of BRMS1L in our cohort and other several databases. We found that BRMS1L levels were significantly lower in GBM compared with low grade glioma (WHO grade II and III) in all cohorts. In survival analyses, our cohort also indicated that the down-regulated expression of BRMS1L correlated with poor outcome in GBM patients and could be used as an independent diagnostic marker. Cross-validation from the CGGA and cross-validation from the GSE16011 and TCGA cohorts also confirmed BRMS1L as a significant biomarker for predicting GBM patients’ outcome.
The invasive property of brain-tumor cells makes an important contribution to the ineffectiveness of current treatment modalities [10]. To further understand biological role of BRMS1L in glioma invasive phenotype, we overexpressed BRMS1L in U87 and U251 GBM cells and detected their invasion change. And we found that ectopic expression of BRMS1L inhibited GBM cells’ invasion ability. However, further in vivo model invasion assay of GBM is needed to prove BRMS1L targeting to be a useful therapeutic strategy, which can impede the malignant characteristics of GBM.
In summary, low levels of BRMS1L is associated with glioma progression and plays as an independent poor prognosis biomarker. The up-regulation of BRMS1L suppresses glioma cells’ invasion. All of those indicate that BRMS1L is a novel prognostic biomarker with potential anti-invasion therapeutic implications in GBM.
